Abstract
Objective
The aim of the study is to examine the association between long-term occupational exposure to Mn and Fe and their health effects in workers.
Methods
108 Mn workers were selected for the Mn exposure groups; 92 non-Mn workers were in the control group. Inductively coupled plasma-mass spectrometry was used to determine the Mn and Fe concentration in the working environment. Graphite furnace-atomic absorption spectroscopy was used to determine the blood Mn concentration of workers. Serum inflammatory factors were measured by enzyme-linked immunosorbent assay.
Results
The blood Mn concentration, positive rate of clinical symptoms and serum inflammatory response in the Mn exposure group was higher than in the control group.
Conclusions
Low levels of Mn exposure may increase blood Mn concentrations, the rate of complaints of neurological symptoms and promote increased serum inflammatory response in workers.
Introduction
Neurodegenerative diseases, such as Parkinson’s disease (PD) and Alzheimer’s disease (AD) are associated with neuroinflammation. The occurrence of neuroinflammation is related to the activation of microglia and astrocytes. Activated microglia and astrocytes are common in brains of PD patients, and neuroinflammaory factors are greatly increased, including nitric oxide (NO), reactive oxygen species (ROS), TNF-α, IL-1β, IL-6 and so on.1–3 AD is characterized by extracellular β amyloid (Aβ) deposition and microglial neuroinflammatory release, inhibiting cerebrolinase and insulin-degrading enzyme. Therefore, adverse stimuli may cause microglia and astrocytes to activate, leading to their dysfunction, releasing inflammatory factors, stimulating and possibly intensifying the inflammatory response, impairing neuronal function, leading to the occurrence of neurodegenerative diseases.4–6
Manganese (Mn) is an essential trace element. It regulates normal growth and homeostasis within the cells and is essential for the activity of multiple enzymes and proteins.7,8 However, excessive Mn exposure in welders, smelters, foundries, finishers and other workers may cause neurological dysfunction. Mn accumulates in the hypothalamus, olfactory bulbs, globus pallidus, hippocampus, and cortex, to name a few regions of the brain. 9 Mn poisoning is characterized by extrapyramidal damage, causing motor dysfunction, “gear-like” increase in muscle tone, micro-tremors in the fingers, abnormal tendon reflexes, agitation, euphoria, and emotional instability, analogous to several of the symptoms inherent to PD. The mechanism of Mn poisoning is associated with inflammation, apoptosis, autophagy, oxidative stress, amino acid metabolism disorders, and abnormal metal metabolism.10–12
Iron (Fe) is one of the most abundant trace elements in human body. Fe plays a key role in brain development and physiology, neurotransmitter synthesis, cytoplasmic protein function, and mitochondrial responses. Recent studies suggest that excess Fe may be detrimental to neurodevelopmental, behavioral, and cognitive functions, especially at extreme ages.13,14 Excessive Fe deposition can cause neurological damage such as neurodegeneration and cognitive impairment, leading to neurological diseases.15,16
Mn poisoning activates immune effector cells (such as microglia) in a resting state of the central nervous system, stimulates their phagocytic function, prompting them to release inflammatory factors.17,18 High levels of Mn tend to accumulate in astrocytes and activate astrocytes, leading to cell enlargement. Activation occurs by recognizing Toll-like receptors and NOD-like receptors that secrete inflammatory cytokines in the central nervous system. The activated astrocytes are mainly marked by increased GFAP activity and can secrete proinflammatory chemokines CCL2 and CXCL1 to generate ROS in NF-κB signaling pathway. 19 Activated astrocytes can also stimulate activated microglia to release neuroinflammatory factors through oxidative stress, and induce neurodegeneration. Thus, microglia and astrocytes play an important role in the development of Mn poisoning. However, the neuroinflammatory mechanisms of Mn toxicity have yet to be fully understood.
Simultaneously, excessive Fe exposure can lead to the accumulation of Fe in the brain, leading to the development of AD or PD. 20 Mn and Fe readily accumulate in the central nervous system and cause neurotoxicity. The toxic mechanism may be related to oxidative stress, mitochondrial dysfunction, apoptosis, autophagy, protein homeostasis imbalance and inflammation.
