Abstract
Fresh frozen plasma (FFP) is an effective reversal agent for hypocoagulable patients. Its proven efficacy continues to prompt its usage as both a prophylactic and a therapeutic therapy. Although published guidelines encouraging the appropriate administration of FFP exist, overutilization continues. The purpose of these ex vivo studies was to determine the effects of succeeding volumes of FFP supplementation on hypocoagulable plasma prothrombin time/international normalized ratio (PT/INR). By analyzing the decline in PT/INR with varying volumes of FFP, a minimal required volume of FFP could be identified representing the optimal volume to administer while still providing therapeutic effect. A total of 497 plasma samples were screened for elevated PT/INR values and 50 samples were selected for inclusion in this experiment. The initial PTs/INRs ranged from 12.5 to 43.4 seconds/1.42 to 4.91. Subsequent declines in PT/INR values were analyzed following addition of 50, 100, and 150 µL of FFP to a fixed volume of 250 µL of plasma (26.4 ± 5.318 seconds/2.99 ± 0.603, 13.3 ± 1.077 seconds/1.51 ± 0.122, 11.2 ± 0.712 seconds/1.27 ± 0.081, and 10.3 ± 0.533 seconds/1.16 ± 0.06, respectively). A nonlinear relationship between decline in INR values and percentage of FFP supplementation was demonstrated. The greatest effect on INR was obtained after supplementation with 50 µL (49%). Doubling and tripling the volume of FFP lead to significantly lower declines in INR (16% and 8%, respectively). Analysis of variance indicated a statistical significance with subsequent volume supplementation of FFP, but marginal clinical benefits exist between the PTs/INRs obtainable with increased FFP volume administration.
Introduction
Hemostasis could be defined as a physiologic equilibrium between coagulation and fibrinolysis. Blood remains in a hemodynamically fluid and inert state unless the coagulation cascade is activated. Virchow first described a triad of factors responsible for disruption of hemostasis leading to thrombosis. 1 Virchow triad describes the 3 primary factors that influence hemodynamics, stasis, endothelial damage, and hypocoagulable states. 1 Primary hemostasis consists of platelet adhesion, aggregation, and activation on the exposed subendothelial collagen following a vascular injury resulting in the formation of a primary hemostatic plug following vascular damage. 1 Secondary hemostasis involves interaction of coagulation factors related to extrinsic and intrinsic coagulation pathways culminating in the deposition of insoluble fibrin, which is further stabilized by crosslinking of the initial fibrin clot. 1 Coagulopathy follows an imbalance between activation of coagulation and compromised inhibition of coagulation and fibrinolysis. 2 Anticoagulant drugs are used to prevent and manage thrombotic disorders and their complications such as stroke. 3,4 Warfarin is the most commonly prescribed oral anticoagulant in the United States for patients with hypercoagulable conditions 5 although a number of other non-Vitamin K antagonists medications are licensed in the United States.
In the United States as reported by the National Collection and Utilization Survey, approximately 5,700,000 units of plasma were manufactured for transfusion in 2009. 5 An estimated 3 million units of fresh frozen plasma (FFP) are administered annually in the United States 6 often to critically ill patients with coagulopathies. Prophylactic FFP is often administered prior to invasive procedures to hemodynamically unstable patients with elevated prothrombin time/international normalized ratio (PT/INR), despite lack of adequate supportive evidence. 7 Most consensus guidelines mention only few clear indications for transfusion of FFP including treatment of bleeding due to multiple factor deficiency, emergency reversal of vitamin K antagonist, and treatment of thrombotic thrombocytopenic purpura (TTP), and other thrombotic microangiopathies. 8 Several published audits of FFP transfusion very often suggested inappropriate excess usage. 9 This excessive usage of FFP, besides incurring unnecessary health care expenditures, is actually etiologic in causing harm to patients due to complications such as transfusion-related acute lung injury (TRALI) and transfusion-associated circulatory overload (TACO). 10 Reports on intensive care unit transfusion practices in Europe and United States indicate an estimated 33% of plasma is transfused to nonbleeding patients, despite lack of evidence of benefit. 6,11,12 This unnecessary use of FFP not only incurs increased health care costs but also consumes considerable resources of blood components, which could either be appropriately used in patients who need them or whose manufacture could be avoided at the outset. Availability and implementation of clearly established transfusion guidelines, which are actually followed by physicians, are desperately needed to prevent such wastage. Based on this background it is hypothesized that the maximum effect on restoration of hemostasis is most apparent after administration of the equivalent of 2 units of FFP in patients with initial PT/INRs above 20 seconds/2.0. In order to evaluate this hypothesis, this ex vivo study was conducted to analyze the effective restoration of normal PT/INR times to hypocoagulable plasma samples after supplementation of 3 varying quantities of FFP.
