Abstract
This study was designed to understand the efficacy and molecular cues of melatonin in cyclophosphamide(CTX)-induced premature ovarian failure (POF) in rats. Female SD rats were used to evaluate the potential effects of melatonin on the ovarian hormonal status, follicular development, and granulosa cells in CTX-treated rats. Here, we found that pretreatment with melatonin before CTX administration preserved the normal sex hormone levels, improved follicular morphology, and granulosa cell proliferation, and reduced apoptosis, as compared to the CTX treatment alone. Additionally, melatonin also up-regulated CYR6 and CTGF at the mRNA and protein levels. A potential mechanism is that melatonin inhibits LATS1, Mps1-One binder (MOB1), and YAP phosphorylation, thereby activating the Hippo signal pathway to promote its downstream targets, CYR61 and CTGF. In conclusion, pretreatment with melatonin effectively protected the ovaries against CTX-induced damage by activating the Hippo pathway. This study lay the foundation for the clinical application of melatonin for cancer patients with CTX treatment.
Introduction
Premature ovarian failure (POF) in female cancer survivors after chemotherapy has aroused widespread public concern. The use of chemotherapeutic drugs in cancer treatment leads to the apoptosis of oocytes and surrounding granulosa cells in the ovary, which leads to premature depletion of follicular reserves and causes POF. 1 About 60-80% of chemotherapy patients may experience POF. 2 CTX, as an alkylating agent commonly used in chemotherapy, induces severe ovarian damage and is a recognized risk of POF.3,4 Studies have shown that CTX causes ovarian follicle failure by activating primal follicles and making them lose a lot.5,6 Hormone replacement therapy, the most commonly used treatment for POF, is ineffective in restoring ovarian function but increases the risk of malignancies such as endometrial cancer and breast cancer.7,8 Therefore, searching for an effective method to treat chemotherapy-induced POF and improve the ovarian function and fertility of the majority of female patients has become an urgent medical problem to be solved.
Melatonin (MT) is an amine hormone produced by the pineal gland of mammals and humans and secreted in tissues such as the ovary and placenta. 1 Melatonin has strong antioxidant activity, scavenging free radicals such as reactive oxygen species (ROS).9–11 It is becoming increasingly clear that melatonin is located in the ovarian filtrate and in the oocyte itself, which protects these cells from oxidative damage and has other beneficial effects during oocyte maturation, fertilization, and embryonic development. 12 Clinical trials have shown that melatonin improves the outcomes of infertile patients who have undergone assisted reproductive technologies including in-vitro fertilization and embryo transfer. 13 Melatonin inhibits ovarian damage caused by radiotherapy and plays an anti-radiation role mainly by scavenging free radicals. 14 When melatonin interacts with -OH, it forms a transitional indolyl, which has low toxicity and can continue to react with -OH to produce 3-hydroxyl melatonin. Melatonin can also increase the level of SOD and GPX to combat ionizing radiation. 15 However, the role and mechanism of melatonin in CTX-induced POF has not been fully understood.
The study was performed on the POF rats model induced by CTX injection. In this article, the effects of melatonin on ovarian reserve capacity, granulosa cells, and related AMH and FSHR were studied. Further, we explored the mechanism by which melatonin alleviated ovarian dysfunction. The data from this study suggested that melatonin provides a promising therapeutic method for patients with POF in the clinic.
Materials and Methods
Animal model
Female SD rats (body weight of 200–220 g) were purchased from Beijing Vital River Laboratory Animal Technology Co., Ltd. and housed in the specific pathogen-free (SPF) condition with temperature control (22 ± 1°C) and humidity control (60 ± 10%) on a 12-hr light/dark cycle with ad libitum access to water and regular rodent chow. All the animal protocols were approved by the Animal Care and Use Committee of the Institute of Radiation Medicine, Chinese Academy of Medical Sciences (Approval No. IRM-DWLL-20022119).
