Abstract
Introduction
This study investigated the molecular mechanism by which HuA influences the expression of pyruvate carboxylase via retinoid X receptor alpha (RXRA), thereby affecting the progression of obstructive sleep apnea (OSA).
Methods
Bioinformatics analysis including screening of differentially expressed genes (DEGs) and searching the downstream target genes of RXRA were conducted. Cognitive function, neuronal damage, oxidative stress, and inflammation were evaluated in chronic intermittent hypoxia (CIH) mouse models. The Morris water maze test was used to assess swimming path length, escape latency, and platform crossing times. H&E and Nissl staining was performed to evaluate pathological changes and neuronal counts in brain tissue. ELISA was utilized to measure the oxidative stress levels and inflammatory cytokines. RXRA enrichment in the pyruvate carboxylase promoter region in CIH was assessed using Chromatin Immunoprecipitation (ChIP), and the effect of RXRA on pyruvate carboxylase promoter activity was analyzed using dual-luciferase assay.
Results
RXRA was identified as a potential regulatory target gene of HuA. Pyruvate carboxylase was identified as a RXRA target gene and a significant DEG in OSA. CIH-induced cognitive impairment, neuronal damage, oxidative stress, and inflammation in mice, while such symptoms were alleviated by HuA treatment. In OSA, suppression of RXRA expression led to reduced pyruvate carboxylase expression. HuA treatment enhanced RXRA expression, thereby promoting pyruvate carboxylase expression. HuA alleviated CIH-induced cognitive impairment, neuronal damage, oxidative stress, and inflammation via the RXRA/pyruvate carboxylase axis.
Conclusion
In summary, HuA alleviates CIH-induced cognitive impairment, neuronal damage, oxidative stress, and inflammation by promoting the RXRA/pyruvate carboxylase axis.
Keywords
Significance statement
Chronic intermittent hypoxia (CIH) mouse models were established in this study to uncover the mechanism through which Huperzine A (HuA) mitigated the pathological responses triggered by OSA via the retinoid X receptor alpha (RXRA)/pyruvate carboxylase axis, with the aim to establish the conceptual groundwork for future investigations into HuA’s neuroprotective and anti-inflammatory properties. Furthermore, the findings indicated that treatment with HuA significantly ameliorated cognitive impairment and neuronal damage arising from CIH, offering promising clinical implications.
Introduction
Obstructive sleep apnea (OSA) is a common sleep disorder characterized by recurrent upper airway collapse during sleep, resulting in intermittent apnea and hypopnea. This condition causes frequent nocturnal awakenings and excessive daytime sleepiness. 1 The global prevalence of OSA is increasing, with an estimated rate of 13% in adult males and 6% in adult females. 2 OSA not only disrupts sleep patterns but also correlates closely with various serious health conditions, such as cardiovascular diseases, metabolic syndrome, and neurological impairment. 3 Chronic intermittent hypoxia (CIH), a primary pathophysiological characteristic of OSA, induces brain hypoxia and alters cerebral blood flow, leading to neuronal damage and cognitive decline. 4 CIH induces increased production of reactive oxygen species (ROS), initiating oxidative stress responses in both systemic and local tissues. 5 This oxidative stress not only causes direct damage to cells and tissues but also activates inflammatory responses, promoting the release of inflammatory mediators through diverse molecular pathways. 6 Together, these oxidative stress and inflammatory reactions contribute to endothelial dysfunction and atherosclerosis, thereby increasing the cardiovascular risks associated with OSA. 7
The traditional Chinese medicine Huperzine A (HuA) has garnered attention for its potential neuroprotective and anti-inflammatory properties. Derived from the traditional Chinese herb Huperzia serrata, HuA, is an alkaloid with a long history of medicinal use. 8 Modern medical research has shown that HuA inhibits acetylcholinesterase (AChE) activity, thereby improving cholinergic neurotransmission and offering significant therapeutic benefits in neurodegenerative conditions such as Alzheimer’s disease (AD). 9 Furthermore, HuA possesses antioxidant and anti-inflammatory characteristics that can alleviate oxidative stress and inflammatory reactions. 10 Recent findings suggest that HuA may hold therapeutic potential for CIH-induced mice, although the specific mechanisms warrant further investigation. 11
Retinoid X receptor alpha (RXRA), a member of the nuclear receptor family, governs a spectrum of biological functions encompassing gene expression, cell proliferation, and metabolism. 12 It forms heterodimers with other nuclear receptors to modulate the transcriptional activity of specific target genes. 13 Notably, RXRA collaborates with peroxisome proliferator-activated receptors (PPARs) to oversee lipid metabolism and energy equilibrium. 14 In our recent investigation, we confirmed pyruvate carboxylase as a direct target of RXRA. Pyruvate carboxylase, a mitochondrial enzyme, facilitates the Adenosine Triphosphate-dependent conversion of pyruvate to oxaloacetate, thus supporting the tricarboxylic acid cycle. 15 In healthy tissue, pyruvate carboxylase plays a vital role in regulating overall energy balance by managing gluconeogenesis in the liver, lipid synthesis in adipocytes, and insulin release from pancreatic β cells. 16 Although RXRA and pyruvate carboxylase show significant alterations in various pathological conditions, their specific implications in OSA necessitate further exploration.
