Abstract
Background and Objective
Ni is an important metal found in the human environment, including air, water, soil, food, kitchenware, and jewelry. It is widely used in electrical engineering, medicine, and industry. Ni compounds, whether insoluble (such as oxides and sulfides) or soluble (such as sulfates, chlorides, and acetates), pose serious risks to the environment, ecosystems, and human health. Such contamination can lead to harmful effects, especially on the respiratory, digestive, and skin systems. This study aims to emphasize the importance of early detection and diagnosis of Ni poisoning, as well as urgent measures and treatments, especially the use of chelators, in addition to the use of nanoparticles to remove nickel compounds from the environment.
Methods and Result
This review article draws on sources from PubMed, Scopus, various citations, and the Toxicologic Emergencies reference book spanning 1996–2025. The findings indicate that high-level exposure to nickel compounds leads to the formation of reactive oxygen species, suppression of DNA repair mechanisms, and disruption of signal transduction pathways. These effects result in genotoxicity, carcinogenicity, immunotoxicity, and poisoning across different species.
Conclusion
A thorough history and examination are essential for evaluating blood and urine nickel levels, as they directly relate to the severity of poisoning. Preventive measures involve avoiding nickel compounds and facilitating removal from the body. Allergic contact dermatitis is treated with antihistamines, corticosteroids, and calcineurin inhibitors like tacrolimus. For nickel poisoning treatment, chelators such as diethyl-dithiocarbamate (DDC) and disulfiram are prescribed based on urine concentration. Recently, nanoparticles have been used to remove nickel compounds from the environment.
Keywords
Introduction
Access to chemical substances and compounds has become easier over the years, leading to increased usage.1,2 Negligent or intentional use of these chemicals can result in poisoning.3,4 Some of these substances include essential trace elements and heavy metals, found in nature, food, industrial compounds, and as nutritional, sports, and medical supplements. Excessive and prolonged exposure to these materials can cause poisoning and harm to human organs.5,6 The use of nickel reaches back to 3500 years BC in ancient times. In the 17th century, the discovery of nickel ore was intertwined with superstitions. German miners, searching for copper ore, found a previously unknown mineral and named it “kupfernickel” (copper-nickel), meaning “evil eye”; today, this is recognized as nickel arsenide or niccolite.7,8 In 1751, Swedish mineralogist Baron Axel Frederick Cronstedt identified nickel by heating copper-nickel with charcoal and observing its distinct properties, leading him to introduce it as a new element and name it “nickel”. 8
Physical - chemical properties and sources of nickel compounds
Ni is the fifth most abundant element on Earth, and its concentration below the crust is 100 times greater. It is the second most abundant element in the Earth’s inner core, following iron. Represented by the symbol Ni, nickel is a solid, silvery-white metal at room temperature with malleable and ductile properties. Additionally, nickel exhibits high electrical and thermal conductivity among transition metals. It has an atomic number of 28, atomic weight of 58.6934, density of 8.912 g per cubic centimeter, melting point of 1455°C, and boiling point of 2913°C. Naturally occurring nickel has five stable isotopes; 58Ni [most abundant (68.077% of the total)], 60Ni, 61 Ni, 62Ni, 64Ni.7,8
Nickel is widely present in the environment, including air, water, soil, household kitchen equipment, electrical appliances, and food.
Air
Airborne nickel exposure varies based on industrial activity. In remote areas, typical levels range from 0.00001 to 0.003 μg/ m3; in urban locations without factories, it ranges from 0.003 to 0.03 μg/m3; and in areas with metallurgical industries, levels can reach 0.07 to 0.77 μg/m3. In nickel-producing or consuming industries, exposure may be as high as 100 μg/day. The primary routes of exposure are inhalation and skin contact, with inhalation involving insoluble nickel compounds in dust, suspended particles, and gaseous forms such as nickel carbonyl.9,10
According to the above, nickel has a wide range of occupational exposures in industries, and it has been shown that workers in various industrial processes, for example, in metal cutting and forming activities, metal spraying, sintering furnace, chemical production, manufacturing of glass, batteries and accumulators and some welding processes, are exposed to high levels of nickel. Based on the findings of Scientific Committee on Occupational Exposure Limits (SCOEL), to protect workers against carcinogenesis, the concentration should not exceed 10 μg/m3 in workplaces
11
(Figure 1). Various available sources of Nickel. “Created by the authors”.
Water
Ni is prevalent in both surface and groundwater due to atmospheric deposits, rain, and soil runoff, which increase its concentration. 10 As a result, environmental waters are rich in nickel, making monitoring for pollutants essential. Water quality is crucial for supporting living organisms. Rivers typically contain about 0.3 ppb, while seawater has 0.5–2 ppb of nickel. In drinking water, nickel concentrations below 10 μg/ L lead to an average daily intake of 7.5–15.0 μg for individuals consuming 1.5 L daily. Excessive nickel intake from drinking water rarely leads to health issues9,12 (Figure 1).
Soil
Ni enters the soil through atmospheric interactions and rainfall, with concentrations reflecting those of the main rock types. 10 Its levels are influenced by industrial and agricultural practices, as well as by soil formation and pollution. Geological studies show that nickel concentrations in soil range from 3 to 1000 ppm, with the lowest levels found in clay, sandstone, limestone, and shale, and the highest in igneous rocks. The accumulation of nickel from sewage waste in urban, industrial, and agricultural areas poses significant health risks to the community 9 (Figure 1).
Food
Key dietary sources of nickel include grains and grain products (such as buckwheat and oats), non-alcoholic beverages (excluding milk), legumes, nuts, oilseeds, certain fruits (like almonds and plums), vegetables (including spinach and peas), cocoa, dark chocolate, sweets, and seafood (such as crab and salmon).10,13,14 Additionally, cooking utensils, particularly stainless steel cookware, can elevate the nickel content in food. 10 Studies indicate that nickel intake varies across countries, with an average daily consumption ranging from 100 to 300 μg 9 (Figure 1).
