Abstract
Organophosphorus (OP) poisoning is a significant cause of morbidity and mortality worldwide. Recent research has explored new approaches to improving treatment options, which present several challenges. This study aimed to evaluate the role of fresh frozen plasma (FFP) as an adjunctive therapy for acute OP intoxication. A prospective single-blinded randomized clinical trial was conducted on patients of both sexes admitted to the Intensive Care Unit (ICU) of the Poison Control Center at Ain Shams University Hospital (PCC-ASUH) with acute OP toxicity during the period from the beginning of August 2022 to the end of July 2023. According to the Peradeniya score, Group I consisted of 48 patients (52%) with moderate OP poisoning, and Group II consisted of 44 patients (48%) with severe OP poisoning. Patients in the moderate group were assigned to receive either standard treatment (Group Ia, n = 24) or standard treatment plus FFP (Group Ib, n = 24). In addition, patients in the severe group were assigned to receive either standard treatment (Group IIa, n = 22) or standard treatment plus FFP (Group IIb, n = 22). A total of 46 patients received FFP transfusion. The authors demonstrated that the early use of a total of nine packs of FFP (250 mL each) over three consecutive days significantly reduced the total doses of atropine and oximes, the total hospitalization period, and the requirement for mechanical ventilation in patients with OP poisoning, both in the moderate and severe groups.
Introduction
Organophosphorus (OP) poisoning is a significant cause of morbidity and mortality worldwide. 1 The risk of exposure to OP is higher in developing countries for several reasons, including inadequate regulatory systems, less public education, less availability of poison information and control centers, poorly maintained or unavailable personal protective equipment, and larger agricultural-based populations. 2 Additionally, OP can be easily absorbed through inhalation, ingestion, and dermal exposure, leading to a large number of reported cases of OP poisoning and high mortality rates. 3
According to the World Health Organization (WHO), OP compounds are the main reason for three million cases of pesticide poisoning every year, causing over 250,000 deaths. 4 Analysis of intoxicated cases presented at Poison Control Center, Ain Shams University Hospitals (PCC-ASUH) in 2019 revealed that insecticide intoxication constituted 30.4% of the total number of non-drug poisoning cases, with OP poisoning accounting for 74% (1958 cases) of these instances. OP poisoning ranked as the second most common exposure substance received at PCC-ASUH, with the highest mortality rate among poisoned patients at 23.8% (34 deaths out of 143 total deaths in that year). 5
There are two types of cholinesterases (ChEs): erythrocyte cholinesterase (AChE) and butyrylcholinesterase (BuChE). AChE is primarily concentrated on RBC membranes, while BuChE is mainly in blood plasma. As inhibitors of ChE, OPs cause an accumulation of acetylcholine, which can lead to symptoms affecting the muscarinic, nicotinic, and central nervous systems (CNSs). 6 In cases of severe poisoning, it can take up to 4 weeks for ChE to recover. Low ChE levels may lead to intermediate syndrome and paralysis of the respiratory muscles. 7
The initial treatment for OP toxicity is to ensure adequate airway and ventilation and establish cardiorespiratory function by reversing excessive muscarinic effects. 8 Additionally, current treatment guidelines recommend the administration of atropine as the definitive treatment for acute OP poisoning, oximes as a follow-up, benzodiazepines for seizure control, and pyridostigmine for prophylaxis. It is worth noting that the treatment for OP poisoning has remained the same for the last five decades; recent research has been exploring new approaches to improve current treatment options, which present several challenges. 9 In severe poisoning, it may take hundreds of milligrams of atropine over several days before the patient improves. 10 Furthermore, in the case of lipophilic OPs, atropine should be continued for several days. Premature termination of atropine treatment may cause a recurrence of cholinergic syndrome. Additionally, atropine does not counteract the nicotinic effects of OP, and it has several adverse effects, such as tachycardia, arrhythmias, fever, and psychosis. The poor ability of oximes to penetrate the CNS and the rapid re-inhibition of reactivated acetylcholinesterase (AChE) by circulating OP are among the reasons why oximes remain controversial, particularly in the case of massive poisoning. 11
Several alternative therapies have been tested on animals, but none have proven beneficial in large clinical trials or widely used in clinical practice. Treatments proposed include using magnesium or clonidine to reduce presynaptic acetylcholine release and fresh-frozen plasma (FFP) as a source of BuChE to scavenge OP pesticides. 11 FFP is a bio-scavenger suggested as a therapy for eliminating free OP; however, its ability to scavenge OP by increasing BuChE levels is controversial.12–14 This study aimed to evaluate the role of FFP as an adjuvant treatment in patients admitted to the Intensive Care Unit (ICU) with acute OP intoxication.