Fresh frozen plasma is a blood component obtained from the centrifugal separation of the fluid component of anticoagulated whole blood, 13 then frozen within 8 hours to at least −18°C. A unit of FFP has a volume of 175 to 250 mL and an inherent INR of up to 1.3. 13 This solution contains the essential coagulation factors, which upon activation promote fibrin clot formation. The FFP consist of vitamin K-dependent factors II, VII, IX, and X, factors V and VIII, fibrinogen, and fibrinolytic/complement factors, which are among the soluble coagulation factors. 13 –15 The FFP is routinely used in the United States for therapeutic reversal of anticoagulation or to stop bleeding in actively hemorrhaging patients, despite scarce evidence from randomized controlled trials to support its use. 16 The FFP is easily produced and relatively inexpensive. Its primary drawbacks include the potential risk of transfusion-transmitted infections, allergic reactions, TRALI, and TACO. In emergency situations, FFP may require significant time to obtain from a regional blood bank and more time to thaw in the Transfusion Service. Furthermore, it may require several hours after administration in order to establish full physiologic anticoagulation reversal. 17,18 Current clinical practices promote excessive usage of FFP, which may result in complications that are better prevented. 19 Ideally, such an excessive volume of FFP may not be necessary for effective anticoagulant reversal.
In light of the lack of proper guidelines, this ex vivo study was designed to determine the minimal required volume of FFP necessary to restore normal clotting times (PT/INR) in patients with altered vitamin K-dependent coagulation factors due to warfarin therapy or altered hepatic function.
Materials and Methods
Deidentified and discarded blood samples were collected from the Loyola University Health System/Trinity Core Clinical Laboratory under an approved IRB protocol. Samples were randomly selected based on observation of icteric plasma. Icteric plasma was taken as an indicator of plasma with strong likelihood of elevated PTs. A total of 497 samples were screened for elevated PT/INRs using Innovin (Dade) on the Automated Coagulation Laboratory 300 Plus (ACL300 Plus, Instrumentation Laboratory). A total of 50 samples were selected for this experiment from the original 497 cohort. Selection criteria for inclusion into this study were based on an initial PT/INR of 12/1.4. The 50 samples’ PT/INR ranged between 12.5 and 43.4 seconds/1.42 and 4.91. A laboratory reference PT time of 9 seconds represented the experimental control value.
The FFP used in this experiment was manufactured in the laboratory by a method, which is equivalent to Blood Center manufacturing steps of licensed FFP. Blood draws from 10 males and 10 females (apparently healthy) were obtained. The blood was then centrifuged after the supplementation of sodium citrate (1part of citrate to 9 parts of blood) at 3000 rpm to obtain platelet poor plasma (PPP). Then, all of the plasma was pooled, mixed, and then aliquoted in 10-mL tubes. All aliquots were then stored at −70°C until used.
Sample Preparation
The following protocol was executed for each of the 50 specimens. From each specimen, 250 µL of plasma was placed into 4 vials individually labeled A, B, C, and D. Vial A was supplemented with 50 µL of saline, vial B was supplemented with 50 µL of FFP, vial C was supplemented with 100 µL of FFP, and vial D was supplemented with 150 µL of FFP, respectively. Saline- and FFP-supplemented vials were analyzed using the ACL300 Plus instrument to determine PT values.
In the design of this in vitro experiment, it is assumed that based on the 5-L blood volume of a patient averaging 70 kg in weight, the approximate plasma obtained based on the normal 45% hematocrit is 2.75 L. Considering administration of 2 bags (500 mL) to such a patient, the new circulating volume of plasma in that patient would be 3.25 L. We have designed this in ex vivo experiments, where we placed 50, 100, and 150 µL of FFP in a fixed volume of 250 µL of plasma and then determined the PT/INR values. The percentage of FFP administered was 16.6% when 50 µL of FFP supplemented; 28.6% when 100 µL of FFP was supplemented; and 37.5% when 150 µL of FFP was supplemented. The thought behind this approach was to identify the minimal volume of FFP necessary to correct PT/INR to avoid complications of circulatory overload while still adequately restoring hemostasis. All PT values for the discarded blood samples included in this study were obtained within 4 hours of their collection from the laboratory.
