Abstract
2,3,7,8 tetrachlorodibenzo-p-dioxin (TCDD) is a potential environmental toxin that has the ability to affect male reproductive tract. Rhamnazin is a naturally present flavone that displays multiple medicinal properties. Therefore, the current study was designed to determine the mitigative role of rhamnazin against TCDD induced reproductive damage. 48 adult male albino rats were randomly separated into four groups: control, TCDD (10 µgkg−1), TCDD + rhamnazin (10 µgkg−1 + 5 mgkg−1 respectively) and rhamnazin (5 mgkg−1). The trial was conducted for 56 days. TCDD intoxication notably affected superoxide dismutase (SOD), glutathione peroxidase (GPx), glutathione reductase (GSR) and catalase (CAT) activities, besides reactive oxygen species (ROS) and malondialdehyde (MDA) concentrations were augmented. TCDD administration also lowered sperm motility, viability, sperm number, while it augmented the sperm morphological (tail, neck/midpiece and head) anomalies. Moreover, it decreased the levels of follicle-stimulating hormone (FSH), luteinizing hormone (LH) and plasma testosterone. Moreover, TCDD reduced steroidogenic enzymes i.e., 17-beta hydroxysteroid dehydrogenase (17β-HSD), steroidogenic acute regulatory protein (StAR) and 3-beta hydroxysteroid dehydrogenase (3β-HSD) as well as B-cell lymphoma 2 (Bcl-2) expressions, but increased the expressions of Bcl-2-associated X protein (Bax) and cysteine–aspartic acid protease (Caspase-3). Furthermore, TCDD exposure also induced histopathological anomalies in testicular tissues. However, the supplementation of rhamnazin recovered all the mentioned damages in the testicles. The outcomes revealed that rhamnazin can ameliorate TCDD induced reproductive toxicity due to its anti-oxidant, anti-apoptotic and androgenic nature.
Introduction
Dioxins are one of the widely present toxic environmental contaminants that are referred to as the so-called “dirty dozen” - a group of harmful materials called as persistent organic pollutants (POPs). 1 2,3,7,8 tetrachlorodibenzo-p-dioxin (TCDD) is an extremely noxious xenobiotic chemical, which can cause damage to multiple organs such as the liver, 2 kidney, 3 heart, 4 reproductive system,5,6 nervous system, 7 endocrine system 5 as well as skeletal system. 8 Due to its high toxicity, International Agency for Research on Cancer has classified TCDD as the potential carcinogenic compound. 9 It is primarily discharged into the atmosphere as a byproduct of different combustion as well as industrial practices, dispersing in the environmental matrices such as, soil and water and eventually it tends to concentrate in plant and animal body. The half-life of TCDD inside the body of organisms is between 7 and 11 years, weeks-years in rodents as well as 25–100 years in the environment. 10
After ingestion, the circulatory system transports the TCDD inside the body, where it is readily stored in the liver and fat cells because of lipophilic nature, slow excretion and metabolism. 11 Previous study shows that it can be transferred across the placental cord into the embryo and fetus 12 and may persist in testes until the 120th postnatal day. 13 The underlying factor the TCDD prompted reproductive toxicity is the overproduction of reactive oxygen species (ROS).14,15 Mounting evidences suggested that TCDD affects male fertility by lowering the weight of testes, 16 late-onset of the process of sexual development, 17 lowered sperm motility, 15 sperm quality 18 as well as quantity 19 and delayed sperm transportation time.20,21 Moreover, TCDD acts as an antiandrogenic compound,22,23 causing impairment of seminiferous tubules, spermatogenic cells apoptosis, tubular necrosis as well as intratubular vacuolization, 24 resulting in metabolic and functional impairments in male germ cells. 15
Flavonoids are a group of naturally occurring compounds renowned for their several pharmacological properties. 25 Rhamnazin is one of such natural flavonoids, commonly present in food, fruits, teas and medicinal herbs. Rhamnazin has also been shown to possess potent antioxidant, 26 anti-inflammatory, 27 antiangiogenic, 28 anticancer properties. 26 Nevertheless, no study has reported its effects on the testicles till the date. Therefore, the present study was aimed to determine the curative role of rhamnazin against TCDD-instigated testicular dysfunction in male albino rats.
Materials and methods
Chemicals
2,3,7,8 tetrachlorodibenzo-p-dioxin and rhamnazin were bought from Sigma-Aldrich, Germany.