While the potential involvement of HuA in modulating pathological processes associated with CIH is acknowledged, its precise mechanisms remain elusive. Existing research predominantly delves into the neuroprotective and anti-inflammatory attributes of HuA, with limited investigations on its impact on regulating RXRA and pyruvate carboxylase expressions, thereby modulating OSA progression. This study sought to elucidate the molecular pathways through which HuA regulates pyruvate carboxylase expression via RXRA, thereby influencing OSA progression.
Materials and methods
Bioinformatics analysis
The downstream target genes of HuA were predicted using the Swiss Institute of Bioinformatics (SIB) database (https://www.swisstargetprediction.ch/). The microarray dataset (GSE38792) for OSA was obtained from the GEO database, which includes 8 normal samples and 10 OSA samples. Differential expression analysis was conducted using the “limma” package in R, with normal samples as the control. The selection criteria for differentially expressed genes (DEGs) were set as |logFC| > 0.585 and p.val < 0.05. The hTFtarget database (https://guolab.wchscu.cn/hTFtarget/#!/tf) was adopted to predict downstream transcriptional regulatory target genes of RXRA. Binding domains in the promoter regions of candidate target genes were predicted using the JASPAR database (https://jaspar.elixir.no/).
CIH mouse model
Male C57BL/6 mice (4-5 weeks old), purchased from Hunan SJA Laboratory Animal Co., Ltd. (Changsha, Hunan, China), were housed under controlled conditions (temperature: 22 ± 2°C, humidity: 50%–60%) with a 12h:12h light-dark cycle. After 1 week of acclimatization, the mice were subjected to CIH model establishment as described previously. 11 Briefly, mice were exposed to cycles of hypoxia (FiO2 reduced from 21% to 9% over 1.5 min, then restored to 21% over 1.5 min) repeated every 3 min (20 times) for 8 h daily for 21 days. Control mice were exposed to normal air (n = 5). Successfully modeled mice were assigned into (n = 5 per group): CIH (mice with CIH treatment), CIH + HuA (mice with CIH treatment and injection of HuA), CIH+HuA+sh-NC + oe-NC (mice with CIH treatment and injection of HuA, as well as negative controls [NCs] of overexpression and silencing lentiviruses), CIH+HuA+sh-RXRA + oe-NC (mice with CIH treatment and injection of HuA, along with lentiviruses of RXRA silencing and NCs of overexpression lentivirus), and CIH+HuA+sh-RXRA + oe-pyruvate carboxylase (mice with CIH treatment and injection of HuA, along with lentiviruses of RXRA silencing and pyruvate carboxylase overexpression) groups. HuA was administered daily (0.1 mg/kg, i.p.) for 21 days. Concurrently, silencing treatments involved intraperitoneal injections of 1 mg/kg sh-RXRA (LentiArray Cas9 lentiviral vector from Beijing TransGen Biotech Co., Ltd.) or its NC, and overexpression treatments involved stereotactic brain injections of pyruvate carboxylase overexpression lentivirus (pLenti6.2 vector from Shanghai GeneChem Co., Ltd.) or its NC (copies/mL: 8.4 × 1011) on the first day of HuA treatment (Supplementary figure: schematic diagram of animal treatment and procedures). Following the Morris water maze test, mice were humanely euthanized using carbon dioxide 17 with their blood and brain tissue samples being collected. The experiment was approved by the Ethical Committee of The Affiliated Changsha Hospital of Xiangya School of Medicine, Central South University (No.: 2024-116) and the experiment was conducted based on the regulations of the Institutional Animal Care and Use Committee.
Morris water maze test
Being first established by neuroscientist Richard G. Morris in 1981, water maze test assesses the hippocampus-dependent learning, including the acquisition of spatial memory and long-term spatial memory. 18
Mice were placed in a water maze facing the wall and allowed to find a hidden platform from one of four starting points. The distance and time taken to find the platform (swimming distance and escape latency) were recorded. After removing the platform, the number of times mice crossed the previous platform location within 2 min was recorded.