In our earlier animal experiments, we found that Mn exposure increased the content of Mn in blood and brain, activated glial cells in thalamus, cortex and hippocampus, and apoptotic neurons in rats exposed to Mn. Levels of inflammatory factors (TNF-α, IL-1β, IL-6, PGE2, etc.) in these tissues were higher than those in the control group, and the serum inflammatory factors (TNF-α, IL-1β, IL-6, PGE2, etc.) in the rats exposed to Mn were higher than those in the control group, suggesting that Mn exposure may cause elevation in brain and serum inflammatory factors.21–23 However, at present, there is scant information on neuroinflammatory factors affected by occupational Mn exposure in workers. According to these studies, Mn can cause increased blood Mn, inflammatory indicators, and globulin globus indices in workers.24,25 In this study, inductively coupled plasma-mass spectrometry (ICP-MS) was used to determine the concentration of Mn in the working environment. Graphite furnace-atomic absorption spectrometry (GFAAS) was used to study the effect of Mn exposure on blood Mn in workers, and enzyme-linked immunosorbent assay (ELISA) was used to investigate the effects of Mn exposure on serum inflammatory factors.
Methods and materials
Study design and participants
The cross-sectional survey was carried out four factories in two cities in Guangxi, three are located in Baise City, Guangxi Province, which is rich in Mn ore, with workers in Mn and Fe smelting and related work (Mn exposure group), the other factory is in Guigang City, Guangxi Province, which mainly engages in sugar production, with workers in sugar making operations (control groups). A total of 200 workers were included (108 workers in Mn and Fe smelting and related work were selected as Mn exposure groups, which were stratified by job type, gender, age and working age; 92 non-Mn workers were used as control group.
Inclusion criteria: Age ≥18 years, worker with no physical illness, consent to participate in the study and sign informed consent. Subjects with current or past central nervous system disorders, such as cardiovascular and cerebrovascular diseases such as transient ischemic attack or brain tumors or other inflammatory diseases, alcohol or drug abusers, were not eligible for inclusion. Those who refused to participate in the survey were not considered for participation.
This study was approved by the ethics committee of Guangxi Medical University. All survey subjects participated voluntarily, and informed written consent was obtained from each participant.
Questionnaires
Basic information, occupational history, occupational disease history, family health history, menstrual history, marriage and childbirth history, symptoms, physical examination, laboratory examination, etc. Data were obtained using semi-structured questionnaires by interview to the participants.
Main reagents
Mn ion standard solution (National Reference Standards Research Center), Palladium chloride (Sinopharm Chemical Reagent Co., Ltd), TritonX-100 (Beijing Tianlai Biomedical Technology Co., Ltd), Nitric acid (superior grade pure), Human tumor necrosis factor α (TNF-α), prostaglandin E2 (PGE2), interleukin 1β (IL-1β), interleukin 2 (IL-2), interleukin 6 (IL-6), Interleukin 8 (IL-8) ELISA Kit (Shanghai Enzyme Exemption Company, China).
Exposure biomarkers and measurements
Blood samples were collected into a suitable vacuum tube by venipuncture of the elbow. 5 mL of biochemical coagulation blood and 5 mL of heparin sodium anticoagulation blood was collected from each worker. The biochemical coagulation blood was centrifuged to obtain the supernatant and stored at ultra-low temperature −80°C. Heparin sodium anticoagulated whole blood was stored in the refrigerator at −4°C, until further experimentation.
Graphite furnace working.

Determination of the detection limit: determination of the blank tube solution was determined for 20 times, followed by calculation of the standard deviation of the blank tube solution. Next, 3 times of the standard deviation of the blank tube solution was divided by the slope of the standard curve to calculate the minimum detection limit of Mn. The standard curve of Mn metal in whole blood was obtained and drawn, and the detection limit, recovery rate and precision of Mn metal in whole blood were determined.