Animals
48 adult albino rats (male) weighing 250 ± 20 g were kept in steel cages in the animal house of University of Agriculture, Faisalabad (UAF). Animals were provided with standard food & water ad libitum as well as 12 h light/12 h dark cycle and standard temperature (23–25°C) was maintained. All the experimental protocols were approved by institutional ethical committee for the handling of laboratory animals.
Experimental design
Rats were randomly separated into four groups ( (1) Group I: Control. (2) Group II: TCDD intoxicated (10 µgkg−1). (3) Group III: TCDD (10 µgkg−1) and rhamnazin (5 mgkg−1) co-treated. (4) Group IV: Rhamnazin (5 mgkg−1) supplemented.
The whole experiment was conducted for 56 days. At the end, the animals were euthanized, blood was collected and centrifugation was performed for 15 min at 3000 r/min. Serum was kept at −20°C after separation for further analysis. Both the testes were excised. The right testis was kept in zipper pouches for 2 days at −80°C and then processed for biochemical assessment, while the left testis was preserved in 10% neutral formalin buffer for histopathological observation.
Biochemical evaluation
The activity of catalase (CAT) was estimated in compliance with the protocol of Chance and Maehly. 29 Superoxide dismutase (SOD) activity was appraised through the approach of Kakkar et al. 30 Glutathione peroxidase (GPx) content was determined by using the procedure of Lawrence and Burk. 31 Glutathione reductase (GSR) activity was computed via the technique of Carlberg and Mannervik. 32 Reactive oxygen species (ROS) and malondialdehyde (MDA) levels were evaluated by using the method of Hayashi et al. 33 and Placer et al. 34 respectively.
Evaluation of sperm indices
The caudal portion of epididymis was used for semen samples. The epididymal portion was crushed in 5 mL of physiological-saline, incubated for 30 minutes at 37°C, to release the sperm. Then sperm motility in percentage was estimated microscopically (400×). 35 Whereas, the viability of sperm was evaluated by nigrosin & eosin stain, followed by microscopic examination. Furthermore, a hemocytometer was used to calculate sperm number. 36 Moreover, morphologically abnormal sperms were determined according to the method of Cao et al. 37
Hypo-osmotic swelling test
Sperm plasma membrane integrity was evaluated as previously demonstrated by Correa and Zavos 38 using HOS test. Firstly, 20 μL of semen was mixed with 180 μL of fructose solution at 80 mOsm/L osmotic pressure for about 20 min. After successive heating and processing, sperms were stained using nigrosin & eosin stain. Lastly, 200 sperms with swollen and non-swollen tails were examined microscopically at 40×.
Evaluation of hormones
Follicle-stimulating hormone (serial number-H101), LH (serial number-H206) and plasma testosterone (serial number-H090) levels were assessed via ELISA kits (Los Angeles, CA, USA) as per company’s instructions. Hormonal level was expressed as ng/ml tissue.
Ribonucleic acid extraction and real-time quantitative reverse transcription-polymerase chain reaction
Primers sequences for the real-time quantitative reverse transcription-polymerase (RT-qPCR).

RT-qPCR: reverse transcription-polymerase chain reaction; 17β-HSD: 17β-hydroxysteroid dehydrogenase; StAR: steroidogenic acute regulatory protein, 3β-HSD: 3β-hydroxysteroid dehydrogenase. Bax: Bcl-2-associated X protein, Bcl-2: B-cell lymphoma 2, Caspase-3: cysteine–aspartic acid protease.
Histopathological evaluation of the testes
After dissection, testicles were isolated from control as well as treated rats, later fixed in 10% formaldehyde solution. In the next step, these tissues were dehydrated in the rising grades of alcohols & fixed in paraffin wax. Then, paraffin-embedded slices were cut into 5-µm sections that were stained using eosin & hematoxylin and observed microscopically (Nikon, 187842, Japan). Then photomicrographs of these sections were captured by Leica LB microscope (Nikon, Japan) and analyzed with Image-J2X software.
Data analysis
The results were shown as Mean ± SE. Tukey’s multiple comparison test was applied to compare complete data after using one-way ANOVA. The level of significance was set at
Results
Effect of rhamnazin on biochemical parameters
Effects of TCDD and rhamnazin on biochemical parameters.

Values are show on the basis of Mean ± SE. Values with different superscripts (a, b and c) in a row are significantly (
Effect of rhamnazin on sperm parameters
Effects of TCDD and rhamnazin on sperm indices.

