Abstract
The sexual maturity status of animals in nonclinical safety studies can have a significant impact on the microscopic assessment of the reproductive system, the interpretation of potential test article–related findings, and ultimately the assessment of potential risk to humans. However, the assessment and documentation of sexual maturity for animals in nonclinical safety studies is not conducted in a consistent manner across the pharmaceutical and chemical industries. The Scientific and Regulatory Policy Committee of the Society of Toxicologic Pathology convened an international working group of pathologists and nonclinical safety scientists with expertise in the reproductive system, pathology nomenclature, and Standard for Exchange of Nonclinical Data requirements. This article describes the best practices for documentation of the light microscopic assessment of sexual maturity in males and females for both rodent and nonrodent nonclinical safety studies. In addition, a review of the microscopic features of the immature, peripubertal, and mature male and female reproductive system and general considerations for study types and reporting are provided to aid the study pathologist tasked with documentation of sexual maturity.
Introduction
Microscopic evaluation of the reproductive system is a critical component of the assessment of the male and female reproductive end points in nonclinical safety studies (note 1). International regulatory guidelines on the conduct of these studies recommend a thorough histopathologic assessment of the reproductive system 1 –4 ; however, the age and reproductive status of animals in nonclinical safety studies can have a significant impact on the evaluation of the reproductive system, interpretation of potential test article–related findings, and ultimately the risk assessment. The age of animals used in nonclinical safety testing varies with study design, species, testing facility practices, and/or animal availability. Across and within studies, the toxicologic pathologist can be presented with a wide range of ages and differing stages of development of the reproductive system. This variability requires a thorough understanding of the normal anatomy, physiology, and histology of the reproductive system at different ages and in a variety of species.
In most general toxicity studies, rodents are sexually mature at the end of the study, while nonrodents may be sexually immature or a mix of immature, peripubertal, and/or mature. As a result, the need for documentation of sexual maturity status in the context of the study is most often encountered when working with nonrodents; however, documentation of reproductive maturity in rodents can also be helpful with juvenile toxicity studies and/or for early decedents before scheduled study termination. Assessment of sexual maturity status of animals within a study is often performed by the study pathologist based on the light microscopic appearance of the reproductive organs, but other study parameters including age, body weight, semen analysis, testis volume, estrous/menstrual cyclicity, and/or hormonal measurements may be included based on the study design and required end points.
Despite the importance of determining the maturity status for the purposes of interpretation of test article–related effects, maturity status is not documented in a consistent manner across the industry. 5 Changes in industry practices, such as increased numbers of juvenile toxicity studies, increased focus on endocrine disrupting agents, and establishment of International Harmonization of Nomenclature and Diagnostic Criteria terminology for histopathologic findings in rodents and key nonrodent species, has increased the awareness of the need for a consistent approach to the documentation of sexual maturity. In addition, with the increasing numbers of biopharmaceuticals and changes to biopharmaceutical regulatory guidance documents, 2 toxicologic pathologists are being asked to evaluate the reproductive system from mature nonhuman primates (NHPs) as a surrogate for more typical fertility-based reproductive toxicity studies, as well as evaluate very young animals for developmental effects in enhanced pre- and postnatal development studies. As a result of the changing regulatory landscape and efforts to increase transparency of study data, a consistent and reliable approach to document sexual maturity based on microscopic evaluation of the reproductive system is needed. Under the direction of the Scientific and Regulatory Policy Committee (SRPC) of the Society of Toxicologic Pathology (STP), the Sexual Maturity Working Group has developed a series of recommended best practices for the documentation of sexual maturity based on microscopic assessment of the reproductive system in nonclinical safety studies for rodent and nonrodent species. This was also reviewed and endorsed by the European Society of Toxicologic Pathology, the British Society of Toxicologic Pathology, and Société Française de Pathologie Toxicologique (the French Society of Toxicologic Pathology).
Scope
A complete microscopic assessment of the reproductive system is best achieved in sexually mature animals, and use of sexually mature animals is widely recommended throughout the literature when microscopic assessment of the reproductive system is needed. 1,6,7 However, this approach is not always practical (eg, limited availability of sexually mature NHPs since this species exhibits a relatively late age and variable onset of sexual maturity) or possible (eg, individual animals or dose groups terminated earlier than planned). Therefore, study data and reports should contain clear documentation of sexual maturity for all animals in any study where a microscopic assessment of the reproductive system is performed to provide context and highlight any challenges in interpretation of potential test article–related findings due to lack of maturity. In rodents and rabbits, age and/or body weight are suitable criteria to make a reasonably informed determination of the likelihood of sexual maturity in individual animals and/or dose groups as the duration of puberty is relatively short, there is limited interanimal variability, and there is suitable historical information available from the literature and suppliers to support this practice. 8 –11 In nonrodents, the use of age and/or body weight alone is not as reliable due to the higher interanimal variability in age and weight at the onset of puberty coupled with a longer peripubertal period. This limitation is less of an issue in dogs and minipigs, where animals are generally expected to be sexually mature at the end of a ≥6-month study or if animals several months older than the industry standard are used at the start of shorter duration studies. In contrast, age and body weight in NHPs are poor indicators of sexual maturity status due to the relatively long duration and variable onset of puberty; therefore, the assessment of sexual maturity in NHPs typically requires additional study end points. 12,13 This may occur during in-life assessment (eg, testicular volume measurement, semen analysis, menstrual cycle evaluation via vaginal swabs, and/or hormonal measurements) as a part of the baseline prestudy evaluation, during the study as an experimental end point, and/or as a part of the microscopic assessment by the study pathologist.
For general toxicity studies, microscopic assessment of reproductive tissues is the most commonly employed method for determining sexual maturity at the time of necropsy. While it is critical that all other available information regarding sexual maturity (body weight, age at necropsy, organ weights, etc) be assessed and used collectively as a weight of evidence approach, the scope of this best practices article is limited to microscopic determination of sexual maturity by the study pathologist in those nonclinical studies that have a microscopic assessment of the reproductive system.
Review of the Microscopic Assessment of Sexual Maturity in Nonclinical Safety Studies
The microscopic determination of sexual maturity requires a weight of evidence assessment of the entire reproductive system. Individual reproductive organs should not be evaluated in isolation. The microscopic assessment is the main end point used by the study pathologist to determine sexual maturity; however, ancillary data such as age, body weight, in-life observations, strain/genetic source, and macroscopic observations should also be considered as part of the weight of evidence.
There are considerable species, breed, and strain/genetic differences in the characteristics, timing of onset, and microscopic features of sexual development. The key features relevant to toxicologic pathology are provided for males (Tables 1 to 4) and females (Tables 5 to 8), and while additional details are beyond the scope of this review, the topic has been extensively described and reviewed elsewhere. 14 –63 The general microscopic features of sexual development are presented along with the basic characteristics of the developing reproductive system in Tables 1 (males) and 5 (females) and the key microscopic features for determination of sexual maturity of each of the major nonclinical species in Tables 2 to 4 (males) and in Tables 6 to 8 (females).
General Features of the Developing Male Reproductive System Across Nonclinical Species.
