Abstract
The chemotherapeutic drug 5-fluorouracil (5FU) is frequently used to treat a wide range of solid malignant tumors, such as colorectal, pancreatic, gastric, breast, and head and neck cancers. Its antitumoral effects are achieved by interfering with the synthesis of RNA and DNA and by inhibiting thymidylate synthase in both malignant and non-malignant cells. Therefore, it can be responsible for severe toxicities in crucial body organs, including heart, liver, kidney, and reproductive system. Given the fact that 5FU-induced reproductive toxicity may limit the clinical application of this drug, in this study, we aimed to discuss the main locations and mechanisms of the 5FU-induced reproductive toxicity. Initially, we discussed the impact of 5FU on the male reproductive system, which leads to damage of the seminiferous epithelial cells and the development of vacuoles in Sertoli cells. Although no noticeable changes occur at the histopathological level, there is a decrease in the weight of the prostate. Additionally, 5FU causes significant abnormalities in spermatogenesis, including germ cell shedding, spermatid halo formation, polynucleated giant cells, and decreased sperm count. Finally, in females, 5FU-induced reproductive toxicity is characterized by the presence of atretic secondary and antral follicles with reduced numbers of growing follicles, ovarian weight, and maturity impairment.
Introduction
5-fluorouracil (5FU) is a well-known chemotherapeutic agent commonly administered for treating a vast number of solid malignant tumors, including epithelial cell malignancies emerging from the genitourinary and gastrointestinal tracts (especially those of colon, pancreas, and stomach), breast, and also epithelial malignancies found in the head and neck. It is also used for treating carcinomas of the ovary, prostate, neck, bladder and oropharynx with less efficacy.1,2 5FU acts by disruption of RNA and DNA synthesis and inhibition of thymidylate synthase in malignant and non-malignant cells.2–4 Since it can interfere with division of noncancerous cells, it may cause severe toxicity in crucial body systems, including heart, liver, and kidney.5–7 It has been also reported that 5FU could lead to notable defects in the reproductive system. 8 Given the fact that 5FU-induced reproductive toxicity may limit the clinical application of this drug, in this study, we have explained the main locations and mechanisms of the 5FU-induced reproductive toxicity.
5FU-induced toxicity in male reproductive system
5FU-induced testicular damage
Drugs used to treat cancer can directly or indirectly damage DNA and interfere with it, causing chromosomal aberrations or strand breaks.9–12 Research has shown that some chemotherapeutic drugs can also cross the blood-testis barrier, which can have negative effects on the testicles. These organs are sensitive to the toxic effects of chemotherapy, which can cause damage to germ cells or lead to and fibrosis of the interstitial tissue.13–16 Previous studies indicate that men who have undergone the MOPP regimen (Mechlorethamine, Oncovin, Procarbazine, and Prednisone) to treat Hodgkin's lymphoma, may experience permanent infertility as a result. 17
The testicular function involves an exocrine function by producing sperms in the seminiferous tubules that make up more than 75% of the testicular mass, and an endocrine function by secreting testosterone by Leydig cells in the interstitial space, and the rest of the testicular structure consists of the supporting and vascular tissues that play the role of nourishing and protecting sperms.18–20 As mentioned, spermatogenesis, which takes place in the seminiferous tubules, which are made up of germ cells and Sertoli cells, is a process that can be divided into three stages: the proliferation of spermatogonia, spermatocyte meiosis, and finally spermiogenesis, which leads to spermatozoa production. 18 This process of spermatogenesis is constantly taking place throughout the seminiferous tubules so that in each area of this tubule 5 to 6 generations of germ cells can be identified and the duration of this process in humans is 64 to 90 days until the release of adult sperms, in different species this time is different.18,21 The process of spermatogenesis, as described, is a complex and always dynamic process that can be affected by cytotoxic agents in various ways, such as damage or loss of a specific cell type in the germinal epithelium or damage during the maturation of sperm and the development of dysfunctional sperm, another effect is to damage the Sertoli and Leydig cells or other supporting components to destroy the suitable environment for sperm production and thereby damaging the process. 22
