Abstract
Purpose
To explore the effect of acacetin on subarachnoid hemorrhage (SAH) and its possible mechanism.
Methods
SAH model of rat was established, and intraperitoneally injected with three doses of acacetin. To verify the role of PERK pathway, we used the CCT020312 (PERK inhibitor) and Tunicamycin (activators of endoplasmic reticulum stress). The SAH score, neurological function score, brain edema content, and Evans blue (EB) exudate were evaluated. Western blot was used to determine the expression of inflammation-associated proteins and PERK pathway. The activation of microglia was also determined through Iba-1 detection. TEM and immunofluorescence staining of LC3B were performed to observe the autophagy degree of SAH rats after acacetin. Tunel/NeuN staining, HE and Nissl’ staining were performed for neuronal damage.
Results
Acacetin increased the neurological function score, reduce brain water content, Evans blue exudation and SAH scores. The microglia in cerebral cortex were activated after SAH, while acacetin could inhibit its activation, and decreased the expression of TNF-α and IL-6 proteins. The pathological staining showed the severe neuronal damage and increased neuronal apoptosis after SAH, while acacetin could improve these pathological changes. We also visualized the alleviated autophagy after acacetin. The expression of Beclin1 and ATF4 proteins were increased, but acacetin could inhibit them. Acacetin also inactivated PERK pathway, which could improve the neuronal injury and neuroinflammation after SAH, inhibit the microglia activation and the overactivated autophagy through PERK pathway.
Conclusion
Acacetin may alleviate neuroinflammation and neuronal damage through PERK pathway, thus having the protective effect on EBI after SAH.
Keywords
Introduction
Subarachnoid hemorrhage (SAH) is one of the three cerebrovascular diseases with high mortality and disability rates, and the main cause of its occurrence is intracranial arterial rupture. 1 After subarachnoid hemorrhage, the body undergoes a series of pathological and physiological reactions, such as blood–brain barrier disruption, oxidative stress, inflammation, neuronal damage, and brain edema.2–4 When subarachnoid hemorrhage occurs, blood pressure and intracranial pressure rapidly increase, while cerebral perfusion pressure rapidly decreases. These pathological changes can exacerbate brain injury. 1 Studies have found that early brain injury (EBI) is the main cause of poor prognosis in SAH. 5 The pathological mechanisms of early brain injury include oxidative stress, inflammatory response, and blood–brain barrier disruption, among which neuroinflammation is an important driving factor for the occurrence of early brain injury. 6 The inflammatory response involves the activation of microglia and the release of pro-inflammatory factors, which may cause damage to adjacent neurons after SAH. 7 Therefore, inhibiting neuroinflammation is an effective treatment for alleviating early brain injury after SAH.
The protein kinase R-like endoplasmic reticulum kinase (PERK) signaling pathway plays an important role in neuronal apoptosis. Cerebral hemorrhage can cause brain damage, disrupt cellular metabolism, activate cellular stress responses, including unfolding protein response (UPR) and endoplasmic reticulum stress (ERS), and the disruption of ER function leads to ERS. 8 ERS can activate autophagy through the PERK/eIF2α signaling pathway. In mouse embryonic fibroblasts, knocking out the PERK/eIF2α gene can inhibit ERS-induced autophagy, indicating that the PERK/eIF2α signaling pathway is necessary for ERS-induced autophagy. 9 In 2018, a study also demonstrated the role of the PERK/Akt pathway in subarachnoid hemorrhage disease. The study showed that tetramethylpyrazine inhibited the PERK/Akt pathway and protected early brain injury in rats with subarachnoid hemorrhage. 10
Acacetin is a natural flavonoid compound containing 5,7-dihydroxy-4′-methoxyflavones, found in plants such as acacetin, chrysanthemum, locust tree, and thistle. It has various biological activities, such as antioxidant, anti-tumor, and anti-inflammatory effects. 11 Numerous literature reports have shown that Acacetin protects against cerebral ischemia-reperfusion injury through multiple pathways. The latest research reports on the treatment of neurological diseases with acacetin, such as improving cognitive function and reducing amyloid accumulation leading to vascular plaque formation and oxidative stress in a mouse model of Alzheimer’s disease. 12 In addition, acacetin can reduce the release of reactive oxygen species (ROS) from intracellular mitochondria, inhibit cell apoptosis signals, enhance adenosine triphosphate (ATP) production, and enhance neuronal cell viability.11,12 In an acute liver injury model, acacetin reduces the expression of ATF6, CHOP, and p-PERK proteins in a dose-dependent manner in vivo and in vitro. 13 At present, there is no research report on the therapeutic effect of acacetin on early brain injury caused by subarachnoid hemorrhage. Based on this, the experiment aims to explore the potential mechanism of acacetin in alleviating neuronal damage and neuroinflammation after subarachnoid hemorrhage through PERK pathway, and further explore its neuroprotective effect.