Subjects and methods
Study setting and design
A prospective single-blinded randomized clinical trial was conducted on patients of both sexes admitted to the ICU of the PCC-ASUH with the diagnosis of acute OP poisoning during the period from the beginning of August 2022 to the end of July 2023.
Ethical consideration
The study complied with the CONSORT guidelines and was approved by the Institutional Ethical Committee of the Faculty of Medicine, Ain Sham University (No: FMASU MD17/2022). This trial was registered at the clinical trial’s government registry (Trial ID: NT06256796). Written informed consent was obtained from the patients or their guardians (if the patient could not participate in the consent process) after an explanation of the purpose and technique of the study. All the data were kept confidential. The present study was carried out according to the World Medical Association Declaration of Helsinki. In Figure 1; the CONSORT Flow chart illustrates enrollment, intervention allocation, and patient follow-up. Flow chart of the patients.
Eligibility criteria
All patients of both sexes diagnosed with acute OP poisoning were admitted to the ICU of the PCC-ASUH during the study period from the beginning of August 2022 to the end of July 2023. The diagnosis of acute OP intoxication was established based on the following criteria: history of exposure to OP compounds, improvement of muscarinic symptoms and signs after atropine administration, and low serum BuChE activity as guided by Lee P. (2001). 15
Exclusion criteria
• Patients under 18 years • Patients with a history of co-ingestion • Patients who received atropine and/or oximes before presentation • Patients with a delay time of more than 24 h • Patients with a history of diabetes, cardiac, respiratory, renal, and hepatic disease • Patients with coagulation disorders or on anticoagulant treatment • Patients with known hypersensitivity to FFP or plasma-derived products
Subject grouping
During the study, the subjects were classified into two groups based on the severity of their intoxication as determined by the Peradeniya score: Group I consisted of 48 patients with moderate OP poisoning (POP scale 4–7), while Group II comprised 44 patients with severe OP poisoning (POP scale 8–11). 1
Randomization and allocation methods
Randomization and allocation concealment were independently conducted by a researcher who was not associated with the care or assessment of the patients. The allocation of patients in this study was performed by computer-generated random numbers, which randomized the patients into two equal subgroups. A single-blind methodology was used, with patients in the moderate group assigned to receive either standard treatment for acute OP poisoning (Group Ia, n = 24) or standard treatment plus FFP (Group Ib, n = 24), and patients in the severe group assigned to receive either standard therapy for acute OP poisoning (Group IIa, n = 22) or standard treatment plus FFP (Group IIb, n = 22). Then, 46 patients received standard treatment only in both groups (Group Ia, n = 24) and (Group IIa, n = 22), and 46 patients received FFP in addition to standard therapy in both groups (Group Ib, n = 24) and (Group IIb, n = 22).
