Abstract
Background
Cyclophosphamide (CP) is a commonly used chemotherapeutic and immunosuppressive alkylating agent. However, cardiac adverse effects of CP interfere with its clinical benefit. Cardio-oncology research is currently an important issue and finding effective cardiopreserving agents is a critical need. For the first time, we aimed to detect if dapagliflozin (DAP) could ameliorate CP-induced cardiac injury and investigated the role of hypoxia inducible factor α (HIF1α)/vascular endothelial growth factor (VEGF)/endothelial nitric oxide synthase (eNOS) pathway.
Methods
Forty male Wistar albino rats were included in the current model. Studied groups are: control group; CP-induced cardiotoxicity group; CP group treated with DAP; CP group treated with DAP and administered a nitric oxide synthase inhibitor; nitro-ω-L-arginine (L-NNA) before DAP to explore the role of eNOS.
Results
Our data revealed that CP could induce cardiac damage as manifested by significant increases in cardiac enzymes, blood pressure, malondialdehyde (MDA), tumor necrosis factor alpha (TNFα), HIF1α, sodium glucose co-transporter 2 (SGLT2) and cleaved caspase-3 levels with toxic histopathological changes. However, there are significant decreases in reduced glutathione (GSH), total antioxidant capacity (TAC), VEGF, and eNOS. On the opposite side, co-administration of DAP showed marked improvement of CP-induced cardiac damage that may be due to its ability to inhibit SGLT2, antioxidant, anti-inflammatory and anti-apoptotic properties. Results showed decreasing the cardioprotective effect of DAP on administration of L-NNA, reflecting the critical effect of eNOS in mediating such protection.
Conclusion
DAP could reduce CP cardiotoxicity based upon its ability to modulate SGLT2 and HIF1α/VEGF/eNOS signaling pathway.
Keywords
Introduction
Cyclophosphamide (CP) is a widely used anticancer alkylating agent. Unfortunately, it has a well-known serious cardiotoxic effects that restrict its dosing regimen, lowering its effectiveness or even leading to cessation of the treatment. Therefore, overcoming this adverse effect is so vital for accomplishing a favorable outcome in cancer patients.1,2 Cardiotoxicity of CP is usually observed after administration of high doses. Although the associated clinical manifestations and cardiotoxic hazards are well-documented, the underlying mechanisms are still not fully understood. Nonetheless, some researchers have shown that CP could disrupt metabolic processes of the cardiac tissue, decrease ATP levels and reduce cellular antioxidants. 3
CP is an inactive prodrug that should be activated by liver cytochrome P450 enzymes to be converted into two molecules; phosphoramide mustard and acrolein. The latter is responsible for occurrence of different CP toxicities including hemorrhagic cystitis, cardiotoxicity, gonadal toxicity, and bone marrow suppression. CP metabolites stimulate oxidative stress and directly damage the endothelial cells resulting in edema, interstitial hemorrhage, and formation of microthrombosis.3,4 Endothelial cell is more susceptible to CP-induced damage than other cells. In addition, CP induces release of reactive oxygen species (ROS) leading to diminishing of nitric oxide availability and decreasing the release of antioxidant molecules with induction of hypoxia.5,6 Decreasing oxygen supply to the myocardium enhances certain molecule; hypoxia inducible factor 1α (HIF1α) that is mandatory in regulating the transcription process of another vascular molecule; vascular endothelial growth factor (VEGF).7–9 It is known that VEGF is a highly effective angiogenic factor released as a defensive mechanism against hypoxia. It improves vascularity, increases blood supply, keeps the tissues against ischemic harmful effect with a great role in several processes of neovascularization, release of vascular endothelial nitric oxide synthase (eNOS) and excessive formation of the potent vasodilating agent; nitric oxide (NO).9,10
Sodium glucose co-transporter 2 (SGLT2) inhibitors are considered as a recent oral antidiabetic drugs that act through preventing renal glucose reabsorption and decreasing blood glucose level, lowering blood pressure, controlling metabolic disorders and decreasing body weight.11,12 In addition, DAP has an antioxidant role with the ability to scavenge reactive oxygen and nitrogen species followed by decreasing cardiac inflammation. Moreover, DAP could control renal renin angiotensin aldosterone system, decrease cardiac and renal complications in diabetic patients, improve cardiac ischemia-reperfusion injury and doxorubicin cardiotoxicity.13,14
Recently, a highlighted link is reported between SGLT2 inhibitors, including DAP, and their cardioprotective effects with the ability to ameliorate acute myocardial infarction and decrease the rate of hospitalization in patients of heart failure. Importantly, these protective properties of DAP have been found to act independently of its lowering effect on blood glucose and the exact underlying mechanism has not been fully elucidated.15–17 Understanding the mechanism of SGLT2 and its selective targets is so important as a new application in treating/preventing heart diseases. The urgent need for finding new preserving agents that could act against CP-induced cardiotoxic damage and the beneficial role of DAP in other models of cardiac injury directed our attention to study DAP possible protective effects in such toxicity.