Role of nickel in biological systems
Ni has been identified and assessed in abundance in all parts of the biosphere. It plays a role as an essential and fundamental element for several biological and biochemical processes. Nickel deficiency causes reduced growth and development, behavioral changes, increased perinatal mortality, as well as reduced iron absorption, leading to anemia. However, nickel deficiency in humans rarely occurs because nickel is abundant in nature. In addition, nickel is present in almost all types of food, which is sufficient to maintain nickel. The nickel requirement in humans is estimated to be between 5 and 50 μg/ day. 15
On the contrary, nickel alone and in combination with other elements in varying amounts is continuously exposed to humans and enters the body through inhalation, ingestion and skin absorption. Due to its usefulness in industries and commercial applications, this metal is found in various alloys as nickel plating, in catalysts, pigments, and in nickel-containing tools. This exposes the workforce to significant amounts of airborne nickel, increasing the occupational nickel hazard. Significant amounts are ingested through drinking water, due to leaching or corrosion of drinking water pipes and containers. In addition, skin contact with many metal objects can cause contact dermatitis and nickel allergy in individuals.15,16
Insoluble nickel particles are phagocytosed and lipid-soluble nickel carbonyls are able to cross the plasma membrane by diffusion or calcium channels. Nickel enters the cell via calcium channels and/or divalent cation transporters (DMT-1) with the help of iron. Nickel is transported in the blood plasma by proteins such as albumin, amino acids such as histidine, and alpha-2-macroglobulin, and is thus distributed throughout the tissues. 15
Adverse effects of the compounds have been shown in 10% of women and 2% of men. It causes changes in metabolism, inflammation, oxidative stress and the production of radical oxygen species, cell proliferation, and cell death. In doses greater than 0.5 gr orally, it causes poisoning in humans, and consumption of nickel close to 1 gr causes death.15,16
The presence of nickel compounds in numerous sources in various locations poses obvious risks to health and the environment. The present study has attempted to based on existing studies, the physicochemical properties, morphology, size and chemical compositions of nickel, its distribution in various body systems, the adverse effects of the compounds and the health and environmental risks that have arisen, and the lack of global coordination with a common regulatory framework for controlling these compounds in the environment, and nickel accumulation in the body also healthcare professionals with the necessary knowledge, enabling them to educate patients and choose appropriate treatments. Ultimately, if poisoning occurs, the focus will be on treating the poisoning, especially the use of chelators, and reducing mortality rates.
Metabolism
Ni is typically accessible to the general public via air, soil, water, plants, and food, with food being the primary source. Occupational exposure primarily affects workers in nickel production and consumption industries. Additional sources of nickel exposure include cooking utensils, jewelry, and certain medical devices such as dental tools and joint prostheses. 17
Inhalation is the primary route of exposure in occupational settings. The absorption of nickel particles in the respiratory system depends on factors such as particle diameter, solubility, surface area, deposition, ventilation rate, and clearance.18–20
Skin absorption of nickel is limited, but Ni and particles can enter through sweat ducts and hair follicles, with smaller particles being more easily absorbed. Skin absorption is also affected by detergents, solvents, and barriers like clothing or gloves.17,21