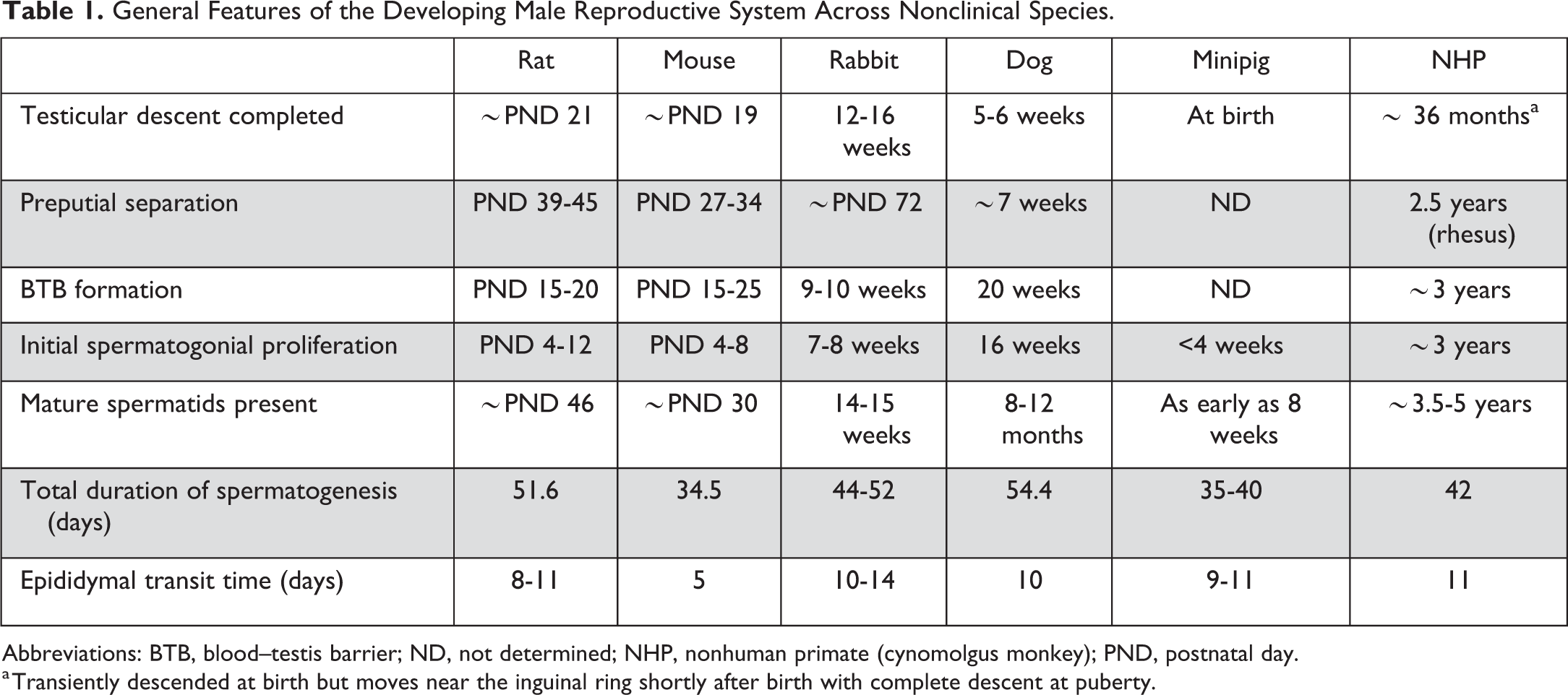
Abbreviations: BTB, blood–testis barrier; ND, not determined; NHP, nonhuman primate (cynomolgus monkey); PND, postnatal day.
a Transiently descended at birth but moves near the inguinal ring shortly after birth with complete descent at puberty.
General Microscopic Features of Immaturity in the Male Reproductive System Across Nonclinical Species.
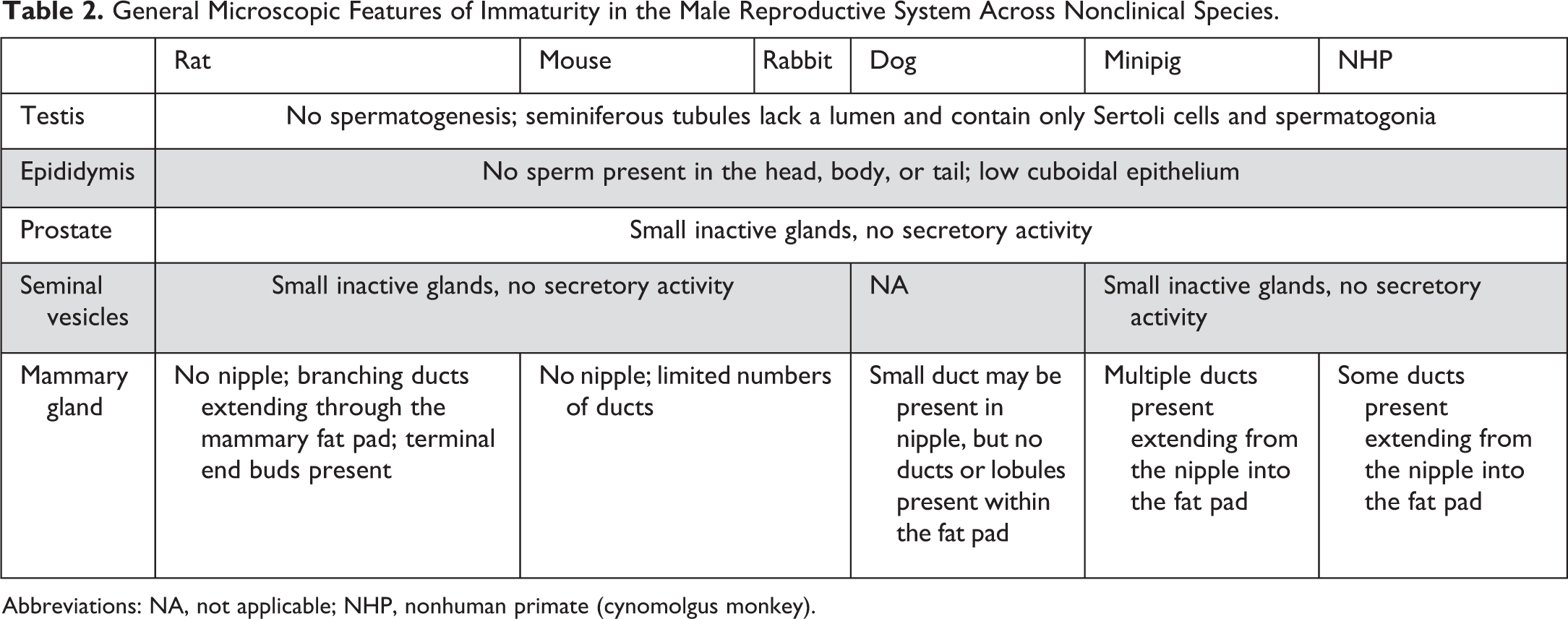
Abbreviations: NA, not applicable; NHP, nonhuman primate (cynomolgus monkey).
General Microscopic Features of the Peripubertal Period in the Male Reproductive System Across Nonclinical Species.