Hematoxylin and eosin (H&E) staining
Sections were rinsed with distilled water, stained with hematoxylin for 3-5 min, differentiated in diluted hydrochloric acid and ammonia water, rinsed in running water, dehydrated in 70% and 90% ethanol for 10 min each, and counterstained with eosin (KFS120, Beijing Baiao Laibo Technology Co., Ltd.) for 2-3 min. After dehydration in absolute ethanol, sections were cleared in xylene and mounted with neutral resin for microscopic examination of histopathological changes.
Nissl staining
Paraffin sections were baked at 55°C for 30 min, dewaxed in xylene for 5-10 min, rehydrated through graded ethanol solutions, and rinsed in distilled water. Sections were stained with pre-warmed Nissl staining solution (MA0129, Dalian Meilun Biotechnology Co., Ltd.) at 35°C for 10 min, rinsed in distilled water, dehydrated briefly in 90% ethanol and 95% ethanol, and cleared in xylene for 5 min. After drying at 37°C, sections were mounted with neutral resin and examined under a light microscope.
Enzyme-linked immunosorbent assay (ELISA)
After behavioral tests, mice were euthanized, and 250 μL of orbital blood was collected. Serum levels of Tumor Necrosis Factor Alpha (TNF-α, PT512, Beyotime), Interleukin-1 Beta (IL-1β, PI301, Beyotime), Interleukin-6 (IL-6, PI326, Beyotime), reactive oxygen species (ROS, S0033S, Beyotime), and lactate dehydrogenase (LDH, C0016, Beyotime) were measured using ELISA kits.
Western blot analysis
Tissue samples were digested with trypsin and lysed in RIPA buffer containing protease inhibitors. Protein concentration was determined using a Bicinchoninic Acid Assay (BCA) assay. Proteins were separated by 10% Sodium Dodecyl Sulfate Polyacrylamide Gel Electrophoresis (SDS-PAGE) and transferred to Polyvinylidene Fluoride (PVDF) membranes. After blocking with 5% bovine serum albumin (BSA), membranes were incubated overnight at 4°C with primary antibodies (RXRA, ab232472, 1:500, Abcam; pyruvate carboxylase, ab126707, 1:1000, Abcam; NLRP3, ab263899, 1:1000, Abcam; GSDMD-N, ab215203, 1:2000, Abcam; Cleaved-Caspase 1, ab238979, 1:1000, Abcam; Cleaved-IL-1β, #83186, 1:500, CST; Glyceraldehyde-3-phosphate Dehydrogenase [GAPDH], ab9485, 1:2500, Abcam). Membranes were then incubated with Horseradish Peroxidase (HRP)-conjugated secondary antibody (ab6721, Abcam) for 1 h at room temperature. Bands were visualized using Enhanced Chemiluminescence (ECL) reagents and analyzed with ImageJ software. GAPDH was used as a loading control.
Chromatin immunoprecipitation (ChIP)
ChIP assays were performed with the help of a ChIP kit (Millipore, USA). Cells (HEK293 T) or brain tissues were fixed with 1% formaldehyde, sonicated to shear DNA, and centrifuged. Supernatant was immunoprecipitated overnight at 4°C with antibodies against RXRA (ab125001, Abcam), RNA polymerase II (positive control), or normal IgG (NC). DNA-protein complexes were captured with Protein A/G agarose beads, which were washed and de-crosslinked. DNA was purified and analyzed by qRT-PCR to determine the enrichment of specific proteins at the pyruvate carboxylase promoter region.
Dual-luciferase reporter assay
HEK-293T cells were co-transfected with luciferase reporter plasmids (Wide type [WT] and Mutant [MUT]), along with pRL-TK (internal control expressing Renilla luciferase). After 48 h, luciferase activity was measured employing the Dual-Luciferase® Reporter Assay System and a Varioskan LUX multimode microplate reader (VLB000D0, Thermo Fisher). Relative luciferase activity was calculated as the ratio of Firefly to Renilla luciferase (FL/RL). Experiments were performed in triplicate.
Statistical analysis
Statistical analyses were completed employing SPSS version 22.0 (IBM SPSS Statistics, Chicago, IL, USA). Data were tested for normality and variance. Normally distributed data were described as Mean ± standard deviation (SD). Unpaired t-tests were utilized for comparisons between two groups, while one-way analysis of variance (ANOVA) followed by Tukey’s post-hoc test for multiple group comparisons. A p-value < 0.05 was noted statistically significant.