Ni distribution and excretion are closely tied to the route of administration and binding to proteins like albumin and metalloproteins. Nickel accumulates in various organs, including the lungs, thyroid, adrenal glands, brain, kidneys, heart, liver, spleen, and pancreas. Most inhaled nickel is excreted through urine, with a small amount in feces. Similarly, nickel absorbed through the digestive system is mainly excreted in urine, while unabsorbed nickel exits through feces. Additionally, hair contributes to the distribution and excretion of nickel, along with sweat and breast milk
17
(Figure 2). Absorption, Distribution, and Excretion of Nickel. “Created by the authors”.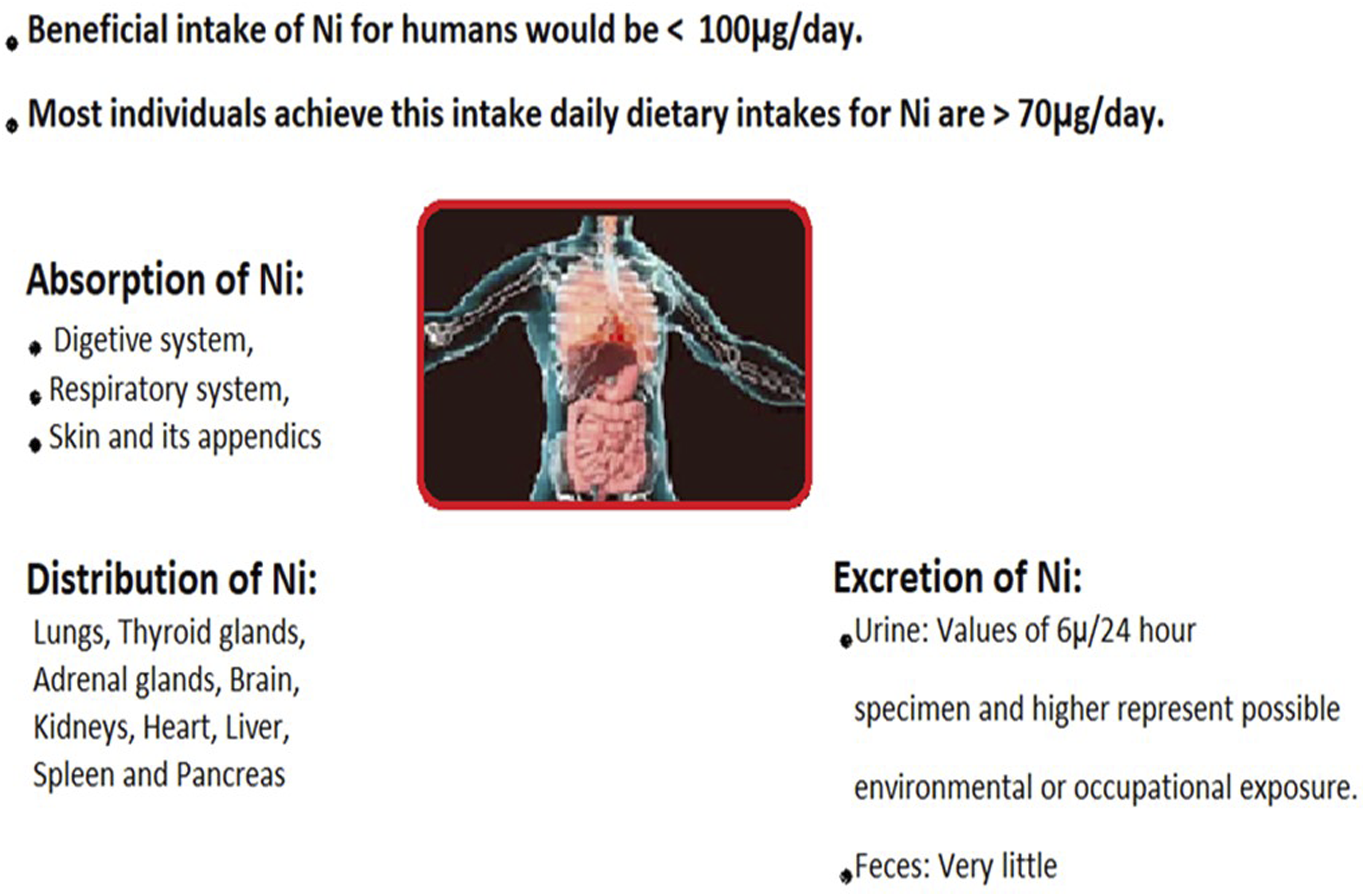
Carcinogenicity of nickel compounds
Due to the abundance of Ni in the environment, human health will always be threatened. This metal is absorbed through the respiratory tract, digestive tract and skin and is always subject to various types of pathological effects. The potential of this metal and its association with the development of cancer in various tissues has been observed in people exposed to nickel compounds. 22 According to the report of the International Agency for Research on Cancer (IARC), soluble and insoluble nickel compounds have been placed in Group 1 (carcinogenic to humans) and nickel and its alloys in Group 2B (possibly carcinogenic to humans). 23
In vitro and in vivo studies have shown that nickel compounds cause toxic effects on the liver, kidneys, lungs, nerves, and immune system, and the possible mechanism behind these diseases is the initiation of increased autophagy, inflammation, stimulation of reactive oxygen species, and apoptosis. 24 Ni compounds cause oxidative stress, which generates reactive oxygen species in cells that damage cellular components and DNA. Studies have shown that DNA damage surveillance mechanisms are disrupted, as DNA damage repair systems are suppressed in various ways.24,25 Cell-level changes due to nickel exposure lead to transcriptional changes, leading to genomic instability. Down-regulation of DNA repair protein pathways and homology-dependent DNA strand break repair (HDR) and mismatch repair (MMR) occur in cells. 26
Another notable point is the lack of regulation in the cell cycle. Studies indicate that the apoptosis pathway is mitochondrial-mediated and passes through the p53-dependent pathway, which will cause G2/M cell cycle arrest. In the meantime, p53 is an important transcription factor that determines whether the cell cycle arrests in G1 phase (G1 and G2 phases act as checkpoints when cells are in S and M phases) or apoptosis occurs. Ni compounds such as Nicl2 and Ni3S2 cause changes in the level of the p53 protein in human cells. This action, by up-regulating Bcl-2-associated protein x (Bax) and down-regulating B-cell leukemia/lymphoma 2 protein (Bcl-2), leads to the increase of p53, which causes the cell to arrest in the G2 phase of the cell cycle.24,27 Another noteworthy point is the imbalance of the Bax/Bcl-2 ratio and depolarization of the mitochondrial membrane action potential in the mitochondrial apoptosis pathway and the activation of some caspases and subsequent cleavage of some caspases, which initiates apoptosis.28,29
The carcinogenicity of nickel compounds leads to transcriptional suppression of DNA repair pathways, and when this incompatibility occurs in the DNA repair processes; this damage is transferred from the mother cells to the daughter cells. Over time, in a multi-step practices, this accumulation of damaged DNA is provided, and on the other hand, the regulatory mechanisms within the cell are gradually destroyed, allowing uncontrolled cell division. This defective transfer will be accompanied by the development of carcinogenesis in organs. 30
Mechanisms of nickel poisoning
Nickel caused oxidative stress
Nickel ions facilitate redox reactions in the human body, leading to the production of reactive oxygen and nitrogen species, as demonstrated in both in vivo and in vitro studies. Nickel compounds, after producing ROS, lead to a decrease in oxygen carrying capacity and changes in the antioxidant defense system, and lower GSH levels. Also, excessive amounts of ROS can lead to DNA oxidaze and inhibition of DNA repair.
