Abstract
Methotrexate (MTX) is a widely used chemotherapeutic agent but its clinical use is challenged with different forms of toxicities including testicular injury. The aim of the current study was to evaluate the potential protective effect of potassium channel opener, nicorandil (NIC) (3 and 10 mg/kg/day) on MTX-induced testicular injury in a rat model. Rats were randomly divided into four groups (nine rats each) and treated for 2 weeks as follows: (I) normal control (CON group) received vehicle, (II) model group (MTX group) given MTX (20 mg/kg) single intraperitoneal (i.p.) injection dose on 11th day, (III) MTX + NLD group treated with NIC (3 mg/kg/day) orally for 2 weeks and MTX (20 mg/kg) single i.p. dose on 11th day, and (IV) MTX + NHD group treated with NIC (10 mg/kg/day) orally for 2 weeks and MTX (20 mg/kg) single i.p. injection on the 11th day. The testicular injury was assessed biochemically via serum testosterone, total antioxidant capacity, testicular oxidative stress parameters, P-glycoprotein, tumor necrosis factor-alpha, and interleukin-1β. Furthermore, histopathological evaluation, endothelial nitric oxide synthase (eNOS) immunoexpression, and detection of p53 expression level using Western blotting were performed. Results showed that MTX induced testicular injury which was proved by both biochemical and histopathological evaluations. Our results concluded that NIC pretreatment attenuated MTX-induced testicular injury via significantly increased eNOS immunoexpression, antiapoptotic, anti-inflammatory, and antioxidant properties. Interestingly, NIC high dose is more protective than low dose.
Introduction
Methotrexate (MTX) is a widely used chemotherapeutic agent. It is a folic acid antagonist which inhibits dihydrofolate reductase and reduces the levels of tetrahydrofolate in the cells. The basis of MTX therapeutic efficacy as well as its toxicities is through inhibition of purine and pyrimidine synthesis. 1 It is commonly used in treatment of wide variety of neoplastic and autoimmune disorders. 2 Unfortunately, besides MTX clinical beneficial uses, it has serious toxicities on different organs as liver, lung, kidney, and testes. 3,4
Several pathways were found to be involved in mediating MTX-induced testicular injury such as inflammation, oxidative stress, apoptosis, and disturbance in vascular homeostasis via attenuating endothelial nitric oxide synthase (eNOS). MTX overdose was reported to cause increase in the release of interleukin-1β (IL-1β) and tumor necrosis factor-alpha (TNF-α). They are the primary inflammatory cytokines produced by macrophages and monocytes in response to a range of stimuli, including activated T cells, immune complexes, various microbial products, and the combined action of other cytokines. They enforce leucocytes to move out of capillaries and accumulate at the sites of injury. In different tissues, the proinflammatory and anti-inflammatory cytokines were at equilibrium. This equilibrium changed in favor of the proinflammatory cytokines released in MTX-induced toxicities. 5,6
Furthermore, MTX causes disruption of eNOS which has a critical role in vascular function homeostasis, and it also generates nitric oxide (NO), that is considered as a key mediator of cellular signaling and it is essential for maintaining organ homeostasis and vascular tone. 7,8 Moreover, oxidative stress plays an important role in the pathogenesis of MTX-induced testicular damage and leading to apoptosis in spermatocytes and atrophy of seminiferous tubules. 4,9
Nicorandil (NIC) is an adenosine triphosphate (ATP)-sensitive potassium channel opener with a nitrate group. It is clinically proved as an antianginal agent and used for treatment and protection of ischemic heart diseases in animal and human models. 10 Previous studies reported that NIC had a scavenging action on free radicals and NO synthase uncoupling in streptozotocin-induced diabetic rats. Moreover, NIC could protect from diabetes through inhibition of the production of ROS stimulated by high blood glucose level. 11 NIC beneficial effects are also through its NO donor, antioxidant and anti-inflammatory properties. 12
The previously mentioned mechanisms attributed to MTX-induced testicular injury directed our attention to study the possible protective role of NIC as an important eNOS stimulator, antioxidant, antiapoptotic, and anti-inflammatory agent in ameliorating MTX-induced testicular toxicity.