The standard protocol for OP detoxification management in PCC-ASUHs involves care of the airway, respiratory, and circulatory systems. The excessive muscarinic effects are then reversed by administering atropine intravenously according to the doubling dose method, beginning with boluses of 1 to 3 mg, to observe clinical responsiveness every 5–10 min of each dose and then double the previous dose until the patient is fully atropinized (which is indicated by a clear chest, a systolic blood pressure greater than 90 mmHg, and a pulse rate greater than 90 beats per minute). Then, patients are switched to an IV infusion of 10% to 20% of the total loading dose per hour (hr) as a maintenance dose, a maximum of 2 mg/hr, until the disappearance of muscarinic symptoms, then gradual tapering by 10%–20% every hour with monitoring for recurrence of the muscarinic symptoms. The patients received an IV obidoxime bolus as early as possible (Toxogonin 250 mg), 1-2 ampoules diluted with 10 mL of 0.9% normal saline initially over 20–30 min, followed by a maintenance dose of 1 amp/8 h in 100 mL of 0.9% normal saline over 30 min. They continued for a maximum of 7 days, 12 h after the atropine infusion was stopped or died. Patients were followed up for a further 24 h. After discontinuing atropine and obidoxime infusions, the patients were discharged. 9
In addition to standard treatment, patients in both group Ib (n = 24) and group IIb (n = 22) received FFP transfusions starting within 1 hour after presentation and lasting for a maximum of 24 h after OP intoxication in a dose of a total of nine packs, each pack 250 mL, over 3 days, as follows: four units on the first day, three units on the second day, and two units on the third day. 16
Study procedures
History-taking, a complete physical examination, 12-lead ECGs, and laboratory investigations were conducted for each patient in the study. The diagnosis of acute OP intoxication was established based on the following criteria: a documented history of exposure to OP compounds, improvement of muscarinic symptoms and signs after atropine administration, and low serum BuChE activity. Treatment, including the total dose of atropine and oximes, was recorded. Patients' outcomes regarding the need and duration of mechanical ventilation, length of hospital stay, and mortality were similarly documented.
Laboratory parameters