31
Reactive oxygen species (ROS) arise within cells through the Fenton and Haber-Weiss reactions. Their levels can be directly measured using spectrofluorometric dichlorofluorescein (DCF) and indirectly assessed by monitoring the reduction of the antioxidant glutathione (GSH). ROS, including free radicals, superoxides, peroxides, and lipid peroxides, can damage cellular proteins, lipids, and DNA10,32 (Figure 3). The figure illustrates the pathways for both soluble and insoluble nickel to enter the cell membrane, along with its distribution and cellular metabolism. Insoluble nickel compounds are phagocytized and stored in cytoplasmic vacuoles near the nucleus, while soluble nickel compounds can enter the nucleus and interact with DNA, potentially causing carcinogenesis. Nickel ions can also enter extracellular fluids via calcium and magnesium channels or transporters like DMT-1, leading to cytotoxicity. Nickel ions can interact with histones, generating reactive oxygen species (ROS), which cause DNA damage, epigenetic changes, and gene silencing. Soluble and insoluble nickel compounds can activate signal transduction pathways, altering gene expression to increase cell survival and proliferation. Nickel compounds induce HIF-1 and its subunits, affecting genes related to energy metabolism, erythropoiesis, angiogenesis, and tumor cell survival. Nickel ions also influence transcription signals like p53, NF-kB, AP-1, TGF-β, and NF-AT, impacting immune responses, proliferation, differentiation, transformation, and apoptosis, ultimately altering cell structure and function. ROS: Reactive oxygen species; DMT-1: Divalent metal transporter-1; HIF-1: Hypoxia inducible factor-1; NF-kB: Nuclear factor kappa-light-chain-enhancer of activated B cells; AP-1: Activating protein-1; TGF-β: Transforming growth factor-β; NF-AT: Nuclear factor of activated T cells; VEGF: Vascular endothelial growth factor. “Created by the authors”.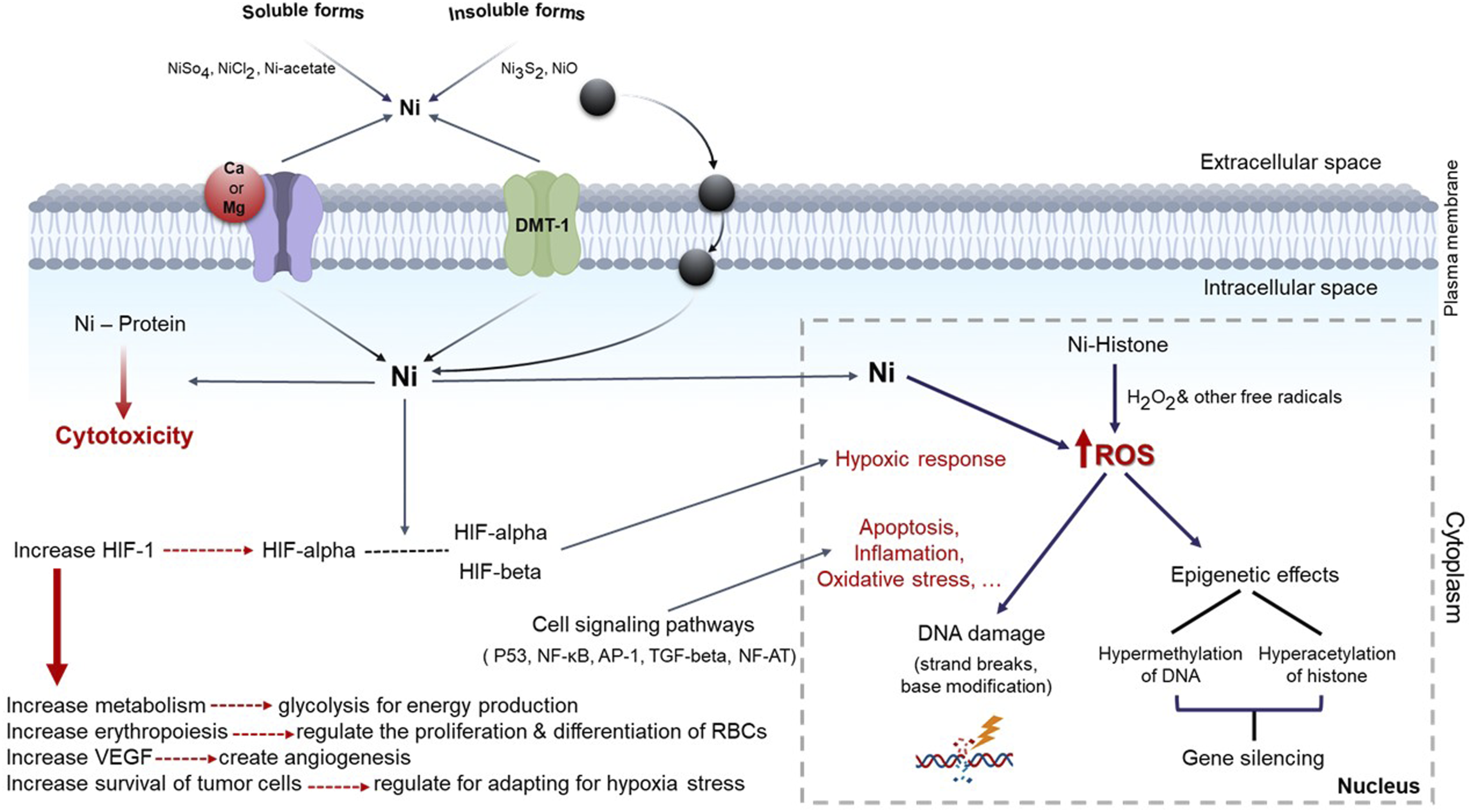
Inhibition of DNA repair
Nickel compounds exhibit weak DNA-damaging properties but can inhibit DNA synthesis. Ni2+ disrupts cell cycle progression and control, leading to a loss of mechanisms for monitoring DNA damage, regulating repair activities, and initiating transcription. Cytometric studies indicate that Ni2+ halts cell division. Even sub-toxic concentrations of NiCl2 can prevent the repair of oxidative DNA damage. Conversely, nickel-nucleic acid histone complexes can induce DNA damage. Nickel binding to DNA chromatin and associated proteins can lead to single-strand breaks and interstrand cross-linking 33 (Figure 3).
