Abstract
Several studies investigated the application of Mesenchymal stem cells (MSCs) for treating spermatogenic disorders. Considering the limitation of MSC application, the present study aimed to compare Wharton’s jelly MSCs secretomes, including condition medium (CM) 10-fold concentrated (CM10), 20-fold concentrated CM (CM20), and extracellular vesicles (EVs) to restore busulfan-induced damage on male mice reproduction. So, Wharton’s jelly MSCs were cultured, CM was collected, and EVs were isolated. Seventy-two mice were randomly assigned to nine groups, including Control, Busulfan 1 month (1M), Busulfan 2 months (2M), CM10, Busulfan + CM10, CM20, Busulfan + CM20, EVs, and Busulfan + EVs groups. Sperm characteristics, DNA maturity, DNA fragmentation index (DFI), and testicular gene expression were evaluated. Data analysis revealed that CM10 significantly improved sperm plasma membrane integrity, sperm DNA maturity, and DFI in the Busulfan + CM10 group compared to the Busulfan 2M group. Although CM20 and EVs showed a non-significant improvement. Gene expression analysis showed busulfan administration significantly decreased the expression of AR, CREB1, and PLCζ genes, while CM10 significantly restored CREB1 gene expression. The present study demonstrated that CM10 is more effective than CM20 or EVs in reducing busulfan-induced reproductive toxicity.
Introduction
Infertility is a public health concern with substantial prevalence in couples all over the world. 1 Impaired spermatogenesis plays an essential role in male infertility. 2 Many factors such as obesity, diet, chronic diseases, tobacco, marijuana, and environmental toxins can cause harmful effects on sperm parameters. 3 Busulfan is a myeloablative alkylating agent that could be used for chronic myelogenous leukemia chemotherapy and as an immunosuppressive agent before bone marrow transplantation.4,5 Busulfan induces detrimental effects on male gonads, resulting in testicular atrophy, oligospermia, and azoospermia. 6 It is frequently utilized to establish oligospermia and azoospermia in animal models.5,7–9
The Mesenchymal stem cells (MSCs) are pivotal in regulating biological and pathological processes, including the maintenance of organ homeostasis, tissue repair, and wound healing. They find applications in regenerative medicine and tissue engineering. 10 However, MSCs can be obtained from different tissues, human umbilical cord MSCs are particularly favored in cell-based treatments and regenerative medicine. Their advantages include a noninvasive isolation method, absence of ethical considerations, lower immunogenicity, and faster self-renewal ability compared to alternative sources. 11 Cell-free therapy, which utilizes cell secretomes, is a therapeutic approach that has gained more attention nowadays. The reason for favoring secretomes over intact cells lies in the finding that a critical mechanism of stem cells operates through their secreted factors. Cell-free therapy offers a way to circumvent the limitations associated with cell-based treatments, including rejection by the immune system, genetic instability due to senescence, and the potential risk of developing malignancies. 12 In addition, the derivatives of MSCs’ secretomes, such as condition medium (CM) or extracellular vesicles (EVs), offer considerable advantages for manufacturing, storage, and handling. 13 The CM comprises a variety of growth factors and agents for tissue regeneration that are produced by the stem cells. Research has tested its efficacy in treating various diseases. 14 The EVs are small vesicles enclosed by a lipid membrane with a size in the nanometer range. They are released by different cells and carry a combination of lipids, proteins, and different nucleic acids from the originating cell. 15 They could be detected in the body fluids like urine, blood, and seminal plasma. 16 The objectives of our study were to investigate the potential protective effects of human Wharton’s jelly Mesenchymal Stem Cell (hWJMSCs)-derived CM or EVs on sperm parameters and testicular gene expression in a busulfan-induced mouse model of male reproductive toxicity.