Chemotherapeutic drugs affect both the functional aspects of the testicles and their fertility and androgen production potential. These drugs can also have an indirect effect on the reproductive system by affecting the hypothalamic-pituitary axis. 13 The process of spermatogenesis is successful if there is a normal balance between the endocrine glands and the inhibition of the glands that can be caused by chemotherapy treatments, which has been found to be toxic to testicular tissue and to increase the risk of infertility.23,24 Usually, this disorder occurs directly with the effect of the drug on stem cells because in later stages the germ cells are more resistant. The administration of alkylating drugs has been observed to cause thickening of the basement membrane, interstitial fibrosis and aplasia of the germinal epithelium, which ultimately leads to reduced fertility in men. 25 Findings from 5FU also suggest that the drug may cause genital damage by inhibiting thymidylate synthetase and its genetic pathways.26–29
The impact of childhood chemotherapeutic drugs on testes remains largely unexplored. Given the physiological changes that occur during this period, including an increase in testicular volume and proliferation of immature Sertoli cells and Leydig cells, it is essential to consider the potential vulnerability of Sertoli cells to chemotherapeutic drugs. 30 Investigations on male childhood cancer survivors have revealed that alterations in Sertoli cells are indirectly reflected in variations in sperm count and follicle-stimulating hormone (FSH) levels. 14 Since Sertoli cells are the primary producer of inhibin in the testis, the inhibin/FSH ratio can also be utilized to assess the abnormality in Sertoli cells due to the absence of quantifiable indicators.31–33 However, it is challenging to utilize simply the inhibin/FSH index because of the strong contact between the gonadotropic system and Sertoli and Leydig cells, so it is preferable to investigate it by taking extra hormonal variables like testosterone and LH into account. 32 Serum testosterone and LH are also used to assess the function of Leydig cells, and hormonal biomarkers such as FSH, LH and testosterone in blood samples are used to evaluate the testes in cases where semen analysis is not possible.23,34,35 Also for detecting subclinical abnormalities of Leydig cells, LH-releasing hormone is used, which highly increases the serum LH level after administration if Leydig cells are abnormal. 20
Testicular atrophy often results from injury to the germinal epithelial cells since seminiferous tubules make up a significant portion of the testicles' bulk. On the other hand, decreased spermatogenesis is marked by a reduction in the quantity and mobility of sperms in each ejaculation. 36 Recent research has also revealed that men with germinal aplasia have lower levels of free testosterone and lower daily production of testosterone, which suggests that the mature testis's germinal epithelium is more vulnerable to chemotherapy than immature testicles are, as well as Leydig cells due to their higher mitotic rate.17,36–39 These cases indicate that people undergoing chemotherapy may develop secondary sexual characteristics despite having oligospermia or azoospermia because the hormones secreted by Sertoli are responsible for their occurrence.40,41 Exposure to chemotherapeutic agents can increase seminiferous tubules without germ cells, along with reduced tubule diameter and vacuolization, which results in decreased testicular weight in rodents, nevertheless, treated animals showed that impaired germ cells can escape abnormalities which cause testes to produce abnormal sperms.42–44
Chemotherapeutic drugs can harm Sertoli cells and decrease their ability to produce two essential proteins, androgen-binding protein (ABP) and transferrin, which play a crucial role in spermatogenesis. Only one study has investigated the impact of doxorubicin exposure on myoid cell proliferation in testicular tissue, and no significant effects were observed.45,46 In relation to Leydig, although it has been observed in pediatric cancer survivors that their function is impaired which subsequently leads to androgen deficiency, there is little information in animal studies and in a study which exposed the testes of premature mice to doxorubicin for 48 h in which there was no change in function or morphology, and in another in vitro study, 24 h of cisplatin, cyclophosphamide, and doxorubicin exposure did not affect Leydig cell density.47–52
Other chemotherapeutic drugs such as paclitaxel and cisplatin have been shown to have minimal effects on germ cell and myoid cell proliferation. However, studies have shown that 6-Mercaptopurine, an anti-metabolite similar to 5FU, can lead to the death of Leydig cells in mice through caspase-3 activation, suggesting that 5FU can also have an impact on testicular function and reproduction.