Disturbances of signal transcription pathways
Nickel carcinogenesis results in transcriptional repression of DNA repair pathways and potentially plays a significant role in nickel-induced carcinogenesis. This repression of DNA pathways will cause the accumulation of damaged DNA in the cell. If the damaged cells survive, they will be transferred to daughter cells, and this process will contribute to potential carcinogenesis.26,33
Transcription factors
Nickel ions trigger signaling pathways through the generation of reactive oxygen species and alterations in gene expression. Mutations in the p53 gene, a crucial tumor suppressor and transcription factor that regulates cell proliferation and apoptosis, are linked to nickel exposure. p53 mutations are the most prevalent genetic alterations observed in human cancers. 34 Nuclear factor kappa-light-chain-enhancer of activated B cells (NF-kB) regulates genes involved in the innate and adaptive immune responses, promoting cell proliferation and preventing apoptosis. NF-kB is often misregulated in various human tumors. 35 Activator protein 1 (AP-1) is another transcription factor that governs gene expression in response to stimuli like bacterial and viral infections, cytokines, and growth factors, while also influencing processes such as differentiation, proliferation, and apoptosis.36,37 Transforming growth factor beta is part of the transforming growth factor family, which includes three isoforms (TGF-β 1, TGF-β 2, TGF-β 3). It plays a crucial role in immune system regulation, stem cell differentiation, wound healing, and various pathologies, including cancer, autoimmune diseases, and infections 38 (Figure 3).
Hypoxia-inducible factor 1 (HIF-1)
Hypoxia-inducing factor-1 (HIF-1) is crucial for energy metabolism, erythropoiesis, angiogenesis, and cell survival by regulating gene transcription. 39 Hypoxia-inducible factor-1 (HIF-1) is another effective transcription factor influenced by nickel. Under normal oxygen levels, HIF-1α is rapidly degraded by proteasomes. However, in hypoxic conditions, this degradation is inhibited, leading to the accumulation of HIF-1β protein. 40 The stability and activity of HIF-1α depend on various post-translational modifications, including hydroxylation, acetylation, and phosphorylation. Reports demonstrate that HIF-1α activation is closely linked to oncogenic pathways and tumor types 41 (Figure 3).
Genotoxicity of nickel compounds
An increase in abnormalities and chromosomal aberrations has been linked to both insoluble nickel (e.g., Ni3S2) and soluble nickel (e.g., NiCl2 and Ni2+ ions), which alter DNA configuration, leading to cross-linking and strand breaks. These compounds induce mutations in the p53 gene, a tumor suppressor and transcription factor. Nickel fosters conditions that create free radicals, causing irreversible protein degradation and inhibiting DNA repair by binding to repair enzymes. The three primary mechanisms for nickel’s genotoxic effects include the generation of reactive oxygen species, interference with DNA repair processes, and epigenetic changes that promote cell proliferation10,32,42 (Figure 3).
Teratogenicity of nickel compounds
Research has shown that nickel compounds, including nickel carbonyl [Ni (CO)4] and nickel sulfide [Ni2S3], are teratogenic in laboratory animals. These compounds can cross the placenta, leading to fetal toxicity and teratogenic effects. Observed outcomes include cystic lung, exencephaly (sometimes with fused ribs), anophthalmia with cleft palate, microphthalmia, fetal death, chromosomal damage during spermatogenesis, hydrocephaly, exophthalmia, clubfoot, umbilical hernia, and various skeletal abnormalities such as reduced ossification in the nasal, frontal, parietal, and supraoccipital bones, incomplete fusion of the sternum segments, and absence of wrist, metacarpal, tarsal, metatarsal bones, and fingers 43 (Figure 3).
Nickel compounds induced apoptosis
Apoptosis, or programmed cell death, involves chemical events and cellular changes such as cell shrinkage, swelling, nuclear fragmentation, chromatin condensation, DNA fragmentation, and RNA degradation. There are two primary pathways of apoptosis: internal (mitochondrial) and external. Internal pathways arise when cellular stress triggers signals for the release of cytochrome C (Cyt C) from the mitochondrial intermembrane space, leading to self-destruction. External pathways are initiated by extracellular ligands that bind to tumor necrosis factor (TNF) receptors, activating cell death signals from other cells. In cases of nickel ion poisoning, Cyt C is released into the cytosol, activating certain procaspases and caspases. This activation affects Poly (ADP-ribose) Polymerase (PARP), ultimately resulting in cytotoxicity, oxidative stress, mitochondrial damage, and apoptosis 43 (Figure 3).
Clinical Manifestations of nickel poisoning
Nickel compound poisoning can be analyzed through both acute and chronic manifestations, considering the impact on each organ.
Acute nickel exposure
Clinical manifestations of acute exposure vary based on the compounds involved and the route of exposure.