Materials and methods
Isolation and culture of hWJMSCs and preparation of hWJMSC-CM
The human umbilical cords were acquired from full-term infants delivered via a caesarian section at Hafez Hospital following the receipt of written informed consent from their parents. All samples were prepared following the Ethics Committee of Shiraz University of Medical Sciences (IR.SUMS.AEC.1400.002). The umbilical cords were gathered in chilled phosphate-buffered saline (PBS) (Shell max, USA) that contained 100 μg/mL penicillin (Gibco, UK) and 100 U/mL streptomycin (Gibco, UK). First, the umbilical vein was opened, and then the endothelium of the vein and amniotic epithelium were scraped, and both arteries were discarded. Subsequently, small explant pieces, approximately 4 by 5 mm and made from the umbilical cord matrix, were placed in a petri dish (SPL Lifesciences, Korea). After 15 min, a completed α-Minimal Essential Medium (α-MEM) (Shellmax, USA) with 10% fetal bovine serum (FBS) (Kiazist, Iran), 1%
Characterization of hWJMSCs
Cell morphology assessment
After the expansion phase of growth, isolated MSCs were evaluated using a light microscope.
Flow cytometry assay
The hWJMSCs from the third passage were utilized to assess cell surface markers via flow cytometry. Cells were separated from the flask (NEST, China) using 0.25% Trypsin-EDTA (Dacell, Iran). The separated cells were then centrifuged for 5 min at 1200 r/min. The cell suspension consisted of 106 cells per ml in 100 μL PBS (Shell max, USA) was prepared and incubated for 30 min at 4°C with anti-human antibodies against CD73-PE, anti-CD144, phycoerythrin–conjugated anti-CD34, and CD90 antibodies (all purchased from Abcam, UK). Cell surface staining was conducted using isotype antibodies conjugated to fluorescein isothiocyanate (FITC) or Phycoerythrin (PE). Flow cytometry (BD, USA) and FlowJo™ software (TreeStar, Ashland, OR, USA) were applied for cell analysis.22,23
Adipogenic and osteogenic differentiation
Cells from the 3rd passage were treated with the adipogenic and osteogenic media for a duration of 3 and 4 weeks, respectively. The medium was refreshed every 3 days. The adipogenic medium consisted of α-MEM (Shellmax, USA) with 10% FBS (Kiazist, Iran), 50 μg/mL indomethacin (Sigma, USA), 50 μg/mL ascorbic acid-2 phosphate (Merck, Germany), and 100 nM dexamethasone (Sigma, USA). Adipocyte differentiation was assessed using oil-red O staining. Firstly, cells were fixed with 4% paraformaldehyde (Merck, Germany). Secondly, they were stained with 0.5% oil-red O (Sigma Aldrich, USA) in isopropyl alcohol (Merck, Germany). The medium for osteogenic differentiation consisted of α-MEM enriched with 10% FBS (Kiazist, Iran), 10 mM β-glycerophosphate (Sigma, USA), 10 nM dexamethasone (Sigma, USA), 50 μg/mL ascorbic acid-2 phosphate, and 2.1604 g/L 6-glycerol phosphate (Merck, Germany). The cells were fixed with methanol (Merck, Germany), and the potential of MSCs to differentiate into osteoblasts was assessed using Alizarin-red S (Sigma Aldrich, USA) staining. The extracellular staining was visualized using an inverted phase-contrast microscope.22,23
EVs isolation
The EVs were isolated from MSCs-CM using the Exocib kit (Cib Biotech, Iran) based on the manufacturer’s instructions. Briefly, the MSCs-CM was collected and centrifuged at 3000 r/min for 10 min to eliminate cellular debris. Then, a 5:1 ratio of supernatant was mixed with Exocib solution and incubated for 12 h at 4°C. The combination was centrifuged for 40 min at 3000 r/min. After that, the supernatant was removed, and the pellet was resuspended in 100 μL of PBS. Finally, the isolated EVs product was stored at −20°C.