While the impact of 5FU on male reproduction is not well understood, the effects of other chemotherapeutic medications, such as paclitaxel, a taxane-based chemotherapeutic drug that reduces testicular volume, or cisplatin, which result in a dramatic decrease in the number of germ cells in both the fetal testis and the pre-pubertal testis is negligible. In the case of 6-Mercaptopurine, which, like 5FU, is an anti-metabolite that activates caspase-3 to eventually kill Lydig cells in mice, which makes it possible to consider 5FU as a factor affecting reproduction and testes.53–55 Mechanisms that have been reported for the pharmacological effects of 5FU in cytotoxicity include inhibition of thymidylate synthetase, which leads to a decrease in thymidine and ultimately reduced DNA production, or direct incorporation into nuclear RNA, which disrupts the formation of cytoplasmic RNA and also damages DNA through the attachment of residues.56–59 And according to the mentioned cases that lead to toxic changes in germ cells, it has been observed that cells in S phase are very sensitive to 5FU toxicity, and this potential 5FU damage to DNA has also been reported in 6-MP, which increases the likelihood of the reproductive system toxicity.60,61 In one study, the tissue of 5FU-treated mice showed destruction of the seminiferous epithelial cells and Sertoli cells vacuolization, while another study found Sertoli cells vacuolization (Figure 1) in the epididymal ducts but no histopathologic changes in the seminal vesicle. Additionally, some studies have reported weight loss in the testicles and defects in spermatogenesis after treatment with 5FU.28,62–65 This morphological effect on Sertoli cells and the observed weight loss can be due to a hormonal imbalance in the serum; as seen in a study in mice, levels of pro-alpha C and GnRH were significantly elevated, while levels of inhibin B, prolactin, activin A, and testosterone faced significant decrease.