Results
HuA mitigated cognitive impairment, neuronal damage, oxidative stress and inflammatory response induced by OSA
One recent study has demonstrated that HuA can ameliorate cognitive impairment induced by CIH,
11
although its mechanisms remain unclear. To explore the regulatory mechanism of HuA in OSA, a CIH mouse model was established, followed by intraperitoneal administration of HuA. The Morris water maze test was employed to assess spatial learning and memory, including swimming path length, escape latency, and platform crossings. Compared to the control group, the CIH group showed significantly enhanced swimming path length and escape latency, with reduced platform crossings. Conversely, the CIH + HuA group exhibited substantially decreased swimming path length and escape latency, with increased platform crossings compared to the CIH group (Figure 1(a)–(c)). H&E staining of brain tissues revealed that, relative to the control group, the CIH group showed significant brain histological damage, but the histological damage was attenuated in the CIH + HuA group (Figure 1(d)). Nissl staining indicated that neuron number significantly decreased in the CIH group compared to the control group, while the neuron viability considerably increased in the CIH + HuA group compared to the CIH group (Figure 1(e)). HuA mitigates cognitive impairment and neuronal damage induced by OSA. Note: (a) Swimming path length in Morris water maze test; (b) Escape latency in Morris water maze test; (c) Platform crossings in Morris water maze test; (d) Brain tissue pathology by HE staining (damaged neurons are indicated by black arrows); (e) Neuronal count by Nissl staining. Asterisks (*) indicate significance compared to control group; hash symbols (#) compared to CIH group. Data are presented as mean ± standard deviation (SD). One-way analysis of variance (ANOVA) was used for multiple comparisons, N = 5. HuA, Huperzine A; OSA, obstructive sleep apnea; RXRA, retinoid X receptor alpha; HE, Hematoxylin and Eosin; CIH, chronic intermittent hypoxia.
To investigate the effects of HuA on CIH-induced oxidative stress and inflammation, we measured the ROS levels using ELISA. Results showed a significant increase in ROS expression in the CIH group compared to the control group, while the ROS expression was significantly down-regulated in the CIH + HuA group compared to the CIH group (Figure 2(a)). Additionally, brain LDH levels markedly enhanced in the CIH group compared to the control group, while significantly decreased in the CIH + HuA group (Figure 2(b)). Serum levels of TNF-α, IL-1β, and IL-6, measured by ELISA, were considerably elevated in the CIH group compared to the control group, and pronouncedly suppressed in the CIH + HuA group (Figure 2(c)–(e)). Western blot analysis of inflammatory markers NLRP3, GSDMD-N, Cleaved-Caspase 1, and Cleaved-IL-1β revealed dramatically elevated expression in the CIH group compared to the control group. In contrast, these levels were considerably decreased in the CIH + HuA group relative to the CIH group (Figure 2(f)). These findings suggest that CIH causes cognitive impairment, neuronal damage, oxidative stress and inflammatory response in mice, which can be alleviated by HuA treatment. HuA reduces oxidative stress and inflammation induced by OSA. Note: (a) ROS levels were detected by ELISA; (b) LDH levels in mouse brain; (c) Serum TNF-α levels were detected by ELISA; D: Serum IL-1β levels were detected by ELISA; E: Serum IL-6 levels were detected by ELISA; (f) Western blot detection of inflammatory markers NLRP3, GSDMD-N, Cleaved-Caspase 1, and Cleaved-IL-1β. Asterisks (*) indicate significance compared to control group; hash symbols (#) compared to CIH group. Data are presented as mean ± standard deviation (SD). One-way analysis of variance (ANOVA) was used for multiple comparisons, N = 5. HuA, Huperzine A; OSA, obstructive sleep apnea; RXRA, retinoid X receptor alpha; ROS, reactive oxygen species; ELISA, Enzyme-Linked Immunosorbent Assay; LDH, lactate dehydrogenase; TNF-α, tumor necrosis factor-alpha; GSDMD-N, Gasdermin D; NLRP3, NOD-like receptor family pyrin domain containing 3; IL-1β, Interleukin-1 beta; CIH, chronic intermittent hypoxia.