Transmission electron microscopy (TEM)
Transmission electron microscopy (LEO 906E, Zeiss, Germany) was employed to characterize the EVs’ size and morphology. The EVs were diluted 1:10 in PBS (Shell max, USA), then fixed onto copper grids, air-dried at room temperature, and analyzed by TEM without staining. The size distribution of EVs was estimated using ImageJ software (Java 1.8.0_112).
Dynamic light scattering (DLS)
The DLS technique can measure the size distribution of particles with diameters ranging from 1 nm to 6 μm. When the laser beam hits the particles such as EVs, they disseminate light. A mathematical model based on Brownian motion and light scattering theory was utilized by analyzing the intensity fluctuations of the scattered light. 24 The average size distribution was calculated by assessment of seven samples of EVs.
Experimental groups
Seventy-two (8‐9 weeks old) BALB/c male mice (29 ± 3g) were supplied by the Experimental Animal Research Center of the Shiraz University of Medical Science. These mice were housed in polypropylene cages at 25 ± 2°C temperature and were subject to a 12 h light-dark cycle at 40 ± 5% relative humidity. They were provided unrestricted access to both water and food throughout the study. All procedures were carried out following the prescribed guidelines set by the Animal Care and Ethics Committee of Shiraz University of Medical Sciences (IR.SUMS.AEC.1400.002) and adhered to the NIH Guide for the Care and Use of Laboratory Animals (NIH publication No. 85–23, revised in 1996).
The mice were allocated at random to nine groups, including Control, Busulfan 1M group, Busulfan 2M, CM10, Busulfan + CM10, CM20, Busulfan + CM20, EVs, and Busulfan + EVs groups (Figure 1). The CM10, CM20, and EVs groups were assigned to explore the effects of these substances on reproductive parameters under physiological conditions. Furthermore, Busulfan + CM10, Busulfan + CM20, and Busulfan + EVs groups were established to assess the therapeutic effectiveness of CM10, CM20, and EVs in the context of busulfan-induced reproductive toxicity. Flow diagram of the study.
Busulfan was diluted in dimethyl sulfoxide (DMSO) to achieve a concentration of 10 mg/mL. Subsequently, 0.2 mL of isotonic saline was added to 0.1 mL busulfan–DMSO solution for additional dilution. 7 Diluted busulfan (40 mg/kg, single-dose) was injected intraperitoneally (IP). Considering that the spermatogenesis cycle in mice lasts 34.5 days,25,26 the Busulfan 1M group was euthanized after 35 days to confirm the establishment of the oligospermia or azoospermia model.27,28
To be sure about the proper diffusion of CM and EVs, a dye was injected into the testes under deep anesthesia by IP administration of ketamine (100 mg/kg) and xylazine (5 mg/kg). After testis dissection, the distribution of the dye was evaluated. Once the suitable injection site was confirmed, mice in the experimental groups were anesthetized, and 10 μL of CM or EVs were injected into each testis for 3–5 min. Busulfan + CM10, Busulfan + CM20, and Busulfan + EVs groups received CM or EVs 35 days after busulfan injection. Also, before euthanasia, the mice underwent anesthesia using an IP administration of ketamine and xylazine.
Sperm and testes collection
Mice were euthanized, and their testes were harvested for gene expression assessments. The caudal part of the epididymis was separated, crashed, and placed in preheated Petri dishes (SPL Lifesciences, Korea) containing sperm washing medium (Inoclon, Iran) supplemented with 10% human serum albumin (Kedrion Biopharma, Italy). The mixture was placed in an incubator for 15 min at 37°C.
Sperm count
The sperm count was determined by diluting the sperm suspension with distilled water at a ratio of 1:10. The sperm count was calculated using a hemocytometer slide. The settled sperm were counted under a light microscope at 400x magnification. 29
Sperm motility
The motility of sperm was evaluated using a light microscope at 400x magnifications. The sperms were classified into three categories: progressive, non-progressive, and immotile sperms, based on the WHO laboratory manual for semen parameters. 30
Sperm morphology
For sperm morphology evaluation, sperm smears were prepared and stained using a commercial Diff-Quick kit (Ideh Varzan Farda, Iran) following the manufacturer’s instructions. Then, under the light microscopy, the percentage of normal and abnormal spermatozoa were determined at 400x magnification.