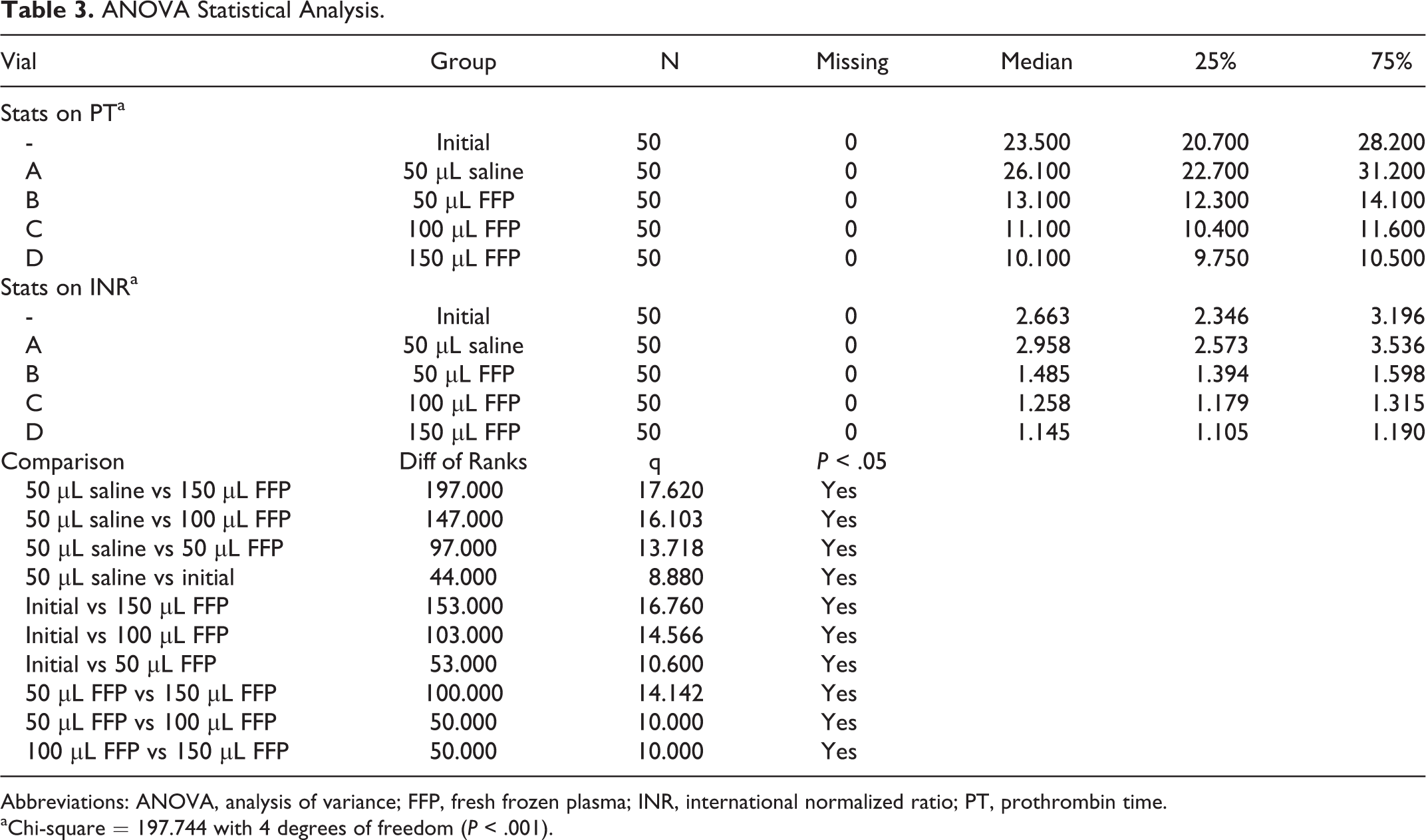
The results are tabulated and graphed. Statistical analysis was performed using Sigmaplot 12.0 statistical programing software. Means and standard deviations of all parameters were calculated for all values. The INRs based on PT values were calculated using a laboratory reference PT of 9 seconds and the International Sensitivity Index (ISI) value of 1.02 for Innovin. Optimal ISI reagents are standardized in clinical laboratories in order not to influence the PT/INR values. After all samples have been analyzed, all data were incorporated into a composite to generate composite means for each supplement group. One-way repeated measure analysis of variance (ANOVA) statistical testing was then performed using Graph Pad Prism. All pairwise multiple comparison procedures were conducted using Student-Newman-Keuls method. The level of statistical significance was established using a P value of <.001.
Results
Analysis of the 50 plasma samples revealed that the initial PT/INR ranged from 12.5 to 43.4 seconds/1.42 to 4.91. The composite initial mean PT/INR for the 50 icteric plasma samples used was 23.9 ± 4.915 seconds/2.71 ± 0.557. In analysis of each plasma sample, a combination of 50 µL of saline, 50 µL of FFP, 100 µL of FFP, 150 µL of FFP placed in vials A, B, C, and D, respectively, with a fixed volume of 250 µL of icteric plasma in each vial, the PT/INR values obtained were 26.4 ± 5.318 seconds/2.99 ± 0.603, 13.3 ± 1.077 seconds/1.51 ± 0.122, 11.2 ± 0.712 seconds/1.27 ± 0.081, and 10.3 ± 0.533 seconds/1.16 ± 0.06, respectively (Tables 1 and 2) (Figures 1 and 2). Fixed volumes in this ex vivo experimental model identified a negative correlation between percentage of FFP supplementation and change in INR. With increasing volumes of FFP supplementation, there was a corresponding decrease in the INR. Following 50 µL of FFP supplementation (representing a 16.6% supplementation), there was a drop in INR from an initial 2.99 to 1.51. With the supplementation of 100 and 150 µL of FFP, the INR dropped to 1.27 and to 1.16. The addition of 100 and 150 µL of FFP represent 28.6% and 37.5% supplementation of FFP to the original 250 µL volume, respectively (Figure 3). The varying volume supplementation in this experiment would correspond to volumes of 550 mL, 1.1 L, and 1.65 L FFP supplementation in a 70-kg adult. The corresponding decline in INR with each sequential increase in percentage of supplementation of FFP was 49%, 16%, and 8%. The decline in PT/INR with subsequent volumes of FFP is nonlinear.