Venous blood samples were obtained for serum BuChE level first when the patients presented to the emergency room for diagnosis and then daily for 3 days after 1 hour of completing the FFP infusion for serum BuChE level follow-up. Serum BuChE was determined using a kinetic colorimetric method. 17 Following the manufacturer’s instructions, the researchers used the pseudo-ChE kit (PCB260, Biochemical Enterprise, Via Toseli, Milano, Italy). BuChE hydrolyzes a substrate, S-butyrylthiocholine iodide, to butyric acid and thiocholine. The liberated thiocholine reacts with DTNB (5,5′-Dithiobis-2-nitrobenzoic acid) to form 5-thio-2-nitrobenzoic acid with a solid yellow color. The color formation rate is directly proportional to enzyme activity measured at 405 nm on a semi-auto analyzer, with a reference range of 4900–11,900 U/L.
Criteria of severity
Peradeniya organophosphorus poisoning (POP) scale.
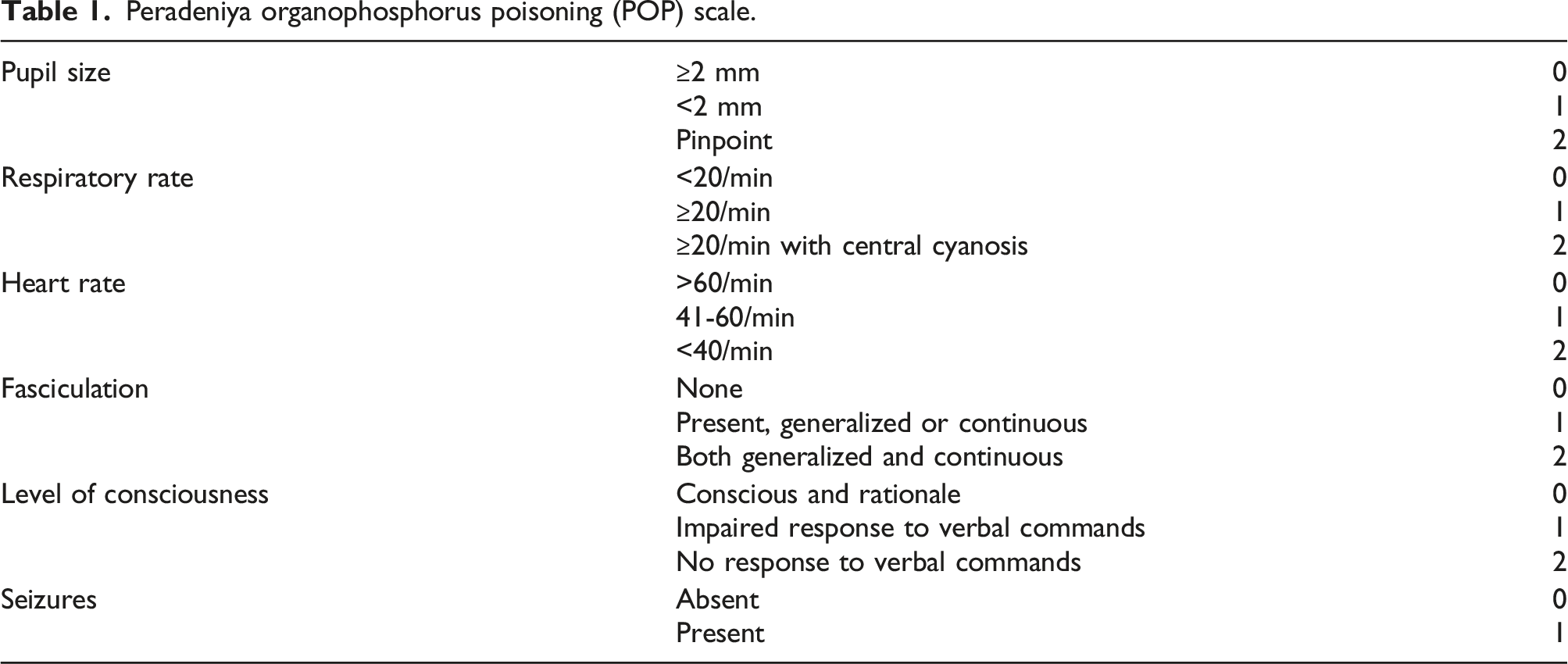
Study outcomes
The primary outcome was in-hospital mortality; secondary outcomes included the total required dose of atropine, the need for and duration of mechanical ventilation, the development of intermediate syndrome, and the total length of hospital stay.
Statistical analysis
The results were statistically analyzed using the SPSS software (Statistical Package for Social Sciences) version 20. Continuous variables were expressed as means ± SD. The chi-square test was used to compare categorical variables between the groups. An independent sample t test and a Mann-Whitney U test were used to compare the mean or median difference between two independent variables. An ANOVA test and a one-way statistical analysis were used to compare more than two groups according to the data distribution. The Friedman test compared more than two mean values in the same group. A p-value less than 0.05 was considered statistically significant for equivalent data.
Sample siz
Using the PASS 11 program for sample size calculation, setting power at 80% and alpha error at 0.05, according to Saad et al., 18 the expected mean duration of hospital stays in the intervention group was 2.43 ± 0.5 days and in the control group was 3.06 ± 1.4 days. The required sample size was estimated to be 92 patients (46 patients per group, either FFP or standard treatment group only).
Results
Classification of the studied patients.

Baseline demographic and toxiologial data.

p-value >.05: Non significant (NS); p-value <.05: Significant (S); p-value <.01: highly significant (HS).
aChi-square test.
bIndependent t test.
cMann Whitney test.
Clinical characteristics and vital data.

p-value >.05: Non significant (NS); p-value <.05: Significant (S); p-value <.01: highly significant (HS).
aChi-square test.
bIndependent t-test.
ECG.findings.

aChi-square test.
Complications among the studied patients.

p-value >.05: Non significant (NS); p-value <.05: Significant (S); p-value <.01: highly significant (HS).
aChi-square test.
Serum Butyryl cholinesterase at baseline, day 1, day two and day 3.

p-value >.05: Non significant (NS); p-value <.05: Significant (S); p-value <.01: highly significant (HS).
aMann Whitney test.
Outcome measures.