Materials and methods
Experimental animals
145 male SD rats, SPF grade, with a body weight of 280-300 g, were provided by Jinan Pengyue Experimental Animal Breeding Co., Ltd. The rats were raised at room temperature (24 ± 2) °C and relative humidity of 50% ± 10%, with light and dark circulation for 12 hours. They were housed in the well-ventilated animal laboratory room and allowed to eat and drink freely. Experimental animal certificate number: SCXK (Su) 2022-0006. The animal experiments were approved by the animal Ethics Committee of Yantaishan Hospital (Ethical number is 2024013). All experiments were performed in accordance with European Union regulations for the handling and use of laboratory animals, and are reported in accordance with the ARRIVE guidelines.
Animal grouping and the construction of SAH model
The mortality rate of rats in each group.

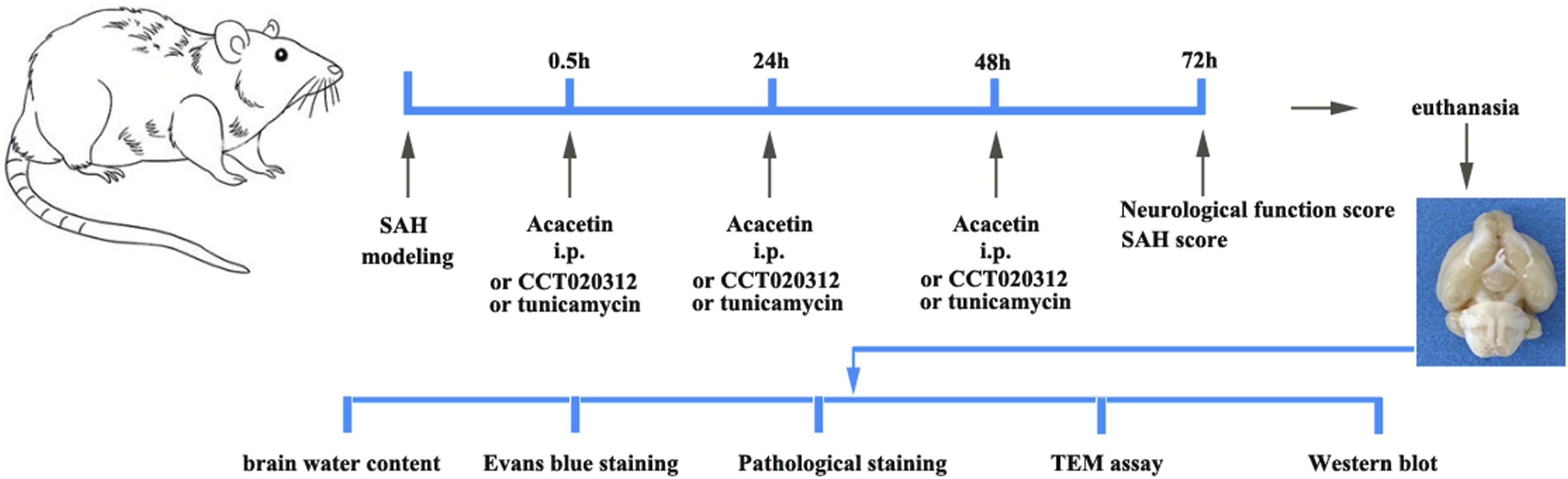
The flowchart of the entire experimental project.