Comparison of the International Normalized Ratio (INR) Values of Native Hypocoagulable Patient Plasma Samples With Saline Supplementation and Fresh Frozen Plasma (FFP)-Corrected Groups.a
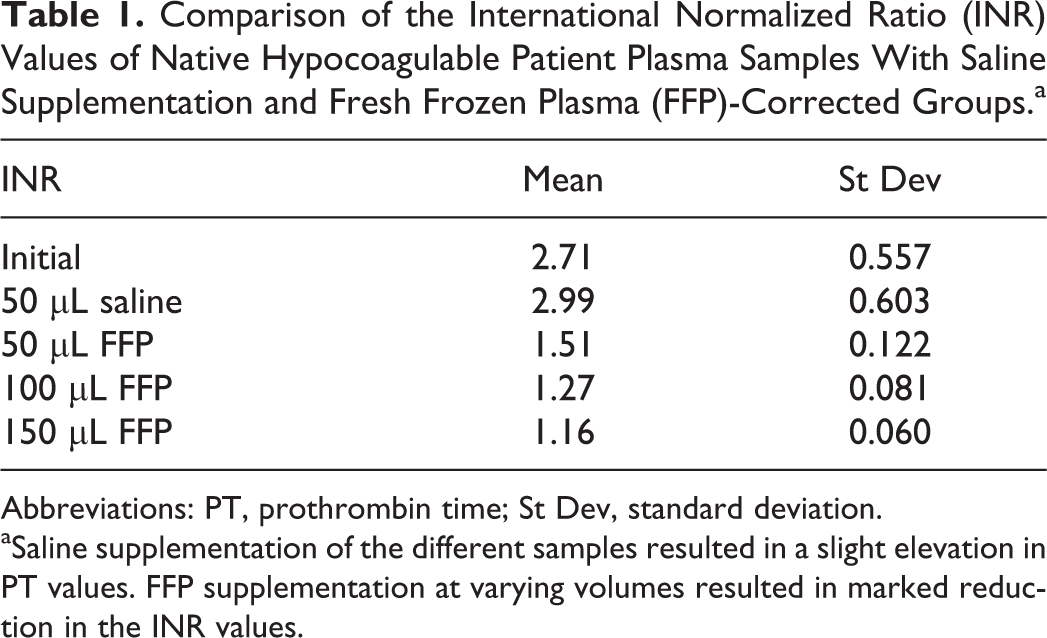
Abbreviations: PT, prothrombin time; St Dev, standard deviation.
aSaline supplementation of the different samples resulted in a slight elevation in PT values. FFP supplementation at varying volumes resulted in marked reduction in the INR values.
Comparison of the PT Values of Native Hypocoagulable Patient Plasma Samples With Saline Supplementation and Fresh Frozen Plasma (FFP)-Corrected Groups.a
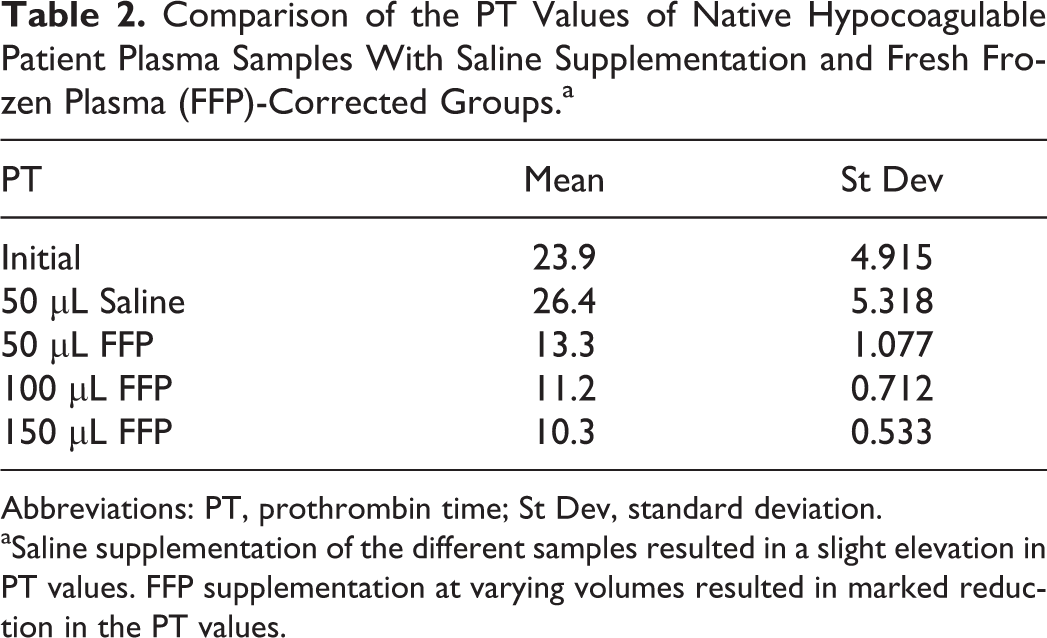
Abbreviations: PT, prothrombin time; St Dev, standard deviation.
aSaline supplementation of the different samples resulted in a slight elevation in PT values. FFP supplementation at varying volumes resulted in marked reduction in the PT values.