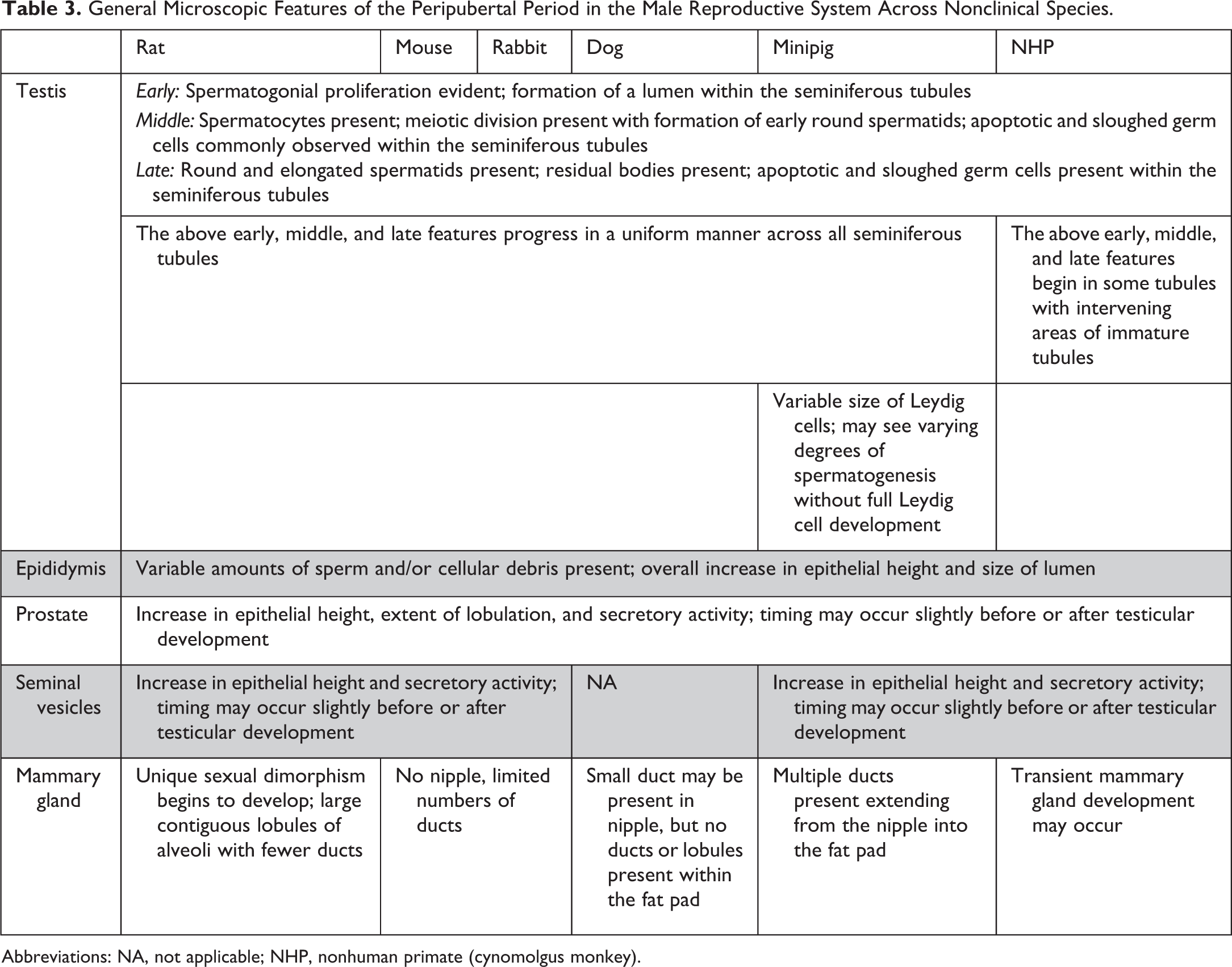
Abbreviations: NA, not applicable; NHP, nonhuman primate (cynomolgus monkey).
General Microscopic Features of Sexual Maturity in the Male Reproductive System Across Nonclinical Species.
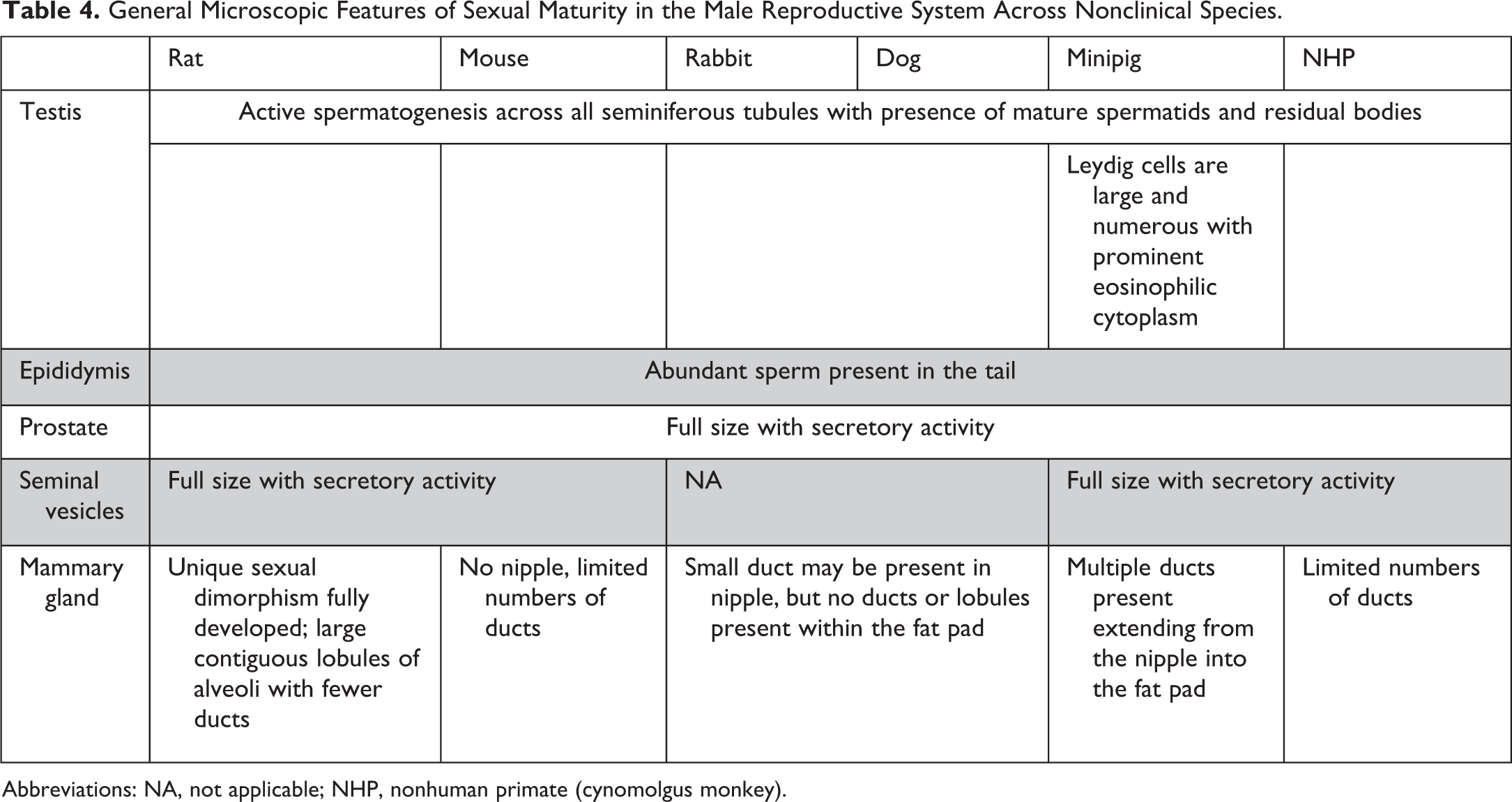
Abbreviations: NA, not applicable; NHP, nonhuman primate (cynomolgus monkey).
General Features of the Developing Estrous/Menstrual Cycle Across Nonclinical Species.