Materials and methods
Ethical issue
Our research was approved in accordance with the research ethical roles of Faculty of Medicine-Research Ethics Committee, “FMREC”, Minia University, Egypt in line with ARRIVE Guidelines and EU Directive 2010/63/EU Guidelines for animal experiments, approval No. 331-4-2022.
Animals
Forty male Wistar albino rats, 8 weeks old, weighing (200–220 g) were obtained from the National Research Center, Giza, Egypt then kept to acclimatize for 2 weeks in a suitable standard housing condition (25°C, 45% humidity, and 12 h light/dark cycle). Rats were randomly divided into three rats per cage with free access to laboratory chow and tap water throughout the experiment.
Chemicals and antibodies
Cyclophosphamide and DAP were obtained from Baxter Co. Germany, AstraZeneca pharmaceutical Co. (Cambridge, US), respectively. Enzyme-linked immunosorbent assay (ELISA) kits of troponin I (CAT# MBS722833), creatine kinase-MB (CK-MB) (CAT# MBS2515061), lactate dehydrogenase (LDH) (CAT# MBS043166), SGLT2 (CAT# MBS1600381), HIF-1α (CAT# MBS2506922) were purchased from MyBiosource (San Diego, California, USA). Total antioxidant capacity (TAC) (CAT# TA2513) was from Biodiagnostic, Egypt.
Cleaved caspase-3 rabbit polyclonal antibody (CAT# PA5-114,687) was from Invitrogen, US. Phosphorylated eNOS (CAT# 9571), total eNOS (CAT# 9572), TNFα (CAT# 3707) rabbit polyclonal antibodies, GAPDH rabbit monoclonal antibody (CAT# 2118) and alkaline phosphatase (AP)-linked goat anti-rabbit polyclonal antibody (CAT# 7054) were from Cell Signaling Technology, Inc., US.
Experimental design
The duration of studying the cardioprotective effect of DAP in CP cardiotoxicity was for 5 days; 3 days before giving CP and 2 days during administration of CP. The toxic drug; CP was i.p. injected on 4th and 5th day.
We randomly allocated the animals into four equal groups (n=10) as follows:
DAP was prepared by dissolving in distilled water; and CP was dissolved in saline immediately before administration. CP toxic dose is comparable to that of human toxic dose. All doses were selected according to our pilot study and previous studies of similar models in rats.
Measurement of blood pressure
Before animal sacrifice, blood pressure (BP) was measured with the tail-cuff method (LETICA, Panlab S.L., Barcelona, Spain). First, we kept each rat at 38°C for 15 min and determine pulsation of rat tail artery then apply tail–cuff and measure BP for five successive times in each one. The values were based on the mean of several successive measurements. 20
Preparation of the samples
At the end of the experiment, each rat was anesthetized by urethane hydrochloride (i.p., 20% in a dose of 1 g/kg). Blood was obtained from the abdominal aorta, centrifuged for 15 min at 5000 rpm (JanetzkiT30 centrifuge, Germany) for separation of the sera that were used for evaluating cardiac enzymes and TAC. Heart of each animal was dissected and washed with normal saline then weighed. Part of the cardiac tissue was homogenized in 5% w/v ice-cold phosphate-buffered saline (0.01 M, pH 7.4), then the homogenate was centrifuged at 3000 rpm for 20 min at 4°C in a cooling centrifuge (Ray Wild TGL-16, Germany) and the supernatant of each sample was stored at −80°C for biochemical analysis. Part of tissue homogenate was stored at −80°C for western blotting; and other parts were stored in 10% formalin for performing histopathological and immunohistochemical examination.
