Abstract
Background
Stereotactic body radiation therapy (SBRT) is a targeted form of radiotherapy used to treat early-stage cancers. Despite its effectiveness, the impact of SBRT on myeloid-derived suppressor cells (MDSCs) is not well understood. In this study, we examined how SBRT affects the differentiation and survival of MDSCs, as well as delved into the molecular mechanisms involved.
Methods and results
SBRT was utilized on bone marrow (BM)-derived MDSCs to investigate its impact on the differentiation and survival of MDSCs using flow cytometry. An animal model of lung cancer was created to assess the anti-cancer properties of SBRT and the role of miR-21 expression in MDSCs. The interplay of miR-21 and Sorbin and SH3 domain-containing protein 1 (SORBS1) in MDSC differentiation was explored through dual luciferase activity assay, RT-qPCR, and Western blot analysis. The findings suggest that SBRT led to an increase in miR-21 levels, inhibited MDSC differentiation, and triggered cell apoptosis in BM cells. Inhibition of miR-21 reversed the effects of SBRT on MDSC differentiation and apoptosis. Additionally, it was revealed that SORBS1 was a downstream target of miR-21 in BM cells, and the miR-21/SORBS1 axis played a role in regulating MDSC differentiation and apoptosis induced by SBRT. Modulating miR-21 levels in vivo impinged on the response to SBRT treatment and the quantity of MDSCs in a mouse model of lung cancer.
Conclusion
Our data indicate that the upregulation of miR-21 induced by SBRT may contribute to the inhibition of MDSC expansion in a lung cancer model.
Introduction
Lung cancer remains a major global health burden, with non-small cell lung cancer (NSCLC) being the most prevalent form and a leading cause of cancer-related mortality worldwide.1,2 Despite advances in various treatment modalities, including targeted therapies and immunotherapy, the overall survival outcomes for NSCLC patients have not significantly improved over the past years.3,4 Over half of NSCLC patients succumb within a year of diagnosis, with a 5-year survival rate as low as 20%. 5 One of the key challenges hampering successful treatment is the development of therapeutic resistance, which can arise from tumor heterogeneity, rapid accumulation of genomic alterations, and a supportive tumor microenvironment.6–8 Radiotherapy, a mainstay treatment for NSCLC, is often limited by the development radio-resistance.9,10
Radio-resistance and toxicity are main obstacles associated with compromised therapeutic outcome and poor prognosis after radiotherapy. 11 Novel techniques have been developed for targeted delivery of high dose irradiation to the tumor sites, reducing damages to surrounding tissues. 12 Stereotactic body radiation therapy (SBRT) is a targeted approach that allows for high-dose radiation to be applied to tumor tissues, which has demonstrated promise in treating early-stage NSCLC.13,14 However, various factors have been reported to influence the efficacy of radiotherapy in NSCLC, including oncogenic mutation, cancer stem cells (CSCs), hypoxia, DNA damage repair, and the tumor microenvironment (TME).15,16
Myeloid-derived suppressor cells (MDSCs) are immune cells derived from the bone marrow that are recruited abundantly to the TME to suppress anti-tumor immunity and promote tumor progression. 17 Emerging evidence suggests that MDSCs are implicated in radio-resistance development and cancer recurrence. 18 For example, radiotherapy may enhance the recruitment of MDSCs to the TME, possibly triggered by inflammation and hypoxia following irradiation.19,20 Nevertheless, most of these findings are based on traditional radiotherapy approaches. Given that SBRT could directly target and destroy tumor tissues, the impact of SBRT on MDSC differentiation and recruitment to the TME warrants further clarification.
MicroRNAs (miRNAs) are a group of non-coding RNAs with 19–25 nucleotides, which regulate gene expression at the post-transcriptional level and play essential roles in tumor progression. 21 These miRNAs are involved in the differentiation, maturation, and proliferation of MDSCs, as well as impacting the radio-sensitivity of cancer cells.22,23 For instance, miR-21 could regulate proliferation and differentiation of MDSCs by targeting multiple signal pathway.24,25 In NSCLC, there is a notable upregulation of miR-21 in radio-resistant cancer cells compared to the radiosensitive ones, and miR-21 overexpression confers resistance to irradiation in NSCLC cells. 26 Despite the implication of miR-21 in dictating radio-sensitivity, the effect of SBRT on miR-21 expression and its role in modulating the differentiation of MDSCs remains to be explored.
