Abstract
Airborne polycyclic aromatic hydrocarbon (PAH) exposure can adversely affect human health by generating reactive oxygen species (ROS) and increasing oxidative stress, which causes changes in mitochondrial DNA copy number (mtDNAcn), a key indicator of mitochondrial damage and dysfunction. This study aimed to determine the effects of atmospheric benzo[a]pyrene (BaP) and 1-nitropyrene (1-NP) exposure on mtDNAcn in humans. One hundred and eight adults living in Cheongju, South Korea, were included in this study. Atmospheric BaP and 1-NP concentrations and urinary 6-hydroxy-1-nitropyrene (6-OHNP), N-acetyl-1-aminopyrene (1-NAAP), and 1-hydroxypyrene concentrations were measured. Blood samples were also collected to assess mtDNAcn. The mean mtDNAcn was 9.74 (SD 4.46). mtDNAcn decreased significantly with age but was not significantly associated with sex, sampling season, or smoking habit. While there was a borderline significant increase in mtDNAcn with increasing ambient total PAH levels, ambient PAH or urinary 1-hydroxypyrene concentrations showed no significant association with mtDNAcn. However, urinary 6-OHNP or 1-NAAP concentrations, 1-NP metabolites, were significantly associated with mtDNAcn. These results suggest that the metabolism of absorbed NPs generates excess ROS, which damages mitochondrial DNA, resulting in increased mtDNAcn.
Keywords
Introduction
Polycyclic aromatic hydrocarbons (PAHs) are compounds consisting of two or more fused benzene rings formed by the incomplete combustion or pyrolysis of organic matter. Metabolic activation of PAHs generates reactive oxygen species (ROS) that contribute to the formation of significantly elevated levels of oxidized guanine in nuclear DNA. 1 Benzo[a]pyrene (BaP) serves as a prototypical PAH and has been associated with adverse effects on neurobehavioral function in adults and neurodevelopment in children due to environmental or occupational exposure.2,3 In particular, the International Agency for Research on Cancer (IARC) has classified BaP as a human carcinogen. 4 Once absorbed into the body, BaP triggers the activation of cytochrome P450 1A1, leading to its conversion to benzo[a]pyrene-7,8-diol-9,10-epoxide (BPDE), 5 which in turn generates an excess of ROS during its metabolism. 6
Diesel exhaust particles (DEPs) are respirable particulate matter characterized by their small particle size and large surface area. 7 This unique structure allows for the adsorption of various substances, including PAHs and nitro-PAHs. Prolonged exposure to elevated levels of diesel exhaust (DE) has been associated with a range of adverse health outcomes, including asthma, respiratory and cardiovascular diseases, allergic sensitization, and even lung cancer.8–10 In 2012, the IARC classified DE as a Group 1 human carcinogen, citing studies that found an increased risk of lung cancer associated with DE exposure.9,11 Researches have shown that 1-nitropyrene (1-NP) contributes to genetic mutations and chromosomal aberrations,12,13 leading IARC to classify it as a Group 2A chemical. 11
Particulate matter, a major air pollutant, contains several PAHs and is found in higher concentrations in South Korea, especially in winter when it is carried across the border by the northwest wind. Cheongju, located in the center of the Korean peninsula, has one of the highest concentrations of air particulate matter owing to its large industrial complexes and three waste incinerators. 14
Mitochondria are susceptible to damage due to their lack of protective histones and reduced ability to repair DNA, and they compensate for damage by replicating mitochondrial DNA (mtDNA) to increase the cell's mtDNA copy number (mtDNAcn).1,15 mtDNAcn analysis provides an integrated assessment of the toxic effects of environmental contaminants, including the effects of unidentified substances and possible interactions between individual contaminants. 16 A correlation between mtDNAcn and oxygen consumption rate has been demonstrated, 17 and studies in humans and animals have confirmed that high absolute levels of wild-type mtDNA genomes can actually counteract the deleterious effects of pathogenic mtDNA mutations.18,19
The ability of PAH to cause damage is even more pronounced in mtDNA, with an affinity for mtDNA that is 40- to 90-fold greater than that for nuclear DNA.20,21 Metabolic activation of PAHs generates ROS that can potentially interfere with electron chain transport, damage mtDNA, and contribute to the formation of significantly elevated levels of oxidized guanine in mtDNA. 22
Given these facts, inhaling airborne particulate matter can expose individuals to harmful PAHs such as BaP and 1-NP. The metabolism of these PAHs can generate ROS, which in turn can lead to changes in mtDNAcn. This study was conducted to investigate the effects of BaP and 1-NP exposure on mtDNAcn in individuals living in Cheongju, South Korea.