HuA promoted RXRA expression to transcriptionally regulate and enhance pyruvate carboxylase expression
To explore the potential regulatory mechanism of HuA in OSA, the downstream target genes of HuA were predicted and 100 potential target genes were identified. Among these genes, we noted the transcription factor, RXRA. To further understand the regulatory mechanisms, we predicted downstream target genes of RXRA and identified 1294 potential targets. Differential expression analysis of GSE38792 identified 71 DEGs in OSA (Figure 3(a)). The intersection of RXRA target genes and DEGs revealed one intersecting gene: pyruvate carboxylase (Figure 3(b)). This gene was not only predicted as a target of RXRA but also exhibited significant differential expression in OSA. Western blot analysis showed markedly reduced RXRA and pyruvate carboxylase expression in the CIH group compared to the control group, but both expressions augmented in the CIH + HuA group compared to the CIH group (Figure 3(c)). JASPAR database predicted two RXRA binding domains in the pyruvate carboxylase promoter region (Table 1). ChIP analysis of RXRA enrichment in the pyruvate carboxylase promoter region in CIH brain tissues showed substantially inhibited enrichment in the CIH group compared to the control group, while the CIH + HuA group exhibited notably increased RXRA enrichment compared to the CIH group (Figure 3(d)). Mutation of the binding sites and subsequent dual-luciferase assay showed significantly inhibited pyruvate carboxylase luciferase activity in the MUT group compared to the WT group (Figure 3(e)). Overexpression of RXRA augmented RXRA and pyruvate carboxylase expression compared to the oe-NC group (Figure 3(f)). ChIP analysis of RXRA enrichment in the pyruvate carboxylase promoter region confirmed notably increased enrichment in the oe-RXRA group compared to the oe-NC group (Figure 3(g)). These results indicate that decreased RXRA expression in OSA leads to suppressed pyruvate carboxylase expression, while HuA treatment promotes RXRA expression, thereby enhancing pyruvate carboxylase expression. HuA promotes RXRA expression to enhance pyruvate carboxylase expression. Note: (a) Heat map of differentially expressed genes in OSA, with gene expression color scale in the histogram at the top right (N: normal, T: treated); (b) Intersection of transcription factor target genes with significantly differentially expressed genes in OSA, the middle section indicates the intersection of the two datasets; (c) Western blot detection of the expression of pyruvate carboxylase ; (d) ChIP analysis of RXRA enrichment in the pyruvate carboxylase promoter region in CIH; (e) Dual-luciferase assay of RXRA’s effect on pyruvate carboxylase promoter activity; (f) Western blot detection of RXRA and pyruvate carboxylase expression levels; (g) ChIP analysis of RXRA enrichment in the pyruvate carboxylase promoter region in different groups. Asterisks (*) indicate significance compared to control or oe-NC group; hash symbols (#) compared to CIH group. Data are presented as mean ± standard deviation (SD). One-way analysis of variance (ANOVA) was used for multiple comparisons, N = 5. HuA, Huperzine A; ChIP, Chromatin Immunoprecipitation; CIH, chronic intermittent hypoxia; OSA, obstructive sleep apnea; RXRA, retinoid X receptor alpha. RXRA binding domains in the PC promoter region.

HuA alleviated OSA-induced cognitive impairment, neuronal damage, oxidative stress and inflammatory response by promoting RXRA and pyruvate carboxylase expression
Then, a CIH mouse model was established, followed by intraperitoneal injection of HuA and simultaneous RXRA silencing or RXRA silencing with pyruvate carboxylase overexpression. Western blot analysis showed significantly suppressed RXRA and pyruvate carboxylase expression in the CIH+HuA+sh-RXRA + oe-NC group compared to the CIH+HuA+sh-NC + oe-NC group, while pyruvate carboxylase expression markedly upregulated in the CIH+HuA+sh-RXRA + oe-pyruvate carboxylase group compared to the CIH+HuA+sh-RXRA + oe-NC group (Figure 4(a)). The Morris water maze test indicated substantially enhanced swimming path length and escape latency, and decreased platform crossings in the CIH+HuA+sh-RXRA + oe-NC group compared to the CIH+HuA+sh-NC + oe-NC group. Conversely, the CIH+HuA+sh-RXRA + oe-pyruvate carboxylase group showed notably decreased swimming path length and escape latency, and increased platform crossings compared to the CIH+HuA+sh-RXRA + oe-NC group (Figure 4(b)–(d)). HE and Nissl staining revealed statistically significantly enhanced brain tissue damage and decreased neuron number in the CIH+HuA+sh-RXRA + oe-NC group compared to the CIH+HuA+sh-NC + oe-NC group. The CIH+HuA+sh-RXRA + oe-pyruvate carboxylase group exhibited clearly reduced brain tissue damage and elevated neuron number compared to the CIH+HuA+sh-RXRA + oe-NC group (Figure 4(e)–(f)). HuA alleviates OSA-induced cognitive impairment and neuronal damage by promoting RXRA and pyruvate carboxylase expression. Note: (a) Western blot detection of RXRA and pyruvate carboxylase expression; (b) Swimming path length in Morris water maze test; (c) Escape latency in Morris water maze test; (d) Platform crossings in Morris water maze test; (e) Brain tissue pathology was evaluated by HE staining; (f) Neuronal count was assessed by Nissl staining. Asterisks (*) indicate significance compared to CIH+HuA+sh-NC + oe-NC group; hash symbols (#) compared to CIH+HuA+sh-RXRA + oe-NC group. Data are presented as mean ± standard deviation (SD). Multiple group comparisons were performed using one-way analysis of variance (ANOVA), N = 5. HuA, Huperzine A; OSA, obstructive sleep apnea; RXRA, retinoid X receptor alpha; ROS, reactive oxygen species; HE, Hematoxylin and Eosin; CIH, chronic intermittent hypoxia.