Materials and methods
Chemicals
NIC and MTX were obtained from Merc Co. and Menapharm Co. (Egypt), respectively. The polyclonal rabbit/anti-rat (eNOS) antibody, detection immunostaining, TNF-α, and IL-1β enzyme-linked immunosorbent assay (ELISA) kits were obtained from Thermo Fisher Scientific Inc./Lab Vision (Fremont, California, USA). Total antioxidant capacity (TAC) kit was purchased from Biodiagnostic, Egypt. P-glycoprotein (P-gp) ELISA kit was obtained from United States Biological (US Biological) Life sciences Co. (Massachusetts, USA). Testosterone ELISA kit was from Cayman Chemicals (USA).
Animals
The current study was conducted on Wistar male albino rats weighing 200–250 g. Rats were housed in cages (three rats/cage) and allowed free access to tap water and standard rat chow diet for 2 weeks before the start of the experiment to acclimatize to the laboratory environment. The facilities were maintained at 24 ± 2°C and 12-h dark:light cycle. The experiment was adherent to the ethical guidelines of the local ethical committee and approved by the board of the Faculty of Medicine, Minia University, Egypt.
Experimental design
Rats were divided randomly into 4 groups (nine animals each) as follows: (I) normal control (CON group) received vehicle (distilled water), (II) model group (MTX group) given MTX (20 mg/kg) single i.p. dose on 11th day, 13 (III) MTX + NLD group treated with NIC (3 mg/kg/day) 8 orally for 2 weeks and MTX (20 mg/kg) 14 single i.p. dose on 11th day, and (IV) MTX + NHD group treated with NIC (10 mg/kg/day) 8 orally for 2 weeks and MTX (20 mg/kg) single i.p. dose on 11th day.
Sample collection and storage
By the end of the experiment, rats were weighed and then euthanized under anesthesia by using urethane hydrochloride (1 g/kg i.p.). Blood from each rat was collected from abdominal aorta and centrifuged (5000 r/min for 10 min) for clear sera collection. Testes were removed, washed with saline solution, and then weighed on Mettler Toledo scale, Switzerland (Glas-Col homogenizer, Rockville centre, New York, USA). One testis was immersed in Bouin’s solution for histopathology and immunohistochemistry. The other testis was frozen in liquid nitrogen, stored at −80°C, and then homogenized in ice-cold Tris HCl buffer for biochemical measurements. Tissue homogenate was centrifuged at 4000 r/min for 15 min at 4°C and then the supernatant was kept at −80°C till used.
Evaluation of serum testosterone and TAC
Serum testosterone was measured using ELISA kit (Cayman Chemicals) according to the manufacturer’s recommendations. It is based on the competition between testosterone and a testosterone acetylcholine esterase conjugate. TAC was colorimetrically assessed by using commercially available kits according to the manufacturer’s instructions.
Evaluation of testicular tissue malondialdehyde and reduced glutathione
Malondialdehyde (MDA) was measured depending on a reaction with thiobarbituric acid. The colored complexes were detected spectrophotometrically at 535 nm and calculated by using the MDA standard curve of 1,1,3,3-tetramethoxy propane. 15 Reduced glutathione (GSH) determination method is based on the Ellman’s reagent reduction by thiol groups of GSH to form a yellow color determined spectrophotometrically at 412 nm. 16
Measurement of testicular TNF-α, IL-1β, and P-gp levels
They were determined by ELISA kit according to the manufacturer’s instructions.
Histopathological assessment
After anaesthesia, testes were dissected, fixed in Bouin’s solution, processed, and then embedded in paraffin wax. Five micrometer serial sections were cut and slides were stained with hematoxylin and eosin. The stained sections were graded semiquantitatively for both histopathological changes affecting the seminiferous tubules according to Cosentino’s score (Table 1) 17 and for spermatogenesis according to Johnsen’s scoring system (Table 2). 18 Evaluation was done with light microscopy (Olympus microscope, Japan). The examination of the slides was performed in a blind fashion.