A SAH model was constructed using intravascular perforation method. Rats were anesthetized by intraperitoneal injection of pentobarbital sodium (40 mg/kg). Disinfect and prepare the skin, cut open the midline tissue of the neck, block the anastomotic branch between the external and internal carotid arteries with an arterial clamp, and then ligate and dissect the external carotid artery; 4-0 nylon thread is inserted into the intracranial carotid artery through an incision in the internal carotid artery; Push the nylon suture forward by about 18 mm until resistance is encountered; Then push the nylon suture further by 3 mm, and a sense of emptiness indicates successful puncture of the blood vessel; After staying for 10 seconds, the suture line was quickly pulled out, the residual external carotid artery was ligated, and the wound was sutured and disinfected. The sham group does not puncture blood vessels, and other steps remain unchanged. Model success criteria: No substantial brain injury after puncture.
Neurological function score
Scoring criteria for balance beam experiments.

Modified garcia scoring rules.

Subarachnoid hemorrhage score
Using the Sugawa scoring system, 17 the basal cistern is divided into 6 segments, ranging from 0 to 3 levels, based on the number of blood clots in the subarachnoid space. Score 0: No subarachnoid hemorrhage; score 1: Minor subarachnoid hemorrhage; score 2: Moderate coagulation; score 3: Blood clots and identifiable arteries. The total score (0–18) is calculated by adding up the scores of all regions. 0–7 points for mild bleeding; 8–12 points for severe bleeding; 13–18 points for severe bleeding.
Measurement of brain water content
Brain water content was determined through the wet-dry method . 18 72 hours after SAH model, the brains were removed, and blood clots on the cerebellum and cerebellar surface were removed. The brain is divided into the left hemisphere (LH), right hemisphere (RH), cerebellum (CB), and brainstem (BS) and weighed separately (wet weight). The tissues were immediately placed in an oven for 24 hours, and then weighed again (dry weight). The water content of brain tissue was calculated in each group of rats according to the following formula: brain tissue water content = (Wet weight-Dry weight)/Wet weight × 100%.
HE staining
The brain tissue of each group of rats was fixed in 4% paraformaldehyde, then embedded in paraffin and sliced (the thickness of 5 μm). After soaking in xylene for dewaxing, ethanol gradient dehydration, hematoxylin & eosin staining, soaking in water, ethanol gradient dehydration, transparent sealing, and observing the brain tissue structure under a microscope (200x).
Nissl’ staining
3 μm of paraffin section was stained using Nissl’ Stain Kit (G1436, Solarbio). After dewaxing and rehydration, the slices were stained with Nissl staining solution for 30 min at 50°C, washed with distilled water, and differentiated with 95% ethanol for 2-3s. Finally, the slices were dehydrated with anhydrous ethanol, transparent with xylene, and sealed with neutral gum. The neuronal damage was observed under a microscope at a magnification of 400X.
Evans blue staining
Three rats were randomly selected from each group, and 2% EB dye (3 mL/kg) was injected into the tail vein to circulate for 1 hour; Perfuse rat heart with 0.9% sodium chloride solution; Brain tissue was homogenized with 50% trichloroacetic acid; Centrifuge the supernatant and mix it with an equal volume of trichloroacetic acid/ethanol solution (1:3); Centrifuge the supernatant and measure the optical density (OD) values of the sample and standard at a wavelength of 620 nm; Calculate the EB level using the standard curve.
Tunel/NeuN staining
The paraffin sections after 5 μm were sealed with 5% BSA containing 0.3% TritonX-100 for 1 hour, and then incubated with NeuN antibody (26975-1-AP, Proteintech, 1:200) at 37°C for 3 hours. The secondary antibody (A0516, Beyotime, 1:200) and TUNEL detection solution (KGA1406, KeyGEN BioTECH) were also dropped onto the slices and incubated in the dark at 37°C for 1 hour. Then, the nuclei were stained with DAPI (1:200), and the TUNEL positive neurons were observed under a confocal microscope (200X).