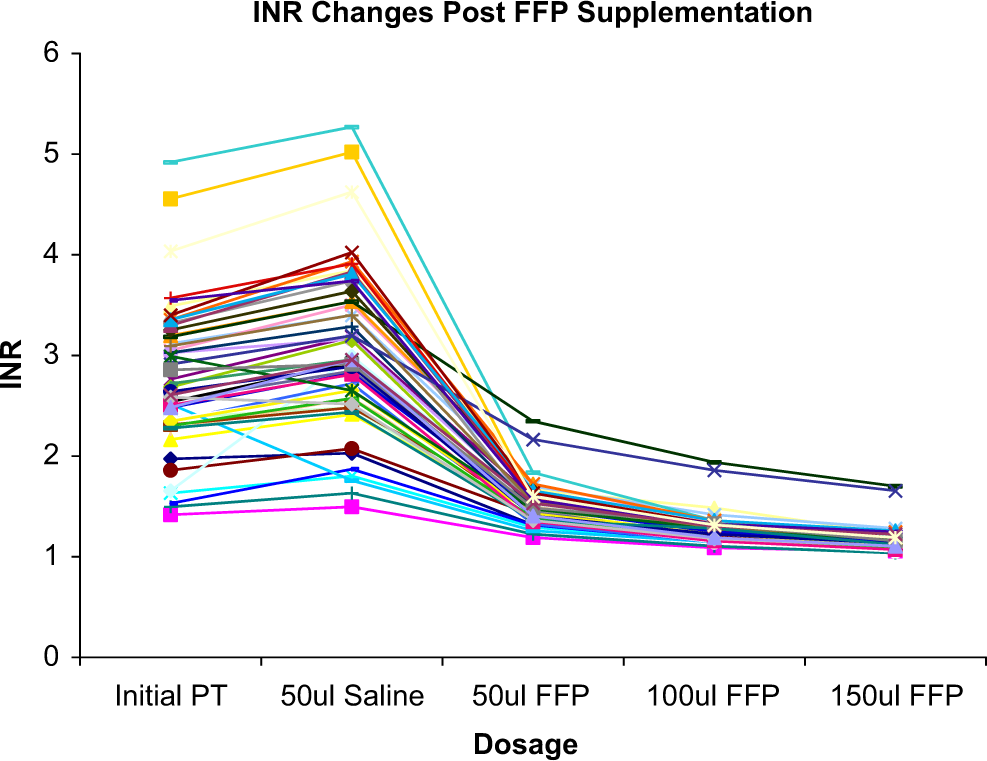
Baseline international normalized ratio (INR) of hypocoagulable patient samples (N = 50). Hypocoagulable patients and the effects of fresh frozen plasma supplementation on the restoration of clotting profile.
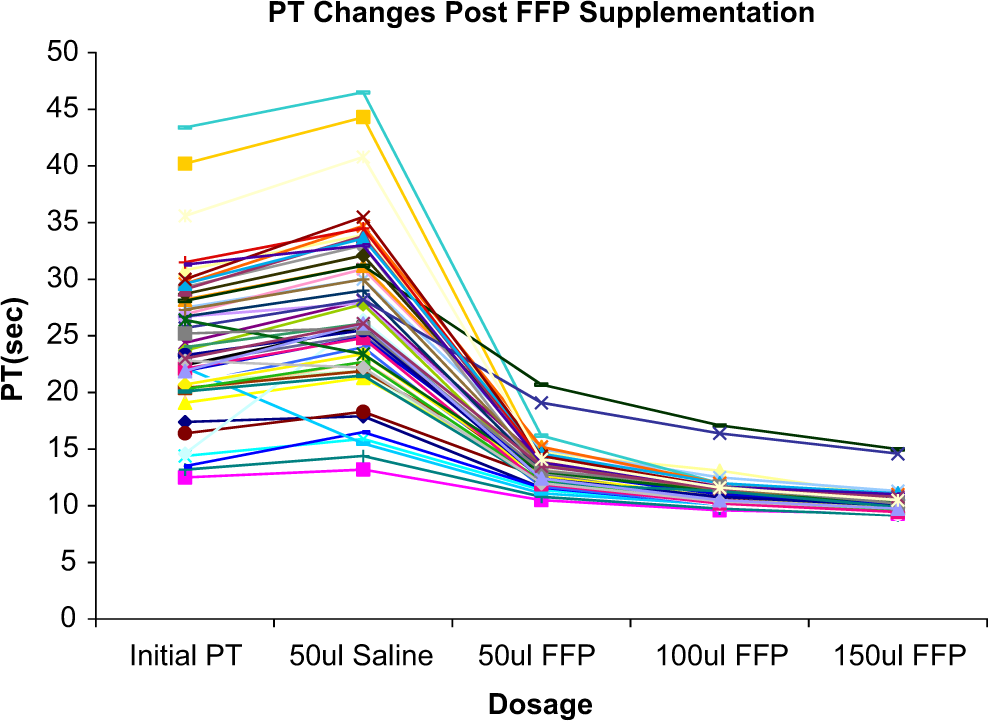
Baseline prothrombin time (PT) of hypocoagulable patient samples (N = 50). Hypocoagulable patients and the effects of fresh frozen plasma supplementation on the restoration of clotting profile.