Biochemical measurements
Measurement of MDA concentration
Malondialdehyde evaluation was performed via a method of Buege and Aust 21 using thiobarbituric acid reacting substance as an equivalent of MDA. 1,1,3,3-tetramethoxypropane standard curve and results were expressed in nmol/g tissue.
GSH and TAC concentrations in cardiac tissue
Glutathione sulfhydryl group could react with Ellman’s reagent forming a yellow color detected at 405 nm by Beckman DU-64 UV/VIS spectrophotometer, USA as µmol/g tissue. 22 The commercial kit of TAC was from Biodiagnostic, Egypt and results were expressed as mmol/L.
Measurement of cardiac enzymes, SGLT2 and HIF1α concentrations
Creatine kinase-MB, troponin I, LDH, SGLT2 and HIF1α concentrations were measured by the available commercial ELISA kits according to the manufacturer's instructions for intra assay detection, MyBiosource (San Diego, California, USA).
Western blotting analysis of caspase-3, TNFα and eNOS
According to previously described methods of western blotting, we measured caspase-3, TNFα
Light microscopic histopathological study
Cardiac specimens of the left ventricle were fixed in 10% neutral buffered formalin for 2 days and processed by paraffin embedding then sectioned. 5 µm sections were stained with hematoxylin and eosin (H&E) to be examined by the light microscopy for general histological, immunohistochemical studies and morphological changes
Microscopic analysis and photography
Slides were assessed blindly by the histologist using light microscopy (Olympus microscope, Tokyo, Japan). Sections were photographed using Olympus digital camera BX51 in Histology and Cell Biology Department, Faculty of Medicine, Minia University. Ten fields of each slide were randomly selected then assessed according to the degree of distorted architecture of the cardiac muscles, loss of muscle striation, inflammatory cellular infiltrate, presence of pyknotic nuclei, and vascular congestion. The severity of the lesion was graded as follows: score 0 was considered normal, score 1 (+) for mild changes, score 2 (++) for moderate changes, and score 3 (+++) for severe changes.24,25
Immunohistochemical expression of eNOS and VEGF
The slides were deparaffinized (through heating at 60°C for 12 h and soaking in xylene, then rehydrated in ethanol (through descending grades of alcohol). Antigen retrieval was performed with citrate buffer (pH 6) at 97°C for 20 min. Immunohistochemical staining was performed using avidinbiotin peroxidase technique for eNOS and VEGF using anti-VEGF monoclonal antibody (A17877, 1:150; ABclonal, USA) and anti-eNOS polyclonal antibody (N3893, 1:100; Sigma Aldich, USA). The surface area fraction and the optical density were calculated using Image-J acquisition software, (AxioVision Release 4.8.2-SP2 Software). Statistical significance was detected from ten non-overlapping randomly selected fields X400 high-power field in each group. 24
Statistical analysis
GraphPad Prism software (version 5.01 for Windows, San Diego, CA, USA) was used in performing the statistical analysis of our data. Results of our experiment were expressed as mean ± SEM. We used One-way analysis of variance (ANOVA) followed by Tukey’s post-test and p-value <.05 was considered significant.
Results
Effect of DAP on cardiac enzymes and heart weight changes
Effect of DAP on cardiac enzymes and heart weight in CP-induced cardiotoxicity.

All parameters are expressed as mean ± SEM of 10 observations. Significant difference is considered when p < 0.05.
DAP= dapagliflozin, CP= Cyclophosphamide, L-NNA= nitro-ω-L-arginine. CK-MB= creatine kinase-MB, LDH= lactate dehydrogenase.
aSignificant difference compared to control group.
bSignificant difference compared to CP group.
cSignificant difference compared to DAP treated group.
Effect of DAP on oxidative stress parameters (MDA, GSH and TAC)
Effect of DAP on cardiac oxidative stress parameters.