26
Also tubular contraction and atrophic cells and abnormal sperms have also been observed, which has ultimately reduced the number of spermatocytes, as well as swelling and shrinkage of the tubules following 5FU administration.66,67 The effect of 5FU on male reproduction. Sclerosis, vacuolization and polynucleated giant cells (PGCs) were observed in seminiferous tubules that are affected by 5FU.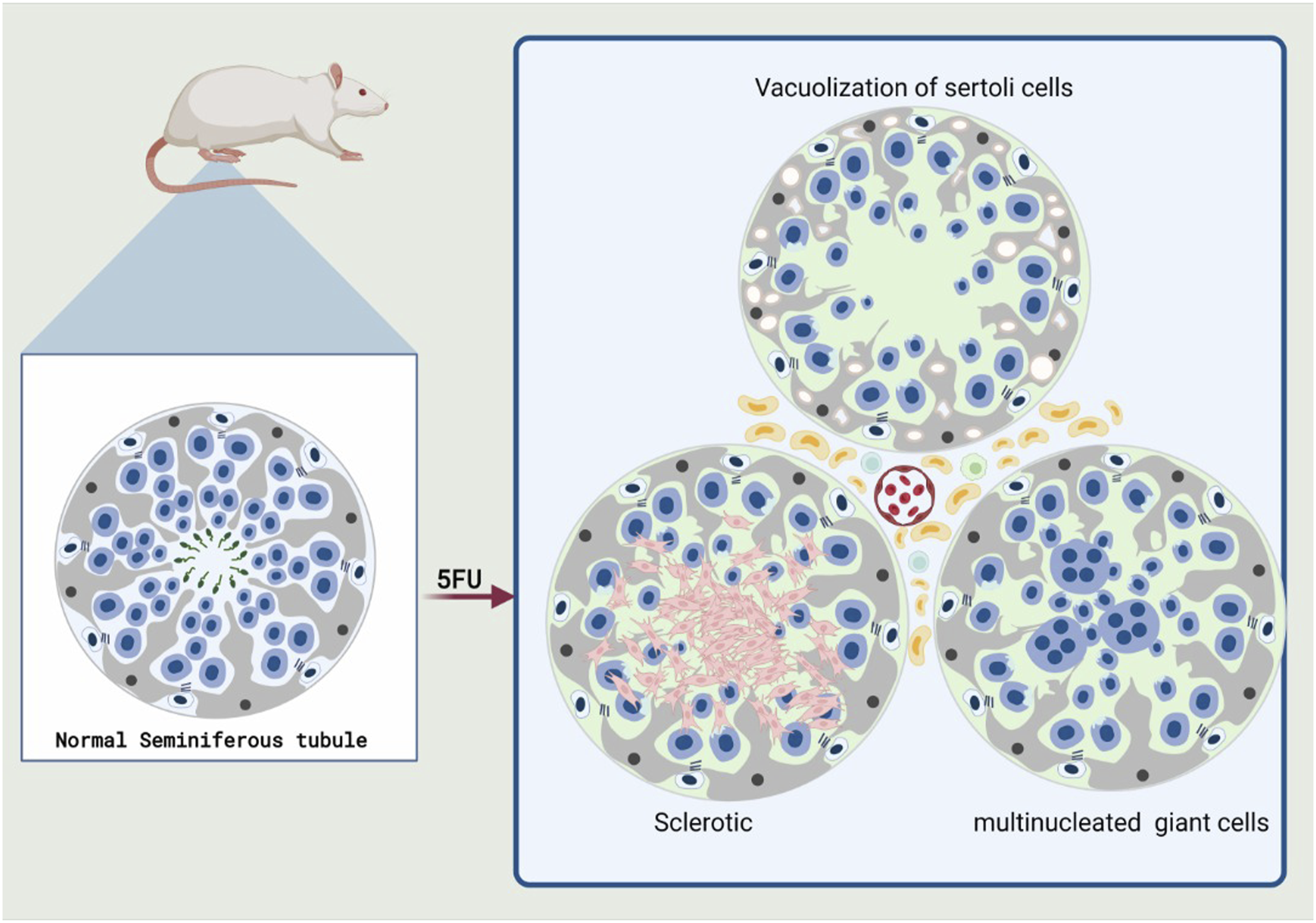
5FU-induced prostate damage
There is not much information about the effect of 5FU on the prostate. In a study by Takizawa et al., It was found that 5FU did not cause any histopathological changes, although it did reduce the relative weight of the prostate. 26 Furthermore, Al-Asmari et al. conducted a study that revealed shrinkage of the prostate gland and the presence of nuclear stratification in the group treated with 5FU. This group also showed an increase in prostate gland secretion. 62 In this study, it was also found that these effects were modified if Tau was administered. In weight loss of the prostate, the role of androgens in regulating its growth can be considered effective, As decreased serum testosterone levels can cause severe weight loss in this organ and it has been observed that in these patients serum levels of inhibin B, prolactin, activin A, and testosterone are significantly reduced, in contrast to serum levels of pro-alpha C and GnRH which showed a significant increase. 68 Nonetheless, in 5FU-treated patients, there were no noteworthy alterations in serum LH and FSH levels observed, although they showed an increase in serum GnRH levels. 26
5FU-induced sperm count and shape abnormalities
Summary of studies with their findings on the effect of 5-FU on male reproductive system.