Determination of standard recovery rate: 3 samples were randomly selected, each sample was divided into 4 small parts, one part was directly determined, and the other three small parts were added to low, medium and high concentration of Mn standard application solution for standard recovery rate determination. The background value of Mn and the content of Mn in the samples were measured, respectively, and the recovery rate was calculated. If the recovery rate is between 95% and 105%, it indicates that the measurement method is accurate. The specific method is as follows: recovery rate = (measured value of added standard sample − measured value of original sample)/added scalar × 100%.
Determination of relative standard deviation: 5 samples were randomly selected, each sample was measured 3 times, and the relative standard deviation (RSD) was calculated, and the relative standard deviation could not exceed 7%, indicating high measurement precision.
Enzyme-Linked Immunosorbent Assay (ELISA): The levels of TNF-α, PGE2, IL-1β, IL-2, IL-6, and IL-8 in serum samples were measured with commercially available ELISA kits. Serum samples collected above were processed and analyzed for TNF-α, PGE2, IL- 1β, IL-2, IL-6, and IL-8 levels, respectively, according to the manufacturer’s protocol.
Atmospheric dust exposure assessment
Adhering to the requirements of the “Workplace Air Hazardous Substances Monitoring and Sampling Specification GBZ159-2004,” atmospheric sampling in each factory was carried out. Fixed-point short-term fixed sampling of each workshop of each factory (including welding, furnace front operation, furnace surface operation, finishing, distribution room, etc.) was carried out, sampling at the breathing height of the worker’s operation at a flow rate of 10 L/min 15 min. Next, the microporous membrane was collected with atmospheric specimens and stored in a membrane cassette. Removing dust microporous membrane filter in the working environment pressure pot, add 3 ∼ 5 mL of nitric acid, 200°C heat, dust has been heated to nitric acid solution clear transparent, digestive juices again transferred to the test tube, the capacity, the use of deionized water editing instrument measurement equation and jamming equation, adjust the internal standard, according to the regression equation of dust Mn and Fe concentration.
ICP-MS atmospheric dust measurements: The Mn and Fe content in the air of the working environment was collected by microporous filter membrane, and the ICP-MS was used for measurement after heating digestion with 3–5 mL of concentrated nitric acid at 200°C. Before measurement, the ICP-MS parameter settings and working conditions were set, and the solution concentration gradient required for determination of Mn and in the air were configured. The concentration of Mn and Fe in dust was calculated according to the obtained regression equation.
Statistical analyses
Excel and SPSS24.0 statistical software are used to process and analyze the data. The data are expressed in
Results
Distribution of exposure
For the work location, position and job type characteristics, 19 air samples of Mn and Fe in the working environment were collected, and the geometric average concentrations of Mn in the working air environment, finishing operation point, smelting operation point, distribution room operation point, welding operation point, were 6 ug/m3, 3 ug/m3, 4 ug/m3, 5 ug/m3, 7 ug/m3, and 1 ug/m3, respectively. The current allowable concentration of Mn time-weighted in the air in China’s workplace (Mn-TWA, GBZ2.1-2007, 150 ug/m3). In addition, we measured the concentration of Fe, where the geometric average concentration in the working air environment, finishing operation point, smelting operation point, distribution room operation point, and welding operation point were 23.4 ug/m3, 19 ug/m3, 21 ug/m3, 13 ug/m3, and 15 ug/m3, respectively.
Participants
The basic situation of the workers
Population characteristics analysis of workers in two groups of occupational groups.

Comparison of the main complaint rate of clinical manifestations of two groups of workers
Comparison of clinical symptom complaint rates between the two groups.

Note: *Compared to the control group, *p < 0.05, **p < 0.01.
Association between exposure to Mn and blood Mn of workers
Graphite furnace - atomic absorption spectroscopy to determine the accuracy and precision of the results
Results of quality assurance.