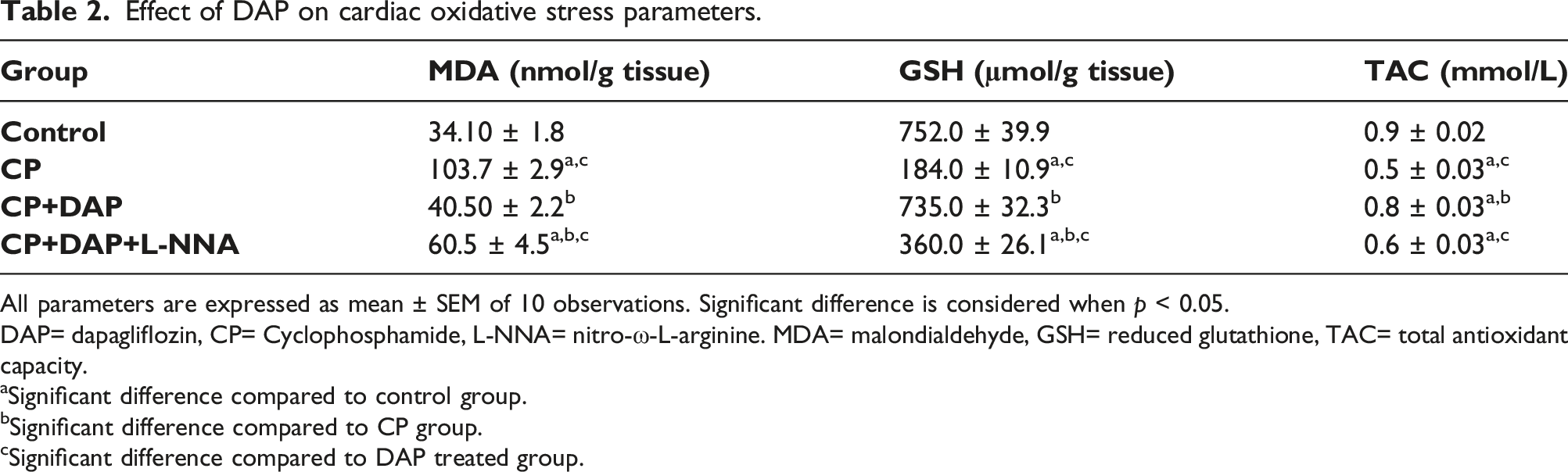
All parameters are expressed as mean ± SEM of 10 observations. Significant difference is considered when p < 0.05.
DAP= dapagliflozin, CP= Cyclophosphamide, L-NNA= nitro-ω-L-arginine. MDA= malondialdehyde, GSH= reduced glutathione, TAC= total antioxidant capacity.
aSignificant difference compared to control group.
bSignificant difference compared to CP group.
cSignificant difference compared to DAP treated group.
Measurement of SGLT2, HIF1α, BP
Effect of DAP on SGLT2, HIF1α and BP.

All parameters are expressed as mean ± SEM of 10 observations. Significant difference is considered when p < 0.05.
DAP= dapagliflozin, CP= Cyclophosphamide, L-NNA= nitro-ω-L-arginine. SGLT2 =sodium glucose co-transporter 2, HIF1α= hypoxia inducible factor α, BP= blood pressure.
aSignificant difference compared to control group.
bSignificant difference compared to CP group.
cSignificant difference compared to DAP treated group.
Evaluation of caspase-3, TNFα and eNOS levels using western blotting
CP given rats showed a significant increase in caspase-3 and TNFα levels and a significant decrease in p-eNOS level compared to control group and DAP treated group. However, CP plus DAP group showed a significant decrease in tissue levels of caspase-3 and TNFα and a significant increase in p-eNOS compared to CP administered group. Interestingly, L-NNA given rats reversed DAP effect and increased caspase three and TNFα tissue levels but decreased eNOS in comparison to both control group and DAP treated group (Figure 1(a)–(c)). (a)–(c): Western blotting analysis of caspase-3, TNFα and eNOS levels. Caspase-3 and TNFα significantly increased but p-eNOS decreased in CP group compared to control group. DAP treated group showed significant decrease of caspase-3 and TNFα but significant increase of p-eNOS compared to CP group. All parameters are expressed as mean ± SEM of 10 observations. Significant difference is considered when p < .05. aSignificant difference compared to control group. bSignificant difference compared to CP group, cSignificant difference compared to DAP treated group. DAP: dapagliflozin; CP: cyclophosphamide.