Additionally, the percentage of abnormal sperms was significantly affected by 5FU treatment at different doses (5, 10 and 15 mg/kg, five consecutive days) (
5FU-induced chromosomal & DNA damage
In a recent study, Fahmy et al. (2020) observed a noteworthy proportion of chromosome aberrations in spermatocytes following the injection of 5FU with different doses; most of them univalent forms. 72 Further, in previous research, the effects of 3 distinct dosages of 5FU—5, 10, and 15 mg/kg—on the male germline cells of Swiss mice were evaluated. The results showed that 4 weeks after the treatment, 5FU significantly increased the number of genetic abnormalities. During week 8 after therapy, gamete morphology analysis did not reveal a statistically significant increase. This showed that there is a progressive reduction in the transfer of 5FU-related chromosomal aberrations from spermatogonia to sperm. This may be because the severely damaged spermatogonial cells are eventually eliminated during the spermatogenesis process. 73
In the seminiferous tubules, 5FU caused PGCs and spermatids with a halo-like appearance.71,74 One of the possible mechanisms for the formation of PGCs can be karyokinesis without subsequent cytokinesis. 75
The genomic expression of the degree of laddering (total DNA fragmentation) and the tail length, was increased in testicular cells after 5FU treatment in comparison with control manifesting single strand DNA damage, while increasing 8-hydroxy-2-deoxyguanosine (8- OHdG) and heat shock protein 70 (HSP-70) were observed. 76
5FU-induced sex hormonal changes
Testosterone is the major sex hormone in males. In Takizawa et al. study, they examined the effects of 5FU on sex hormones, and a decreasing trend in testosterone was observed in the treated-group with 5FU (0, 20, 30 mg/kg/day orally for 2 and 4 weeks). The serum level of GnRH which controls the production of LH and FSH had a statistically significant increase in the 5FU group. Inhibin B is a hormone that inhibits the synthesis of FSH which 5FU treatment lowers its level. The rats with vacuolation of Sertoli cells, had the lowest level of this hormone. In this study, 5FU caused no significant changes in serum levels of LH and FSH. Nonetheless, Activin A and Prolactin were decreased in treated groups. Contrarily, Pro-alpha C, a subunit of inhibin B was increased in the group treated with 20-mg/kg/day dose of 5-FU for 4 weeks (Table 1). 26
Primary and secondary effects of 5-FU on the hypothalamic-pituitary-gonadal (HPG) axis.

ABP, Androgen-binding protein; FSH, Follicle-stimulating hormone; GnRh, Gonadotropin hormone-releasing hormone; HPG, Hypothalamic-pituitary-gonadal; LH, Luteinizing hormone.
*This table summarizes the cases mentioned in the studies and the opinions of the authors, but many of the reactions are not presented due to the many hormonal interactions.
5FU-induced female reproductive toxicity
Effect of 5FU on ovarian size
Summary of studies with their findings on the effect of 5-FU on female reproductive system.

ICR: Institute of Cancer Research.
Effect of 5FU on ovarian function and secondary follicles
In order to examine the ovarian function, Naren et al. (2021) made both of their 5FU-treated and control groups super-ovulated by hormonal stimulation at the end of the 4th day of 5FU injection. The collected ovaries of the control group showed enlarged ampullas and a significant number of MII oocytes after hormone-induced super-ovulation. Unlike the control ovaries, no enlarged ampulla and mature oocyte was seen in the ovaries of 5FU-treated mice (Table 3). 8
As secondary and antral follicles have rapidly dividing cells, including oocytes and granulosa cells, they are more vulnerable to 5FU-induced toxicity. Although observing growing follicles being atretic in the ovary is a normal physiological phenomenon, in a prior intervention, the 5FU-treated ovaries demonstrated a considerable elevation in the amount of morphologically atretic secondary and antral follicles (Figure 2).