Skin
The most well-known clinical manifestation associated with nickel poisoning is allergic contact dermatitis (ACD), which can be diagnosed by skin patch testing. 44 Nickel dermatitis affects workers exposed to nickel compounds and those with allergic contact dermatitis. The use of nickel-containing items like jewelry, watches, and belt buckles can also induce sensitivity. There are two types of nickel dermatitis: primary and secondary. Primary dermatitis manifests as an eczematous reaction with erythematous papules that may develop into lichenification, accompanied by itching. Secondary dermatitis arises from alternative exposure methods, such as ingestion, transfusion, inhalation, or implantation of nickel-containing medical devices, known as systemic contact dermatitis. Nickel compounds, including nickel carbonyl, can oxidize to Ni2+ in tissue, becoming soluble and diffusible in the outer skin layer, thereby penetrating intact skin and triggering allergic reactions. The allergic response is a type 4 delayed hypersensitivity, occurring in two stages: initial sensitization upon exposure to nickel, followed by an allergic reaction during subsequent contact. 45
Nickel exposure can lead to adverse health effects, including allergies, kidney and heart issues, lung fibrosis, and nasal and lung cancers. While the exact molecular mechanisms of nickel toxicity remain unclear, it is thought that mitochondrial dysfunction and oxidative stress significantly contribute to its harmful effects. 46
In the digestive system
The permissible nickel level in drinking water is 0.1 mg/liter according to the US Environmental Protection Agency (USEPA) and 0.2 mg/liter according to the World Health Organization (WHO), European Union (EU), and Australia. High levels of nickel compounds, can cause nausea, vomiting, diarrhea, weakness, headache, and pulmonary symptoms like cough and shortness of breath after 48 h. The estimated ingested dose of nickel ranges from 0.5 to 2.5 g. At even higher levels, symptoms may include decreased consciousness, nuchal rigidity, mydriasis, erythema, tachycardia, and acute lung injury.45,47
Also, recent studies have reported Irritable Bowel Syndrome (IBS) -like symptoms in people whose diets contain a lot of nickel. This sensitivity is due to men being exposed to more nickel in their jobs, and in women, especially at younger ages, due to increased use of jewelry. The mechanism behind this disorder is a malfunction in the regulation of the “brain-gut axis” which is associated with motility, abnormal sensitivity, and autonomic reactivity, in chronic conditions of low-grade inflammation in the intestinal wall and altered immune function. This IBS-like symptom is characterized by nausea, pyrosis, meteorism, abdominal pain, diarrhea, and constipation, along with typical systemic superficial lesions.48,49
Liver
The liver is the main organ of nickel accumulation and toxicity. Excessive nickel intake has been shown to cause liver damage in laboratory animals. On the other hand, increased aspartate and alanine aminotransferase (AST, ALT) have been observed in the blood of these animals, which in the long term increases the risk of developing liver cirrhosis. This is due to the production of ROS from oxidative stress, which leads to lipid peroxidation, reduced hepatic glutathione levels, and nickel accumulation in the liver. This is followed by an increase in cyclooxygenase 2 (COX-2) protein expression, a decrease in glutathione peroxidase 4 (GPX4), nuclear receptor activator 4 (NCOA4) protein, and mRNA expression level. This process is accompanied by mitochondrial damage and ferroptosis, which causes liver damage.50–52
Pancreas
Animal studies suggest that acute and subchronic nickel exposure increases blood glucose. The increase in plasma glucose after nickel chloride injection within 30 min and the decrease in insulin to glucagon plasma ratio, and the concomitant administration of exogenous insulin, indicate this. The reason is the induction of glycogenolysis, increased blood glucagon, decreased blood insulin or gluconeogenesis. In the laboratory tests, the levels of Oral Glucose Tolerance Test (OGTT), Fasting blood glucose (FBG), and glycated haemoglobin (HBA1c) were significantly higher in people exposed to nickel compared to people not exposed to nickel.53,54
Recent studies have implicated nickel as an environmental contaminant in pancreatic cancer, including pancreatic ductal adenocarcinoma. Studies have shown that cellular and molecular changes occur in pancreatic tissue following nickel exposure, with major changes in target gene expression. Observations have shown that nickel levels are significantly elevated in cancerous pancreatic tissue, indicating that nickel is carcinogenic in the pancreas. 55
Kidneys
There is new and persuasive evidence that nickel, even at low doses and with repeated and chronic exposure, has the potential to cause significant kidney damage, due to accumulation through reabsorption in the kidneys. In the early stages, it presents as acute kidney injury (AKI) with neutrophil leukocytosis, leukocyturia, and anemia. Observations have shown that the acute renal event is initiated by an exogenous proinflammatory factor. Early pathology indications acute tubulointerstitial nephritis with focal inflammatory infiltrates at the corticomedullar junction. This is followed by accelerated renal disease progression through cytotoxicity, indirect injury (particularly to renal tubular epithelial cells), and immunological dysregulation, leading to chronic kidney disease (CKD) and end-stage renal disease (ESRD). Laboratory evidence of a decrease in estimated glomerular filtration rate (eGFR) of less than 60 mL/min/1.73 m2 for 3 months or more is expressive of this.56,57
Male & female reproductive
Ni causes disruption of the male reproductive system, which is caused by the production of reactive oxygen species (ROS) that contribute to nickel toxicity. It affects zinc metabolism, which is essential for sperm viability. It has a profound effect on the structure of DNA-binding proteins such as protamines, which can affect sperm function. This has been observed in a dose-dependent reduction in human chorionic gonadotropin (hCG) and cAMP-stimulated testosterone production in interstitial Leydig cell cultures in experimental animals. 58
Studies have shown that nickel compounds lead to changes in ovarian tissue, uterine tone, inflammatory responses, and cellular damage. Damage caused by nickel compounds in the ovaries leads to a decrease in ovarian hormones, which, through negative feedback, increases serum levels of follicle- stimulating hormone (FSH) and luteinizing hormone (LH). Prooxidative changes caused by nickel in the human placenta lead to a decrease in placental viability. Embryotoxicity and teratogenicity, Ni has direct cytotoxic effects on the placenta and on the fetus. Female workers exposed to nickel have been shown to have an increased incidence of spontaneous abortion.59,60