Values are show on the basis of Mean ± SE. Values with different superscripts (a, b and c) in a row are significantly (
Effect of rhamnazin on gonadotropins (FSH and LH) and androgen (Testosterone)
Effects of TCDD and rhamnazin on hormonal level.

Values are show on the basis of Mean ± SE. Values with different superscripts (a, b and c) in a row are significantly (
Effect of rhamnazin on the expression of steroidogenic enzymes
2,3,7,8 tetrachlorodibenzo-p-dioxin intoxication markedly ( Demonstrates the effects of rhamnazin on (a) StAR, (b) 3β-HSD, (c) 17β-HSD expression. Graphs are presented on the base of Mean ± SE values. Different superscripts (a, b and c) indicate significant (
Effect of rhamnazin on apoptotic proteins
2,3,7,8 tetrachlorodibenzo-p-dioxin administration substantially ( Shows the effects of rhamnazin on (a) Bax, (b) Bcl-2 and (c) Caspase-3 expression. Graphs are presented on the base of Mean ± SE values. Different superscripts (a, b and c) indicate significant (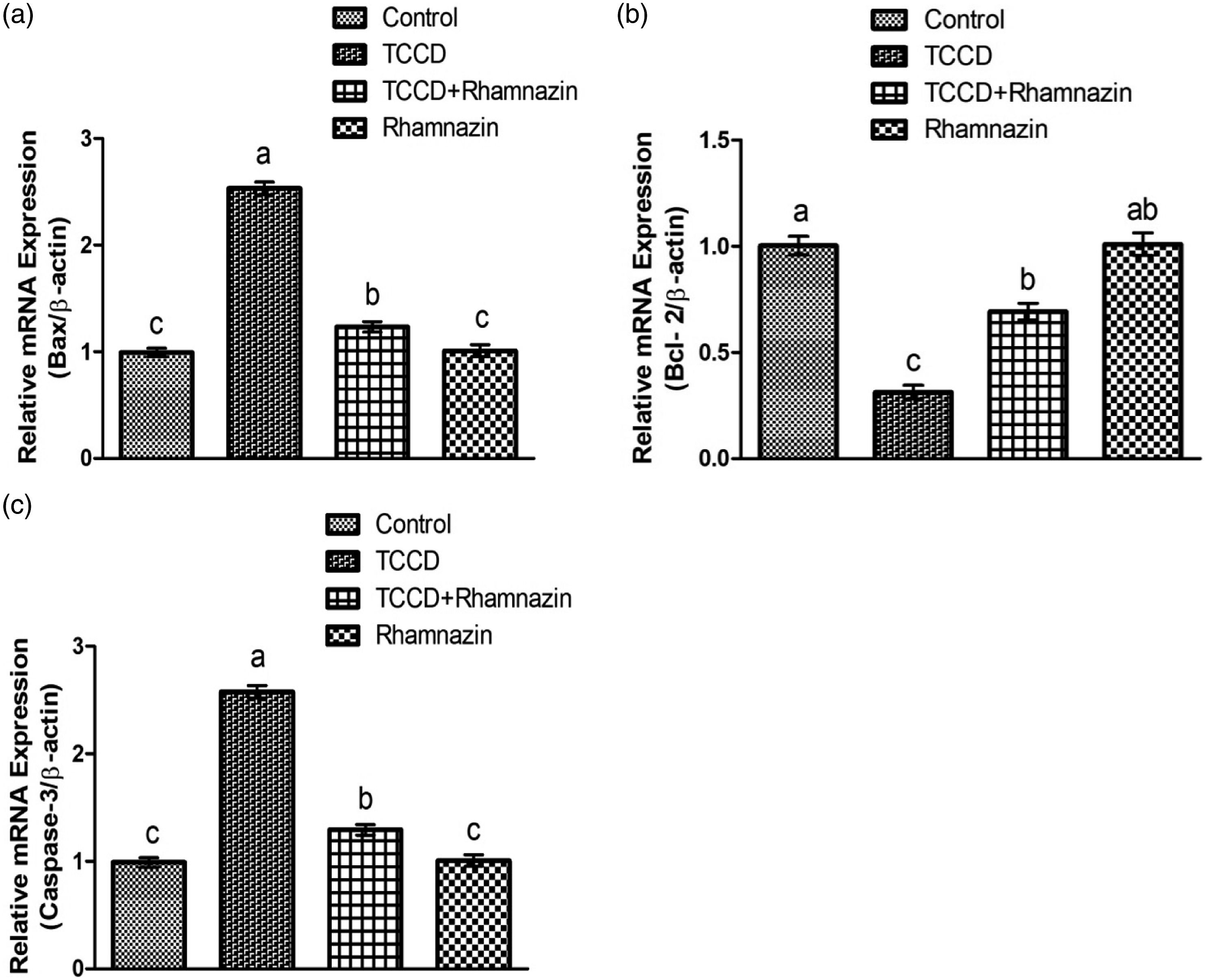
Effect of rhamnazin on testicular histopathology
Effects of TCDD and rhamnazin on the histopathological profile of testes.