Blood Mn in workers
Effect of manganese exposure on blood manganese concentration in workers (
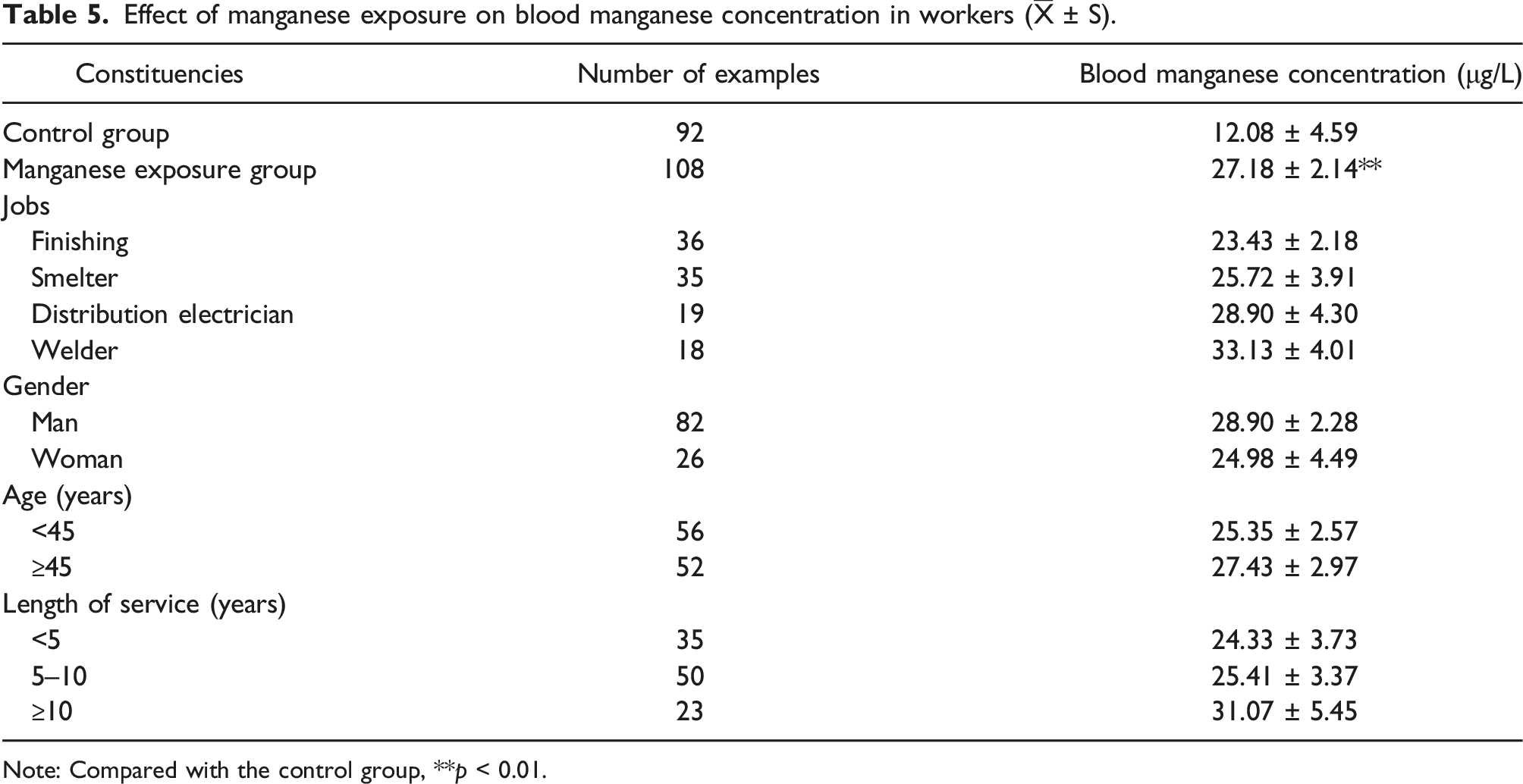
Note: Compared with the control group, **p < 0.01.
Binary logistic regression analysis of factors influencing blood Mn levels in workers
Logistic regression analysis of influencing factors of blood manganese levels in workers.