In the respiratory system
The inhalation dose of nickel affecting the respiratory system is 0.5 mg/cm2 per week. Exposure to nickel solutions at a concentration of 5.869 mg/liter for 75 min causes compression and adhesion in the tracheal epithelium. Inhalation of nickel results in various respiratory issues: (1) local alveolar inflammation, characterized by reduced FEV1 and FEF25%–75%, (2) clinical conditions like asthma, chronic bronchitis, emphysema, pulmonary fibrosis, and pulmonary edema, all leading to decreased lung function, and (3) an association with lung cancer. Additionally, nickel can trigger hypersensitivity reactions.61–63
In the central nervous system
The widespread industrial uses of nickel ion (Ni2+) and its oxidation have been proven. Ni2+ can release dopamine and inhibit glutamate receptors, and its consequences have a significant impact on gamma-aminobutyric acid (GABA) levels and serotonin neurotransmission. Researches have shown that excessive exposure to nickel is prominent in mitochondrial dysfunction, oxidative stress generation, neurochemical disorders and brain structure. Nickel neurotoxicity is characterized by several neurological symptoms such as headache, fatigue, lethargy, dizziness, ataxia and changes in cognitive function, motor activities, mood including anxiety-like behaviors and depression.64,65
Chronic nickel exposure
Chronic inhalation of nickel leads to damage and specific changes in the nasopharynx and respiratory epithelial tissue, including rhinitis, polyps, asthma, and pulmonary fibrosis. Moreover, nickel exposure significantly increases the risk of sino-nasal cancers, such as squamous cell carcinoma, anaplastic carcinoma, and adenocarcinoma. The use of nickel-containing jewelry and tools also heightens the risk of dermatitis. Therefore, implementing appropriate safety measures and protective equipment in environments where nickel is present is essential to mitigate these risks.66,67
Diagnostic evaluation
In patients suspected of nickel poisoning, a thorough history and clinical examination are essential. Poisoning typically occurs via inhalation or ingestion, warranting consideration of respiratory and digestive issues. Nickel distributes across various organs and fluids, with serum and urine samples being the preferred initial analyses. The average serum nickel concentration is 0.3 μg/L, but may exceed 8 μg/L in exposed workers. Urine tests for nickel concentration are more common, typically showing levels below 10 μg/L.66,67 For diagnosing and treating nickel-induced dermatitis, tests like the dimethylglyoxime spot test, a type of patch test, are utilized. A positive reaction is indicated by the appearance of a pink color.45,66
Patients exposed to nickel undergo CX-Ray, revealing pleural effusion and irregular linear and nodular shadows, indicating lung involvement. The ECG shows abnormal findings, including sinus tachycardia and ST segment and T wave changes, suggesting myocardial damage. These cardiac manifestations indicate cardiovascular involvement, necessitating further evaluation and treatment. 66
Treatment
Primary supportive treatment
To treat medical issues related to nickel compounds, first identify and eliminate the source of exposure. Remove any contaminated clothing and decontaminate the skin. For allergic contact dermatitis, avoid using jewelry and contact devices. If food and drinking water exceed permissible nickel levels, avoid consumption. Monitor for pulmonary symptoms from inhalation; if hypoxia occurs, provide supplemental oxygen. Treat bronchospasm with bronchodilators and corticosteroids. In severe cases, mechanical ventilation may be necessary. Since nickel binds strongly to proteins, hemodialysis is ineffective; instead, administering intravenous fluids can increase diuresis and lower nickel levels.45,66,68
Treatment of allergic contact dermatitis
Allergic contact dermatitis can be triggered by various factors, with nickel compounds being one of the most common. This condition arises from direct and prolonged skin contact with nickel and can lead to pain, inflammation, and discomfort. It results in a delayed allergic reaction (type 4), known as the induction or sensitization phase, which varies in duration among individuals. Importantly, this type of dermatitis is not life-threatening and does not lead to anaphylactic shock. It is noteworthy that if the skin is already damaged, sensitivity to nickel develops more rapidly and at lower concentrations. Nickel ions released from nickel compounds, metals, and various alloys are responsible for allergic contact dermatitis. 17
The therapeutic goals of allergic contact dermatitis are: identify and avoid nickel compounds, treat skin inflammation, and restore and protect the skin barrier.
Identify and avoid nickel compounds
(see primary supportive treatment section).
Treat skin inflammation
Eczematous changes and itching are the primary symptoms of nickel-induced allergic contact dermatitis. While topical corticosteroids may help alleviate itching and prevent recurrence, they are not approved by any health organizations, including the US FDA. For severe cases with widespread lesions and intense itching, oral steroids for a few days, along with antihistamines, wet dressings, and emollients, may be prescribed. In steroid-resistant cases, local calcineurin inhibitors like pimecrolimus and tacrolimus can be effective.69–71
Restoration of the skin barrier and protection of the skin
Prevention begins with surveillance, necessitating monitoring of the production lines for clothing and devices using nickel alloys. These should be replaced with safer alternatives. Community-level training and information dissemination by the media and healthcare teams are essential. Additionally, thick protectors for cell phone cases, plastic instead of metal buttons on garments, and emollients to strengthen the skin barrier for those with allergic contact dermatitis should be considered.69,72
Chelation therapy
Nickel chelating agents.

aIt is not currently used to chelate nickel compounds.
bIt has slow performance and is not commonly found.
cChelators are valuable and should be administered immediately after exposure. In severe cases, it is important to monitor urine concentration levels for nickel.
dIf DDC is unavailable, it should be used immediately after exposure. Prolonged use can lead to an increase in liver enzymes, so regular monitoring of these enzymes is necessary to check for hepatotoxicity.
Treatment protocol for using DDC according to urinary nickel concentration in nickel poisoning.

Dosage adapted from Goldfrank’s Toxicologic Emergencies (Ref# 45) & Gates A et al. Nickel toxicology (Ref#66).
Treatment protocol for using Disulfiram according to urinary nickel concentration in nickel poisoning.

Dosage derived from Bowman N et al. Clinical toxicology (Ref# 76).