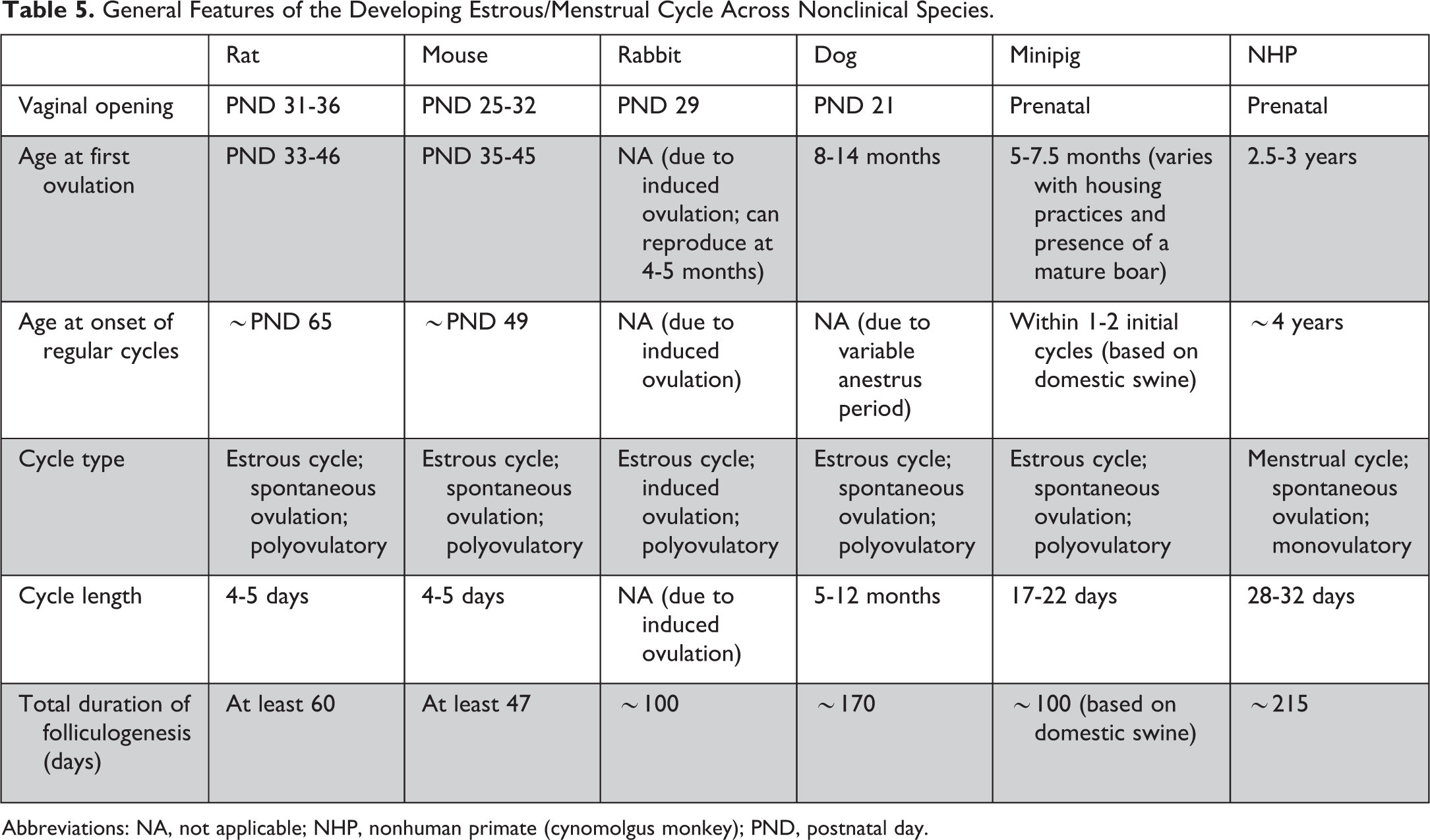
Abbreviations: NA, not applicable; NHP, nonhuman primate (cynomolgus monkey); PND, postnatal day.
General Microscopic Features of Immaturity in the Female Reproductive System Across Nonclinical Species.a
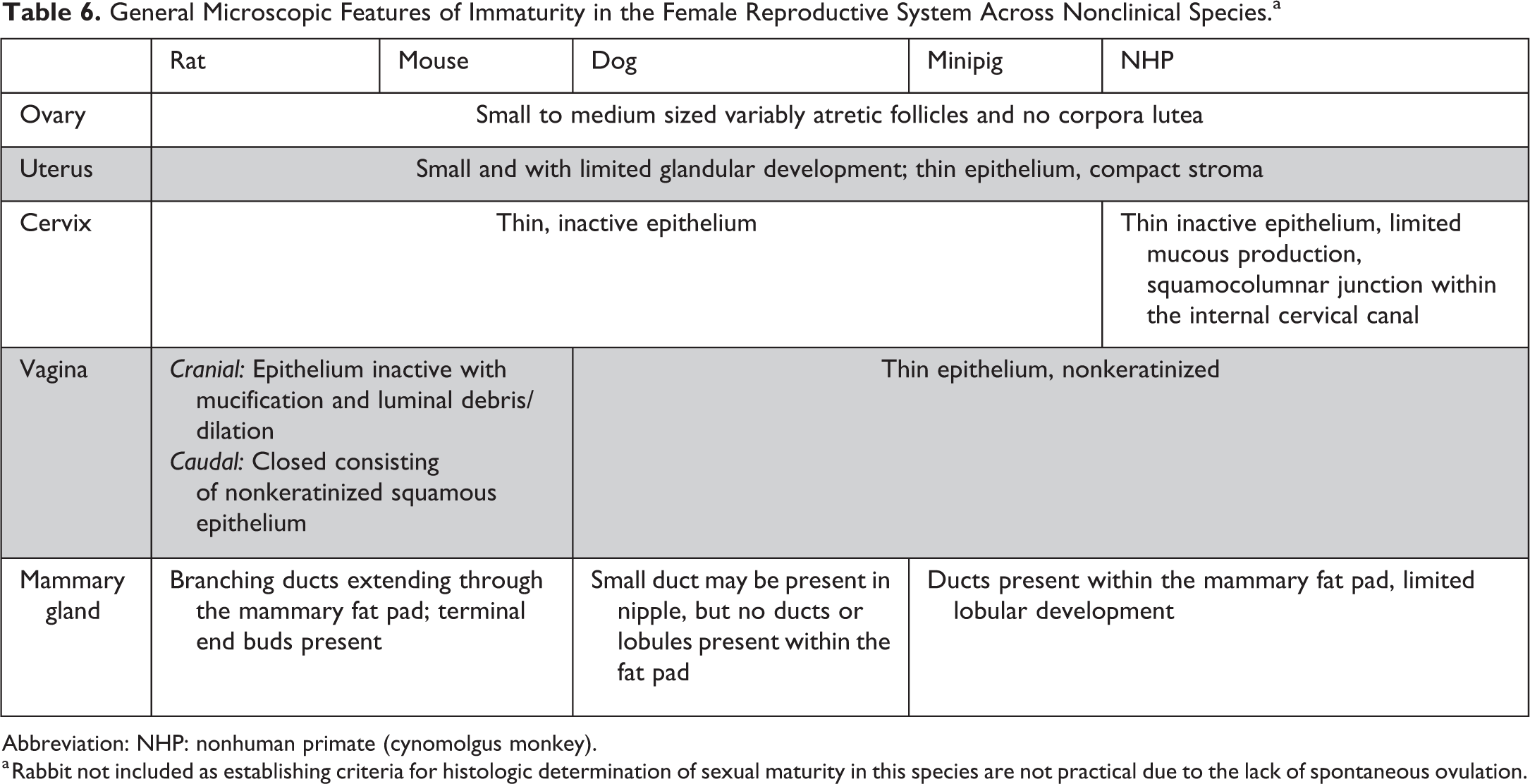
Abbreviation: NHP: nonhuman primate (cynomolgus monkey).
a Rabbit not included as establishing criteria for histologic determination of sexual maturity in this species are not practical due to the lack of spontaneous ovulation.