Hypo-osmotic swelling (HOS) test
The Hos test was employed to assess sperm plasma membrane integrity. Therefore, 10 µL of sperm suspension was mixed with 50 µL of hypoosmotic solution (50-mOsm NaCl) and incubated for 10 min at 37°C. Subsequently, the percentage of sperms with a swollen coiled tail, considered as live sperms with intact membrane, was calculated using a light microscope at 400x magnification.31,32
Sperm chromatin maturation assay (SCMA)
To evaluate the maturity of chromatin, SCMA kit (Ideh Varzan Farda, Iran) was used. The sperm sample was centrifuged in PBS (Shell max, USA) at 300 g for 10 min. Then, a thin smear of sperms was prepared and permitted to air dry. Then, the smear was treated according to the manufacturer’s instructions. The percentages of immature sperms (blue) and mature sperms (pink) were calculated using a light microscope at 400x magnification.
Acridine orange (AO) test
A thick sperm smear was fixed by Carnoy's fixative (methanol: acetic acid, 1:3) (both from Merck, Germany) for at least 2 hours. Subsequently, 0.19% acridine orange in phosphate-citrate buffer (all from Sigma Aldrich, USA), pH = 2.5, was added to the smear for 10 minutes, and then, gently rinsed with water. The sperms were then counted under a fluorescence microscope to estimate the number of sperms with a green head (double-stranded DNA) and a red or yellow head (denatured or single-stranded DNA). 33
Sperm chromatin dispersion (SCD) test
The SCD test or Halo sperm test is a valuable technique used to detect sperm DNA fragmentation. In this test, sperms are exposed to an acid solution before being treated with a lysis buffer. In spermatozoa with fragmented DNA, this process results in either a complete absence or minimal halo formation. Conversely, sperm without DNA fragmentation exhibit big or medium halos of dispersed DNA. These differences can be observed by bright field microscopy or fluorescence microscopy. 34 This test was conducted using a commercially available kit (Ideh Varzan Farda, Iran) according to the manufacturer’s instructions, and the samples were assessed using a light microscope.
Real-time polymerase chain reaction (real-time PCR)
Primer sequences used for quantities real-time PCR.

Statistical analysis
The Statistical Package of Social Science (SPSS) software version 21 was utilized for statistical analyses. The normality of the sperm parameters and sperm DNA data was assessed by the Kolmogorov-Smirnov test. After confirming the normal distribution of data and homogeneity of variances, One-way analysis of variance (ANOVA) with Tukey’s post-hoc test was employed to determine inter-group differences. In addition, PCR data analysis was carried out using the Kruskal-Wallis test. A p-value <.05 was considered statistically significant.
Result
Characterization of hWJMSCs
Light microscopy evaluations showed that monolayer MSCs were homogenous and spindle-shaped (Figure 2). Flow cytometric analysis confirmed the high expression level of MSC markers (CD73, CD90) and low expression level of hematopoietic markers (CD34, CD144) in isolated cells (Figure 3(a)). Furthermore, Oil-Red O staining detected lipid droplets formation in the differentiating MSCs (Figure 3(b)), and Alizarin Red staining showed deposited calcium that confirmed the differentiation of osteoblasts (Figure 3(c)). Morphology of hWJMSCs under light microscope. Characterization of hWJMSCs: (a) Flow cytometry analysis of hWJMSC surface markers. (b) Oil-Red O staining of adipogenic differentiation of hWJMSCs. (c) Alizarin Red staining of Osteogenic differentiation of hWJMSCs.

Characterization of isolated EVs
Transmission electron microscopy
Under the TEM microscope, groups of round or discoid-shaped particles were observed (Figure 4). The TEM imaging. (a) The TEM image of EVs. (b) The size distribution of EVs according to TEM image assessed by ImageJ software.