Establishment of the POF rats model and melatonin treatment
The rats (n = 48) were randomly divided into four experimental groups (12 animals per group): 1 control group, 2 POF group, 3 10 mg/kg melatonin (MT10), and 4 20 mg/kg melatonin (MT20). The CTX-induced POF rats model was established according to the previous studies.16,17 The rats of the POF, MT10, and MT20 groups were intraperitoneally injected with 50 mg/kg of CTX on the first day and then 8 mg/kg for 15 consecutive days, while the control group was injected with 0.9% saline instead of CTX. The rats of the MT10 and MT20 groups were injected with melatonin (Sigma, St. Louis, MO, USA) at 10, or 20 mg/kg body weight (i.p.), respectively, once daily, and before 30 min of CTX injection. All rats were sacrificed under anesthesia and blood and ovarian were collected for further study.
Serum levels of hormone measurement
Blood samples of rats were taken from postcava and centrifuged to get the serum. The ELISA kits (Greenleaf, CN) were used to survey the levels of estradiol (E2), follicle stimulation hormone (FSH), luteinizing hormone (LH), and anti-Müllerian hormone (AMH) according to the instructions of manufacturers.
Ovarian follicle count and morphological analysis
To analyze the effect of melatonin administration on ovarian development of CTX-induced POF rats, the wet ovarian weight of each sacrificed rat was recorded and further analyzed with the ovarian index (ovarian weight/body weight ×100%).
The ovaries were embedded in 4% paraformaldehyde for 12 hours and then embedded in paraffin. Paraffin tissues were sectioned continuously (6μm thick) and stained with H&E. Ovarian follicles were counted under an optical microscope (Olympus) according to the method described in a previous study. In short, 5 slides were randomly selected from each group, and 5 non-repeating views were selected from each slide for statistical analysis. Only follicles containing oocytes with clearly visible nuclei were counted. According to the method described earlier, follicles are classified as primary, primary, secondary, and atretic follicles.
Immunohistochemical staining
HE staining was administered for formalin-fixed, paraffin-embedded (FFPE) specimens. Anti-CTGF (Cell Signaling Technology) and anti-CYR61 (Cell Signaling Technology) monoclonal antibodies were applied for IHC staining as previously described. The expression of CYR61 and CTGF was independently calculated by two authors blinded to section treatment. The immunoreactivity scores of CYR61 and CTGF were derived by multiplying the percentage of immunoreactive cells and the intensity of IHC staining, which ranged from 0 to 12. As a result, specimens with a final staining score of ≥6 were labeled as high expression, while those of <6 were classified as low expression.
Western blot analysis
Ovarian tissues were collected from each group to prepare tissue homogenates. Tissue homogenate was centrifuged at 12,000 ×g at 4°C for 10 min. Total proteins were extracted with RIPA (1%PMSF) and protease inhibitors and were measured by the BCA Protein Assay Kit (Beyotime). Total protein was separated by 10% SDS–PAGE (Beyotime) gel electrophoresis and then electro transferred onto polyvinylidene difluoride (PVDF) membranes (Millipore, Billerica, MA, USA), which was sealed with serum blocking solution. Subsequently, at 4°C, it was combined with the primary antibody and incubated overnight. After three washes, the second antibody was added to the membrane and incubated at room temperature for 1 h. Then the immunoresponse signal was detected using the ECL Plus immunoblotting system (Thermo Fisher Scientific, USA). Image J software was used to calculate the gray value of each band.
In this study, anti-MOB1 (Cell Signaling Technology), anti-phospho-MOB1 (pMOB1, Cell Signaling Technology), anti-LATS1 (Cell Signaling Technology), anti-phospho-LATS1 (pLATS1, Cell Signaling Technology), anti-YAP (Cell Signaling Technology), anti-phospho-YAP (pYAP, Cell Signaling Technology), anti-CTGF (Cell Signaling Technology), anti-CYR61 (Cell Signaling Technology) and anti-GAPDH (Cell Signaling Technology, Danvers, MA, USA) monoclonal antibodies were used.