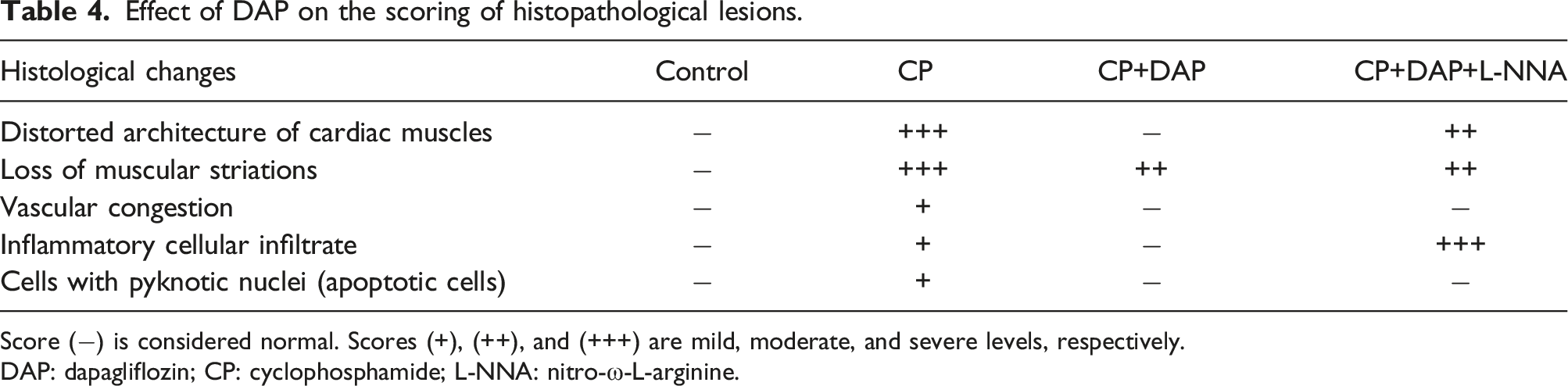
Effect of DAP on histopathological changes
Figure 2: Figure 2(a) Histopathological examination of cardiac tissue. Representative photomicrographs for the histological structure of heart tissues showing: (a) Effect of DAP on the scoring of histopathological lesions. Score (−) is considered normal. Scores (+), (++), and (+++) are mild, moderate, and severe levels, respectively. DAP: dapagliflozin; CP: cyclophosphamide; L-NNA: nitro-ω-L-arginine.
Immunohistochemical results
Effect of DAP on eNOS immunoexpression
The control group showed positive expression in capillary endothelium among cardiac muscle fibers (A). Both CP and CP+DAP+L-NNA groups showed negative expression with significant decrease in surface area fraction and optical density of eNOS immunoreactivity compared to the control group and DAP treated group (B,D). However, CP+DAP given group revealed strong positive expression with significant increase in both surface area fraction and optical density of eNOS immunoreactivity compared to CP group (C) (Figure 3). Effect of DAP on eNOS immunoexpression. Representative photomicrographs of heart tissues immune stained for eNOS represents: (a) It represents control group with positive expression in the endothelium of capillaries (arrows) among myocytes. (b) It shows CP group with negative eNOS expression. (c) It reveals CP+DAP group that shows increased eNOS expression (arrows). (d) It is CP+DAP+L-NNA group that shows negative expression of eNOS. (e) It reveals negative control. (f) Surface area fraction and optical density of eNOS immunoreactivity. IHC counterstained with H x400, scale bar = 50 µm. All parameters are expressed as mean ± SEM of 10 observations. Significant difference is considered when p < .05. aSignificant difference compared to control group. bSignificant difference compared to CP group, cSignificant difference compared to DAP treated group. DAP: dapagliflozin; CP: cyclophosphamide.