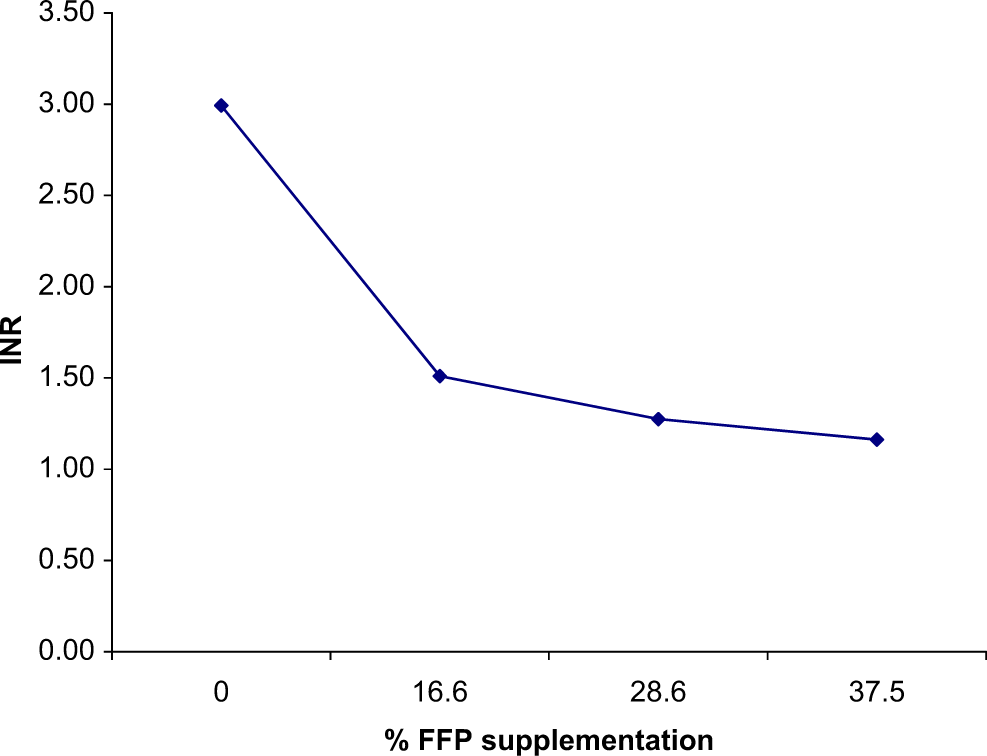
Evaluation of the correlation between percentage of fresh frozen plasma (FFP) supplementation to a set volume of plasma and the corresponding reduction in international normalized ratio (INR) values.
Discussion
Fresh frozen plasma represents one of the various therapeutic options available for the correction of anticoagulable states and treatment of hemorrhages. Its composition of essential clotting factors and proteins necessary for the activation of the intrinsic/extrinsic pathways of the coagulation cascade make it a useful blood component for manipulation of a patient’s coagulation profile. This experiment was designed considering a possible clinical scenario. Many patients present to emergency departments daily in hypocoagulable states, which are also highly prevalent in critically ill patient populations. Actively bleeding patients require immediate intervention. While conducting primary evaluations, routine blood draws are sent to the laboratory and patients are typically started on normal saline to maintain hemodynamic stability. Of the many preliminary laboratory tests requested, the coagulation profile is of particular importance when determining the most appropriate direction of treatment for correction of hypocoagulable states. Once the coagulation profile is available, it is evident whether the patient is anticoagulated/warfarinated based on elevations in PT time. The recommended therapeutic range for warfarin therapy is an INR between 2 and 3. The FFP is more commonly used initial option to correct elevated PT/INR in warfarinated or bleeding patients. However, the risk/benefit profile of FFP use, which has limitations and a very narrow therapeutic window, is not widely appreciated or considered by ordering physicians.
The use of warfarin depletes vitamin K-dependent coagulation factors that are necessary for the activation of the extrinsic coagulation pathway. Also, patients with liver pathology (liver insufficiency or end-stage failure) present with elevated PT/INR due to the liver’s inability to generate vitamin K-dependent factors, especially factor VII. The lack of available essential clotting factors coupled with the increased total blood volume secondary to intravenous fluid supplementation further elevates the PT/INR in these hypocoagulable patients. The 50 µL of saline that was supplemented to the 250 µL of patient plasma samples in our experiment is equivalent to the addition of 550 mL of normal saline to a 70-kg adult with an estimated plasma volume of 2.75 L. The effect of repleting lost volume to a bleeding patient causes a secondary dilutional effect on the PT/INR (Figures 4 and 5).