ELISA showed that ROS levels substantially increased in the CIH+HuA+sh-RXRA + oe-NC group compared to the CIH+HuA+sh-NC + oe-NC group (Figure 5(a)). LDH levels in the brain also significantly augmented in the CIH+HuA+sh-RXRA + oe-NC group (Figure 5(b)). Serum levels of TNF-α, IL-1β, and IL-6, measured by ELISA, were elevated in the CIH+HuA+sh-RXRA + oe-NC group compared to the CIH+HuA+sh-NC + oe-NC group (Figure 5(c)–(e)). Western blot analysis of NLRP3, GSDMD-N, Cleaved-Caspase 1, and Cleaved-IL-1β revealed markedly elevated expression in the CIH+HuA+sh-RXRA + oe-NC group compared to the CIH+HuA+sh-NC + oe-NC group and decreased expression in the CIH+HuA+sh-RXRA + oe-pyruvate carboxylase group when compared with the CIH+HuA+sh-RXRA + oe-NC group (Figure 5(f)). These findings suggest that HuA alleviates CIH-induced cognitive impairment, neuronal damage, oxidative stress and inflammatory response by promoting the RXRA/pyruvate carboxylase axis. HuA alleviates oxidative stress and inflammation in OSA by increasing RXRA expression. Note: (a) ROS levels were assessed by ELISA; (b) Brain LDH levels; (c) Serum TNF-α levels were assessed by ELISA; D: Serum IL-1β levels were assessed by ELISA; (e) Serum IL-6 levels were assessed by ELISA; F: Western blot detection of inflammatory markers NLRP3, GSDMD-N, Cleaved-Caspase 1, and Cleaved-IL-1β. Asterisks (*) indicate significance compared to CIH+HuA+sh-NC group. Data are presented as mean ± standard deviation (SD). One-way analysis of variance (ANOVA) was used for multiple comparisons, N = 5. HuA, Huperzine A; OSA, obstructive sleep apnea; RXRA, retinoid X receptor alpha; ROS, reactive oxygen species; ELISA, Enzyme-Linked Immunosorbent Assay; LDH, lactate dehydrogenase; TNF-α, tumor necrosis factor-alpha; GSDMD-N, Gasdermin D; NLRP3, NOD-like receptor family pyrin domain containing 3; IL-1β, Interleukin-1 beta; CIH, chronic intermittent hypoxia.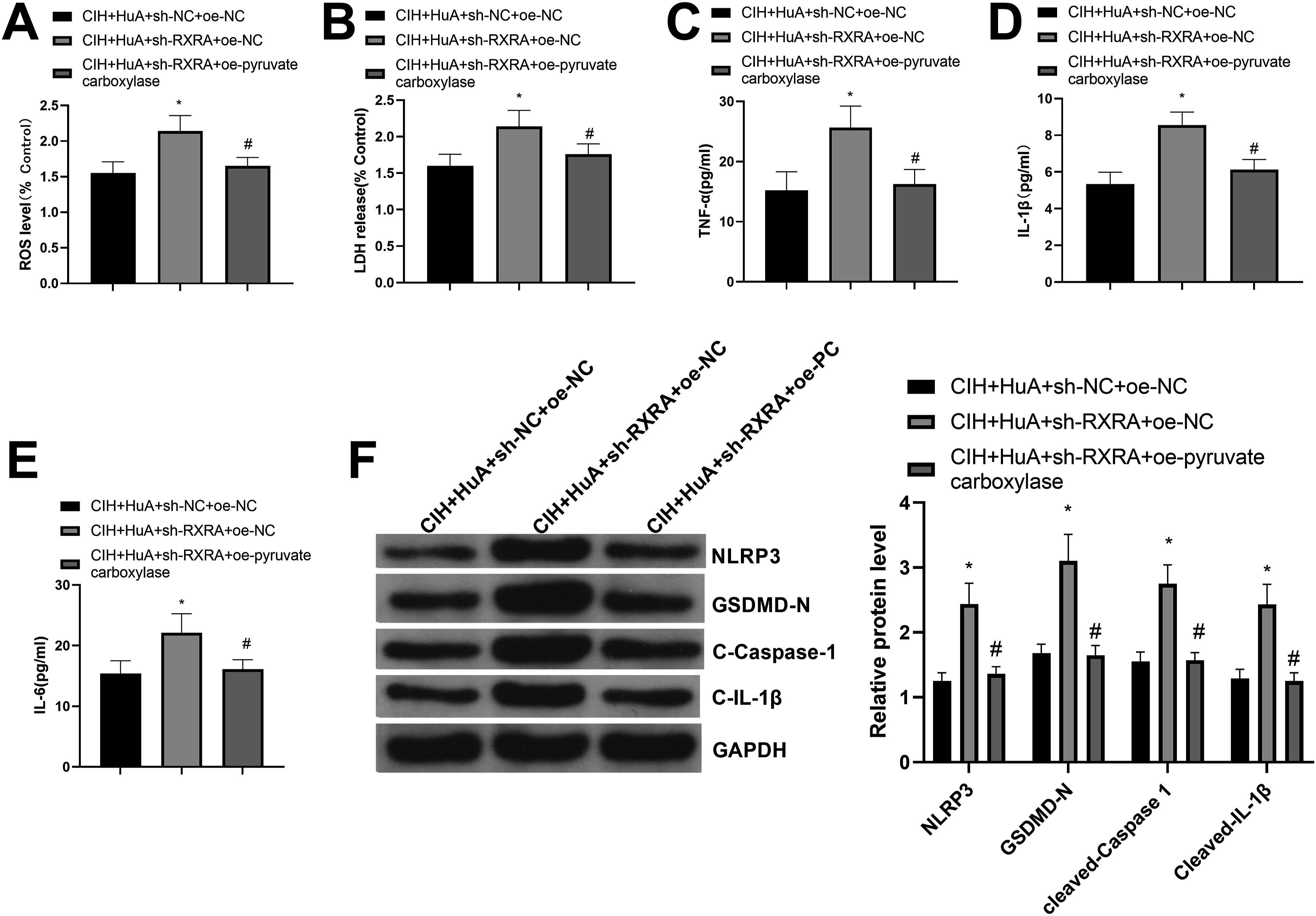
Discussion