Association between exposure to Mn and serum inflammatory factors in workers
Effects of manganese exposure on serum inflammatory factors in workers (
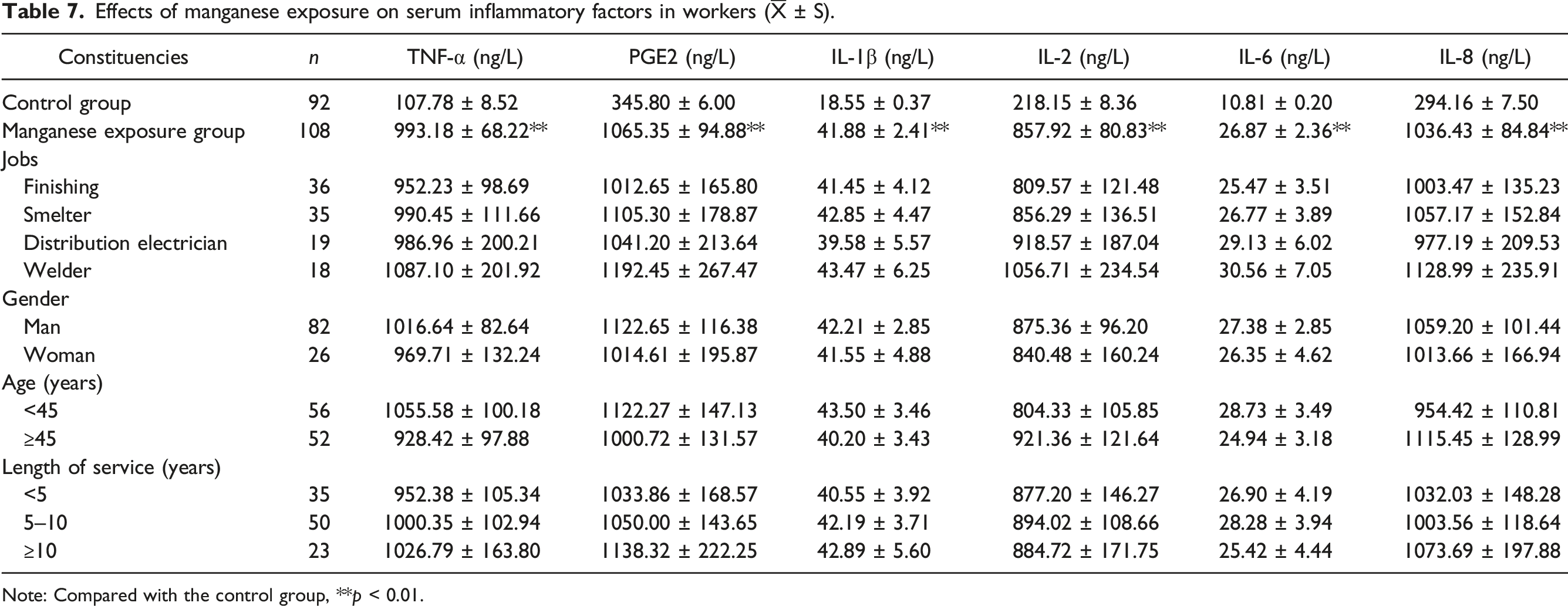
Note: Compared with the control group, **p < 0.01.
Analysis of the correlation between duration of work, age, blood manganese concentration and inflammatory factor concentration of manganese exposure workers (n = 108).