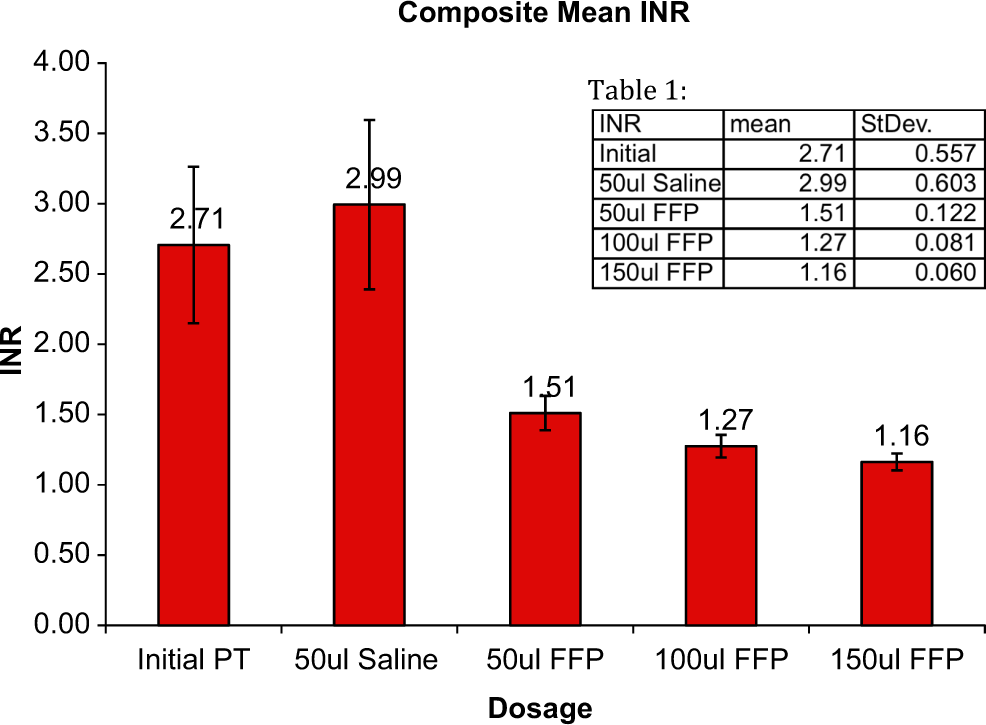
Comparison of the international normalized ratio (INR) values of native hypocoagulable patient plasma samples with saline supplementation and fresh frozen plasma (FFP)-corrected groups. Saline supplementation of the different samples resulted in a slight elevation in INR values. FFP supplementation at varying volumes resulted in marked reduction in the INR values.
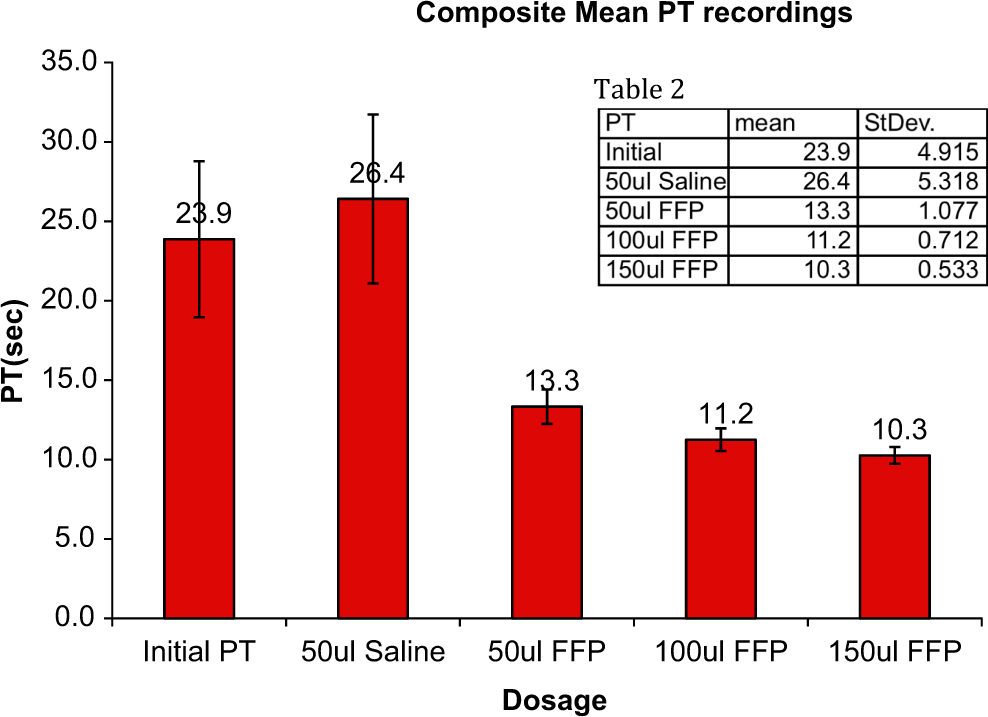
Comparison of the PT values of native hypocoagulable patient plasma samples with saline supplementation and fresh frozen plasma (FFP)-corrected groups. Saline supplementation of the different samples resulted in a slight elevation in PT values. FFP supplementation at varying volumes resulted in marked reduction in the PT values. PT indicates prothrombin time.