The objective of current study was to explore the effect of SBRT on MDSC differentiation and survival, and to investigate the underlying molecular mechanism. We applied SBRT in bone marrow (BM)-derived MDSCs to study the impact of SBRT on the differentiation and survival of MDSCs by flow cytometry. We also extended our investigation in the animal model of lung cancer to evaluate the anti-cancer effect of SBRT and its impact on MDSCs. The interplay of miR-21 and Sorbin and SH3 domain-containing protein 1 (SORBS1) in MDSC differentiation of MDSCs was examined.
Methods
Bone marrow (BM) cell isolation and culture
Six-to eight-week-old BALB/c male mice were acquired from Beijing Weitong Lihua Experimental Animal Technology Co., Ltd. (Beijing, China). Bone marrow (BM) cells were isolated from the mouse femurs by flushing with cold PBS and treated with red blood cell lysis buffer (Beyotime Biotech, Beijing, China). The remaining BM cells were filtered through a 40-µm cell strainer (Zeye Biotech, Shanghai, China). Subsequently, 2 × 10^6 cells were seeded into a 6-well plate with RPMI-1640 medium (Lonza, Basel, Switzerland) supplemented with 10% fetal bovine serum (Procell, Wuhan, China), 2 µL 2-mercaptoethanol, 200 μg streptomycin (Sigma, Shanghai, China), and 100 μM sodium pyruvate (Procell, Wuhan, China). To induce myeloid-derived suppressor cell (MDSC) differentiation, recombinant interleukin (IL)-6 and granulocyte macrophage-colony stimulating factor (GM-CSF) (Procell, Wuhan, China) were added to the medium at a concentration of 40 ng/mL. The BM cells were cultured in an incubator at 37°C, 5% CO2, and 95% humidity for 4 days prior to subsequent analyses. Differentiation experiments for all conditions were conducted three independent times. For each independent trial, the BMs were pooled from 5 to 8 mice and divided into different conditions.
Radiation delivery in cell culture
BM cells were cultured and irradiated in 6-well Nunc™ culture plates (ThermoFisher Scientific, Waltham, MA, USA). The cell culture medium was changed to PBS before irradiation to ensure homogenous radiation delivery. A 6 MV beam was applied to the cells in the plate using the TrueBeam linear accelerator (Varian Medical Systems, Palo Alto, CA, USA). The calculation of the dose was performed in the Eclipse Treatment Planning System, v. 15.6 (Varian Medical Systems, Palo Alto, CA, USA). The radiation was delivered via the photon beam with a flattening filter to apply a total dose of 6 Gy to the cells in 3 consecutive fractions of 2 Gy as a dose scaling approach described before. 27 After irradiation, PBS was replaced with fresh RPMI medium for further culture.
Cell transfection
The synthetic controls (miR-NC and inhibitor NC), miRNA mimic and inhibitor were procured from MedChemExpress (Shanghai, China). The empty vector (pcDNA3.1) and SORBS1 expression vector were synthesized by Yunzhou Biotech (Guangzhou, China). Lipofectamine 2000 reagent (Invitrogen, Shanghai, China) was utilized in cell transfection experiment in line with specific instructions. 48-h post-transfection the cells were harvested for further functional assays.
Flow cytometry analysis of MDSC markers
Functional differentiated MDSCs were detected by flow cytometry by surface staining of CD11b and Gr-1.28,29 BM-MDSCs cell suspensions were first incubated with an anti-FcR monoclonal antibody (catalog #14-0161-82, 1:100, eBioscience, Thermo Fisher Scientific, San Diego, CA, USA) to block nonspecific binding. Afterward the cells were labeled with anti-CD11b (catalog #101208, clone M1/70, 1:100, phycoerythrin (PE) fluorophore, BioLegend Inc., San Diego, CA, USA) and anti-Gr-1 (catalog #108412, clone RB6-8C5, 1:100, Allophycocyanin (APC) fluorophore, BioLegend Inc.) antibodies. The cells were washed twice with PBS and the stained cells were analyzed on the BD FACScanto II flow cytometer (BD Biosciences, Franklin Lakes, NJ, USA). Data analysis was conducted using FlowJo X 10.0.7 software (BD Biosciences, Franklin Lakes, NJ, USA).
CCK-8 proliferation assay
CCK-8 assay kit (MedChemExpress, Shanghai, China) was used for measuring cell growth in line with specific protocols. After transfection, cells were inoculated into the 96-well plates for indicted culture periods. CCK-8 reagent (15 μL) was added into every well and the plate was incubated for another 3-h period under 37°C. The measurement of absorbance (OD) value was conducted using a microplate reader at 450 nm.