Values are show on the basis of Mean ± SE. Values with different superscripts (a, b and c) in a row are significantly (

Micro-photographs of mature rat testicles (H&E, 400×): (a) Control group showing compact germinal epithelium exhibiting germ cells in addition to tapered luminal area holding sperms; (b) TCDD-exposed rats presenting vacant lumen, shedding of the epithelial layer in addition to damaged IS area; (c) TCDD + rhamnazin administrated group demonstrating re-establishment in epithelial part as well as TL packed with ST and recovered IS; (d) Rhamnazin treated rats showing compacted seminiferous tubules, luminal part packed with germ cells and less IS. TCDD: 2,3,7,8 tetrachlorodibenzo-
Discussion
2,3,7,8 tetrachlorodibenzo-p-dioxin is among the top listed pollutants that provokes deleterious impacts on humans and animals. 41 Among the multiple TCDD-instigated damages, reproductive damage is notorious in both males as well as females.42,43 In males, TCDD disrupts the reproductive function of male gonads that are the site of sperm and androgen synthesis. 44 TCDD induces reproductive dysfunctions by disturbing anti-oxidant balance that leads to oxidative stress (OS). 45 Previous studies documented that OS is a major culprit of various ailments linked with male fertility. 46 Thus, keeping in mind the various routes of TCDD exposure and its harmful impacts on animals and human health, research on treatment against TCDD-induced damages is desired. Nowadays, nutritional supplements containing antioxidant properties are used as empirical therapies for improving sperm function. 47 Therefore, the current study was formulated to assess the potential protective effects of rhamnazin against TCDD induced testicular damages in male albino rats.
Antioxidant enzymes (SOD, CAT, GPx, and GSR) are crucial for the maintenance of cellular redox homeostasis in male reproductive tract. 48 SOD protects sperms from superoxide anion (O2−) via catalyzing the transformation of superoxide into hydrogen peroxide (H2O2). However, CAT transforms the H2O2 into H2O and oxygen. 49 On the other hand, GPx/GSR system degrades the H2O2 and lipid hydroperoxide with the help of reduced glutathione (GSH). However, in the current study, TCDD supplementation markedly lowered anti-oxidant enzymes activities i.e., SOD, GSR, GPx as well as CAT, whereas ROS and MDA (a potential indicator of LP) levels were increased. According to Ijaz et al. 50 ROS has cytotoxic effects on spermatozoa, which directly attacks the cellular constituents that produce OS by altering the antioxidant/oxidant balance. This OS, as feedback, attacks the poly unsaturated fatty acids (PUFAs) that are present in spermatozoa plasma membrane due to notably limited volume and localization of the cytoplasm. 51 The sperm membranes then undergo permeability alterations as evident from glutathione exhaustion and heightened LP. 52 Conversely, this lowered antioxidant enzymes activity in rat’s testicular tissues was improved by the supplementation of rhamnazin, that may be attributed to its anti-oxidant potential. This antioxidant property of rhamnazin is due to its structure, particularly the presence of a hydroxy group at carbon-4′. 53
2,3,7,8 tetrachlorodibenzo-p-dioxin exposure considerably lowered sperm motility, number, viability along with a decline in the number of sperm, while caused anomalies in sperm tail, midpiece and head. TCDD exposure leads to ROS production that instigate adverse effects on male reproductive tract and decreased sperm count & motility. 20 Additionally, TCDD exposure remarkably reduces the production of adenosine triphosphate (ATP) by affecting the functions of mitochondria. 54 Reduced synthesis of ATP in sperms damages the flagellar activity and induces the sperm immobility. 55 However, the supplementation of rhamnazin recovered all the spermatogenic anomalies due to its potential free radical scavenging nature.