TUNEL staining
In situ Cell Death Detection Kit (Roche, USA) was used to assess apoptosis of granulosa cells. After dewaxing, the sections were incubated with protease K (20mg/mL) in a humidifier for 15 min and treated with 3% H2O2 for 10 min to block endogenous peroxidase activity. Then, sections were incubated with TdT labeling buffer at 37°C in a wet chamber for 1 hour. These parts are then re-stained by DAPI. TUNEL-positive cells were stained brown and the nuclei were stained with DAPI (blue). Five random fields per slide (five slides per animal, eight animals per group) were examined. TUNEL positive granulosa cells and total granulosa cells in sinus follicles were counted. Image-Pro Plus 6.0 software was used to analyze the rate of TUNEL-positive granulosa cells (%) in sinus follicles.
Statistical Analysis
SPSS 22.0 software was used to analyze the experimental data, and the average ± of SEM was used to represent the statistical data. Data distribution was analyzed by one-way analysis of variance (ANOVA) followed by Duncan’s multiple range post hoc analysis. p <0.05 indicates statistical significance.
Results
Protective effects of melatonin on the HPG axis and AMH expression in CTX-induced POF model rats
CTX was used to induce the POF model in rats, and ELISA was used to test the serum FSH and LH, progesterone (P), E2, and AMH levels. As shown in Figure 1, the POF rats had markedly increased serum FSH and LH levels (p < 0.05 and p < 0.01), but decreased progesterone, E2, and AMH levels (p < 0.01) compared to those in the control. However, melatonin treatment significantly decreased the serum FSH and LH levels (p < 0.05) and increased the serum P, E2, and AMH levels (p < 0.01). Protective effects of melatonin on the HPG axis and ovarian AMH. The serum FSH (a), LH (b), E2 (c), P (d), and AMH (e) levels were tested in each group. Ten rats per group were examined. *p < 0.05 and **p < 0.01 vs the control group, #p < 0.05 and ## p < 0.01 vs the POF group.
Effect of melatonin on follicular development in the POF model
First, the ovarian weight of rats was assessed. As shown in Figure 2(a), the body weight of rats in the POF group was remarkably lower in comparison with the NC group. Meanwhile, melatonin administration increased the ovarian weight of POF rats. Next, H&E staining showed that most healthy follicles were observed in the ovaries of the control group, including primary, primary, secondary, and atretic follicles. In contrast, the ovaries of POF rats withered along with most of the mesenchymal cells in the fibrous stroma. The granulosa cells were arranged irregularly, and lymphocyte infiltration was observed in follicles. The morphological changes in the melatonin treatment group were better than those in the POF group (Figure 2(b)). Follicle counting was performed after H&E staining. The follicle classification was based on the characteristics proposed by Hirshfield & Midgley (1978). Significantly more follicles were counted at the different developmental stages of maturation in the melatonin treatment group than in the POF model group (Figure 2(c)). Effect of melatonin on follicular development in the POF model. (a) ovarian weight. (b) H&E staining of ovaries (midline sections). Asterisks: the corpora lutea; Red arrow: atretic ovarian follicle. Scale bars, 200 μm. The follicle classification was based on the characteristics proposed by Hirshfield and Midgley. Magnification × 100. Scale bar: 200 μm. (c) The numbers of normal and atretic ovarian follicles from the ovaries of each group were summarized. ***p < 0.001 vs the control group, #p< 0.05, ##p < 0.01, ###p < 0.001vs the POF group.