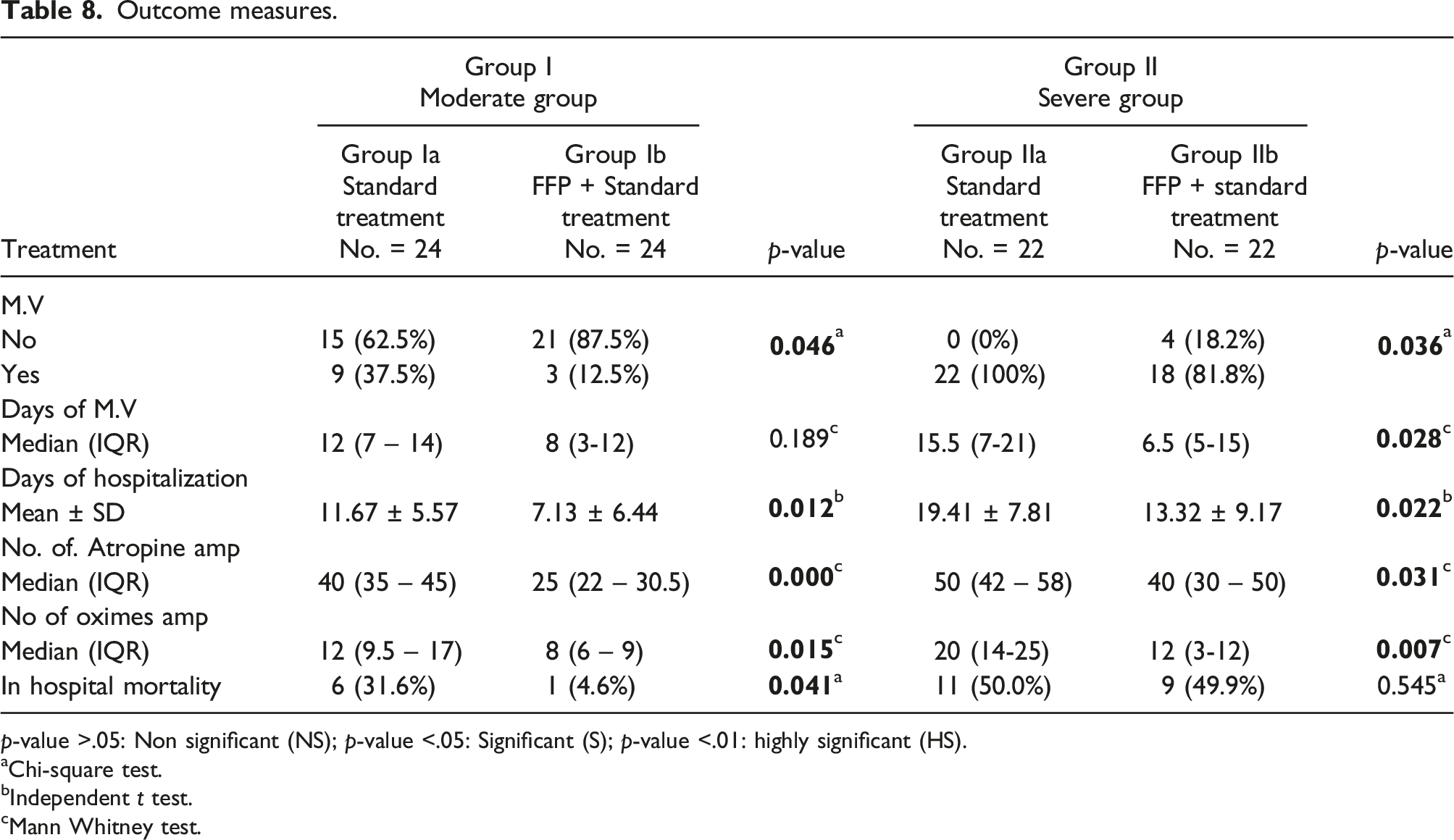
p-value >.05: Non significant (NS); p-value <.05: Significant (S); p-value <.01: highly significant (HS).
aChi-square test.
bIndependent t test.
cMann Whitney test.
Discussion
In this randomized controlled trial (RCT), it was demonstrated that using a total of nine packs of FFP (250 mL each) on three consecutive days in patients with moderate and severe grade OP poisoning significantly reduced the total doses of atropine and oximes, the total hospitalization period, and the need for mechanical ventilation. FFP significantly reduced the intermediate syndrome and mortality rates among patients with moderate-grade poisoning. FFP transfusions significantly improved serum BuChE levels in patients with OP poisoning. The hypothesis underlying the use of FFP in OP poisoning was that FFP acts as a bioscavenger to facilitate various catalytic and stoichiometric detoxification mechanisms of OP and lower its concentrations in the plasma.19,20 Fulton and colleagues have suggested that the positive effects of administering varying amounts of fresh frozen plasma (FFP) units in OP poisoning may be due to the volume resuscitation effects of the total amount of proteins, primarily albumin, found in the transfused plasma. 21
BuChE is a natural scavenger that binds OPs and inactivates them in blood before binding to AChE in target sites, thus preventing toxic effects on the neuromuscular junction. However, no human trials have been conducted using BuChE due to the high costs and large quantity required to inactivate OPs. An alternative approach is to use cholinesterase rich FFP. BuChE activity in banked blood is maintained for up to 7 days in FFP, and its half-life is 10 days in healthy individuals who received 460 mL of plasma. The exact amount of FFP needed for OP-poisoned patients is not yet clearly defined.