Apoptosis analysis
Cells under different experimental conditions were washed twice with PBS, and re-suspended in the cell staining buffer. The analysis of apoptotic cells was performed using the FITC Annexin V Apoptosis Detection Kit (BD Biosciences) according to the manufacturer’s protocols. In brief, 1 μL Annexin V-FITC and 1 μL Propidium-Iodide reagent were mixed with 1 million cells for 15-min incubation in the darkness at ambient temperature. The stained cells were centrifuged and washed twice with the washing buffer, and the samples were analyzed on the BD FACScanto II flow cytometer (BD Bioscience).
Dual luciferase reporter assay
The luciferase reporters containing wilde type (WT) or mutated binding sites (MUT) were prepared by the GenePhama (Shanghai, China). The reporters were delivered into the cells by Lipofectamine 2000 reagent (Invitrogen, Shanghai, China) together with miR-NC or miRNA mimic. After 48 h, the Luciferase Reporter Gene Detection Kit (LUC1-1KT, Sigma, Shanghai, China) was used for the determination of luciferase activities in line with the supplier’s protocols.
RT-qPCR
Total RNA samples were isolated from the cells using TRIzol Reagent (Invitrogen, Shanghai, China) in line with specific protocols. PrimeScript II 1st Strand cDNA Synthesis Kit (TaKaRa, Tokyo, Japan) was employed for synthesizing cDNA from 1 μg of total RNA. For the quantification of miRNA, cDNA synthesis was performed using Mir-X miRNA first-strand synthesis kit (Takara, Tokyo, Japan) following the manufacturer’s protocol. Afterward, by adopting SYBR Premix Ex Taq II kit (Takara, Tokyo, USA) together with corresponding primers, CFX96 Touch Real-time PCR detection system (CFX96, Bio-Rad, Hercules, CA, USA) was adopted for qPCR analysis. 2−△△CT approach was utilized to determine the relative expression level of target genes, with U6 as the internal reference and GAPDH as the endogenous control.
Western blot (WB)
RIPA buffer containing phosphatase and proteinase inhibitors (KeyGen, Beijing, China) was employed to collect protein sample Thereafter, the Enhanced BCA Protein Assay Kit (Beyotime, Beijing, China) was used for protein concentration determination. Aliquots of proteins were separated through 10% SDS-PAGE and transferred on PVDF membranes (Millipore, Darmstadt, Germany). Thereafter, 5% bovine serum albumin (Sigma, Shanghai, China) was utilized to block the membranes for 1 hour, followed by the incubation with the target antibodies (Abcam, Cambridge, UK): anti-SORBS1 (1:1000; Abcam, ab224129) and anti-β-actin (1:1000; Abcam, ab8226) overnight under 4°C. Afterward, HRP-labeled secondary antibody (1:10,000; 61-6520, Invitrogen, Shanghai, China) was utilized to incubate the membranes for 1 hour at room temperature. After signal development, protein blots were visualized and quantified by ImageJ Software Version 1.53t (NIH, Bethesda, MD, USA).
Animal experiment
Murine lung cancer cell line (LLC) was obtained from Hongshun Biotech (Shanghai, China). To the lung cancer mouse model, eight-week-old BALB/c male mouse was subcutaneously injected with 200 μL LLC cells suspended in PBS (2 ×106 cells per mouse). When the diameter of the tumor reached about 5 mm (after 7 days), SBRT (6 Gy) was applied to each tumor for irradiation treatment for 4 days (6th, 7th, 8th, 9th day), with the TrueBeam linear accelerator system (Varian Medical Systems, Palo Alto, CA, USA). For the administration of miRNA mimic and inhibitor, the synthetic oligos was delivered at 1 mg/kg/day on the 8th, 11th, 14th, and 17th day using EntransterTM-in vivo-RNA transfection reagent (Engreen Biosystem Co, Ltd., Beijing, China) according to the instructions of the manufacturer. All the mice were executed by cervical dislocation on the 18th day, and the tumor samples were collected for further analyses. The animal protocol was approved by the animal research ethics committee of Kunming Medical University.
Hematoxylin and eosin (H&E) staining
H&E staining was conducted in the tumor tissue sections using H&E Stain Kit (ab245880, Abcam). After the deparaffinization and hydration, the tumor sections were incubated in adequate Hematoxylin Mayer’s solution to cover tissue section for 5 min. After the rinsing with the distilled water, the Bluing Reagent was applied to incubate the tissue sections for 1-2 min. Then the sections were dehydrated in absolute alcohol, and further stained using the Eosin Y Solution for 2-3 min. The images were collected under an Leica AM6000 microscope (Leica, Wetzlar, Germany).