Protective effect of melatonin on the proliferation of ovarian granulosa cells in ovarian tissues of POF rats
Immunofluorescence of Ki-67 demonstrated that proliferation of granulosa cells in secondary and antral follicles was observed in all experimental groups, but the percentage of Ki-67 positive granulosa cells in the POF group was significantly less than that in the control group, and the percentage of Ki-67 positive granulosa cells in the ovary after MT treatment was significantly increased, especially in MT20 group (Figure 3(a)). Apoptosis in ovarian tissue was assessed by a TUNEL assay. As illustrated in Figure 3(b), there were few cells with DNA damage in the ovarian tissue of the control group. The percentage of TUNEL-positive granulosa cells in the POF group was significantly increased compared with the control group. However, melatonin treatment significantly prevented the increase in TUNEL-positive cells, suggesting a protective effect on ovarian cell apoptosis. Furthermore, the Western blotting analysis demonstrated that POF rats showed reduced Bcl-2 and Ki-67 protein levels and increased Bax protein levels compared with the control group (p < 0.01). After melatonin treatment, Bcl-2 and Ki-67 protein levels were extensively increased (p < 0.01), and the Bax expression levels were significantly decreased as revealed in Figure 3(c). Effect of melatonin on POF-induced apoptosis of ovarian cells. (a) Immunofluorescence of Ki-67 (brown) represents granulosa cell proliferation in the ovary. Magnification × 200. (b) Apoptosis of granulosa cells in ovarian tissues was measured using TUNEL staining. Apoptotic cells are shown as brown. The nucleus was stained with DAPI, shown as blue fluorescence. Magnification × 200. Five random fields per section (five sections per tissue from a mouse) were examined in each experiment. The TUNEL-positive granulosa cells and total granulosa cells in the antral follicles were counted. (b) Western blotting was conducted to analyze Bcl-2, Ki-67, and Bax protein levels. *p < 0.01 and **p < 0.01 vs the control group; #p < 0.01 and ##p < 0.01 vs the POF group.
Melatonin upregulates the expression of CYR61/CTGF in POF rats
The expression and cellular locations of the cysteine-rich angiogenic protein 61 (CYR61) and connective tissue growth factor (CTGF) proteins were examined using RT-PCR, Western blotting, and immunohistochemistry. The CYR61 and CTGF mRNA and protein expression levels were significantly lower in the POF group than in the control group (p < 0.01, Figure 4(a) and (b)), whereas the expression levels were partially and significantly increased in the melatonin treatment group (p < 0.01 and p < 0.05, respectively). The