22
FFP treatment can be used as an alternative approach for patients with acute OP poisoning due to BuChE replacement, albumin content, and volume restoration.
23
In this study, BuChE levels significantly increased in each group after treatment, possibly due to the oximes used, which are cholinesterase enzyme reactivators and should be given as soon as possible in OP poisoning. However, the poor ability of oximes to penetrate the central nervous system (CNS) and the rapid re-inhibition of reactivated acetylcholinesterase (AChE) by circulating OP could lead to a continuous re-inhibition of regenerated AChE. Therefore, patients can only benefit from the regeneration of AChE, when the OP concentrations have dropped below a toxic level. In the present study
The present study explored important findings regarding the therapeutic efficacy of FFP transfusion in OP poisoning. Consistent with other studies, FFP therapy reduced the incidence of respiratory failure, the requirement of mechanical ventilation among moderate and severe groups, and the duration of mechanical ventilation among the severe group only. The benefit of ventilator-free management is always significant in OP poisoning, as ventilator-associated factors contribute to morbidity and mortality. In their study, Tang et al. (2016) 25 noted that mechanical ventilation was a significant risk factor for mortality in patients with chlorpyrifos intoxication. Patients with severe OP poisoning often require invasive positive pressure ventilation, as respiratory failure is the most life-threatening effect and requires immediate intervention. OP poisoning inhibits respiration through central and peripheral mechanisms, with central mechanisms being more predominant. Atropine can impede the production of secretions in pulmonary airways; however, it cannot help remove secretions. 26
This study noted that adding FFP to standard treatment reduced the incidence of shock among patients in the severe group. However, the current investigation did not demonstrate any significant difference regarding ECG findings between patients who received FFP plus standard treatment and those who received standard treatment only. The most important clinical consequence of the acute cholinergic crisis is hypovolemic shock, with all its negative metabolic consequences. Organophosphorus poisoning induced shock is a complex condition that can cause a combination of various types of shocks, including hypovolemic, cardiogenic, and distributive. Hypovolemic shock can occur due to symptoms like vomiting, diarrhea, and sweating that cause fluid loss. Cardiogenic shock is rare, but conventional therapy is ineffective in severe cases. Distributive shock occurs due to various pathophysiological mechanisms, such as the decrease in peripheral vascular tone caused by organophosphorus poisons acting on cholinergic receptors, inhibition of baroreflex due to the blockage of sympathetic and parasympathetic ganglionic nicotinic transmission, and histamine-induced hypotension 27 FFP transfusion may act as a form of volume resuscitation and have a positive effect on OP-intoxicated patients, according to Fulton J et al. (2005). 21
Another important impact of FFP transfusion in the present study was a significant shortening of the hospital stay. This might be attributed to the reduced incidence of respiratory failure, requirement of mechanical ventilation, and duration of mechanical ventilation. We observed a lower incidence of pneumonia and intermediate syndrome among patients who received FFP transfusion. Shorter hospital stay saves costs, staff, and bed capacity.
The results of the present research agreed with the results of Guven et al. (2004), which included 33 patients admitted to the ICU with OP poisoning and detected that using two packs of FFP daily from the second day of treatment in 12 patients significantly reduced the intermediate syndrome and mortality rate when compared with 21 patients receiving conventional therapy. 22 Dayananda et al. (2016) conducted a randomized study to investigate the effects of administering decreasing amounts of FFP during the first 3 days after hospitalization to 40 ICU hospitalized patients with OP poisoning, 16 demonstrating a significant reduction in ventilation time and mortality for those who received additional FFP therapy compared to the control group. According to Saad et al. (2017), treating OP toxicity early (30 min after hospitalization) and with a high dose of FFP (20 mL/kg) could be an effective method. 18 FFP containing BuChE can help improve clinical outcomes by reducing mortality rates, the duration of hospital stays, and the need for ICU admission. Late administration of FFP can lead to the complete absorption and distribution of OP through systemic circulation, which can reach the target tissues. In contrast, early administration of FFP is more effective in blocking free OP.