Methods
Study subjects
This study was reviewed and approved by the Institutional Review Board of Chungbuk National University (IRB approval number: CBNU-201708-SDBR-0075, CBNU-202010-HRBR-0163). A total of 114 subjects were recruited, including farmers, homemakers, yogurt drink delivery and sales workers, intercity bus terminal workers, city cleaners, and white-collar workers living in Cheongju. Of these, six subjects were excluded from the final analysis because their blood was not collected; therefore, data from 108 subjects were included in the statistical analysis. After receiving detailed information about the study, subjects signed informed consent forms, provided urine the morning after air sampling, and had blood drawn. The interviews were conducted by an interviewer using a questionnaire on demographic characteristics, work environment, smoking habits, and diesel vehicle use.
Measurement of airborne PAH concentrations
Sampling of air and biological material was conducted during summer or winter from November 2017 to August 2021, with sampling seasons categorized as winter (November to February) and summer (June to August). Air sampling was performed using a personal air sampler (Apex Standard; Casella CEL, Bedford, England) at a flow rate of 3 L/min for 24 h. Subjects wore the sampler with a filter holder attached to their collar. A PTFE filter (Teflo; Pall Corporation, Ann Arbor, MI, USA) with a 2-μm pore size was used for sampling. After collection, the filters were placed in a flask and mixed with 2 mL of dichloromethane. The contents of the flask were shaken and subjected to ultrasonic waves to extract PAHs and nitro-PAHs. The resulting extracts were evaporated to dryness and then redissolved in acetonitrile.
To quantify PAHs and nitro-PAHs, aliquots of the solution were injected into a two-dimensional high-performance liquid chromatography (HPLC) system equipped with a fluorescence detector (FD). 23 The injected sample was first passed through a clean-up column (Cosmosil, 5NPE, 150 × 4.6 mm i.d., 5 μm; Nacalai Tesque, Kyoto, Japan) with its guard column (10 × 4.6 mm i.d.), and then nitro-PAHs were reduced to their amino derivatives using a reduction column (NPpak-RS, 10 × 4.6 mm i.d.; JASCO, Tokyo, Japan) at 80°C. The mobile phase for the clean-up and reduction columns consisted of ethanol/acetate buffer (pH 5.5) (95/5, v/v) at a flow rate of 0.2 mL/min.
A fraction of the amino derivatives and unchanged PAHs eluted with the mobile phase from the reduction column was mixed with 30 mM ascorbic acid at a flow rate of 1.6 mL/min and then collected using a concentration column (Spheri-5 RP-18, 30 × 4.6 mm i.d., 5 μm; PerkinElmer, Waltham, MA, USA). The concentrated fraction was further separated using two separation columns (Inertsil ODS-P, 250 × 4.6 mm i.d., 5 μm; GL Sciences, Tokyo, Japan) with their guard column (10 × 4.6 mm i.d.) in tandem. All columns, except the reduction column, were maintained at 20°C. The columns were subjected to a programmed gradient elution using 10 mM imidazole buffer (pH 7.6) as eluent A and acetonitrile as eluent B. Finally, the separated analytes were detected using a two-channel FD at their optimal excitation and emission wavelengths.
Ten PAHs, including fluoranthene (Flu), pyrene (Pyr), benz[a]anthracene (BaA), chrysene (Chr), benzo[b]fluoranthene (BbF), benzo[k]fluoranthene (BkF), benzo[a]pyrene (BaP), benzo[ghi]perfluorine (BpF), dibenz[a,h]anthracene (DBA), benzo[ghi]perylene (BghiP), and indeno[1,2,3-cd]pyrene (IDP) were measured as analytes in this study. In addition, 18 nitro-PAHs were measured, including 1,3-, 1,6-, and 1,8-dinitropyrene (1,3-, 1,6-, and 1,8-DNP); 2-nitrofluorene (2-NF); 9-nitrophenanthrene (9-NPh); two- and 9-nitroanthracene (2- and 9-NA); 1-, 2-, and 3-nitrofluoranthrene (1, 2, and 3-NFR); 1, 2, and 4-nitropyrene (1, 2, and 4-NP); 7-nitrobenz[a]anthracene (7-NBaA); 6-nitrochrysene (6-NC); 6-nitrobenz[a]pyrene (6-NBaP); 1-nitroperylene (1-NPer); and 3-nitroperylene (3-NPer).