Immunofluorescence double staining
After fixation in 4% paraformaldehyde solution, the tissues were dehydrated and embedded in 30% sucrose solution. The frozen sections (6 μm) are reheated at room temperature for 2 hours, fixed with acetone, washed with phosphate buffer (PBS), dripped with immunofluorescence transparency and blocking solution, and incubated overnight at 4°C with primary antibodies rabbit anti-rat LC3B (bs-2912R, Bioss, 1:200) and Iba-1 (ab283319, Abcam, 1:2000). Next day, the slices were washed with PBS and incubated with secondary antibodies Cy3-labeled Goat Anti-Rabbit IgG (H+L) (A0516, Beyotime, 1:200) at 37°C for 1 hour. The cell nuclear was stained with DAPI staining (1:200, C1002, Beyotime). After sealing with anti-fluorescence quenching agent. The expression and localization of NLRP3 were observed under a microscope (200x).
TEM detection
The brain tissue was fixed at 4°C for 2h using 2.5% glutaraldehyde and 1% citric acid, followed by acetone gradient dehydration, embedding, sectioning, and staining with uranyl acetate and lead citrate. Then, we observed the mitochondrial ultrastructure using HT7800 transmission electron microscope, and take photos using the Hitachi TEM system.
Western blot
Brain tissue from each rat were lysed, and total protein was extracted using low-temperature ultrasound homogenization, and quantified using BCA protein kit. The protein sample (30 μg) was separated by protein electrophoresis, and transferred onto a polyvinylidene fluoride (PVDF) membrane after electrophoresis. Then membranes were placed in TBST containing skim cow milk and blocked at 4°C for 2 h. Primary antibodies p-PERK (bs-3330R, Bioss, 1:1200), t-PERK (bs-2469R, Bioss, 1:1500), p-eIF2α (#9721, Cell signaling technology, 1:1000), t-eIF2α (#9722, Cell signaling technology, 1:1000), Beclin1 (#3738, Cell signaling technology, 1:900), ATF4 (bs-1531R, Bioss, 1:1200), Iba-1 (ab283319, Abcam, 1:1000), TNF-α (17590-1-AP, Proteintech, 1:1500), IL-6 (BA4339, Boster, 1:1000) overnight at 4°C; After washing with phosphate buffered buffer with Tween (PBST), secondary antibody (1:5000) was incubated with PVDF membranes for 2h at 4°C. The protein plots were visualized through enhanced chemiluminescence (ECL), and taken under gel imager. Each protein band was analyzed using Image J, and the relative expression of the above proteins was normalized to GAPDH.
Statistical analysis
This experiment used SPSS 16.0 software for statistical analysis, and GraphPad 6.0 software for statistical graph production. The data is expressed as mean ± standard deviation (SD), and analyzed using one-way ANOVA. Multiple comparisons are conducted to compare whether there is a statistical difference between the groups (p < .05).
Result
Acacetin can improve neurological function and inflammatory response in rat after SAH
The representative images of brains were showed in Figure 2(A). In order to investigate whether acacetin can alleviate neurological damage after SAH, we selected 24h after SAH to evaluate neurological damage, using the balance beam score and the modified Garcia score for evaluation. The balance beam score results were shown in Figure 2(B). Compared with the Sham group, the SAH group showed a decrease in balance beam score. After intraperitoneal injection of 20 mg/kg, 40 mg/kg, and 80 mg/kg acacetin, the balance beam score significantly increased in a dose-dependent behavior. The modified Garcia score was consistent with the balance beam score, with a decrease in score in the SAH group. After intraperitoneal injection of three doses of acacetin, the score increased, and the most effective dose was 80 mg/kg (Figure 2(C)). In addition, we also evaluated the SAH scores of all rats. SAH model group showed a significant increase in bleeding volume and a significant increase in SAH grade score. After intraperitoneal injection of Acacetin, it was demonstrated that acacetin can significantly reduce bleeding volume and SAH score (Figure 2(D)). Further, we investigated whether acacetin has the improving effect on blood–brain barrier permeability (Figure 2(E)) and brain edema (Figure 2(F)) after SAH, and selected 72h after SAH for measuring brain water content. Meanwhile, we also determined the inflammatory factors (IL-6, and TNF-α) in brain tissues by western blot (Figure 2(G)). After SAH, these proteins IL-6 and TNF-α in brain tissues were upregulated, while after intraperitoneal injection of acacetin, these proteins decreased in a concentration-dependent manner. We also evaluated the activation of microglia in the brain tissue of SAH rats using the Iba-1 index. The result of western blot (Figure 2(H)) indicated that SAH rats exhibited the higher expression of Iba-1 than other groups, while acacetin treatment could decrease its expression in brain tissues. The images of immunofluorescence staining in each group were listed in Figure 2(I), and the result was consistent with that of western blot. Acacetin ameliorated SAH-induced neurological deficits, brain edema, blood–brain barrier disruption and inflammation. (A) Brain images after SAH. Neurological function score: (B) Beam balance assay and (C) Modified Garcia score. (D) SAH score; (E) EB content determination. (F) Brain water content. (G) Pro-inflammatory cytokines TNF-α and IL-6 in brain tissues of SAH rats were detected by western blot. (H) Western blot and (I) Immunofluorescence staining for Iba-1 was carried out to assess the activation status of microglia. The immunofluorescence image represents the expression of Iba-1 in the cerebral cortex. The scale bar of immunofluorescence images was 20 μm. *p < .05, **p < .01, ***p < .001.