Histopathological grading of seminiferous tubules (Cosentino’s score).

Spermatogenesis grading system (Johnsen’s score).

Immunohistochemistry of eNOS
Immunohistochemistry was performed on 5 µm thick sections placed on positively charged slides. Tissue sections were de-paraffinized by xylene, rehydrated in descending grades of ethyl alcohol. Endogenous peroxidase was blocked with 3% hydrogen peroxide for 30 min, washed in phosphate-buffered saline (PBS) solution (pH 7.4). Then, slides were boiled in citrate buffer (pH 6.0) in microwave for antigen retrieval. Slides were incubated in a humidity chamber using eNOS primary antibody (dilution of 1:100) overnight and then rinsed in PBS. The biotinylated secondary antibody was applied for 30 min after that sections were washed in PBS and incubated with streptavidin-biotin complex reagent for 30 min. After rinse in PBS, 3,3-diaminobenzidine tetrahydrochloride was applied for 10 min and then washed in distilled water. Lastly, slides were counterstained with Mayer’s hematoxylin, dehydrated in ascending ethyl alcohol, cleared in xylene, mounted, and cover slipped. For negative control, PBS was added with omission of the primary antibody. Screening and scoring of immunoexpression was performed with light microscope (Olympus). Staining of the cytoplasm by eNOS was considered positive and graded using a semiquantitative score; 0, 1, 2, and 3 for negative, weak, moderate, and strong positivity, respectively. 19
Detection of p53 using Western blotting technique
For estimation of the level of p53 in testicular tissue, Western blotting was performed as previously described. 20 Briefly, the tissue was homogenized, total protein was estimated, and then an equal amount of protein from each tissue homogenate was loaded per lane and separated on a 10 % sodium dodecyl sulfate (SDS)–Tris glycine polyacrylamide gel electrophoresis (PAGE) gel. Protein bands were transferred to a nitrocellulose membrane using a semi-dry blotter (Bio-Rad, CA, USA). This blot was subsequently blocked by using Tris-buffered saline-Tween (TBS-T) buffer containing 5% skim milk powder for 1 h at room temperature. Then, the blot was probed overnight at 4°C with rabbit polyclonal anti-p53 (Neo Markers Co., CA, USA) or mouse monoclonal anti-β-actin antibodies (Sigma-Aldrich Co., St. Louis, MO, USA). Subsequently, incubation with alkaline phosphatase (AP)-coupled anti-rabbit or anti-mouse secondary antibodies (Sigma-Aldrich Co., St. Louis, MO, USA) was performed for 1 h at room temperature. Finally, blots were analyzed using 5-bromo-4-chloro-3′-indolyphosphate and nitro-blue tetrazolium colorimetric detection method (Sigma-Aldrich Co., St. Louis, MO, USA). Protein bands on the blots were analyzed using Image-J (1.48v, Wayne Rasband, National Institutes of Health, USA) and GraphPad Prism-6 for Windows (Version 6.01, San Diego, California, USA).
Statistical analysis
Statistics were expressed as means ± standard error of mean. The results were analyzed using the one-way analysis of variance followed by the Tukey test. The values of p were considered significant if less than 0.05. Statistical calculations were performed by GraphPad Prism-6 for Windows.
Results
Effect of NIC on testicular weight, serum testosterone, testicular TNF-α and IL-1β levels
There was a significant decrease in testicular weight and serum testosterone level along with a significant increase in testicular TNF-α and IL-1β levels in the MTX group as compared to the CON group. Pretreatment with NIC resulted in a significant increase in testicular weight and serum testosterone level accompanied with a significant reduction in testicular TNF-α and IL-1β levels as compared to the MTX group. The MTX + NHD group showed more improvement than the MTX + NLD group (Table 3).
Effect of NIC on testicular weight, serum testosterone, TNF-α, and IL-1β.a

NIC: nicorandil; TNF-α: tumor necrosis factor-alpha; IL-1β: interleukin-1β; CON: control; MTX: methotrexate; NLD: nicorandil low dose; NHD: nicorandil high dose; SEM: standard error of mean.
a Data are presented as mean ± SEM (n = 9).
b Significant difference from the CON group.
c Significant difference from the MTX group.
d Significant difference from the MTX + NLD group.
e Significant difference from the MTX + NHD group.