Statistics
SPSS software 19.0 version (IBM, New York, NY, USA) was used for statistical analysis. Data were summarized as mean ± SD. Unpaired student's t-test was used to compare continuous variable between two samples. The analyses of data from multiple groups were conducted via one-way ANOVA. *, p < .05; **, p < .01; ***, p < .001 and ****p < .0001 represent different degrees of statistical significance.
Results
SBRT inhibits MDSC differentiation and induces cell apoptosis in BM cells
To explore the impact of SBRT on MDSC growth and differentiation, BM cells were induced to MDSCs by GM-CSF and IL-6. We applied SBRT at 2 Gy (low), 4 Gy (medium) and 6 Gy (high) to the induced MDSCs. It was found that high dose of SBRT totally arrested the growth of induced MDSCs, while the medium dose showed mild inhibition (Figure 1). We therefore adopted 4 Gy dose (medium) for the following experiments. We performed flow cytometry analysis of Gr-1+CD11b + MDSC cells after GM-CSF and IL-6 induction. Compared with the sample without SBRT, the proportion of Gr-1+CD11b + MDSC cells significantly decreased upon SBRT treatment (Figure 2A). Accordingly, there was a significant increase of apoptotic events in the differentiated MDSCs after SBRT treatment (Figure 2B). We also measured the change of miR-21 and found that SBRT treatment caused a significant upregulation of miR-21 in the induced MDSCs (Figure 2C). These data promoted us to further investigate the role of miR-21 in MDSC differentiation and survival upon SBRT treatment. SBRT suppresses cell growth in MDSC differentiation. BM cells were induced to MDSCs by GM-CSF and IL-6, and SBRT was applied at 2 Gy (low), 4 Gy (medium) and 6 Gy (high) to the induced MDSCs. CCK-8 proliferation assay in the induced MDSCs. N = 3 independent experiments. *p < .05; **p < .01; ***p < .001; ****p < .0001. SBRT inhibits MDSC differentiation and induces cell apoptosis in BM cells. (A) Flow cytometry analysis of Gr-1+CD11b + MDSC cells after GM-CSF/IL-6 induction and SBRT treatment. (B) Flow cytometry analysis of apoptotic events in the differentiated MDSCs with or without SBRT treatment. (C) RT-qPCR of miR-21 expression in the differentiated MDSCs with or without SBRT treatment. N = 3 independent experiments. *p < .05; **p < .01; ***p < .001; ****p < .0001.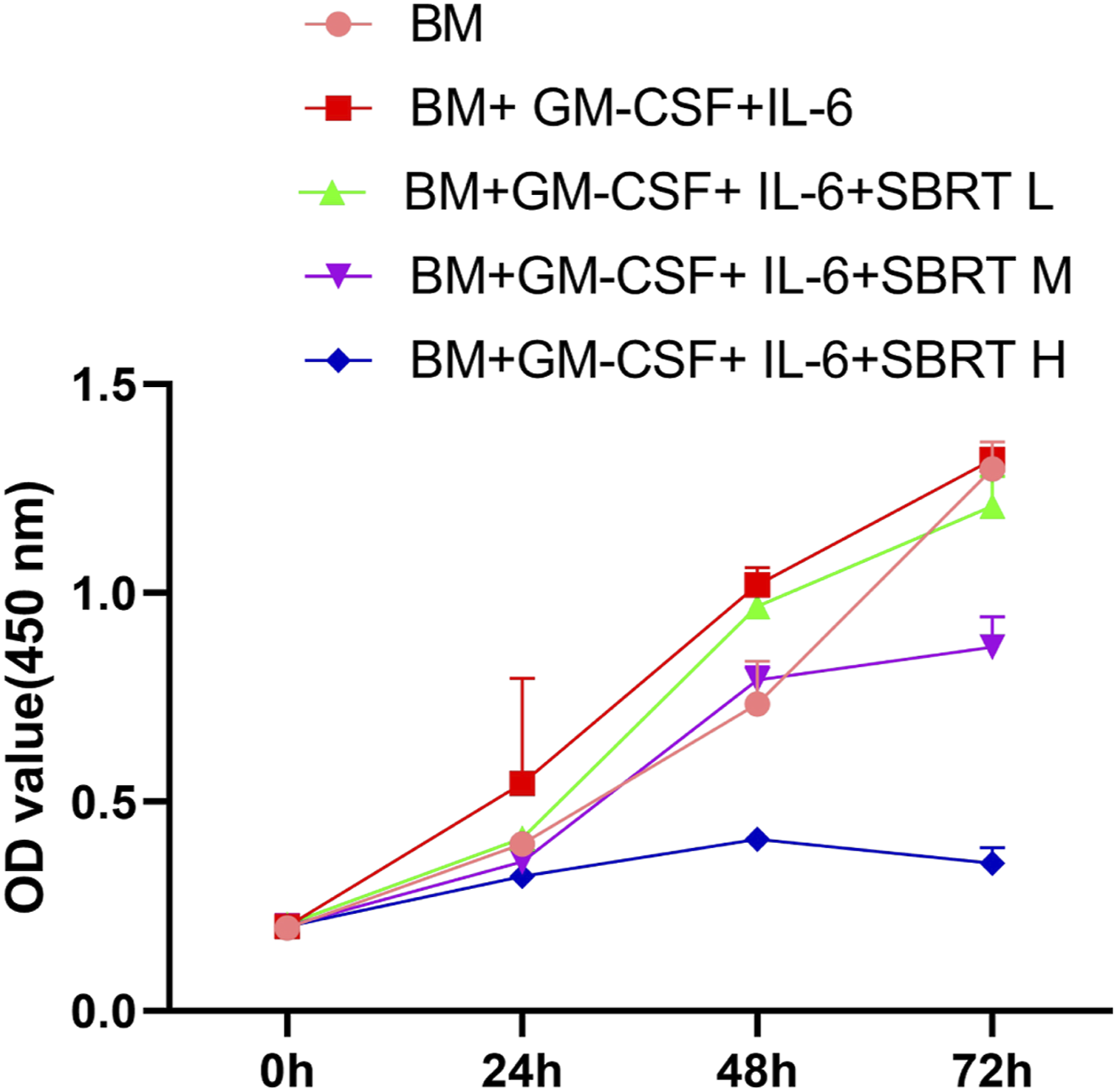

miR-21 inhibition suppresses the effect of SBRT on MDSC differentiation apoptosis induction