CYR61 and CTGF proteins were mainly located in the ovarian interstitial cells (Figure 4(c)). Effects of melatonin on CYR61 and CTGF expression and location. (a) CYR61 and CTGF mRNA expression were quantitatively analyzed by RT-PCR (n = 8). (b) CYR61 and CTGF mRNA expression were quantitatively analyzed by Western blot (n = 8). (c) The cellular locations of these proteins were observed using immunohistochemistry. *p < 0.01 and **p < 0.01 vs the control group; #p < 0.05 and ##p < 0.01 vs the POF group.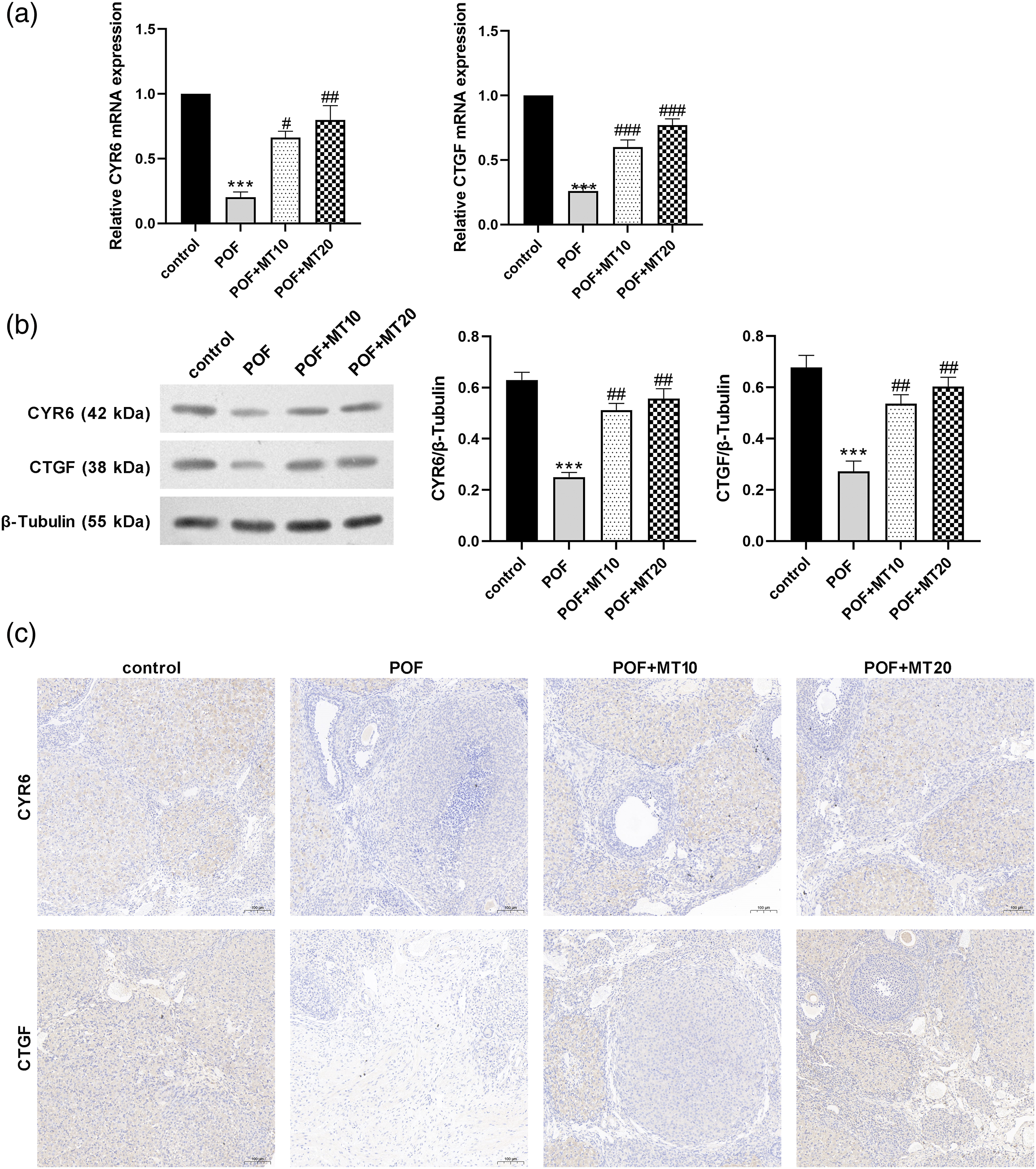
Melatonin activates the Hippo signaling pathway in POF rats
We investigated the potential mechanisms involved in the effects of melatonin on POF-induced ovarian aging. The expression levels of Hippo signal pathway-associated markers were assessed by Western blotting (Figure 5). Total LATS1, MOB1, and YAP protein expression levels were markedly lower in the POF group than in the control group and were significantly higher in the melatonin group than in the POF group (p < 0.01). However, the p-LATS1、p-MOB1 and p-YAP levels were markedly higher in the POF group than in the control group (p < 0.01) and were significantly lower in the melatonin group than in the POF group (p < 0.01). Our results suggest that melatonin can prevent Hippo pathway disruption induced by CTX. Effect of melatonin on the expression of Hippo signaling pathway-related proteins in ovarian tissues. The LATS1、p-LATS1、MOB1、p-MOB1、p-YAP、YAP protein expression levels in ovarian tissues were observed using Western blotting (a). The LATS1、p-LATS1、MOB1、p-MOB1、p-YAP、YAP protein expression levels were quantitatively analyzed (n = 8) (b). Data are shown as the mean ± s.d. Statistical significance: *p < 0.01 and **p < 0.01 vs the control group; #p < 0.05 and ##p < 0.01 vs the POF group.
Discussion
In the past few decades, in addition to traditional hormone replacement therapy, several new clinical treatments for chemotherapy-induced POF patients, including stem cell therapy, ovarian cryopreserved, and donor oocyte in vitro fertilization (IVF), have been used to treat POF-related symptoms. 18 However, due to the complex pathogenesis of POF, the clinical treatment of chemotherapy-induced POF patients remains unsatisfactory. In this study, our results illustrate melatonin maintains the ovarian hormonal status, enhances the ovarian index, promotes follicular development, and also inhibits granulosa cell apoptosis in the CTX-induced rat POF model. Additionally, melatonin activates the Hippo pathway and increases the expression of its downstream effectors CYR61 and CTGF. This study lay the foundation for the clinical application of melatonin for cancer patients with CTX treatment.