General Microscopic Features of the Peripubertal Period in the Female Reproductive System Across Nonclinical Species.a
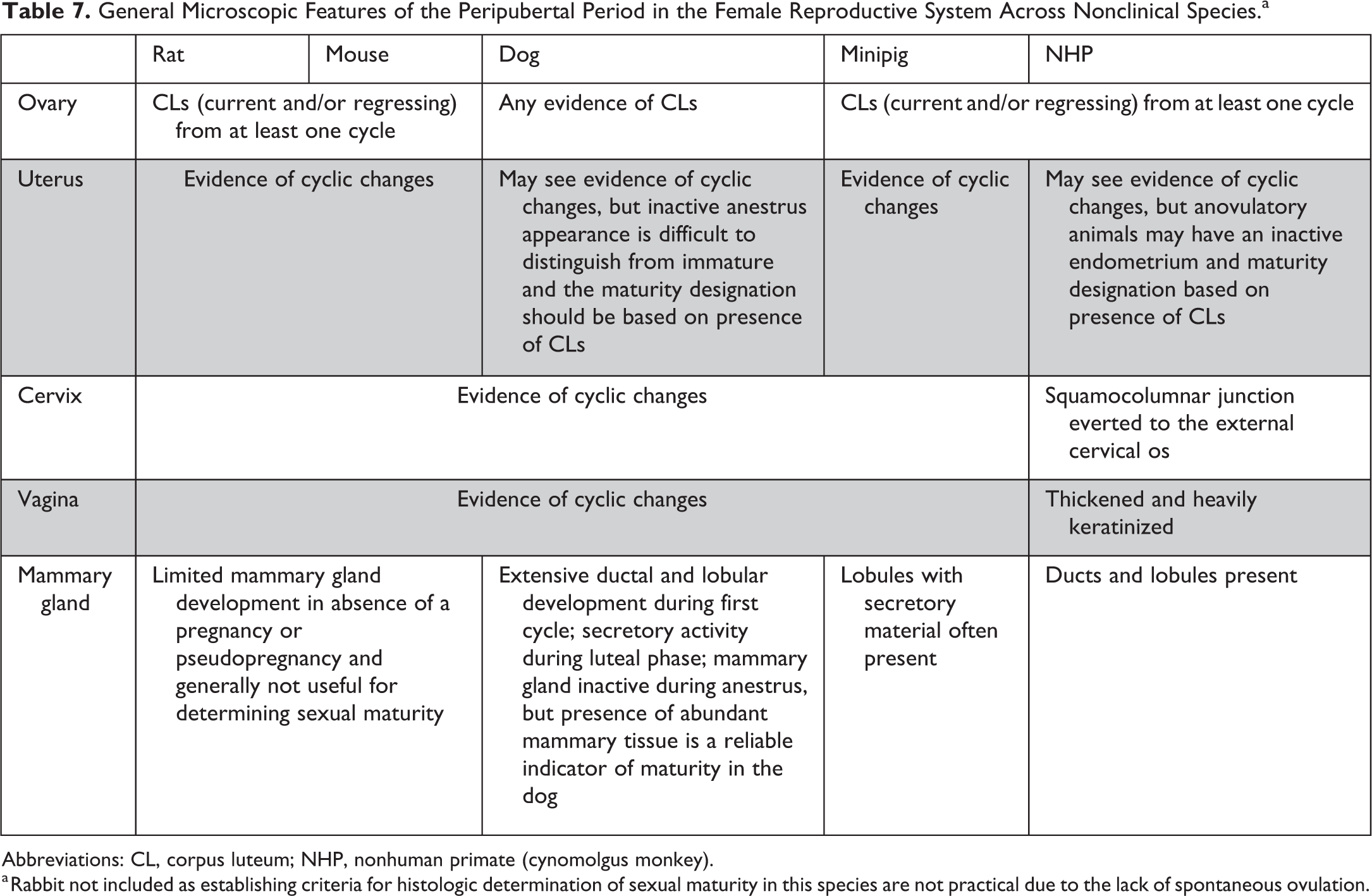
Abbreviations: CL, corpus luteum; NHP, nonhuman primate (cynomolgus monkey).
a Rabbit not included as establishing criteria for histologic determination of sexual maturity in this species are not practical due to the lack of spontaneous ovulation.
General Microscopic Features of Sexual Maturity in the Female Reproductive System Across Nonclinical Species.a
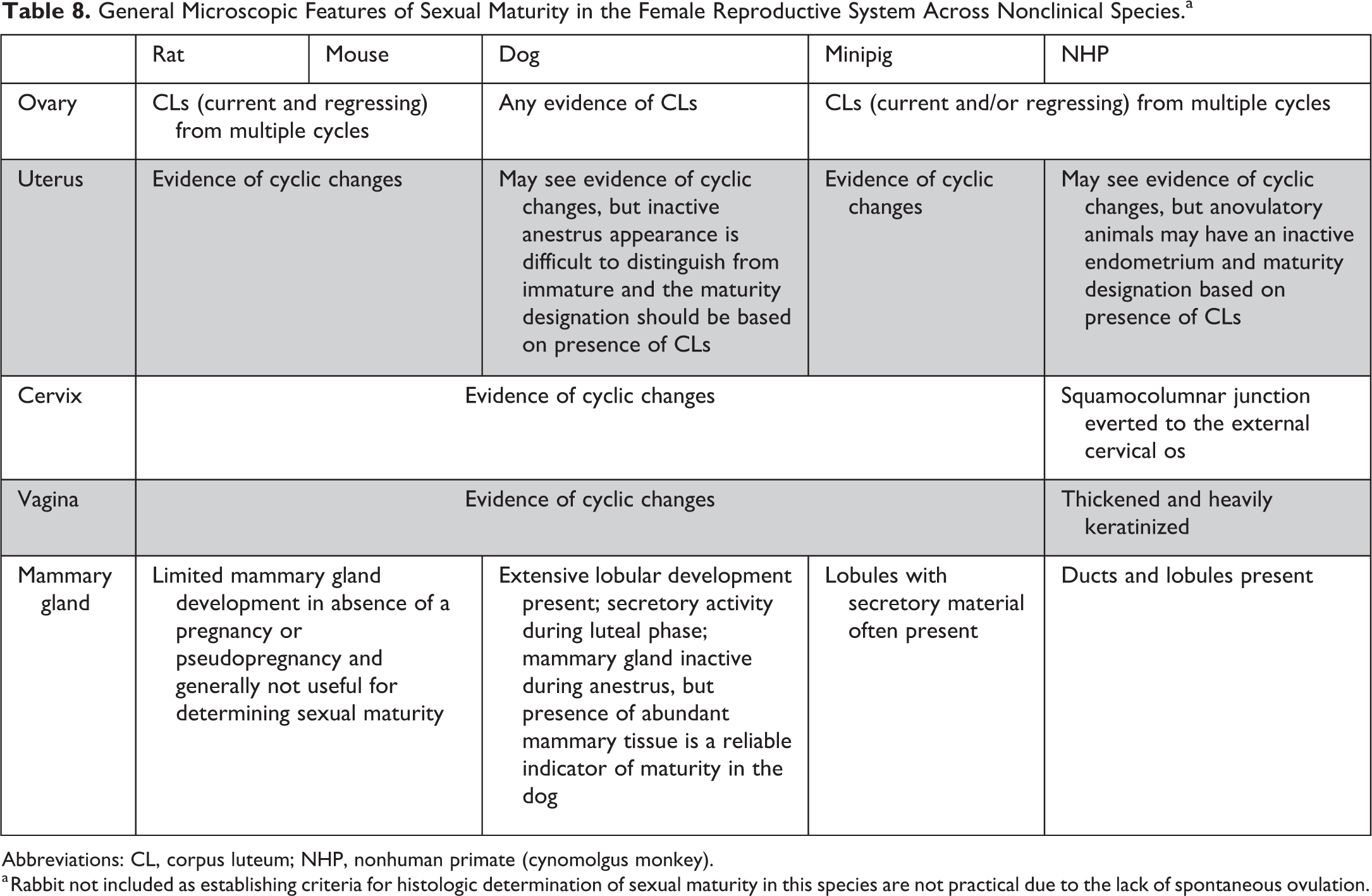
Abbreviations: CL, corpus luteum; NHP, nonhuman primate (cynomolgus monkey).