Acacetin reduces neuronal apoptosis after SAH
Next, we analyze the neuronal damage caused by SAH through H&E staining (Figure 3(A)). SAH led to early brain damage. In the SAH group, severe pathological changes were found in the cortical area of rats, including nuclear folding, vacuolization, and neuronal cell deformation. Acacetin significantly improved the damage present in brain tissues, and reduced the number of damaged neurons in a dose-dependent manner. Nissl’s staining was also conducted (Figure 3(B)), and the result was consistent with the result of H&E assay. In this study, we used dual-fluorescence staining of Tunel/NeuN to observe cell apoptosis in rat brain tissue slices 72 hours after SAH (Figure 3(C)). TUNEL/NeuN/DAPI immunofluorescence staining showed that after SAH injury, the number of TUNEL-positive neurons was significantly increased. After administration of acacetin, the number of TUNEL-positive neurons was significantly reduced. Acacetin reduces neuronal apoptosis after SAH. (A) H&E staining. (B) Nissl’s staining. (C) Double fluorescence staining of TUNEL/NeuN. After 72h of SAH, the brain tissues were collected and fixed. 3 μm slices were stained. The immunofluorescence image represents the apoptosis of neuronal cells in the cerebral cortex. All images were captured at the magnification of 200X. The scale bar of HE and Nissl’s staining was 50 μm. The scale bar of immunofluorescence images was 20 μm. **p < .01, ***p < .001.
Acacetin inhibits excessive autophagy after SAH injury
The ultrastructure of mitochondria was observed using a transmission electron microscope. Compared with the Sham group, the SAH group had a large amount of mitochondrial swelling and mitochondrial cristae rupture, indicating that the mitochondrial structure of neurons had been damaged after SAH (Figure 4(A)). Acacetin treatment promoted the formation of mitochondrial phagosomes and maintained the normal mitochondrial morphology of neurons. Additionally, we also determined the autophagy-marker LC3B in brain tissues through immunofluorescence staining (Figure 4(B)). The experimental results indicated that SAH led to a high expression of LC3B, but when given acacetin, LC3B expression was significantly reduced. Acacetin inhibits decreased neuronal cell autophagy induced by SAH. (A) Representative TEM images. The scale bar is 2 μm. (B) Immunofluorescence staining for LC3B (red) in brain tissues. The fluorescence images were captured at the magnification of 200X (Scale bar was 20 μm). The mean fluorescence intensity of LC3B was quantitatively analyzed using Image J software. *p < .05, **p < .01, ***p < .001.
Moreover, we also analyzed some proteins in PERK pathway by western blot (Figure 5). We found that SAH activated PERK pathway, manifested with the increased phosphorylated-PERK and phosphorylated-eIF2α, further upregulating the expression of autophagy-related proteins Beclin 1 and ATF4. However, acacetin administration reduced the ratio of p-PERK to PERK and p-eIF2α to eIF2α, respectively, and decreased the expression of Beclin 1 and ATF4 proteins in a dose-dependent behavior. Acacetin decreased the expression of autophagy related proteins Beclin1 and ATF4, some proteins in PERK pathway. The protein bands were visualized through ECL reagent. These proteins were quantified by Image J software. *p < .05, **p < .01, ***p < .001.