Dynamic light scattering
To measure the size of EVs, DLS has been used. The mean value of the EVs diameter was 18.37 nm, 10% of EVs had diameters of less than 13.75 nm, 50% less than 17.75 nm, and 90% less than 24.41 nm.
Effects of hWJMSCs-derived CM or EVs on sperm count, motility, morphology, and sperm plasma membrane integrity
Sperm count
Sperm count significantly decreased in the Busulfan 2M group compared to the control group (p < .001). Sperm concentration non-significantly improved in busulfan-treated groups (Busulfan + CM10, Busulfan + CM20, Busulfan + EVs) compared to the Busulfan 2M group. Notably, the sperm concentration in busulfan-received mice, even after treatment with CM or EVs, remained lower than the control group (p < .001) (Figure 5(a)). Comparison of sperm parameters in experimental Groups: (a) sperm count, (b) motility, (c) morphology, and d sperm plasma membrane integrity. Different letters indicate statistically significant differences (p < .05). Bars show the mean ± SE.
Sperm motility
The data indicated that busulfan administration significantly reduced the percentage of motile sperms compared to the control group (p < .001). However, treatment of busulfan-treated mice with CM10 or EVs enhanced the percentage of motile sperms (p < .05). The results showed that CM10 was more effective than EVs to ameliorate busulfan-induced side effects on sperm motility (p < .001). Despite these improvements, none of the treatments were able to increase sperm movement to the level observed in the control group (p < .001) (Figure 5(b)).
Sperm morphology
Regardless of the type of treatment, the busulfan administration led to a significant decrease in the percentage of sperms with normal morphology compared to the control group (p < .001). All the treatments non-significantly enhanced the percentage of the sperms with normal morphology, and there remained a marked difference between these groups and the control group (p < .001) (Figure 5(c)).
Sperm plasma membrane integrity
The HOS test was employed to evaluate sperm plasma membrane integrity. Findings showed that busulfan notably disrupted sperm plasma membrane integrity (p < .001). The CM10 significantly enhanced the percentage of sperms with intact plasma compared to the Busulfan 2M group (p < .05). Furthermore, the treatments were not able to restore sperm plasma membrane integrity to the same level as the control group (p < .001) (Figure 5(d)).
Effects of hWJMSCs-derived CM or EVs on sperm DNA maturity and fragmentation
Sperm chromatin maturation assay
Assessment of sperm chromatin maturity status revealed that after 2 months of busulfan administration, the number of immature sperms significantly increased compared to the control group (p < .001). Sperm maturity significantly increased in the Busulfan + CM10 group compared to the Busulfan 2M group (p ≤ .001). However, even in this group, the percentage of immature sperms was significantly lower than the control group (p < .001) (Figure 6). Sperm chromatin maturation assay. Different letters indicate statistically significant differences (p < .05). Bars show the mean ± SE.
Acridine orange
The percentage of sperms with single-strand or denatured nuclear DNA was significantly lower in the control group compared to the Busulfan 2M group (p < .001). Although CM10 administration led to a significant improvement in the percentage of the sperms with green fluorescent-stained heads in the Busulfan + CM10 group compared to the Busulfan 2M group (p < .05), it did not normalize as compared to the control group (p < .001). The CM20 or EVs administration just non-significantly decreased the sperm DNA fragmentation index (DFI) (Figure 7(a)). Sperm DNA fragmentation: (a) Acridine Orange, (b) Sperm Chromatin Dispersion. Different letters indicate statistically significant differences (p < .05). Bars show the mean ± SE.
Sperm chromatin dispersion test
Halo sperm test result.

Data are presented as means ± SE.
ap < .05 compared to the control group.
bp < .05 compared to the Busulfan 2M group.