Given the escalating global prevalence of OSA and its associated risks to health and productivity, 19 this study examined the impact of HuA on regulating the RXRA/pyruvate carboxylase axis to address the underlying molecular mechanisms of cognitive dysfunction, neuronal impairment, oxidative stress, and inflammation triggered by OSA. The findings revealed that HuA upregulated RXRA expression, consequently enhancing the expression of its downstream target gene pyruvate carboxylase in CIH mouse models. This modulation aided in attenuating diverse pathological responses elicited by CIH. These results present a novel perspective for further exploration into the potential therapeutic mechanisms of HuA in managing OSA.
In this current study, HuA exhibited remarkable efficacy in mitigating the pathological changes induced by CIH. The findings of this study suggested that HuA intervention led to substantial amelioration of cognitive impairments, neuronal injuries, oxidative stress, and inflammation in CIH-induced mouse models. Importantly, cognitive impairment is well-documented as a significant complication of OSA, affecting various cognitive domains such as memory, attention/vigilance, and executive function. 20 Our findings were consistent with previous studies, emphasizing the neuroprotective potential of HuA.21,22 For instance, Friedli et al. demonstrated that HuA enhanced cognitive function and mitigated neuronal damage in a mouse model of AD. 8 A study on AD explained that the protective role of HuA on neurons can be ascribed to reducing the loss of acetylcholine and preventing apoptosis against Aβ induction. 23 Consistently, HuA exhibits disease-modifying properties, including protective effects on neurons against oxidative damage induced by amyloid-β and mitochondrial dysfunction. 24 Similarly, the current study also found the neuron-protective role of HuA, as evidenced by attenuated brain tissue damage, increased neuron viability, reduced ROS expression and inflammatory cytokine secretion in response to HuA treatment. Recent findings also confirmed that HuA holds promise as a therapeutic agent for alleviating cognitive deficits in CIH-exposed mice. 11 But more evidence is required to fully understand the mechanism of HuA-mediated cognitive impairment in CIH-exposed mice.