a Rabbit not included as establishing criteria for histologic determination of sexual maturity in this species are not practical due to the lack of spontaneous ovulation.
Males
Males are typically considered sexually immature until the onset of spermatogenesis, which is considered the beginning of the peripubertal period. At the start of the first wave of spermatogenesis, the pathologist can observe the beginnings of spermatogonial mitotic activity, the first emergence of early spermatocytes, and the formation of a lumen within the seminiferous tubules. This peripubertal period typically lasts until spermatogenesis is completed (presence of mature spermatids and residual bodies) across the majority of seminiferous tubules, and there is a full complement of sperm within the tail of the epididymis and the accessory sex glands are fully developed, at which time the male is considered sexually mature.
Females
Females are typically considered sexually immature when there is no microscopic evidence of ovulation (no corpora lutea) and no evidence of cyclical glandular/epithelial development of the reproductive tubular tract. 30 During the peripubertal period, the histologic features are not easily defined and can have overlap with both immature and mature animals and documentation of the peripubertal period may not be possible in all cases. There may be corpora lutea or remnants of corpora lutea and/or development of glandular and epithelial elements in the tubular tract, but these features can be variable and may not correlate well on an individual animal basis with physical hallmarks of sexual development such as vaginal opening or menarche. 30 Following the first ovulation, there is a variable period where estrous/menstrual cycles are irregular, which may complicate assessment of the reproductive tract. 10 This peripubertal period of cycle irregularity occurs in all species, but the length and types of changes encountered differ across species and individual animals.
In rats, the timing of vaginal opening generally coincides with the onset of estrous cyclicity with vaginal opening typically occurring during proestrus of the first cycle. 30 As a result, the peripubertal period based on microscopic findings is short, and following the second or third ovulation, the pathologist would not be able to discern a newly cycling young rat with irregular cycles from a more mature regularly cycling rat. 30 In mice, the time between vaginal opening and first estrus and the onset of regular cyclicity is inconsistent and may vary across strains or with husbandry practices. 64 –66
In female rabbits, the features described for other species do not apply. Female rabbits are induced ovulators and it is normal for nonmated rabbits to lack corpora lutea regardless of age. 67 While there is some development of the uterus and vagina as rabbits mature, without the presence of corpora lutea and/or clear histologic evidence of cyclicity, it is difficult to establish consistent criteria for microscopic determination of sexual maturity. Therefore, age and/or weight are the only available end points for determining sexual maturity in female rabbits.
In the dog, there is typically one set of corpora lutea present (whether active or regressing) that can be used to determine sexual maturity. This is due to the long estrous cycle with variable anestrous periods. While occasionally an additional set of remnant corpora lutea may be present, 36 the age of animals and study duration in standard nonclinical safety studies makes this unlikely.
In the minipig, the reported age of sexual maturity varies across references with differences observed between individual females and housing practices. 34,68 Female minipigs have often been considered sexually mature as early as 4 to 5 months of age, 69 but when considering the presence of corpora lutea in the ovary, the majority of minipigs would be considered to be mature at ≥6.5 months. 22,34 Differences in housing practices may contribute to this variability as absence of contact with mature boars may prolong the onset of sexual maturity.
In the NHP, the peripubertal period is especially variable and long where lengthy anovulatory periods are common. 70 Ideally, the key histologic features to establish that a given female is sexually mature include evidence of ovulation from multiple cycles (ideally corpora lutea from current and/or previous cycles) and full development of the reproductive tubular tract; however, recognition of both features is not always possible across all species.
Due to the lack of a clear distinction between the peripubertal period and sexual maturity in females, often the presence of any corpora lutea becomes the only clear determination of sexual maturity. 30 Therefore, for practical purposes in most nonclinical safety studies, it is recommended that females are considered immature when there are no corpora lutea present and sexually mature when there is clear evidence that ovulation has occurred and corpora lutea are present. It is recognized that there will be some animals that are considered sexually mature that are still likely in or toward the end of pubertal development. Additional features like mammary gland development may be helpful in nonrodents (especially in the dog where mammary gland development occurs during the first estrous cycle), 33,34,71 but because of the dynamic and often variable nature of the female reproductive cycle, evidence of ovulation remains the clearest microscopic end point for use in determining maturity of the endocrine axis and functionality of the female reproductive system.
Despite these challenges, pathologists will occasionally encounter females that are considered peripubertal where the reproductive system is neither immature nor definitively mature such as rats during the first estrous cycle, dogs in first proestrus, NHPs with evidence of endometrial bleeding but no corpora lutea present, and so on, but it is anticipated that the peripubertal window will not be identified as frequently in females as in males.
Review of Existing Practices for Sexual Maturity Documentation
Historically, documentation of sexual maturity by the study pathologist has been inconsistently performed across laboratories, species, and sex. 5 A frequently used method by study pathologists has been to document sexual maturity only when an animal was clearly immature, with the assumption that if not stated otherwise, the animal was mature. This approach may be acceptable when evaluating rodents, as age and/or body weight are a reliable indicator of sexual maturity, but in nonrodents, this can create uncertainty as a study with all mature animals will have nothing documented with regard to reproductive tract status, leaving downstream readers with no way to verify if the animals were mature or not. A further complication is that it has been more common to document sexual maturity (or immaturity) for males than for females, 5 likely due to general familiarity with the histopathologic features of immature testes and study-specific requirements for stage-aware evaluation of spermatogenesis. Even when laboratories clearly documented the different stages of reproductive development, the approach to how the data were recorded could differ. For example, some laboratories document sexual maturity status as a morphologic diagnosis under the gonad (eg, testis: immature, present). While this approach has the advantage of easily formatting results into tables and historical control data summaries, once maturity status is used as a morphologic diagnosis, it is no longer possible to simultaneously document the gonads as normal in most laboratory information management systems. It is also possible that a diagnosis of “immature” could be interpreted as the reproductive tract being abnormal or not suitable for evaluation even though assessment at a younger age may be part of the intended study design.