We next transfected miRNA inhibitor negative control oligo (NC) or the synthetic miR-21 inhibitor into the BM cells before MDSC differentiation and SBRT treatment. RT-qPCR analysis showed that miR-21 inhibitor suppressed miR-21 upregulation induced by SBRT (Figure 3A). We found that miR-21 inhibition rescued SBRT-induced cell growth arrest of differentiated MDSCs (Figure 3B). In the meanwhile, SBRT-induced apoptosis was suppressed by miR-21 inhibitor (Figure 3C). There was a also significantly higher percentage of Gr-1+CD11b + MDSC cells in SBRT-treated cells population upon miR-21 inhibition (Figure 3D). Therefore, our data suggest that miR-21 is a negative regulator of MDSC differentiation and survival. miR-21 inhibition suppresses the effect of SBRT on MDSC differentiation apoptosis induction. miRNA inhibitor negative control oligo (NC) and the synthetic miR-21 inhibitor were applied into the BM cells before MDSC differentiation and SBRT treatment. (A) RT-qPCR analysis of miR-21 expression. (B) CCK-8 proliferation assay in the differentiated MDSCs. (C) Flow cytometry analysis of apoptotic events in the differentiated MDSCs. (D) Flow cytometry analysis of Gr-1+CD11b + MDSC cells. N = 3 independent experiments. *p < .05; **p < .01; ***p < .001; ****p < .0001.
miR-21 negatively regulates SORBS1 expression in BM cells
To identify the mRNA target of miR-21, we searched TargetScan database to predict the mRNA containing potential binding sites for miR-21. Sorbin and SH3 domain-containing protein 1 (SORBS1) mRNA 3′ UTR contains complementary sequences to miR-21 (Figure 4A). To further investigate the interaction between miR-21 and SORBS1 mRNA, we constructed wild-type (WT) and mutant (MUT) SORBS1-3′ UTR luciferase reporter. The luciferase activity assay revealed that that miR-21 mimic transfection significantly suppressed luciferase activity of the WT reporter in BM cells, while miR-21 mimic showed no inhibition in MUT reporter (Figure 4A). qRT-PCR and Western blot analysis demonstrated that the overexpression of miR-21 by miRNA mimic reduced SORBS1expression in BM cells, while the inhibition of miR-21 caused the upregulation of SORBS1 (Figure 4B and C). These results indicate that miR-21 negatively regulates SORBS1 expression in BM cells. miR-21 negatively regulates SORBS1 expression in BM cells. (A) TargetScan database to predict the SORBS1 mRNA 3′ UTR containing potential binding sites for miR-21. Luciferase reporter assay in wild-type (WT) and mutant (MUT) SORBS1-3′ UTR luciferase reporter with miRNA NC and miR-21 mimic. (B) qRT-PCR of miR-21, and (C) western blot analysis of SORBS1 protein level in BM cells after the transfection of miR-21 mimic or inhibitor. N = 3 independent experiments. *p < .05; **p < .01; ***p < .001; ****p < .0001.