CIH is recognized as a primary pathological mechanism underlying OSA-associated cognitive impairment through the initiation of neuroinflammation. 25 Emerging evidence indicates that the detrimental effects of hypoxia on cognitive impairments may be associated with changes in ion channel function, the release of glutamate-induced excitotoxicity, the overactivation of oxidative stress, and the upregulation of pro-inflammatory mediators. 26 Accumulating evidence has documented the critical role of neuroinflammation in the onset and progression of cognitive dysfunction associated with OSA. 27 Inflammation induced by OSA can lead to vascular endothelial cell impairment, thereby altering vascular structure and function; and endothelial dysfunction is implicated in various systemic morbidities, including cardiovascular disease, metabolic dysfunction, and importantly, neurocognitive impairment. 28 In supportive of the participation of HupA in anti-inflammation and neuroprotection, 29 our study demonstrated that HuA effectively diminished ROS production and inflammatory reactions triggered by CIH, resulting in reduced expression of inflammatory cytokines including TNF-α, IL-1β, and IL-6. HuA exerts its anti-inflammatory and neuroprotective effects through AChE inhibition and activation of nicotinic cholinergic receptors. Activation of these receptors by HuA reduces the release of cytokines like IL-1β and TNF-α, and inhibits NF-κB activation, thereby providing anti-inflammatory benefits and protection against oxidative stress. 30
Subsequently, we conducted an investigation into the potential regulatory mechanism of HuA in OSA. Our findings established that HuA had the capability to augment RXRA expression to the promotion of pyruvate carboxylase expression, thus mitigating various pathological manifestations triggered by CIH. Notably, RXRA conditional knockout mice exhibit normal behavior but demonstrate deficits in tasks associated with social memory. 31 In line with our results, recent research focusing on the retinoid X receptor (RXR) agonist bexarotene has validated its anti-inflammatory potential in myocardial infarction models. 32 RXRs are nuclear receptors (NRs) that regulate gene transcription in a ligand-dependent manner, functioning as homodimers or heterodimers with other NRs such as peroxisome proliferator-activated receptor γ (PPARγ) and liver X receptors (LXRs). 33 RXRA, in particular, forms heterodimers with various NRs to modulate the transcriptional activity of specific target genes. 13 Our findings also highlighted the modulation of pyruvate carboxylase via RXRA, which required further exploration. Pyruvate carboxylase is known to be transcriptionally regulated by PPARγ; specifically, increased PPARγ expression enhances pyruvate carboxylase levels and vice versa.34,35 Previous evidence has clarified that the inhibition effect of HuA on the secretion of inflammatory moleculers induced by lipopolysaccharide is PPARγ-dependent. 36
Collectively, this study uncovered the mechanism through which HuA mitigated the pathological responses triggered by OSA via the RXRA/pyruvate carboxylase axis, establishing the conceptual groundwork for future investigations into HuA’s neuroprotective and anti-inflammatory properties. Furthermore, the findings indicated that treatment with HuA significantly ameliorated cognitive impairment and neuronal damage arising from CIH, offering promising clinical implications. Although it elucidates the mechanism by which HuA functions through the RXRA/pyruvate carboxylase axis, uncertainties persist regarding the signaling pathways and involved molecules. Subsequent research endeavors should deepen the exploration of its mechanisms of action, and furnish a more comprehensive scientific foundation for addressing OSA.
Supplemental Material
Supplemental Material - In silico and in vivo experiments of Huperzine A modulating the development of obstructive sleep apnea by transcriptionally regulating pyruvate carboxylase expression via retinoid X receptor alpha
Supplemental Material for In silico and in vivo experiments of Huperzine A modulating the development of obstructive sleep apnea by transcriptionally regulating pyruvate carboxylase expression via retinoid X receptor alpha by Juan Huang, Hui Li, Qin Huang, Li Wang, Ying Wu and Xin Tan in Human & Experimental Toxicology
Footnotes
Ethical statement
Author Contributions
Xin Tan contributed to the conception of the study. Xin Tan, Juan Huang and Hui Li performed the experiment and contributed significantly to analysis. Qin Huang and Li Wang contributed significantly to manuscript preparation and performed the data analyses. Juan Huang and Hui Li wrote the manuscript. Qin Huang and Li Wang revised the manuscript. All authors approved the final version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is granted by Changsha Natural Science Foundation (Project No.: kq2202011), Natural Science Foundation of Hunan Province (Project No.: 2025JJ80480) and Health Commission of Hunan Province (Project No.: 202206012964).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The original contributions presented in the study are included in the article, and further inquiries can be directed to the corresponding author.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