In an attempt to circumvent these issues, some labs instead place the maturity status as a tissue comment under the gonad (eg, testis [immature]: no abnormality detected). This allows the pathologist to clearly record the gonad as normal, but the ability to easily generate tabular and historical control data is lost. An additional challenge with placing these data as a morphologic diagnosis or as a tissue comment under gonad is that there are often comments or confusion from downstream readers as to why the gonad has a designation as “immature” or “mature,” but the other reproductive organs do not have a sexual maturity designation. Taken together, the inconsistent recording and varied documentation processes across the industry can create confusion and, in some instances, unnecessary reexamination of study data, prompting the need for a new, more transparent approach. The ideal approach would allow for documentation of sexual maturity status for the animal as a whole including all available reproductive organs, be easily tabulated and summarized in tables and historical control/reference data, allow the pathologist to record routine histopathology on individual reproductive organs as normal or abnormal, and fit within the framework of the Standard for Exchange of Nonclinical Data (SEND) and enable future data mining activities.
The Working Group Recommendation: Summary Tissue Approach for Sexual Maturity Documentation
Documentation of sexual maturity based on the microscopic evaluation of the reproductive organs differs from other microscopic data in 2 important ways. First, the documentation of sexual maturity is a recording of a normal feature of the tissue rather than an abnormality or test article–related change. As such, the data should be separate from the histopathology results but still allow for tabulation and/or querying. The second is that the assessment is often based on the evaluation and interpretation of multiple tissues within the reproductive tract. In order to document normal features of multiple tissues, use of a summary tissue in data capture systems is recommended. A summary tissue is an additional tissue that is added to the list of histopathology tissues in the data entry system to facilitate documentation, in this case, of the overall sexual maturity assessment across multiple tissues. The concept of a summary tissue has also been referred to as a “pseudotissue,” but since collection of data in this manner is still consistent with microscopic assessment of actual tissue, it is recommended to use the term “summary tissue.”
When documenting sexual maturity based on the microscopic assessment of the reproductive organs for males and females, the recommendation is to the use of “Reproductive Tissue, Maturity” or other similar terms as the summary tissue in the data entry system. This term would be included in addition to the full complement of individual reproductive organs and tissues expected as part of the routine microscopic evaluation of the male and female reproductive systems. The typical organs would be handled and diagnosed individually as usual with a designation of “normal” or use of an appropriate morphologic diagnosis for a finding. Following review of the individual reproductive tissues for maturity, one of the following terms to indicate the maturity status of the reproductive system as a whole would be entered for the summary tissue: immature, peripubertal, mature, or indeterminate. Although there may be some variability in how individual pathologists or laboratories define each of these terms across sexes and species, it is recommended that these terms be used without a severity grade or other descriptors. The terms “immature,” “peripubertal,” and “mature” are features of normal development and should not be used as morphologic diagnoses to indicate abnormalities in individual organs. Occasionally, macroscopic observations such as “small” or “reduced size” may be observed for individual reproductive organs due to normal sexual development. In these cases, it is recommended to microscopically diagnose the individual organ as “normal” with no microscopic correlate of the macroscopic finding and include the appropriate designation of “immature” or “peripubertal” in the summary tissue. If needed, additional information can be included in a comment under the individual reproductive organs and/or summary tissue. Any differences in incidence across dose groups or impact on organ weights can be discussed as needed in the pathology report. If there are significant test article–related or spontaneous findings in the reproductive system that preclude the pathologist from making a determination of sexual maturity, then “indeterminate” should be used. In times when “indeterminate” is used, it is anticipated that there will also be appropriate morphologic diagnoses for one or more of the individual reproductive organs.
Since the concept of a normal entry such as “within normal limits” or “no visible lesions” does not exist for maturity assessments, one of the 4 terms (immature, peripubertal, mature, or indeterminate) must be entered for this summary tissue. This is important for the pathologist to recognize during data entry as this may impact the study setup and/or use of a global designation to indicate all tissues as normal. The summary tissue should have different rules for entry and associated glossaries to prevent the inadvertent assignment of a normal entry. In addition, software vendors and SEND compliance teams will need to accommodate the handling of a summary tissue differently for SEND output and utilize a separate “test” for sexual maturity in the SEND Microscopic Findings data set that will differentiate them from the current test results for microscopic abnormalities.
The use of the summary tissue is the recommended default approach for any study requiring documentation of sexual maturity as part of a microscopic assessment of the reproductive system (see below for additional information on study recommendations). This approach allows for the determination of sexual maturity status to be representative of the animal as a whole after considering the microscopic appearance of all evaluated reproductive organs and should be included for all animals in a given study. This is critical during puberty where organs may not transition from immaturity to maturity at the same rate, 17,30,36 and designating each organ individually may give the false impression that there is a mismatch or abnormality. While this “summary tissue” approach allows for ease of entry and provides a clear, fit-for-purpose documentation of the sexual maturity status of the animal, it is recognized that there is no electronic traceability of the maturity status of the individual organs that were used to make this determination. If needed, comments about individual reproductive organ development and/or relationship to macroscopic observations can be included under the individual reproductive organs and/or summary tissue.
Study Types and Reporting
The assessment of sexual maturity and documentation using a summary tissue is an important component of a comprehensive microscopic assessment of the reproductive system. However, it is important to note that documentation of sexual maturity is not a requirement for all studies and inclusion of such data will vary with the goals of the study, program needs, international guidelines, regulatory requests, and so on. In addition, documentation of sexual maturity by the study pathologist is not needed for all studies. For example, microscopic assessment of the reproductive system in studies with breeding end points already requires use of sexually mature animals at the study start, so further documentation by microscopic assessment offers little value. A full accounting of all potential study types is not within the scope of this review and use of sexually mature animals is expected to vary with program and regulatory needs, but general guiding examples on when documentation of sexual maturity by the study pathologist may be helpful are provided in Table 9.