83
At last, according to almost all the previous studies, it is allegedly true that 5FU leads to a significant reduction in the amount and a notable deficiency in the maturation of growing follicles.8,78,84 The effect of 5FU on female reproduction. The amount of morphologically atretic secondary and antral follicles was significantly increased in ovaries treated with 5FU. 5FU causes a considerable decrease in the number of growing follicles, ovarian weight and, a notable impairment in their maturity.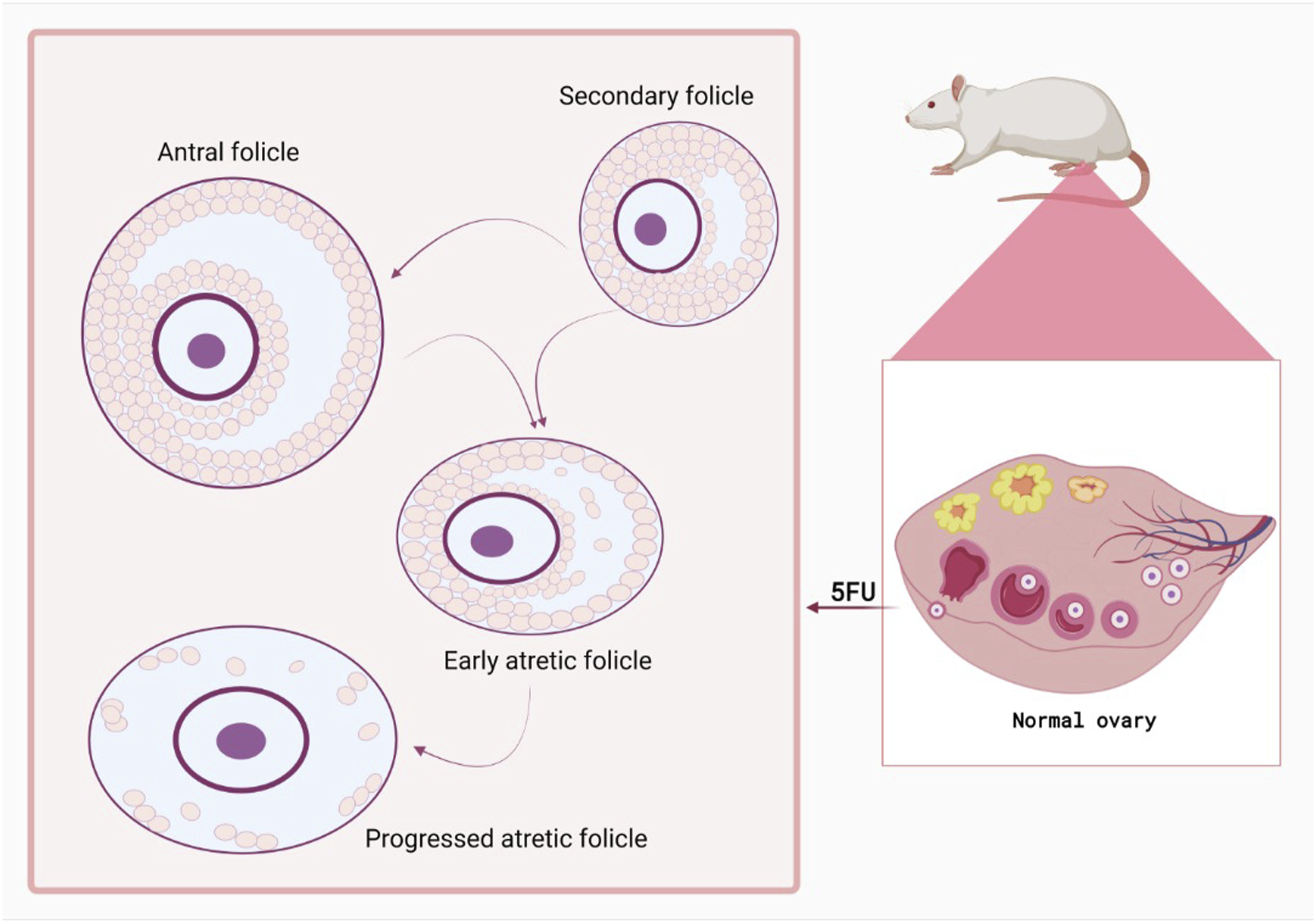
Effect of 5FU on primordial and primary follicles
It should be stated that, in some studies, the count of primordial and primary follicles had no remarkable differences between 5FU-treated and control ovaries which meant that 5FU did not significantly affect primordial and primary follicles.78,83 However, Almeida et al. (2021) observed a significant depletion (
Effect of 5FU on ovulation
In former research, corpus luteum quantification was performed in 5FU and saline treated mice, in order to distinguish whether 5FU treatment interfered with ovulation or not. The count of corpora lutea faced a considerable depletion in all 5FU-treated ovaries. 78 In the study of Lambouras et al. (2018), there were not any notable changes in the amount of corpora lutea between saline and 5FU-treated mice in 12-h-treated groups, however, outstandingly less corpora lutea were detected on the 7-th day after 5FU treatment in 5FU-treated ovaries, compared to controls 83 (Table 3).