Effects of hWJMSCs-derived CM or EVs on testicular gene expression
The real-time PCR demonstrated that expressions of AR, CREB1, and PLCζ in testicular tissue significantly decreased in the Busulfan 2M group compared to the control group (p < .001). In the busulfan-received mice, CM10 significantly enhanced the expression level of CREB1 compared to the Busulfan 2M group (p < .05), and there was no statistically significant difference compared to the control group. The CM10 had no impact on AR and PLCζ expressions. The other treatments did not have any significant impact on the gene expression levels (Figure 8). Real-time RT-qPCR analysis of the expression of (a) AR, (b) CREB1, and (c) PLCζ mRNAs. Different letters indicate statistically significant differences (p < .05). Bars show the mean ± SE.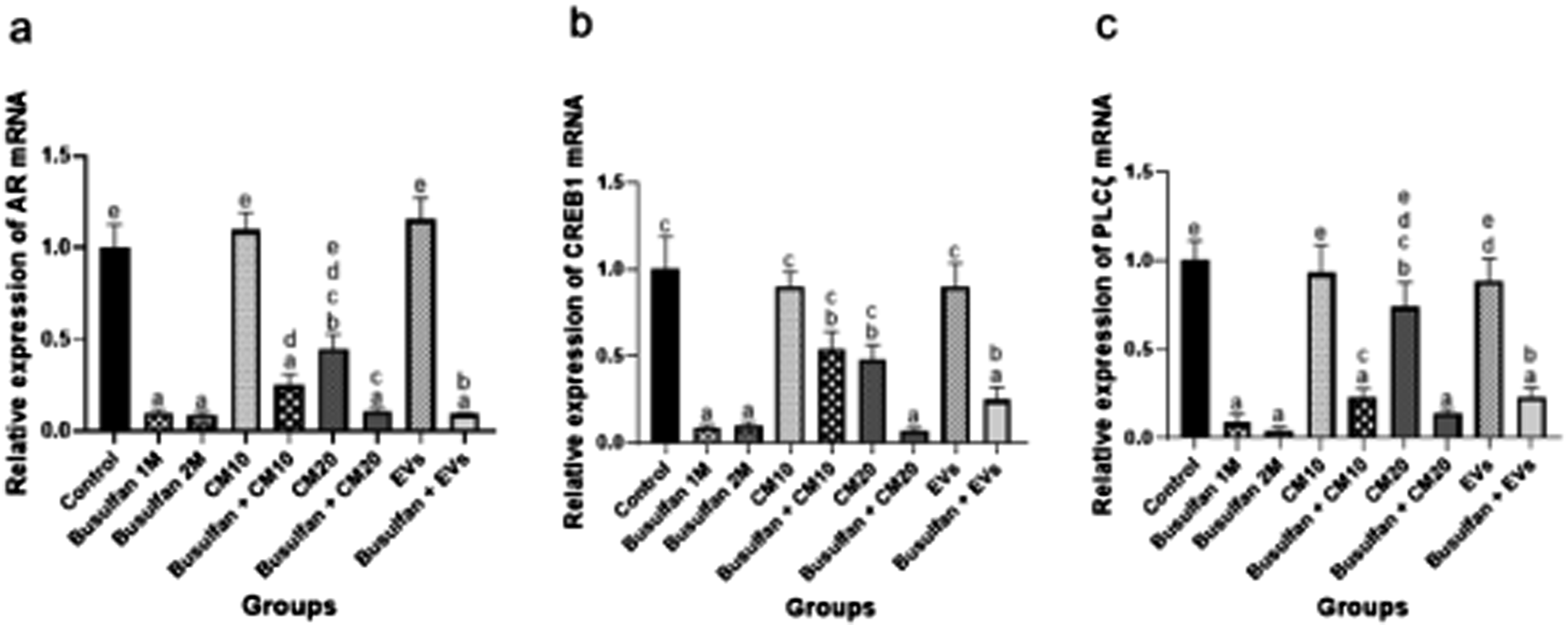
Discussion
To the best of our knowledge, this is the first in vivo study that compares the effectiveness of cell-free therapy methods, including CM and EVs, in an animal model of male reproductive toxicity. Our findings demonstrated that busulfan impairs all mentioned sperm parameters and testicular tissue mRNA gene expressions of AR, CREB1, and PLCζ. Among the treatments, CM10 showed superior effects on sperm plasma membrane integrity, maturity, DNA fragmentation, and the CREB1 expression level in busulfan-received mice.