miR-21/SORBS1 axis is implicated in the regulation of MDSC differentiation and apoptosis induction by SBRT
To further dissect the functional interaction between miR-21 and SORBS1 in MDSC differentiation and survival, cells were co-transfected with miR-21 inhibitor with si-NC (negative control siRNA) or si-SORBS1 (siRNA targeting SORBS1). It was found that SBRT downregulated SORBS1 level, and miR-21 inhibition restored its expression. The co-transfection of si-SORBS1 further reduced SORBS1 protein level (Figure 5A). Cell proliferation assay showed that the rescue effect of miR-21 inhibition on SBRT-induced cell growth arrest was abrogated upon SORBS1 silencing (Figure 5B). Further, SORBS1 silencing promoted SBRT-induced cell death in the presence of miR-21 inhibition (Figure 5C). After SORBS1 silencing, the differentiation of Gr-1+CD11b + MDSC cells was also largely suppressed (Figure 5D). Therefore, miR-21 mediates MDSC differentiation and survival upon SBRT treatment by regulating SORBS1. miR-21/SORBS1 axis is implicated in the regulation of MDSC differentiation and apoptosis induction by SBRT. To further dissect the functional interaction between miR-21 and SORBS1 in MDSC differentiation and survival, cells were co-transfected with miR-21 inhibitor with si-NC (negative control siRNA) or si-SORBS1 (siRNA targeting SORBS1). (A) Western blot of SORBS1 protein levels; (B) CCK-8 proliferation assay in the differentiated MDSCs. (C) Flow cytometry analysis of apoptotic events in the differentiated MDSCs. (D) Flow cytometry analysis of Gr-1+CD11b + MDSC cells. N = 3 independent experiments. *p < .05; **p < .01; ***p < .001; ****p < .0001.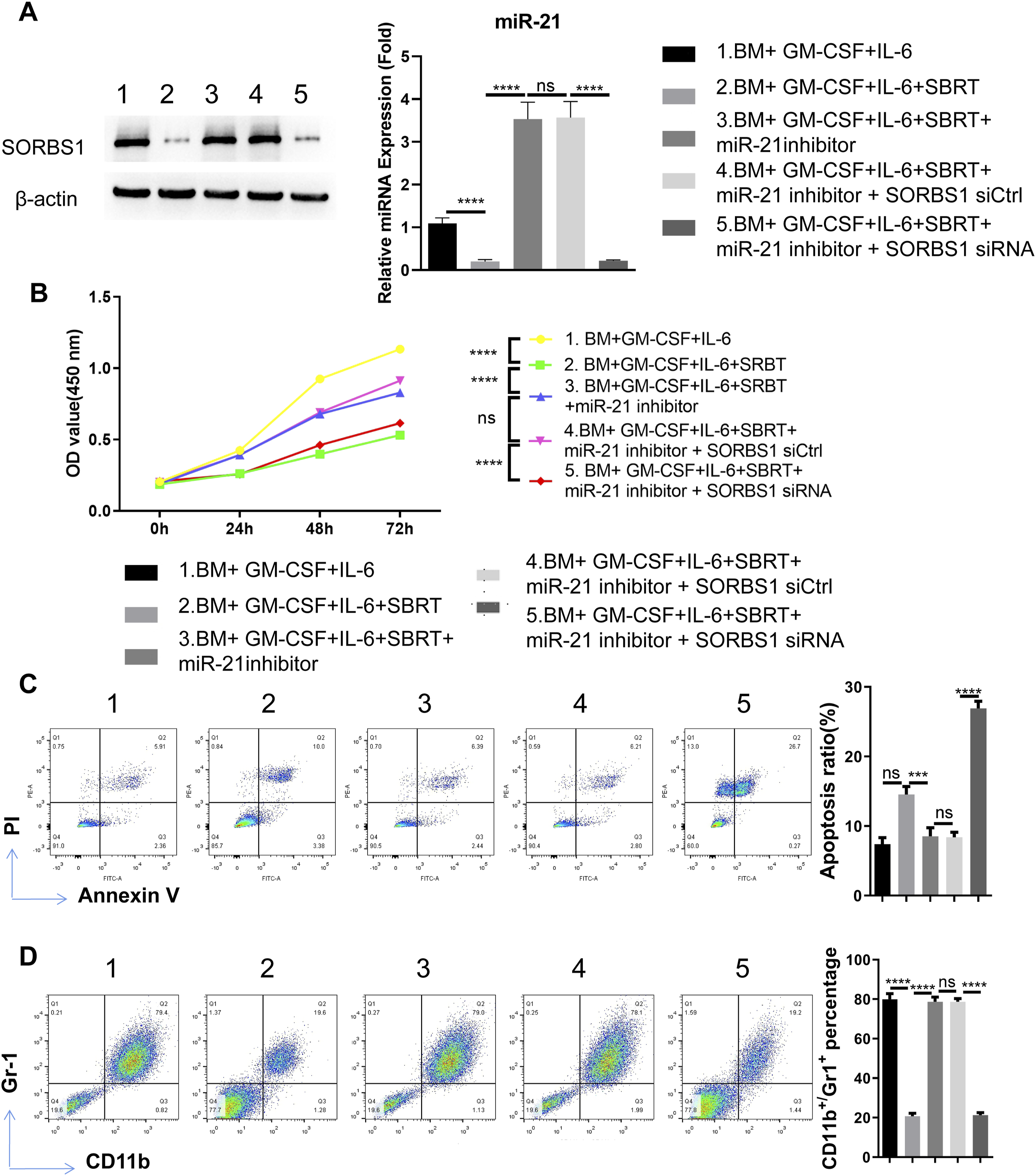
Manipulating miR-21 alters the response to SBRT therapy and the abundance of MDSCs in the mouse model of lung cancer