The mechanism of 5FU-induced reproductive toxicity
Although the exact mechanism of 5FU-induced reproductive toxicity, in female, has not been completely revealed, there are a few reactions that could explain this phenomenon to some extent.
5FU induces cell apoptosis
5FU can terminate the proliferation of cancer cells and induce apoptosis. This is thought to be accompanied by mitochondrial state changes and increased caspase-9 activity. 86 Moreover, it has been reported that 5FU induces apoptosis through upregulation of some proapoptotic proteins, like FAS and Bax, and activation of caspase-3. 87 Furthermore, the 5FU-induced apoptosis was detected to be accompanied by DNA degradation and disruption of mitochondrial membrane capacity, and overexpression of p21 protein and caspase-8 and caspase-9. 88
5FU causes DNA and RNA damage
As mentioned previously, 5FU is a potent antimetabolite and performs its main inhibitory function through inhibition of thymidylate synthase (TS). However, enzymatic changes on the nucleotide (including ribosylation and phosphorylation) are needed to perform the cytostatic activity of 5FU.
89
Among all the derivatives of this chemotherapeutic agent, fluorodeoxyuridine monophosphate (FdUMP) is the most active one which creates a covalent ternary complex in combination with TS, thus, disrupting its ordinary activity by blocking the binding of its natural substrate.
90
5-fluorouridine-50 -triphosphate (FUTP) and 5-fluoro-20 -deoxyuridine-50 -triphosphate (FdUTP) are the other major metabolites of 5FU that are respectively inserted into RNA and DNA and lead to the disruption of RNA processing and function, and inhibition of DNA synthesis and function (Figure 3).90–92 Damage to DNA and RNA is aggravated by 5FU. Thymidylate synthase (TS) is inhibited by 5FU. The cytostatic action of 5FU depends on its enzymatic conversion to the nucleotide, including ribosylation and phosphorylation. Combining with TS, fluorodeoxyuridine monophosphate (FdUMP) forms a covalent ternary complex. The other two primary metabolites of 5FU are 5-fluorouridine-50-triphosphate (FUTP) and 5-fluoro-20-deoxyuridine-50-triphosphate (FdUTP), which respectively impair RNA and DNA production and function.
5FU disrupts cellular function by oxidative stress
Although, reactive oxygen species (ROS) have a fundamental role in main cellular activities, including signal transduction and bactericidal effect of phagocytes, apoptosis and cellular injury can result from their accumulation.93–95 Besides, a significant part of the chemotherapy-related oxidative damage has been reported to be played by an increase in ROS.96–98 5FU, as a widely used chemotherapeutic agent, can also produce mitochondrial ROS through the p53-dependent route, and accordingly, it can cause neoplastic cell death. 99 But, 5FU can also bring about some serious adverse events in which ROS overproduction and oxidative stress play a crucial role (Figure 3). 100 It could be predicted that these mechanisms might be of vital importance in 5FU-induced reproductive toxicity in females. Undoubtedly, future studies should aim to determine the uncertain mechanism behind this phenomenon.
Conclusion
Despite being widely used in various types of solid tumors, 5FU can result in severe forms of multi organ toxicities. Importantly, reproductive toxicity is one of the major complications that may limit the clinical application of this drug. In the current review, we explained and discussed 5FU-induced reproductive toxicity in an organ-based method. First, we explained 5FU-induced toxicity in male reproductive system, which, in summary, showed destruction of the seminiferous epithelial cells and Sertoli cells vacuolization. Although, 5FU does not cause any histopathological changes, it reduces the relative weight of the prostate. The chemotherapy agent also causes severe sperm abnormalities such as germ cells shedding, spermatid halo appearance, and polynucleated giant cells along with reduction in sperm count. 5FU-induced female reproductive toxicity is mainly characterized by morphologically atretic secondary and antral follicles, along with decrease in the number of growing follicles, ovarian weight and, a notable impairment in their maturity.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Correction (December 2023):
Article updated online to correct “5-flourouracil” to “5-fluorouracil” throughout the article.
Data Availability Statement
The data that supports the findings of this study are available from the corresponding author upon reasonable request.