Spermatogenesis is a cascade of events that is regulated under the influence of hormones of hypothalamic pituitary gonadal axis (HPG).56–58 Hypothalamus discharges the gonadotropin-releasing hormone (GnRH) that instigates the liberation of FSH and LH (gonadotropins), which are crucial for reproduction. FSH is responsible for the proper functioning of Sertoli cells (SCs) that make and discharge regulatory molecules and nutrients required for spermatogenesis. 59 LH promotes the formation and liberation of testosterone from Leydig cells (LCs). This testosterone then diffuses into seminiferous tubules and stimulates the germ cell maturation. Therefore, an appropriate level of these hormones is required for sperm production, whereas TCDD administration considerably decreased the gonadotropins and androgen levels, possibly due to disturbed hypothalamic pituitary testicular axis. Lowered level of FSH is unable to instigate SCs, while reduced LH is not capable to regulate LCs to produce testosterone. 60 Moreover, lowered levels of FSH & testosterone are unable to stimulate spermatids growth and sperms ejaculation. 61 However, rhamnazin administration recovered the hormonal changes induced by TCDD probably via regulating the normal functioning of HPG axis. Moreover, our outcomes are further in compliance with the previous study, which stated that flavonoids have the potential to regulate the level of hormones. 62
To explore the philosophy responsible for the lowered level of testosterone following TCDD administration, StAR, 3β-HSD & 17β-HSD expressions were appraised. As evident, testosterone production is regulated by multiple enzymes and proteins i.e., StAR, a mitochondrial membrane protein, which plays an important role in cholesterol transportation from the cytoplasm to mitochondria. 63 StAR acts as a rate limiting protein in steroidogenic process. Cholesterol conversion to testosterone occurs in multiple reactions regulated by17β-HSD and 3β-HSD. 64 The findings of current study revealed that TCDD exposure considerably decreased the expressions of above stated steroidogenic enzymes. Thus, the reduced level of testosterone is probably due to the effect of TCDD on steroidogenic mechanism. Conversely, rhamnazin significantly escalated the repressed level of testosterone by augmenting the expression of steroidogenic enzymes that might be attributed to its androgenic nature.
2,3,7,8 tetrachlorodibenzo-p-dioxin intoxication upregulated the pro-apoptotic protein i.e., Caspase-3 & Bax expressions, on the other hand lowered anti-apoptotic protein (Bcl-2) expression. According to Venkatadri et al. 65 apoptosis is regulated by anti-apoptotic & apoptotic proteins. The imbalance in these proteins prompts apoptosis. 66 Bax is an apoptotic protein that instigates apoptosis, whereas Bcl-2 is an anti-apoptotic protein, which performs a pivotal role in the suppression of apoptosis. 67 Besides, Caspase-3 belongs to Caspases that split the cellular proteins and prompts apoptotic cell death. Hence, the down regulation in Bcl-2 and escalated Bax expression potentially change the mitochondrial membrane’s permeability and increased the liberation of cytochrome C into the cytoplasm. 68 Overproduction of cytochrome C in cytoplasm stimulates Caspase-3 that prompts apoptosis. 69 Nevertheless, rhamnazin co-treatment remarkably scaled down the expression of Caspase-3 and Bax and elevated the expression of Bcl-2 in contrast to TCDD-exposed group. Our study demonstrates that the protective role of rhamnazin against apoptosis may be attributed to its anti-apoptotic nature.
According to our results TCDD exposure prompted severe histopathological abnormalities in rat’s testes such as, reduction in the seminiferous tubular diameter along with height and reduced tunica propria height, on the other hand, escalated tubular lumen’s diameter and interstitial spaces. Moreover, TCDD lowered the count of germ cell. It is reported that spermatogenesis is an androgen dependent process. 70 So, the lowered production of testosterone results in structural irregularities. 60 However, rhamnazin administration markedly alleviated the histopathological anomalies induced by TCDD. These ameliorative effects of rhamnazin might be ascribed to its androgenic, anti-oxidant & anti-apoptotic nature.
Conclusion
The outcomes of our study have shown that rhamnazin has potential palliative role against TCDD prompted testicular toxicity. The supplementation of rhamnazin considerably restored anti-oxidant enzymes activities, regulated ROS & MDA levels, steroidogenic enzymes and apoptotic markers expressions as well as histological architecture. Taken together, it can be proposed that rhamnazin can be used as a potential therapeutic agent against TCDD instigated testicular damage.
Footnotes
Author contributions
MUI and SM designed the study. SM & AH executed the experiments. QUA, AH and SA performed the statistical analysis. MUI & SM wrote the manuscript. All the authors read and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data Availability Statement
Data will be available on reasonable request from the corresponding author.