We then wondered whether the manipulation of miR-21 levels in vivo could impinge on the response of cancer cells to SBRT therapy and MDSC abundance. To this end, murine lung cancer cell line (LLC) was subcutaneously injected into BALB/c mice and SBRT was applied at 6 Gy when the diameter of the tumor reached about 5 mm. miRNA mimic and inhibitor was delivered every 3 days for 4 times. Compared to the tumor model group without SBRT treatment, SBRT application arrested tumor growth. miRNA mimic further suppressed tumor growth upon SBRT treatment and while miR-21 inhibitor rescued tumor growth after SBRT treatment (Figure 6A). H&E staining in the tumor tissues revealed that SBRT or SBRT + miR21 mimic induced cell dramatic damages in tumor tissues, while miR21 inhibitor attenuated the effect of SBRT (Figure 6B). Manipulating miR-21 alters the response to SBRT therapy and the abundance of MDSCs in the mouse model of lung cancer. Murine lung cancer cell line (LLC) was subcutaneously injected into BALB/c mice and SBRT was applied at 6 Gy when the diameter of the tumor reached about 5 mm. miRNA mimic and inhibitor was delivered every 3 days for 4 times. (A) The images of tumor tissues in each experimental group. (B) H&E staining of tumor tissues in each experimental group. (C) RT-qPCR of miR-21 expression level in the bone marrow samples. (D) Western blot of SORBS1 protein levels in bone marrow samples. E. Flow cytometry analysis of Gr-1+CD11b + MDSC cells in the bone marrow samples. N = 5 animals in each group. *p < .05; **p < .01; ***p < .001; ****p < .0001.
We also collected the bone marrow samples for molecular and cellular analysis. As expected, SBRT upregulated miR-21 expression; the delivery of miR-21 mimic further enhanced miR-21 level and miR-21 inhibitor suppressed miR-21 expression (Figure 6B). SBRT treatment reduced SORBS1 protein level in bone marrow, and miR-21 mimic further decreased its expression. The delivery of miR-21 inhibitor restored SORBS1 expression (Figure 6C). Besides, compared to the mice without tumor engraftment (control), the bone marrow samples of the tumor model mice showed an significant expansion of Gr-1+CD11b + MDSC cells. The application of SBRT and miR-21 mimic both suppressed the expansion of Gr-1+CD11b + MDSC cells, while the delivery of mR-21 inhibitor abrogated the effect of SBRT treatment (Figure 6D).