Determination of urinary 1-hydroxypyrene concentration
For analysis, 45 mL of urine was mixed with 4.5 mL of 1 M sodium acetate buffer (pH 5.0). Beta-glucuronidase/aryl sulfatase (450 µL) from Helix pomatia (type H-2: β-glucuronidase activity, 100,000 units/mL; sulfatase activity, 7500 units/mL) was added to the mixture. In addition, 50 mg of blue rayon was added. The resulting solution was then incubated at 37°C for 16 h. After incubation, the blue rayon was removed, washed with water, and eluted with 2 mL of methanol, followed by 2 mL of methanol +1% NH3 solution. Thereafter, the eluate was evaporated to dryness using N2 gas and redissolved in 400 µL of methanol. For analysis, 50 µL of this eluate was used to determine the concentration of 1-hydroxypyrene (1-OHP), while the remaining eluate was used to determine the concentrations of 6-hydroxy-1-nitropyrene (6-OHNP) and N-acetyl-1-aminopyrene (1-NAAP).
The determination of 1-OHP concentration was performed using an HPLC system (LC-20AD; Shimadzu, Kyoto, Japan) equipped with a 250 × 4.6 mm reversed-phase column (J'sphere ODS-H80; YMC, Kyoto, Japan) and an FD (RF-20A; Shimadzu). The mobile phase was 65% acetonitrile, and the flow rate was set at 1 mL/min. The excitation/emission wavelength of the FD was set at 242/388 nm.
Measurement of urinary concentrations of 1-NP metabolites
The remaining blue rayon eluate was subjected to a nitro-reduction reaction by passage through an on-line reduction column (NPpak-RS). The same HPLC system, column, and mobile phase used for measuring 1-OHP concentrations were used to measure 6-hydroxy-1-aminopyrene (6-OHAP) concentrations. The FD was set to an excitation/emission wavelength of 285/428 nm.
After the nitro-reduction reaction, an amidation reaction was performed for 1 h at room temperature by adding 200 µL of acetic anhydride to the nitro-reduced fraction. We used an amidation method to convert 1-aminopyrene (1-AP) to 1-NAAP, which means that the 1-NAAP concentration measured in this study represents the combined concentration of 1-AP and 1-NAAP.
The measurement of 1-NAAP concentration was performed using the same HPLC system, column, and mobile phase as those used to measure 1-OHP concentration. The FD was set at an excitation/emission wavelength of 273/385 nm. 24
Determination of the copy number of mitochondrial DNA in blood cells
DNA was extracted from 200 μL blood using a QIAamp DNA Blood Mini Kit (Qiagen, Hilden, Germany) according to the manufacturer's instructions.
mtDNAcn was determined using a quantitative PCR method by real-time PCR, as described by Wu et al. (2019). 25 One hundred nanograms of the DNA was used as the template. Primers specific for mitochondrial genes were used: ND1 forward primer (5′-cctagccgtttactcaatcct-3′), ND1 reverse primer (5′-tgatggctagggtgacttcat-3′), ND2 forward primer (5′-aacccgtcatctactctaccatct-3′), and ND2 reverse primer (5′-gcttctgtggaacgagggtttatt-3′). In addition, 18s rRNA primers were included for reference: 18s rRNA forward primer (5′-cttagagggacaagtggcgttc-3′) and 18s rRNA reverse primer (5′-cgctgagccagtcagtgtag-3′).