Acacetin displayed the protective effect on SAH rats and controlled ERS-induced autophagy response via PERK/eIF2α pathway
To validate the role of PERK/eIF2α pathway in SAH, we applied the PERK activator CCT020312, and ERS activator tunicamycin was also given into SAH rats. We found that the improvement effect of acacetin on neuronal damage was disrupted by CCT020312 and tunicamycin (Figure 6(A) and (B)). Meanwhile, Iba-1, the marker of microglia, was determined by immunofluorescence assay (Figure 6(C)) and western blot (Figure 6(D)). The decrease in Iba-1 caused by acacetin was reversed by the above two reagents. At the same time, the expression of pro-inflammatory cytokines TNF-α and IL-6 was also altered after CCT020312 and tunicamycin injection (Figure 6(E)). Acacetin alleviated neuronal damage and neuroinflammatory response in brain tissues of SAH rat through PERK pathway. (A) H&E staining. (B) Nissl’s staining. The scale bars of HE and Nissl’s images were 50 μm. (C) Representative fluorescence images of Iba-1 in the cortical regions of the brain. The mean fluorescence intensity was analyzed by Image J software. The scale bar was 20 μm. (D) Protein band of Iba-1 was exposed to ECL and quantified by Image J software. (E) TNF-α and IL-6 proteins were determined by western blot. **p < .01, ***p < .001.
Next, TEM images showed that both CCT020312 and tunicamycin can exacerbate the malignancy of autophagy (Figure 7(A)). The fluorescence images of LC3B also showed similar results to TEM results, and the above two activators were able to upregulate the expression of LC3B (Figure 7(B) and (C)). After administration of CCT020312 and tunicamycin, the expression of autophagy-related proteins Beclin 1 and ATF4 significantly increased compared to 80 mg/kg acacetin. The inhibitory effect of acacetin on the phosphorylation of PERK and eIF2α was also weakened (Figure 7(D)). Acacetin inhibits endoplasmic reticulum stress-induced autophagy in SAH rats through the PERKE pathway. (A) TEM images showed the mitochondrial ultrastructure. The scale bar is 2 μm. (B) and (C) Immunofluorescence staining of LC3B in the cortical regions of SAH rats and histogram of LC3B-positive percentage which analyzed by Image J software. The scale bar was 20 μm. (D) The phosphorylated PERK and eIF2a proteins in PERK pathway and autophagy-associated proteins Beclin 1 and ATF4 in brain tissues were determined by western blot. **p < .01, ***p < .001.
Discussion
Subarachnoid hemorrhage (SAH) is a complex clinical syndrome caused by various cerebrovascular diseases, which can have a catastrophic impact on the central nervous system. Its high disability and mortality rates significantly affect the recovery of patients’ neurocognitive function, increasing family and socio-economic burden. Among them, intracranial aneurysm rupture and bleeding are the most common causes of SAH (85%). 19 In recent years, multiple studies have shown that early brain injury (EBI) is an important factor leading to poor prognosis in subarachnoid hemorrhage. 5 After the rupture of blood vessels on the surface or bottom of the brain, the blood entering the subarachnoid space causes a strong neuroinflammatory response in the body, manifested as activation of microglia, recruitment of peripheral immune cells, microcirculation disorders, disruption of the blood–brain barrier, and neuronal apoptosis.20,21 Our experimental data showed a severe neuronal damage in cerebral cortex, and the activation of microglia, and increased EB exudation, and excessive autophagy after SAH.
There are currently many effective monomeric drugs for the treatment of subarachnoid hemorrhage, such as Minocycline, 22 β carotene, 23 Trehalose, 24 Clodronate, 25 Cilostazol, 26 et al. Acacetin is a dihydroxy and monomethoxy flavonoid found in various plants. 27 Many literatures showed that acacetin exhibits a range of pharmacological potential, such as the prevention of cerebral damage caused by ischemia/reperfusion, 11 neuroinflammation induced by spinal cord injury (SCI), 28 lipopolysaccharide (LPS), 29 and 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine hydrochloride (MPTP), 30 LPS 31 and sepsis 32 induced lung injury, collagen induced arthritis, 33 as well as obesity. 34 However, the efficacy of acacetin in treating early brain injury (EBI) after SAH is not yet clear. Our study found that acacetin improved early brain injury after subarachnoid hemorrhage, inactivated microglia, and mitigated neuroinflammatory response, and inhibited the ERS-induced autophagy. The improvement effect of acacetin on EBI after SAH is dependent on drug concentration. So the potential mechanisms of acacetin’s protective effect on brain injury in SAH rats and its inhibitory effect on the ERS-induced autophagy are also worth exploring.