Effect of DAP on VEGF immunoexpression
The control group showed positive expression in capillary endothelium (A); however, CP given group showed faint expression with significant decrease of VEGF immunoexpression compared to control group and CP+DAP group (B). On the contrary, CP plus DAP co-administered group revealed strong positive expression with significant increase in both surface area fraction and optical density of VEGF immunoexpression compared to CP toxic group (C). CP+DAP+L-NNA group showed mild expression with significant decrease in both parameters of VEGF immunoexpression compared to the control and DAP co-administered groups (D) (Figure 4). Effect of DAP on VEGF immunoexpression. Representative photomicrographs of heart tissues immune-stained for VEGF shows: (a) It is the control group with positive expression in capillary endothelium (arrows). (b) CP group shows decreased VEGF expression (arrow) in capillary endothelium. (c) CP+DAP group shows extensive strong positive expression in capillary endothelium (arrows). Notice scattered cells with positive expression (arrowheads) among myocytes. (d) CP+DAP+L-NNA group shows scattered capillaries with faint positive expression (arrow). (e) It reveals negative control. (f) Surface area fraction and optical density of VEGF immunoreactivity. IHC counterstained with H x400, scale bar = 50 µm. All parameters are expressed as mean ± SEM of 10 observations. Significant difference is considered when p < .05. aSignificant difference compared to control group. bSignificant difference compared to CP group, cSignificant difference compared to DAP treated group. DAP: dapagliflozin; CP: cyclophosphamide.
Discussion
It is well known that administration of CP in high doses or for long duration is complicated by serious cardiotoxic effects. Till now, there is no efficient treatment that could protect the myocardium from these cardiotoxic hazards.26,27 Thus, we aimed in the current model to evaluate the possible cardioprotective effect of DAP in CP-induced cardiotoxicity. We found that CP administration in high toxic dose (150 mg/kg/day) i.p. on 4th, 5th days successfully induced cardiac injury as manifested by significant elevations in BP and cardiac enzymes including troponin-I, CK-MB, LDH with severe cardiac damage in the histopathological features. Furthermore, there is induction of oxidative stress in the form of elevation of MDA but decrease in GSH and TAC. In addition, there is modulation of HIF1α/VEGF/eNOS pathway, TNFα and caspase-3 tissue levels reflecting occurrence of vascular injury, spasm, inflammation, oxidative stress and apoptosis. However, co-administration of DAP could ameliorate the biochemical and histopathological changes of CP.
Detection of cardiac enzymes is essential in evaluating heart performance including serum troponins, CK-MB, LDH. Cardiac troponins are included in the contractile apparatus of myocardial cells, expressed mainly in the heart, and considered as the specific marker of cardiac tissue injury. Moreover, the most sensitive cardiac enzyme reflecting cardiac lesion is CK-MB. CP-induced cell damage causes leakage of cardiac enzymes to the blood stream followed by elevation of the cardiac enzymes in the serum serving as a diagnostic marker to detect the severity of heart injury. This occurs due to the associated release of ROS, oxidative stress and membrane lipid peroxidation with cell damage and release of cardiac enzymes.28,29 In our study, the levels of serum troponin I, LDH, and CK-MB dramatically increased in CP given group indicating the occurrence of cardiotoxicity in rats. Moreover, the histopathological results showed marked cardiac injury including hemorrhage, necrosis, and inflammatory cell infiltration in CP given group. These results are in line with different previous studies.30–32
Release of free radicals and stimulation of oxidative stress associated with imbalance between oxidants and antioxidants leads to membrane lipid peroxidation which was detected by measuring its sensitive indicator, MDA level. Furthermore, intracellular antioxidants, such as GSH, serve as the first line of defense system to rescue the myocardium and prevent oxidative injury. 33 Our model found significant decrease of GSH and TAC in CP group compared to control group. This occurs due to depletion of antioxidants by the released free radicals and stimulation of oxidative stress that is in accordance with others.34–36
Another essential signaling cascade during CP cardiotoxicity is the apoptotic pathway. Apoptosis is considered as the major focus in the primary process of programmed cell death in different tissues. CP stimulates release of ROS associated with induction of apoptotic as well as the non-apoptotic pathways. One of the most essential factors in mediating apoptosis is caspase-3 that induces DNA fragmentation with destruction of cytoskeletal proteins. Moreover, mitochondrial functions are markedly affected by CP with reduction of ATP production causing cardiotoxicity37,38 and this is in agreement with our results.