Accupower 2X GreenStar qPCR Master Mix (Bioneer, Daejeon, Korea) and DEPC-DW (Bioneer) were combined to give a final volume of 50 μL for the PCR reaction. Real-time PCR was performed using a CFX Opus Real-Time PCR System (Bio-Rad Laboratories, Hercules, CA, USA). PCR cycling conditions were as follows: initial denaturation at 95°C for 5 min, followed by 40 cycles of 95°C (15 s), 54°C (20 s), and 72°C (20 s) and denaturation at 95°C for 15 s. Both mtDNA and 18s rRNA, CT values were obtained from the real-time PCR software, and the following formula was used to determine ND1 mtDNA and ND2 mtDNA content relative to 18s rRNA DNA.: (a) ΔCT = (18s rRNA CT – mtDNA CT) (b) Relative mitochondrial DNA content = 2 × 2ΔCT
The relative content of ND1 mtDNA and ND2 mtDNA was calculated, and the mean value was used as the mtDNAcn value. 26
Statistical analysis
To avoid skewing the results of statistical analysis by extreme values, measurements larger than the mean plus three standard deviations were replaced with values equivalent to the mean plus three standard deviations. Correlation analysis was performed to determine the correlations of the exposure levels of total PAH, total nitro-PAH, BaP, 1-NP, and their corresponding urinary metabolite concentrations with mtDNAcn.
A general linear model (GLM) was used to statistically analyze the effects of BaP or 1-NP exposure on mtDNAcn while controlling for potential confounding factors such as age, sex, and smoking habits.
SAS OnDemand (SAS Institute, Cary, NC, USA) was used for statistical analyses. A p-value less than 0.05 was considered statistically significant, indicating a high probability that the observed associations were not due to chance.
Results
General characteristics of the study participants.
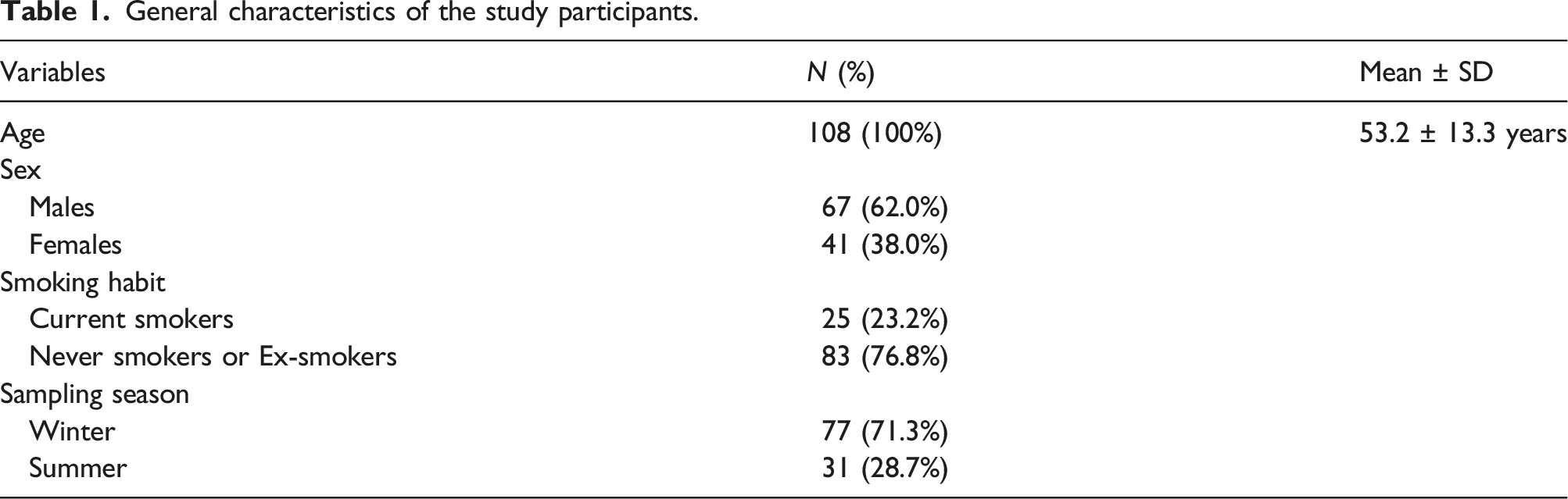
General characteristics of the study participants.

The concentration of 1-OHP in the urine of the participants was 16.1- and 166.5-fold that of 6-OHNP and 1-NAAP in urine.