However, the current results contrasted with two negative randomized studies conducted by Pazooki et al. (2011), who discovered that administering four packs of FFP as a starting dose did not significantly improve the clinical outcome of patients poisoned with OP, and they did not measure the BuChE enzyme levels. 28 Furthermore, Pichamuthu et al. (2010) conducted a study involving 60 OP poisoning patients; 20 received albumin, 19 received FFP, and 19 received saline. 29 They reported that even though FFP could enhance the scavenger effects of BuChE, it may not be sufficient to influence the inhibitory effect of OPs at the nerve site. ChE activity in FFP may decrease by up to 30% due to the duration of preservation and freezing methods, as revealed in various studies. 18
In the present study, the type of OP involved could not be identified because patients and their guardians lacked information. The most common OP-ingested compounds are off-label, readily available, inexpensive, locally made, and liquid in small glass bottles. This finding aligns with previous research. There are varying degrees of toxicity among different types of OPs. Some, like phorate, are highly toxic and belong to the WHO Class IA, while others, such as dimethoate, are less toxic and belong to Class II. The WHO stated that FFP may be more effective against Class IA OPs than Class II OPs; additionally, fenthion and chlorpyrifos can significantly inhibit BuChE compared to dimethoate. However, it is unclear whether FFP is equally effective against all types of OPs, as the studies evaluated did not specify the specific type of OP. 23 In this research, adverse events associated with FFP infusions were predictable and did not require treatment discontinuation. Two patients developed urticaria, which was treated with antihistamines.
The total case mortality rate in this study was 29%, with 74% of the mortalities occurring in patients with severe-grade poisoning. The overall mortality following OP poisoning described in various studies varies between 4% and 30%, but in a recent report from India, it has reached 33%. Mortality among mechanically ventilated patients can be above 50%.6,30,31 The high mortality rate in this study may be attributed to the fact that the researchers included patients admitted to the ICU only, where 52% and 48% of patients had moderate and severe poisoning, respectively, and no patients with mild-grade poisoning were included in this study. Mortality rates are relatively low for mild to moderate cases of OP poisoning. However, severe cases carry an exceptionally high mortality rate 1 FFP significantly reduced the mortality rate among patients with moderate-grade poisoning. Among patients with severe-grade poisoning, mortality was lower in the FFP group (9 of 24) than the standard treatment-only group (11 of 24) but was not statistically significant.
Overall, these discrepancies in results between studies regarding FFP efficacy in OP poisoning could be attributed to different factors, including variable FFP administration time; early administration of FFP is more effective in blocking free OP; various dosages of FFP in these studies (either four packs of FFP as a starting dose or a reducing dose (total of nine packs) over three consecutive days); as well as different types of used OP compounds, as it is unclear whether FFP is equally effective against all kinds of OPs.
Conclusion
In this RCT, it was demonstrated that early use of a total of nine packs of FFP (250 mL each) over three consecutive days in OP-poisoned patients of both moderate and severe groups significantly reduced the total doses of atropine and oximes, the total hospitalization period, and the requirement for mechanical ventilation. In addition, it decreased the incidence of the intermediate syndrome among patients in the moderate group. The authors reported a survival benefit from adding FFP to standard treatment in the moderate group only. However, a larger multicenter randomized clinical trial with stratification of patients according to the severity of intoxication and type of ingested OP compound is necessary to confirm and clarify the magnitude of the benefit of adding FFP to standard treatment for OP poisoning.
Limitations of the study
The study had limitations as it was conducted at a single center with a relatively small sample size, and the authors could not identify the specific type of OP compound involved. A larger multicenter RCT with stratification of patients according to the severity of intoxication and type of ingested OP compound was needed to ensure the efficacy of FFP in OP poisoning before considering its routine use in treatment.
Footnotes
Author contribution
all authors equally contributed.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and/or analyzed during this study are available from the corresponding author upon reasonable request