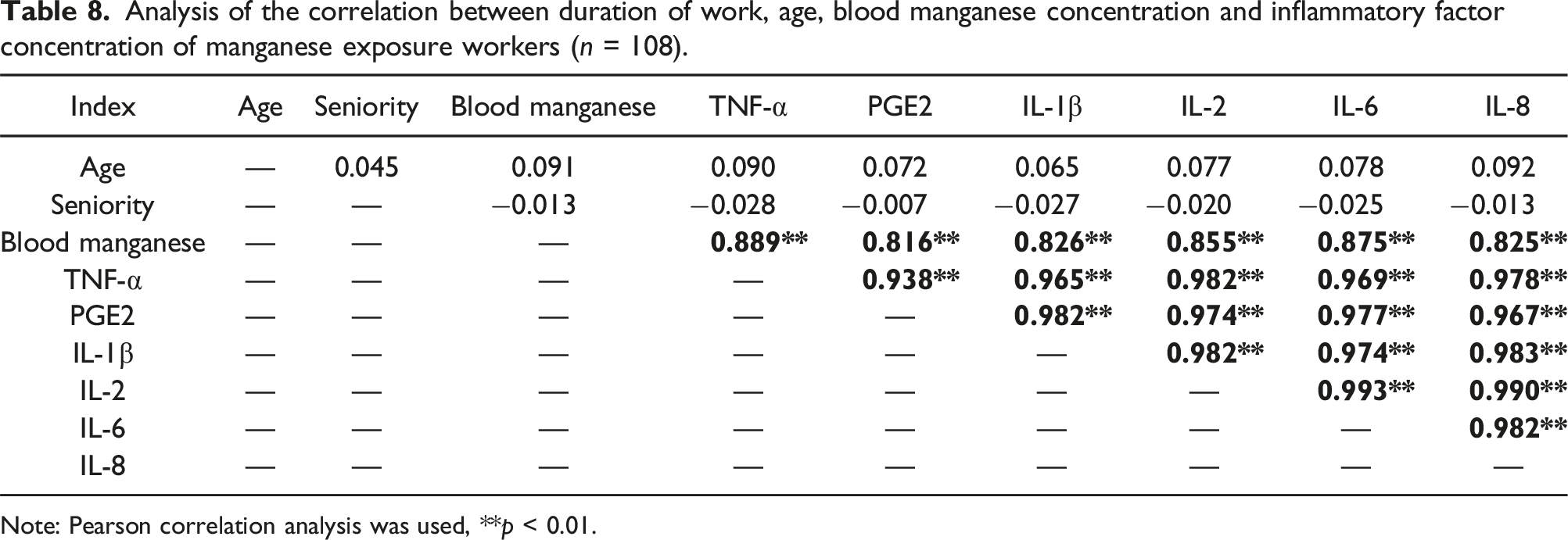
Note: Pearson correlation analysis was used, **p < 0.01.
Results of multiple linear regression analysis of blood manganese concentration and serum inflammatory factors (n = 108).

Discussion
Long-term exposure to low Mn concentrations may cause its brain accumulation, damaging the central nervous system, especially the extrapyramidal system, affecting learning, memory, movement and cognition.26–29 Mn exposure is common in mining, crushing, transportation, processing and smelting industries, as well as welding.
This study showed that the average geometric concentration of Mn in the working environment air was 6 ug/m3 in the working environment air Mn geometrical concentration, which was significantly lower than the current Mn time-weighted average allowable concentration (PC - TWA, 150 ug/m3). The Mn exposure concentration from high to low was in order of welding, power distribution room, smelting and finishing operations, which were all higher than those in non-Mn working points (control group). We also analyzed the average geometric concentration of Fe in the air of the working environment, which was 23 ug/m3, 3.8 times that of Mn exposure, however, at present, China has not established occupational health standards for Fe. The positive rates of dizziness, headache, memory loss, distraction, fatigue and blood Mn levels in Mn exposed workers were higher than those in the control group. The exposure level of Mn in the ambient air of welders, electricians, smelters and finishing workers increased with blood Mn level. Analysis of occupational populations showed that no differences in the composition of the sex, age and working age of the survey, hence it was speculated that the binary logistic regression model of the influencing factors of blood Mn level contributed little to the influence on blood Mn levels. Combined with the on-site Mn operation conditions, the Mn exposed (welder, distributor, smelter, finisher) and the control workers (cleaner, canteen worker, fitter, etc.) were included in the binary logistic regression model, establishing a 5.32-fold risk for increased blood Mn levels on the Mn-exposed versus non-exposed workers. There was no difference in blood Mn level related to gender, age and length of service, failing to support the notion that the longer the Mn exposure, the higher the blood Mn level.30,31 We also found a trend suggesting that the higher the working age of the Mn exposed workers, the higher their blood Mn level; however, the lack of statistical significance suggests an insufficient sample size. The positive rate of complaints of discomfort in the Mn-exposed workers was higher than in the control workers, consistent with results of occupational epidemiological investigations.32,33
Mn neurotoxicity is closely related to inflammation.34–36 Mn has been shown to activate microglia to produce a variety of inflammatory factors, such as TNF-α, IL-1β, NO, PGE2, causing an inflammatory response, which in turn affects neuronal function.37–40 Mn also activates astrocytes, releasing neurotoxic cytokines and promoting pro-inflammatory responses, with overexpression of TNF-α and IL-1β as well as release of NO. Upon Mn exposure, the levels of blood Mn, inflammatory factors (TNF-α, IL-1β, IL-6) and pallidal index (PI) in the nonexposed group were lower than in the exposed group,41,42 and neurological symptoms were less frequent. 27 Here, we found that concentrations of serum inflammatory factors TNF-α, PGE2, IL-1β, IL-2, IL-6 and IL-8 in the Mn exposed group were higher than those in the control group. Interestingly, our previous study found that Mn exposure induced increased ROS levels and expression of TNF-α and IL-1β in BV2 cells. 