Many studies have shown that CTX causes ovarian dysfunction including accelerated loss of ovarian reserve, folliculogenesis dysregulation, and steroid disorder.3,4,16 We found that the serum FSH and LH levels were significantly increased and the E2 and P levels were decreased in rats treated with CTX. The number of atretic follicles was increased by the CTX treatment; conversely, the decreased number of primordial, primary, and secondary follicles resulted in a decreased number of total follicles, which was in line with a previous report. AMH is a very important early marker of ovarian aging and reflects the size of the ovarian follicle pool. Lower AMH expression has been detected long before normal menopause and during the normal course of aging in mice. We also found that serum AMH expression was significantly decreased by CTX. Thus, the use of CTX successfully induced the rat POF model in the present study.
Melatonin is generally produced in various tissues and acts as a universally acting antioxidant in cells. 19 Melatonin is now more widely used in various biological processes as an adjuvant during chemotherapy.20,21 Melatonin is found at a high concentration in human follicular fluid, and it increases in proportion to follicular growth. 12 Melatonin has been demonstrated to exert a protective effect on ovarian function. Several studies revealed that melatonin reduces oxidative stress in ovarian follicles via its antioxidative action, thereby protecting oocytes and granulosa cells.13,22,23 Several studies reported that melatonin protects granulosa cells and oocytes from ROS by reducing oxidative stress.24,25 Tamura et al. 12 showed that melatonin blocked the inhibitory effect of H2O2 on oocyte maturation via reduction of ROS in oocytes. It was also found that melatonin promoted folliculogenesis and reduced apoptosis in murine ovarian cells. Consistent with these studies, we confirmed the protective effect of melatonin on the ovarian hormonal status and follicular development in a CTX-induced POF rat model. We found that melatonin decreased the FSH and LH levels and increased the E2, P, and AMH levels in the rat POF model. Melatonin increased the total number of follicles with the numbers of primordial, primary, and secondary follicles. These data suggest that melatonin regulates the reproductive endocrine function and promotes follicular development and maintains primordial follicles.
Previous studies have shown that the Hippo pathway is involved in ovarian physiological and pathological processes.26,27 The hippo signaling pathway participates in the regulation of ovarian follicle development. 28 In both mouse and human ovaries, several Hippo signaling genes are expressed in follicles at different stages. 29 In the ovary, when primordial follicles are activated to grow, Hippo signaling of follicular cells near the cortical area could be disrupted due to the stiff niche, leading to increased YAP activity and CCN growth factor secretion followed by cell proliferation and follicle growth into the secondary stage. As follicles grow larger, they move into the softer medullar region and subsequent resumption of Hippo signaling could slow down follicle growth. Herein, we found that melatonin treatment upregulated total LATS1、MOB1, and YAP1 in the POF rats, but reduced the phosphorylation levels of these proteins, indicating the activation of the Hippo signaling pathway. In turn, the activated Hippo pathway promotes downstream effector expression, including CYR61 and CTGF. CYR61, also known as CCN1, is involved in processes of angiogenesis within reproductive systems, thereby potentially contributing to diseases such as endometriosis and disturbed angiogenesis in the placenta and fetus.30,31 In the ovary, CTGF, also known as CCN2, is the key factor for follicular development, ovulation, and corpora luteal luteolysis, and its deletion leads to fertility defects.32,33 This explains why melatonin protects follicles at molecular levels.
Conclusions
Taken together, we presented evidence that melatonin protects ovarian function through modulation of the Hippo signaling pathway. These findings indicate that melatonin has the potential to serve as a chemotherapeutic adjuvant to preserve ovarian function during CTX chemotherapy.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by the Medical Leading Talents Fund of Yunnan Province (No. L-2017016), the National Natural Science Foundation of China, Regional Fund (No. 81760037) and the Yunnan Science and Technology Department (No. 2020LCZXKF-SZ10 and No. 2017ZDKFKT004).
Ethical approval
All animal care and experimental procedures were approved by the Animal Care and Use Committee of the Institute of Radiation Medicine, Chinese Academy of Medical Sciences (Approval No. IRM-DWLL-20022119).