Diethyl dithiocarbamate and disulfiram influence nickel compounds but do not affect elemental or inorganic nickel treatment. If using DDC or disulfiram, they should be administered immediately after exposure, as delays can increase nickel toxicity. High doses of disulfiram can lead to nickel retention in the brain and raise the risk of death. Despite numerous experimental claims about these chelators for nickel, more controlled clinical studies are needed to confirm their effectiveness and safety for humans.77,78
Hemodialysis, peritoneal dialysis and High volume continuous venovenous hemofiltration (HV-CVVHF)
Hemodialysis is utilized for individuals lacking proper kidney function to purify substances and excrete waste. However, it cannot replicate all the complex functions of the human kidney, and its filters often fail to adequately process various substances, potentially leading to the accumulation of some elements in the blood while excessively removing others. Research indicates that in cases of nickel poisoning, hemodialysis is ineffective at reducing serum nickel levels, which may even raise post-treatment, causing issues like allergic contact dermatitis and acute nickel poisoning in some patients. Similar findings have been observed with peritoneal dialysis, where nearly half of the patients’ experience elevated urinary nickel levels. Therefore, monitoring serum and urinary nickel levels and other elements is essential.79,80
Eliminating the overuse of drugs and toxins is essential for treatment. This includes supportive care, preventing toxin absorption, enhancing toxin excretion, using antidotes, and extracorporeal treatments. One such method is continuous venous hemofiltration, which employs convection therapy principles. Compared to intermittent techniques, continuous methods have lower blood and output flow rates, resulting in less clearance over the same time period. Additionally, continuous techniques address limitations like hemodynamic changes, allowing for prolonged toxin removal and preventing sudden increases in plasma toxin concentration and distribution. 81
Hemofiltration utilizes a transmembrane pressure gradient to extract a significant volume of plasma water from the dialyzer membrane, compensating for the loss by administering intravenous fluids. Both plasma water and solutes are removed under the applied pressure. The international association defines high-volume hemofiltration as a continuous treatment exceeding 50 ml/kg per hour over 24 h. Very high-volume hemofiltration involves rates of 100–120 mL/kg/h for short periods of 4–8 h, followed by standard continuous veno-venous hemofiltration. 82 When removing nickel compounds, high-volume hemofiltration effectively addresses a variety of substances with differing molecular weights due to its high sieving coefficients, primarily targeting nickel transported by albumin and other lower molecular weight proteins. Additionally, if DDC and disulfiram are unavailable or if high doses are limited, alternative methods of elimination can be advantageous. With two veins available, high-volume hemofiltration can eliminate both chelated and non-chelated nickel through an extracorporeal route, facilitating the excretion of extracellular and intracellular nickel via urine and bile. 83
Use of nanoparticles in the removal or reduction of nickel compounds in nature
Nickel is a ubiquitous metal with increasing applications in industrial and non-industrial applications and its presence in air, water, soil, and kitchen utensils, Also an environmental pollutant, excessive exposure and its accumulation in the body raise significant concerns for human health. 65 Nanoparticles have a structure with unique properties and are used in a wide range of applications because their size is small, up to 100 nm. They are classified into three main types based on their materials: inorganic, carbon-based, and organic, and include various materials such as proteins, lipids and their derivatives, polymers, metal ions, magnetic minerals, etc. Many scientific reviews mention the precise application of nanoparticles in specific fields, such as industry, food products, drug delivery, antimicrobial activities, gene delivery, cosmetics, imaging, wound healing, diabetes, cancer treatment, etc. 84
In recent years, much work has been done using nanoparticles to remove pollution from air, wastewater, and soil. Nanoparticles address issues that were previously difficult to address with traditional methods. Air pollutants are a major threat and one of the greatest environmental challenges. Nanomembrane technology is used to separate and purify pollutant gases and vapors in various sectors and to prevent their discharge into the environment. Nanosensors detect harmful and dangerous gases even in small quantities and provide necessary warnings.85,86 Nanoparticles, due to their high adsorption capacity and catalytic activity, are effective in the absorption and removal of heavy metal ions, especially nickel (II) ions, which accumulate in nature and are not biodegradable, from wastewater through particle size control and surface modification.87,88 Also, synthesized nanoparticles show good performance for the removal of nickel from contaminated soil compared to chemical synthesis. 89
Conclusions
Nickel compounds are widely present in our environment, which means that both humans and animals are continually exposed to them. This exposure raises concerns about potential adverse health effects, as well as risks to the environment. Thus, this article has attempted to address various aspects, such as how nickel enters the body through different routes and its amount, and to estimate the required amount and its metabolism in the human body, using scientific resources from recent years.
Additionally, there is a noticeable lack of global coordination in establishing a common regulatory framework to manage these risks, particularly in the industrial and trade sectors. Accordingly, this article focuses on carcinogenicity, mechanisms of toxicity such as oxidative stress, inhibition of DNA repair, transcription factors, genotoxicity, and teratogenicity, that commercial and industrial sectors, as well as healthcare organizations can adopt a different approach.
In addition, this article reviews the clinical manifestations of nickel poisoning in different systems so that the healthcare team can have a better understanding of nickel poisoning. The article also presents aspects of first aid, skin protection, and treatment of damage to this system. Other points of this article include the use of chelators at the right time and the necessary measures for different conditions, the use of continuous venovenous hemofiltration, and the recent mention of nanoparticles in the reduction of nickel in nature.
In this way, educating the community about these contaminated sources by a trained and experienced professional team, rapid diagnosis, and correct treatment will lead to performing tasks with high and desirable standards. Performing these tasks will decrease the effects of poisoning, reduce mortality, and complications caused by nickel compounds.
Footnotes
Acknowledgments
We extend our gratitude to the library staff at Beheshti Hospital and Zahravi Central Library at Babol University of Medical Sciences. We also appreciate our families for their inspiration, patience, and encouragement in our work.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