Bader and colleagues reported that incubation of human spermatozoa with Adipose-Derived Mesenchymal Stem Cell CM (AD-MSC-CM) didn’t improve sperm morphology but preserved sperm viability. 35 Additionally, an in vitro study of AD-MSC-CM revealed that sperm total motility and plasma membrane integrity did not improve 2 hours after incubation of semen samples with different doses of CM. 36 In contrast, some research reported greater efficacy of MSC-CM for improving sperm parameters. It has been shown that administration of AD-MSC-CM to busulfan-treated mice improved sperm count and motility. 27 Additionally, Sertoli cell–CM enhanced the number of testicular cells, increased sperm motility, and reduced sperm abnormal morphology in busulfan-induced azoospermia mice model. 28 Prihatno et al. applied the secretome of the human fetal umbilical cord MSCs in a rat model of testicular damage caused by cisplatin. The result showed that the secretome increased sperm count and motility. 37 Furthermore, rat bone marrow MSC-derived microvesicles (MVs) were used for rat sperm cryopreservation. The results demonstrate that sperm samples treated with a higher concentration of MVs had a higher percentage of viable cells and progressive motile sperms after thawing. 38 Additionally, exosomes isolated from dogs AD-MSC-CM were employed for dog sperm cryopreservation. Post-thawing, sperm treated with exosomes demonstrated improved membrane integrity, acrosome integrity, motility, and live sperm percentage. 39 These divergent outcomes compared to our findings may arise from variations in MSC sources, dosages, or method of treatment administration. Besides, elevated sperm DFI level is associated with male infertility, reduced fertilization and pregnancy rates, increased miscarriage rates, poor embryo quality, and worse ART outcomes. 40 The three main reasons for sperm DNA fragmentation are incomplete protamination, apoptosis, and the action of reactive oxygen species. 41 In consonance with the current study, Bader and colleagues found that incubating human spermatozoa with AD-MSC-CM decreased sperm DNA fragmentation rates, as assessed by the Halo sperm test. 35 Also, the usage of Sertoli cell-CM in azoospermic mice resulted in a reduction of TUNEL-positive testicular cells. 28 Furthermore, Chromomycin A3 staining revealed that adding exosomes to dog sperm cryopreservation medium significantly reduced the percentage of protamine-deficient sperm after thawing. 39 Moreover, the investigation of adipose mesenchymal stromal cell-derived exosomes on testicular damage following torsion revealed these exosomes via activating the PI3K/AKT and MAPK/ERK1/2 signaling pathways inhibit spermatogenic cells apoptosis. 42
The genes AR, CREB1, and PLCζ play crucial roles in spermatogenesis and fertilization.43–45 Androgens and AR are essential for male reproduction. 46 Mutations and polymorphisms in the AR gene and protein are significantly associated with decreased spermatogenesis and idiopathic male infertility. 45 Also, CREB1 is involved in the development of testis, spermatocytogenesis, and spermiogenesis. 44 Studies have suggested that inhibiting the CREB/cAMP Response Element Modulator (CREM) pathway may lead to a reduction in spermatogenesis. 47 Dysregulation of PLCζ is related to various forms of male infertility. 48 Alteration of the level and localization patterns of PLCζ is related to conditions such as globozoospermia, 43 abnormal sperm morphology, higher sperm DFI, and abnormal embryogenesis. 48 Our study confirms that the expression of these three genes is altered based on sperm characteristics. Other research has also investigated the alternation of gene expression following CM or EVs application. Abdollahifar et al. showed that CM enhanced mRNA expression levels of the genes engaged in the transport of cholesterol to the mitochondria and steroidogenesis (StAR and PBR genes) in the testicular tissue. 27 During rat sperm cryopreservation, bone marrow MSC-derived MVs enhanced sperm adhesive markers, including CD29, CD44, ICAM-I, and VCAM-I. These markers play crucial roles in sperm fusogenic and signaling processes. 