General Examples of Nonclinical Safety Study Types That May Benefit From Inclusion of Microscopic Determination and Documentation of Sexual Maturity.a
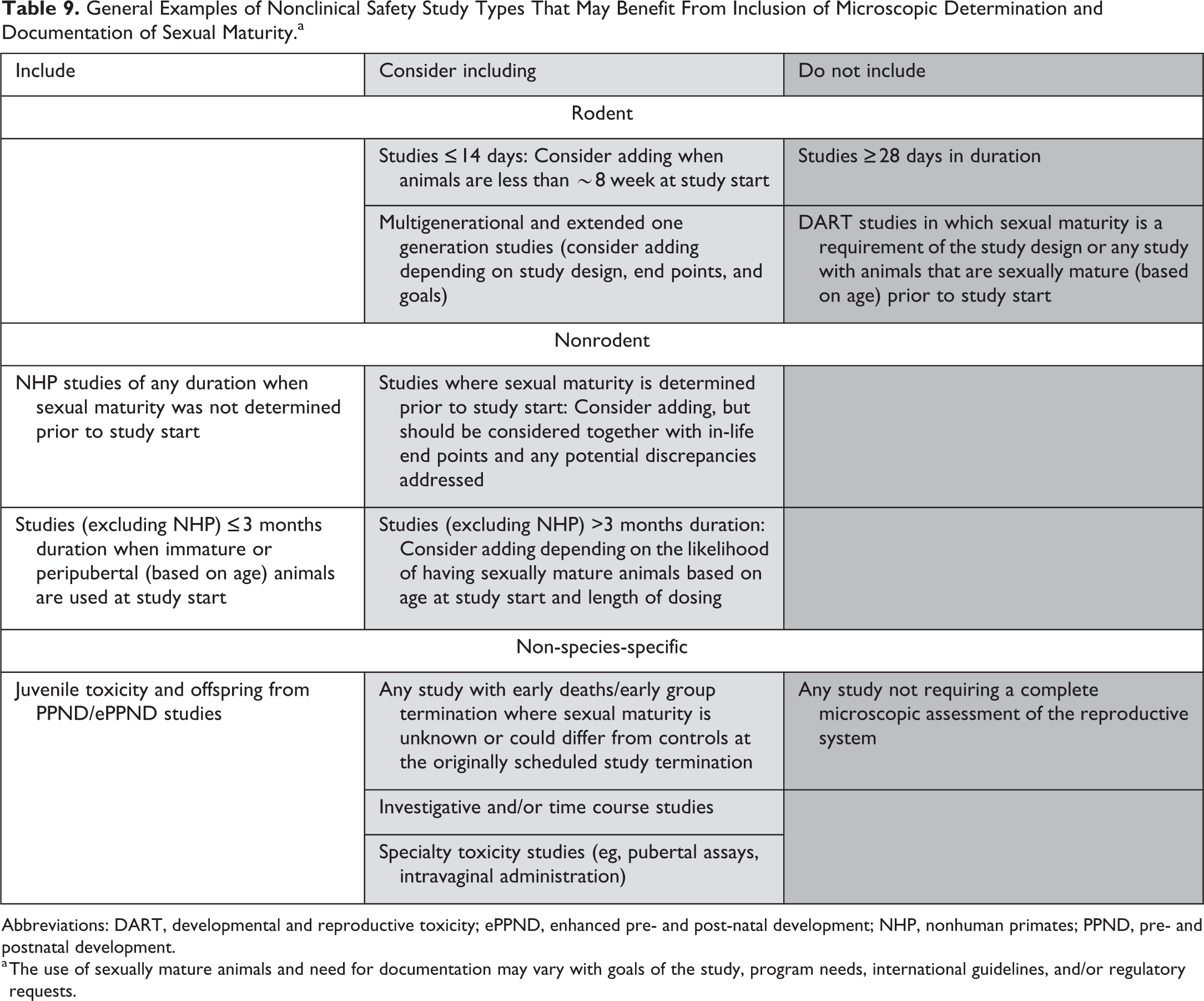
Abbreviations: DART, developmental and reproductive toxicity; ePPND, enhanced pre- and post-natal development; NHP, nonhuman primates; PPND, pre- and postnatal development.
a The use of sexually mature animals and need for documentation may vary with goals of the study, program needs, international guidelines, and/or regulatory requests.
Laboratories should assess their needs based on study types, species, and workflow and incorporate these practices into standard operating procedures (SOPs) and/or study protocols/plans as appropriate. While it is recognized that individual laboratories will require some degree of flexibility in use of SOPs and study protocols/plans, the Working Group recommends that enough detail be provided in study protocols/plans and reports so that toxicologists, study sponsors, clinicians, regulatory reviewers, and so on, can clearly understand if and/or how sexual maturity was determined and documented.
Since the assessment and documentation of sexual maturity is a characterization of normal developmental features, it is anticipated that in most studies there will not be a need to include a summary or description of the sexual maturity in the results section of the of the pathology report. In certain cases where the sexual maturity status of an animal or animals impacts the stated goals of the protocol and/or interpretation of the histopathology findings, additional detail regarding individual animal and/or group sexual maturity may need to be provided in the pathology report. Due to the potential combinations of study types, species, ages of animals, and so on, a list of specific situations or recommendations is not practical. However, the Working Group recommends that when required, the study pathologist provide the downstream reader with a clear understanding of how the sexual maturity status may have impacted the assessment. For example, in a study where stage aware evaluation of spermatogenesis is required, but not all animals were sexually mature due to test article nontolerability and early group termination, the report should state that the lack of complete spermatogenesis in early death animals may have impacted the ability to critically assess the testis as per protocol. It is recommended that any such discussion be focused and not overly detailed, for instance, “Males were terminated early and were considered peripubertal with incomplete spermatogenesis, which limited the microscopic assessment of the male reproductive system in this dose group.” When discussing such considerations in the pathology narrative, it is advisable to avoid phrases such as “Males and females were sexually immature, which precluded microscopic evaluation.” Statements such as these are misleading since there are some findings that can be readily detected in immature animals (eg, administration of estrogen receptor agonists 72 –74 ). In addition, such statements may give the appearance that the reproductive system was not evaluated due to the immaturity.
Conclusion
The SRPC of the STP convened an international working group to develop best practices for documentation of sexual maturity by microscopic assessment in males and females on nonclinical safety studies in rodent and nonrodent species. This was also reviewed and endorsed by the European Society of Toxicologic Pathology, the British Society of Toxicologic Pathology, and Société Française de Pathologie Toxicologique (the French Society of Toxicologic Pathology).
Best Practice Recommendations are as follows:
Microscopic assessment of reproductive tissues is the primary method for determining sexual maturity at the time of necropsy; however, ancillary data such as age, body weight, in-life observations, strain/genetic source, macroscopic observations, organ weights, and/or other nonroutine techniques (eg, semen analysis, testis volume, estrous/menstrual cyclicity, and/or hormonal measurements) should also be considered as part of the weight of evidence.
The microscopic determination of sexual maturity in males and females is based on the microscopic review of the reproductive system as a whole and individual reproductive organs should not be evaluated for maturity in isolation.
When documenting sexual maturity based on the microscopic assessment of the reproductive organs for males and females, it is recommended to use “Reproductive Tissue, Maturity” (or equivalent) as a summary tissue in the data entry system for all animals on the study with one of the following terms: immature, peripubertal, mature, or indeterminate.
In studies where sexual maturity is documented based on the microscopic assessment, most will not require a summary or description in the pathology report as documentation of sexual maturity status is a characterization of normal features and not a histopathologic diagnosis. In some cases, additional detail may be needed in the results and/or discussion section(s) of the pathology report explaining any potential impact of the maturity status on the ability to detect and/or interpret potential findings in the reproductive system.
Additional points to consider:
In studies requiring sexually mature animals, enough detail should be provided in study protocols/plans so that study scientists can clearly understand how sexual maturity will be determined and documented.
Study data and/or reports should contain clear documentation of sexual maturity either as age/body weight (rodents and rabbits), in-life determination prior to study start (nonrodents), and/or by microscopic determination (all species for both sexes except female rabbits) by the study pathologist in any study where a microscopic assessment of the reproductive system is part of the study design.
In rodents and rabbits, age and/or body weight are suitable criteria to make a reasonably informed determination of the likelihood of sexual maturity in individual animals and/or dose groups and microscopic documentation of sexual maturity in rodents and rabbits (males only) is generally not helpful in routine general toxicity studies but may be used as needed (eg, juvenile toxicity studies, early deaths/early group termination, etc.).
Female rabbits are induced ovulators and normally lack corpora lutea, so it is difficult to establish criteria for microscopic determination of sexual maturity. Therefore, age and body weight are the only available end point for determining sexual maturity female rabbits.
In dogs and minipigs, age and/or body weight can be helpful in establishing a likely determination of sexual maturity, but depending on the age at study start and time to necropsy, microscopic documentation of sexual maturity can be valuable.
In NHPs, the use of age and/or body weight alone are generally not reliable indicators of sexual maturity, so when sexually mature animals are required, confirmation of sexual maturity should be employed as a part of the baseline prestudy evaluation and/or as a part of the microscopic assessment by the study pathologist.
Footnotes
Authors’ Note
All authors contributed to conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The recommendations in this article are those of the authors and should not be construed to represent the policies, positions, or opinions of the Food and Drug Administration or their employers.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