The endoplasmic reticulum (ER) is crucial for the synthesis and modification of intracellular proteins, and when ER function is impaired, it is called endoplasmic reticulum stress (ERS). In many pathological and physiological states, ERS can lead to cell apoptosis.35,36 Multiple studies have shown that ERS plays an important role in the occurrence and development of many neurological diseases, such as cerebral ischemia, Alzheimer’s disease, Parkinson’s disease, etc.35,37,38 When the function of the endoplasmic reticulum is damaged, the synthesized abnormal proteins will trigger endoplasmic reticulum stress and produce an unfolded protein response, inducing endoplasmic reticulum autophagy (ER-phagy). ERS mainly includes three signaling pathways mediated by membrane channel proteins, namely the PERK pathway, IRE1 pathway, and ATF6 pathway. In a stress-free state, the three membrane channel proteins mentioned above are tightly bound to the GRP78 protein. 35 Under specific conditions, due to the competitive binding of unfolded or misfolded proteins to GRP78 in the endoplasmic reticulum, GRP78 dissociates from PERK, leading to self-phosphorylation activation of PERK. The activated PERK phosphorylates eIF2α, ultimately activating the transcription of C/EBP homologous protein (CHOP) genes, thereby further mediating downstream cell apoptosis signaling pathways. 39 Research has also shown that phosphorylated PERK/eIF2α is crucial for the activation of endoplasmic reticulum stress-related autophagy. Once eIF2α is phosphorylated, it can promote the conversion of microtubule associated protein 1A light chain 3-I (LC3-I) to LC3-II, thereby activating the autophagy system. 40 Our research indicates that the PERK/eIF2α pathway is activated after SAH, leading to excessive activation of autophagy caused by endoplasmic reticulum stress, upregulation of ATF4 and Beclin1 expression, and promotion of neuronal apoptosis. However, acacetin can inhibit the activation of this signaling axis, downregulate the expression of phosphorylated PERK and eIF2α proteins, and inhibit ER phagocytosis, manifested by decreased expression of ATF4 and Beclin 1 proteins.
In addition, microglia-mediated neuroinflammation plays a crucial role in EBI after SAH. Animal experiments have shown that microglia immediately aggregate after SAH and diffuse from the cortex near the perforation site to the distal motor cortex area. Microglia exhibit sustained and significant accumulation from day 1-10 after SAH. 20 In this study, we also investigated the activation of microglia in the SAH model, and the experimental results are consistent with previous reports.
In summary, the results of this study indicate that acacetin can improve neurological behavior after SAH, alleviate brain edema, and inhibit the activation of microglia after SAH, and reduce the expression of pro-inflammatory cytokines. Importantly, acacetin exerted the significant inhibitory effect on SAH-induced excessive ER-phagy, inactivating the PERK/eIF2α pathway and downregulating the expression of ATF4 and Beclin 1 proteins. Our result suggests that acacetin may alleviate neuroinflammation after subarachnoid hemorrhage by inhibiting the activation of microglia and ER-pahgy through PERK/eIF2α pathway, thus having a protective effect on EBI after subarachnoid hemorrhage.
Footnotes
Author contribution
All authors conceived and designed research. Ying Liu: Material preparation, data curation, formal analysis, funding acquisition, writing original draft. Jianhua Tang: material preparation, data curation and formal analysis, investigation and methodology. Yiwei Hou and Lu Li: material preparation, investigation and methodology. Wenna Li, Ling Yu and Xue Wang: project administration and resources. Changbai Sui reviewed and revised the article. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical statement
Data availability statement
The data that support the findings of this study are not publicly available due to confidentiality but are available from the corresponding author.