There is a close relation between two important processes oxidative stress and inflammation, one easily induces the other. So that, it is difficult to determine which one is the cause or the result. Both processes could be simultaneously detected in many pathological conditions followed by activation of apoptosis, DNA damage and tissue death. 39 We noticed a significant increase in one of the most important pro-apoptotic and pro-inflammatory mediators; TNFα in CP group.
HIF-1α gene is a transcription factor that is so important in mediating O2 homeostasis and regulation of cardiovascular function via releasing HIF-1α during hypoxic states and cancer biology in areas of energy metabolism, vascularization, angiogenesis, cell survival and tumor invasion. HIF-1α is responsible for regulating the synthesis process of different angiogenic factors such as VEGF which is an active protein molecule that stimulates the formation of new blood vessels with increasing blood flow to restore oxygen requirement.36–38 VEGF plays an essential role in angiogenesis and it is highly visible in carcinomas, where it is currently an important target for cancer therapy. Cardiovascular toxicity is associated with suppression of VEGF expression followed by alterations in eNOS and NO release.39–42 This is in agreement with our results as we found significant increase of HIF1α but decreases of VEGF, eNOS expression in CP given group. This leads to the associated increased BP, myocardial damage, necrosis, cell death with impaired vascularization, blood flow and tissue hypoxia. However, CP plus DAP given group could reverse this harmful effect with decrease in HIF1α, up-regulation of VEGF/eNOS pathway. This signaling cascade keeps the myocardium from CP induced tissue hypoxia, improves blood flow and normalizes cardiac tissue and blood pressure. Inhibition of eNOS in CP+DAP+L-NNA group decreased VEGF/eNOS expression with suppression of the cardioprotective effect of DAP reflecting the essential role of eNOS in mediating such protection. This is in accordance with previous studies of DAP in other cardiotoxic models.43–47 In addition, VEGF is not only expressed in vascular endothelium, but also it was reported to be expressed in macrophages around the infarcted tissue and it has essential roles in myocardial repair.40–42,48–50
SGLT2 is a new class of oral anti-diabetic agents used mainly for lowering blood glucose levels in type 2 diabetic patients. This drug group can act on the proximal tubules, decrease renal glucose reabsorption and increase glucose excretion. Interestingly, this drug group has a great cardioprotective effect, but the mechanism is still unclear.48–51 SGLT co-transporters are highly expressed in the heart tissue and it is suggested that they have an indirect effect on the cardiac tissue. This may be via controlling renin angiotensin aldosterone system, decreasing cardiac remodeling and hypertrophy, ameliorating volume overload and decreasing blood pressure.47,51 In addition, DAP decreases calcium overload, diminishes oxidative stress and ROS production.47,51 In our model, we found that co-administration of DAP could improve CP-induced biochemical and histopathological changes. DAP could modulate HIF1α/VEGF/eNOS pathway and inhibit inflammation, oxidative stress and apoptosis processes as it decreased TNFα, MDA and csaspase3 levels but increased GSH and TAC. These findings were in line with previous studies.41–46
These protective properties of DAP were detected in other forms of cardiac injuries including diabetic cardiomyopathy, doxorubicin cardiotoxicity, ischemia reperfusion models, myocardial infarction and heart failure.51–53 These effects were based upon its ability to inhibit SGLT2, antioxidant, anti-inflammatory and anti-apoptotic properties and regulation of RAAS with preservation of the myocardium.
Conclusion
Dapagliflozin ameliorated cyclophosphamide-induced cardiotoxicity. This may depend on its ability to modulate HIF1α/VEGF/eNOS pathway, anti-apoptotic, antioxidant and anti-inflammatory activities. Further studies are in a critical need to confirm its different effects in patients receiving cyclophosphamide.
Data availability statement
All data is available.
Footnotes
Author contributions
Dr. Marwa and Dr. Sayed selected the point, performed the experimental part, wrote and revised the manuscript. Dr. Nahla and Dr. Sahar performed and wrote the histopathology, immunohistochemistry and revised the manuscript. Dr. Asmaa performed and wrote the western blotting part and revised the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Animal handling, medications, and animal sacrifice were carried out following the guidelines for the care of experimental animals and approved by the Institutional Ethical Committee, Faculty of Medicine, Minia University, Egypt according to ARRIVE Guidelines and EU Directive 2010/63/EU Guidelines for animal experiments.