Figure 1 shows the correlation of age, atmospheric PAHs, and their urinary metabolites with mtDNAcn. Correlations of age (a), atmospheric total PAHs (b), benzo[a]pyrene (c), total nitro-PAHs (d), 1-nitropyrene concentrations (e), urinary 1-hydroxypyrene (f), 6-hydroxy-1-nitropyrene (g), and N-acetyl-1-aminopyrene concentrations (h) with copy number of mitochondrial DNA.
As age increased, the value of mtDNAcn decreased significantly. (p-value <.0001) Total PAHs and nitro-PAHs and airborne BaP and 1-NP concentrations were not significantly correlated with mtDNAcn. However, leukocyte mtDNAcn increased significantly as urinary 1-OHP, 6-NP, and 1-NAAP concentrations increased.
General linear model for mitochondrial DNA copy number.

aControlled for age, sex, season, smoking habit; bbold values: P -value < 0.05; 1-OHP, 1-hydroxypyrene; 6-OHNP, 6-hydroxy-1-nitropyrene; 1-NAAP, N-acetyl-1-nitropyrene.
mtDNAcn tended to increase significantly with increasing urinary concentrations of 6-OHNP or 1-NAAP. The beta values for the mtDNAcn of 6-OHNP and 1-NAAP were 0.683 and 5.438 per ng/g creatinine, approximately 122- and 969-fold that of 1-OHP, respectively.
Pearson correlation coefficients of mitochondrial DNA copy number and exposure markers with age.

p < .05, **p < .01.; 1-OHP, 1-hydroxypyrene; 6-OHNP, 6-hydroxy-1-nitropyrene; 1-NAAP, N-acetyl-1-nitropyrene.
In winter, 1-NP exposure decreased significantly with increasing age, while total airborne nitro-PAHs decreased with borderline significance (p = .061). Urinary 1-OHP, 6-OHNP, and 1-NAAP concentrations also decreased significantly with increasing age. Age significantly and negatively correlated with mtDNAcn. However, no exposure biomarker significantly correlated with age in summer. Nevertheless, a significant negative correlation between age and mtDNAcn was observed in summer.
Discussion
In a study of Danish individuals, Mengel-From et al. (2014) reported that mtDNAcn did not change significantly before the age of 50. However, after the age of 50, mtDNAcn decreased by 0.54 for each additional year of age, and individuals with lower mtDNAcn had worse outcomes in cognitive performance, physical strength, self-rated health, and all-cause mortality compared with those with higher mtDNAcn even after adjusting for age as a potential confounding factor. 27 The present study similarly showed a significant decrease in mtDNAcn with increasing age, but the beta was −0.149, which is 28% of that of the Danish study. This is because the Danish study targeted a different gene than ours, and their mean was 47.37, which is approximately 5-fold the mean of our study (9.74).
Kim et al. (2014) reported a general increase in mtDNAcn after exposure to PAHs in a dose- and time-dependent manner. 28 These results suggest that a loss of the ability to compensate for high levels of oxidative stress was induced by high PAH concentrations. Pavanello et al. (2013) found that workers exposed to PAHs above 3 μmol 1-OHP/mol creatinine had significantly higher mtDNAcn in peripheral blood leukocytes compared with that in the controls, and mtDNAcn also positively correlated with urinary 1-OHP concentrations and the amount of anti-BPDE DNA adducts in nuclear DNA. 29 In our study, 1-OHP and mtDNAcn were not significantly related. This difference in significance is due to the difference between our study and that of Pavanello et al. (2013). The latter focused on the industrial PAH exposure of workers, whereas our investigation focused on individuals exposed to environmental PAHs. The average urinary 1-OHP concentration among the participants in our study was 0.02 µg/g creatinine (0.0104 µmol/mol creatinine), which is exceedingly low relative to the values in the study by Pavanello et al. (median value of the coke-oven workers: 3 μmol 1-OHP/mol creatinine). Furthermore, none of our participants were exposed to PAHs above a concentration of 3 μmol 1-OHP/mol creatinine. If the urinary concentration of anti-BPDE, a specific exposure marker for BaP, were measured in this study, a significant association would likely be observed.