Discussion
In this study, we showed that SBRT suppressed MDSC differentiation and induced cell apoptosis in BM cells. This effect was associated with the upregulation of miR-21. miR-21 inhibition suppressed the effect of SBRT on MDSC differentiation and apoptosis induction. We further demonstrated that miR-21 negatively regulated SORBS1 expression in BM cells, and miR-21/SORBS1 axis was implicated in the regulation of MDSC differentiation and apoptosis induction by SBRT. The manipulation of miR-21 altered the response to SBRT therapy and the abundance of MDSCs in the mouse model of lung cancer. These findings suggest that SBRT could potentially reduce the immunosuppressive MDSCs in the TME, thereby enhancing anti-tumor immunity.
miR-21 has been implicated in the radio-sensitivity of non-small cell lung cancer cells (NSCLC). For example, significant upregulation of miR-21 was reported in the radio-resistant NSCLC, and the forced overexpression of miR-21 rendered A549 NSCLC cells resistant to radiation. 26 miR-21 also suppressed the proliferation of colon cancer cells by negatively targeting Cdc25A. 30 However, its role in MDSC differentiation and survival is unclear. We showed that SBRT induced the upregulation of miR-21 in differentiated MDSCs from the BM cells. This is in agreement with the previous report that ionizing radiation significantly increased miR-21 expression level in hematopoietic cells. 31 We further demonstrated that miR-21 overexpression upon SBRT suppressed MDSC differentiation from BM cells. miR-21 inhibition rescued the effect of SBRT on MDSC apoptosis, suggesting miR-21 as a negative regulator of MDSC differentiation and stability. A previous report showed that the infiltration of CD4+ and CD8 + activated T cells and a reduction in PD-L1 + monocytes in solid tumours accounted for tumor regression. Decreased miR-21 expression was associated with the loss of anti-tumor immune suppression. 24 In Lewis lung-cancer-bearing mice, miR-21 was reported to regulate immunosuppression mediated by MDSCs by targeting RUNX1-YAP interaction. 25 Thus, miR-21 may serve as a novel factor implicated in the differentiation and functional stability of MDSCs. Our study highlights the potential of combining miR-21 inhibitors with SBRT to preserve the functional MDSCs and improve treatment efficacy.
SORBS1 was identified as a downstream target of miR-21, and SBRT-induced miR-21 overexpression suppressed SORBS1 expression and MDSC differentiation. Additionally, silencing SORBS1 abrogated the effect of miR-21 inhibition in MDSC differentiation and survival. These findings reveal a novel mechanism by which SBRT modulates MDSCs, suggesting potential therapeutic strategies targeting this axis to enhance SBRT efficacy. SORBS1 has been characterized as a protein which functions in the signaling pathway of insulin, which is required for insulin-stimulated glucose transport. 32 Emerging evidence has unveiled its potential implication in immune regulation. For instance, SORBS1 and its family member can downregulate the expression of pro-inflammatory cytokines in myeloid-derived leukocytes. 33 In bladder cancer, SORBS1 expression level was positively correlated with the signatures of tumor infiltrated macrophages. 34 This recent evidence is in line with our findings that SORBS1 is required for the differentiation of immunosuppressive MDSCs.
There are several limitations and multiple open questions to be further addressed. First, clinical samples in NSCLC patients before and SBRT treatment need to be collected to validate the role of miR-21 in MDSC differentiation and survival. Further, the mechanism by which SBRT induces miR-21 upregulation remains unclear. In addition, how SORBS1 orchestrates MDSC differentiation in vitro and in vivo warrants future investigation. Answers to these questions may offer novel perspectives into the development of strategies to improve SBRT outcome.
Conclusion
In summary, miR-21 expression was elevated in MDSCs and animal model after SBRT treatment. Silencing of miR-21 could promote MDSC differentiation upon SBRT treatment. miR-21 negatively regulates SORBS1 to suppress MDSC differentiation. Our findings provided strong evidence that the application of miR-21 mimic might boost the therapeutic efficacy of SBRT by dampening MDSC expansion. This underscores the clinical potential of investigating miR-21-based therapies in combination with SBRT to overcome immunosuppression and improve outcomes in lung cancer patients.
Footnotes
Acknowledgements
The following funds are gratefully acknowledged for supporting this study: the Basic Research Program of Yunnan Province-Joint Project of Kunming Medical University (NO. 202101AC070460).
Author contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Congbo Yang, Lingli Zhou, Jinli Peng and Tianwen Zhang. The first draft of the manuscript was written by ChunFang Zhao and Qi Tang. Ya Li and Shaoqiang Zhou were mainly responsible for the conception of the experimental scheme and the review of the final manuscript and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Basic Research Program of Yunnan Province-Joint Project of Kunming Medical University. (Grant numbers 202101AC070460).
Ethical statement
Data availability statement
The data generated in this study are available upon request to the corresponding author.