38 Additionally, canine sperms treated with exosomes derived from AD-MSC-CM during cryopreservation exhibited a higher expression level of genes associated with the plasma membrane repair (FN1, ANX1, and DYSF) and chromatin material (HMGB1 and H3). 39 However, in the present study, neither CM20 nor EVs injection did not influence gene expression, CM10 enhanced the CREB1 expression level. It seems that CM administration is dose-dependent and a higher concentration of CM ingredients may interfere with cell signaling and physiological processes. Also, it’s worth noting that CM contains more biomolecules than EVs isolated from it. 49
Legiawati et al. compared concentrated and non-concentrated AD-MSC-CM with minoxidil for hair regeneration therapy in androgenetic alopecia of men. They found that the non-concentrated AD-MSC-CM exhibited the most impact on hair growth and total follicular units; however, the difference wasn’t statistically significant. 50 Furthermore, in vitro assessment showed that the proliferation of periodontal ligament stem cells was inhibited by exposure to high concentrations of CM. 51 On the other hand, another study has shown an increase in CM concentration leading to increased tissue regeneration. 52 Soukup and colleagues conducted a study comparing CM, EVs, and the soluble protein fraction (PF) for the treatment of inflamed tenocytes. The CM, PF, and EVs showed the most protein and particle concentrations, respectively. Notably, CM led to the most significant difference in gene expression in comparison to the inflamed control and demonstrated better therapeutic efficacy than EVs. 53 Another study compared the content of pro-inflammatory and anti-inflammatory cytokines in MSC-CM and MSCs-derived exosomes isolated from peripheral blood mononuclear cells. Interestingly, MSC-CM exerted a greater immunoregulatory impact on monocytes in contrast with exosomes. 54 Additionally, Giannasi et al. evaluated the therapeutic potential of AD-MSC-CM and EVs in an in vitro model of osteoarthritis using human articular chondrocytes. Their findings revealed that AD-MSC-CM exhibited greater therapeutic efficacy than EVs. Specifically, only AD-MSC-CM significantly decreased matrix metalloproteinase activity. 55 Moreover, the anti-inflammation effects of the CM and its components, including EVs and soluble culture fraction (SF), were studied in an in vitro model of cell inflammation. The CM exhibited strong anti-inflammation and metalloproteinase inhibition effects, while EVs and SF showed these effects to a lesser extent. 56 Similarly, in the present study, CM10 was more effective than EVs in ameliorating male reproductive toxicity.
Conclusion
The present study suggests that CM derived from hWJMSCs could enhance sperm quality. In an animal model of male reproductive toxicity, CM10 showed superior effects on improving sperm characteristics compared to CM20 or EVs. Furthermore, CM10 enhanced the expression level of CREB1 in the testicular tissue. Considering the faster and more accessible production process of CM compared to EVs, they could be introduced as a promising method of cell-free therapy for male infertility treatment. However, more in vitro and experimental studies are required to ensure the safety and efficacy of CM for clinical applications.
Footnotes
Author contributions
All authors contributed to the conceptualization and design of the study. Experiments were conducted by SB, MD, and AS. Data analysis, interspersion, and preparation of figures and tables done by S.B., S.A., Z.K., and T.T. S.A., Z.K. supervised the study. T.T. and S.M. provided advice on the study methodology. S.B., S.A., and Z.K. collaborated on writing and editing the first draft of the manuscript. All authors provided feedback on earlier drafts of the manuscript and all authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Shiraz University of Medical Sciences, Shiraz, Iran (Grant No. 23122).
Ethical statement
Data Availability
The datasets generated and/or analyzed during the current study are available from the corresponding authors upon reasonable request.