The BaP concentrations in the current study are similar to those in a Croatian study (cold period: 0.057–1.526 ng/m3; warm period: 0.009–0.111 ng/m3), 30 but lower than the average value of 2 ng/m for airborne BaP in urban and suburban areas in the Czech Republic from 2010 to 2018. 31 The average airborne BaP concentrations in three Chinese cities—Beijing, Jinan, and Shanghai—measured in the winter of 2014–2015 were 13.3, 13.5, and 1.8 ng/m, respectively; our results were much lower. 32
An investigation conducted in Kanazawa, Japan, in 1994 documented atmospheric 1-NP levels of 32 pg/m3, a significantly lower concentration than we observed in our current study. 33 In contrast, urban areas in China showed significantly elevated 1-NP levels compared with those in our study. 34 For example, Shenyang, China, recorded approximate 1-NP levels of 80 pg/m3 in 2010. 35
While acknowledging that no single analyte can perfectly capture the intricate and variable composition of diesel particulate matter in all scenarios, measuring 1-NP concentrations could be a potentially accurate measure of DE exposure. In addition, such an approach may provide a more accurate representation of the carcinogenic potential of DE. 36 Measurement of the urinary concentrations of 1-NP metabolites, including 6-OHNP, 1-aminopyrene (1-AP), and 1-NAAP, is a valuable strategy for assessing environmental exposure to both 1-NP and DEPs.
1-NP or total nitro-PAH exposure and mtDNAcn were not significantly associated, but urinary 6-OHNP or 1-NAAP concentrations showed a significant positive correlation with mtDNAcn. These results suggest that exposure to 1-NPs by itself does not increase ROS, but 1-NP metabolism leads to ROS production. In particular, the beta value for urinary 1-NAAP concentration was much larger than that for urinary 6-OHNP concentration, suggesting that more ROS are generated when 1-NP is metabolized to 1-NAAP than when it is metabolized to 6-OHNP. In the present study, the average 1-NP concentration in the air significantly affected mtDNAcn, even though it was only 1/48 of the average BaP concentration in the air. The finding that mtDNAcn was increased by such a low level of environmental exposure suggests that the levels of ROS generated by the metabolic activation of 1-NP are much greater than those generated by the metabolic activation of other PAHs. To our knowledge, this is the first study to investigate the association between 1-NP exposure and mtDNAcn.
The correlation analysis of age and exposure markers showed significant negative correlations between age and urinary exposure markers in winter but not in summer. This may be due to decreased activity with age, especially in winter, when older adults are less likely to be outdoors, reducing their exposure to particulate matter and thus their exposure to PAHs and nitro-PAHs. Urinary 6-OHNP and 1-NAAP concentrations were significantly associated with mtDNAcn even after adjusting for age and seasonal effects, suggesting that these two exposure biomarkers are independent determinants of mtDNAcn.
Kim et al. (2014) reported that PAH exposure in cells was closely associated with increased mtDNA copy number, and that Lamin-A/C isoform 3, talin-1, and annexin A1 were universal biomarkers of PAH exposure. 28 Consistent with this rationale, the simultaneous assessment of biomarkers of toxicant exposure and mtDNAcn provides a robust means to measure the escalation of exposure to a specific toxicant and to detect changes in mtDNAcn in response to increased exposure. For example, simultaneous quantification of mtDNAcn in peripheral blood cells along with urinary concentrations of BPDE and 6-OHNP can be used to determine whether exposure to BaP or 1-NP has increased and to identify which substance exerts a substantial influence to induce a shift in mtDNAcn.
Individuals with higher levels of baseline mtDNAcn have a higher risk of developing lung cancer.37,38 In addition, alterations in mtDNAcn are associated with impaired apoptosis and subsequent increased cellular proliferation.39 Therefore, the results of this study suggest that exposure to 1-NPs could potentially alter mtDNAcn, which may contribute to lung cancer development.
A limitation of this study is that we did not measure historical atmospheric PAH or nitro-PAH concentrations. While the PAH concentrations measured here are not necessarily an accurate reflection of past exposure levels, they are a good approximation of average exposure levels. Another limitation of this study is that the number of participants was not large enough to be representative of Korean exposure to PAHs and nitro-PAHs.
In summary, mtDNAcn decreased significantly with increasing age, and as the absorbed 1-NP was metabolized, mtDNA became damaged, resulting in an increase in mtDNAcn. The results of the present study confirm that exposure to 1-NP at concentrations as low as environmental exposure levels can lead to a significant increase in mtDNAcn.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Professor Heon Kim was supported by National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (2020R1A2C1014531).