Fresh frozen plasma contains all the key coagulation factors necessary to restore hemostasis. It is administrated as a colloid, and volume remains chiefly intravascular, expanding the patients’ total plasma volume. The effects of FFP as an anticoagulation reversal agent are volume dependent and time dependent. By increasing the quantity of FFP administrated, an INR goal is obtainable. The duration of FFP supplementation is limited by the individual half-lives of the coagulation factors that constitute the mixture. Factor VII in particular has the shortest half-life of 4 to 6 hours. A patient with iatrogenic or pathologic vitamin K deficiency needs continued vitamin K supplementation after the administration of FFP to sustain the reversal effects on hypocoagulability provided by FFP. An average healthy individual not on anticoagulation therapy has an approximate PT/INR of 11 to 14 seconds/0.9 to 1.1. 20 For patients anticoagulated with warfarin, for example, the target INR is in the range of 2 to 3. In this study, plasma samples within the range of normal-, therapeutic-, and supratherapeutic INR values were tested to identify the effects of supplementation with varying volumes of FFP on the INR.
Current excessive usage of FFP suggests that PT/INR correction to a desired point depends upon volume, and it is assumed that a linear relationship exists between plasma dosage and PT/INR. In fact, Figure 3 illustrates a more negative exponential relationship between a decline in INR and an increase in volume supplementation, with the greatest effect on reversal of an elevated INR being obtained with the initial volume of FFP supplemented to a plasma volume with a therapeutic INR. The effects of this initial volume of 50 µL supplementation to 250 µL correlates with 550 mL of FFP supplementation to a 70-kg adult. The 1.51 INR obtained after this initial supplementation of FFP represents a 49% drop in INR. Doubling this volume only leads to a marginal 16% decline in INR. Tripling the initial FFP volume further drops the INR by 8%. With further FFP supplementation, the slope of the line continues to flatten. Although our ANOVA indicates statistical significance is present with additional FFP supplementation, marginal clinical benefits of further FFP supplementation are not likely once an INR of 1.5 has been achieved (Table 3). Current consensus guidelines also support that an INR of 1.5 is considered safe to perform a variety of invasive procedures, thus also negating the value of further FFP supplementation. 21
ANOVA Statistical Analysis.
Abbreviations: ANOVA, analysis of variance; FFP, fresh frozen plasma; INR, international normalized ratio; PT, prothrombin time.
aChi-square = 197.744 with 4 degrees of freedom (P < .001).
Hemostasis is a complex physiologic process. The construction of any experiment to modify any particular aspect of this complicated system has inherent limitations. All of the samples used in this experiment were obtained from a single institution thus potentially limiting the population variability. Our experiments only considered coagulation status. No consideration was taken into the status of platelet function or count, both of which might additionally increase risks of clinical bleeding. Also, the variable of time and timing of the dose was not investigated. Thus, the duration of elevated INR reversal using FFP was not established. To analyze the overall reduction in risk of bleeding complications with FFP supplementation, these limitations, which have clinical importance, would need to be separately considered and tested.
Conclusion
The therapeutic benefits of FFP are experimentally and statistically evident for the reversal anticoagulation. Supplemental FFP’s effects are volume dependent. Current clinical usage of FFP lacks unequivocal guidelines and thus excessive quantities are habitually administered to hypocoagulable/anticoagulated patients. This study illustrates that the greatest reduction in elevated PT/INR is obtained after supplementation with 550 mL of FFP to the average-sized adult who’s initial INR is >2. Our results support that administration of an initial bolus of 2 units possibly followed by initiation of a third unit of FFP drip with monitoring provides substantial reverses of PT/INRs above 17.8 seconds/2 and appears to provide appropriate treatment while minimizing the excessive administration of this blood component.
Two clinical trials, one called The TOPIC trial (http://www.clinicaltrials.gov website, NCT00953901)—aimed at demonstrating that omitting prophylactic FFP transfusion in critical ill patients with coagulopathy is safe in terms of bleeding complications—and another called EPICC Trial (http://www.clinicaltrials.gov website, NCT00302965)—studying the effect of prophylactic FFP use in actively bleeding patients—have both been recently completed. The publication of the results of these trials are eagerly anticipated to establish an improved evidence base for use of FFP in hypocoagulable patients.
Footnotes
Acknowledgments
The authors gratefully acknowledge the encouragement and support of Dr Eva Woijck, chairperson department of pathology. We are also thankful to Dr Debra Hoppensteadt for her scientific advice and logistic input for these studies. The authors are thankful to Dr Gail Hecht, assistant dean for medical students research, and Ms Kate Peterson, program coordinator of the STAR program, for their support in the completion of these studies.
Authors’ Note
This study was carried out as part of the STAR program at the Stritch school of medicine.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