43 However, the levels of ROS and inflammatory factors induced by Mn and Fe combined exposure were lower than those of the Mn alone group, but higher than those of the Fe alone group, suggesting that Fe can antagonize the Mn-induced oxidative stress and neuroinflammation.43,44 Correlation analysis showed that blood Mn concentration was positively correlated with serum TNF-α, PGE2, IL-1β, IL-2, IL-6, and IL-8 concentrations. In addition, the concentrations of various serum inflammatory factors (TNF-α, PGE2, IL-1β, IL-2, IL-6, and IL-8) were positively correlated with each other. Blood Mn concentrations, TNF-α, IL-6, and IL-8 contributed to the regression model. These results indicated that Mn exposure increased Mn concentration in the blood of workers and also led to increased serum TNF-α, PGE2, IL-1β, IL-2, IL-6 and IL-8 levels in workers, whether Mn exposure was related to Fe exposure was not clear. A positive correlation between blood Mn and serum inflammatory factors was noted, suggesting that low levels of Mn exposure promote increased serum inflammatory response in workers. In the present study, we mainly explored the association between Mn exposure and inflammatory factors, and did not address possible interactions between Mn and Fe. Therefore, more studies are needed to clarify the exact mechanisms of the interaction between Mn and Fe.
Conclusion
This novel study showed that low-level Mn exposure not only caused increased blood Mn concentrations and positive rates of neurologic symptoms in workers, but were also associated with high serum TNF-α, PGE2, IL-1β, IL-2, IL-6, elevated IL-8 levels and a positive correlation between blood Mn and serum inflammatory factors. Our study suggests that low-level Mn exposure may lead to increased serum inflammatory levels in workers, requiring further investigation.
Limitations
Firstly, in this novel study, our findings indicated that Mn exposure may elevate serum inflammatory levels in workers. However, due to the low concentrations detected in the workplace, we did not explore the relationship between Fe and serum inflammatory cytokine levels. Secondly, the relevant data on the symptoms experienced by workers were obtained through self-reported questionnaires, which may introduce bias when evaluating these diseases. Lastly, our research design is cross-sectional and lacks relevant sample sizes, which limits our ability to verify the effects of long-term or low-level Mn exposure on serum inflammation in workers. In addition, it is still necessary to construct in vitro and in vivo models to verify how Mn exposure leads to elevated levels of inflammatory factors in workers, and to obtain relevant results through long-term follow-up through cohort studies.
Footnotes
Author contributions
Dianyin Liang: conceptualization, investigation, formal analysis, writing—original draft, writing—review and editing. Jianchao Peng and Bingyan Xie: investigation, formal analysis, writing—original draft, supervision, writing—review and editing. Wenxia Qin: investigation, methodology, formal analysis, data curation. Michael Aschner: revised and polished the paper. Shiyan Ou: Investigation, Supervision. Yueming Jiang: supervision, project administration, funding acquisition, writing—review and editing. Dianyin Liang, Jianchao Peng and Bingyan Xie have contributed equally to this article. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by grants from the National Natural Science Foundation of China (NSFC 81973094, 81460505).
Ethical statement
Informed consent
Participation was voluntary, and written informed consent was obtained from all parents of the participants before enrollment.
Data availability statement
All data generated or analyzed during this study are included in this published article.