Effect of NIC on testicular oxidative stress parameters and P-gp level
The MTX group showed a significant increase in testicular MDA level along with a significant decrease in testicular P-gp level, TAC, and GSH activity compared to the CON group. Pretreatment with NIC significantly decreased testicular MDA level with a significant rise in testicular P-gp, TAC, and GSH compared to the MTX group (Table 4). The MTX + NHD group showed a significant improvement than the MTX + NLD group.
Effect of NIC on testicular oxidative stress parameters and P-gp level.a

NIC: nicorandil; P-gp: P-glycoprotein; MDA: malondialdehyde; TAC: total antioxidant capacity; GSH: reduced glutathione; CON: control; MTX: methotrexate; NLD: nicorandil low dose; NHD: nicorandil high dose; SEM: standard error of mean.
a Data are presented as mean ± SEM (n = 9).
b Significant difference from the CON group.
c Significant difference from the MTX group.
d Significant difference from the MTX + NLD group.
e Significant difference from the MTX + NHD group.
Histopathological evaluation
As shown in Figure 1, the CON group (Figure 1(a)) showed preserved testicular architecture with regular seminiferous tubules outlines which are lined by Sertoli cells and germinal epithelium representing stages of spermatogenesis. The MTX group (Figure 1(b)) showed remarkable disruption of testicular architecture in the form of irregular tubules with marked reduction in the number of germinal epithelium, sloughed germinal cells with pyknotic nuclei, and arrest of spermatogenesis. The MTX + NLD group (Figure 1(c)) showed moderate restoration of seminiferous tubules architecture in comparison to the MTX group. The MTX + NHD group (Figure 1(d)) showed obvious reduction of MTX-induced damage in the form of remarkable improvement of both testicular architecture and the number of germinal epithelial layers.

Histopathological evaluation. CON group (a) showing preserved testicular architecture with regular seminiferous tubules outlines lined by Sertoli cells and germinal epithelium. MTX group (b) featuring disruption of testicular architecture, sloughed germinal cells with pyknotic nuclei (black arrow), and arrest of spermatogenesis (blue arrow). MTX + NLD group (c) showing moderate restoration of seminiferous tubules architecture with preservation of spermatogenesis mixed with few disorganized tubules containing sloughed necrotic germinal cells (green arrow). MTX + NHD group (d) showing an obvious reduction of MTX-induced damage in form of marked improvement of testicular architecture and the number of germinal epithelium layers with preservation of spermatogenesis. (e) Histopathological scoring according to Johnsen’s score. (f) Histopathological scoring according to Cosentino’s score. Data are presented as mean ± SEM (n = 9). a,b,c,dSignificant difference from the CON group, MTX group, MTX + NLD group, and MTX + NHD group, respectively. CON: control; MTX: methotrexate; NLD: nicorandil low dose; NHD: nicorandil high dose; SEM: standard error of mean.
Histopathological scoring according to Johnsen’s score and Cosentino’s score
Johnsen’s score was significantly decreased in the MTX group in comparison with the CON group. On administration of NIC, results showed a significant improvement of the score compared to the MTX group (Figure 1(e)). On pretreatment with high dose of NIC (NHD) (10 mg/kg/day), there was a significant increase in the score than in low dose of NIC (NLD) (3 mg/kg/day). According to Cosentino’s score, the MTX group showed a significant increase in the damage affecting seminiferous tubules in comparison with the CON group. Pretreatment with NIC, there is a significant improvement of such damage and decrease in the Cosentino’s score (Figure 1(f)).
Evaluation of eNOS immunoexpression
As shown in Figure 2, the CON group (Figure 2(a)) showed weak immunoexpression of eNOS; the MTX group (Figure 2(b)) revealed negative immunoexpression; the MTX + NLD group (Figure 2(c)) showed moderate immunoexpression of eNOS; while the MTX + NHD group showed strong positive immunoexpression (Figure 2(d)).
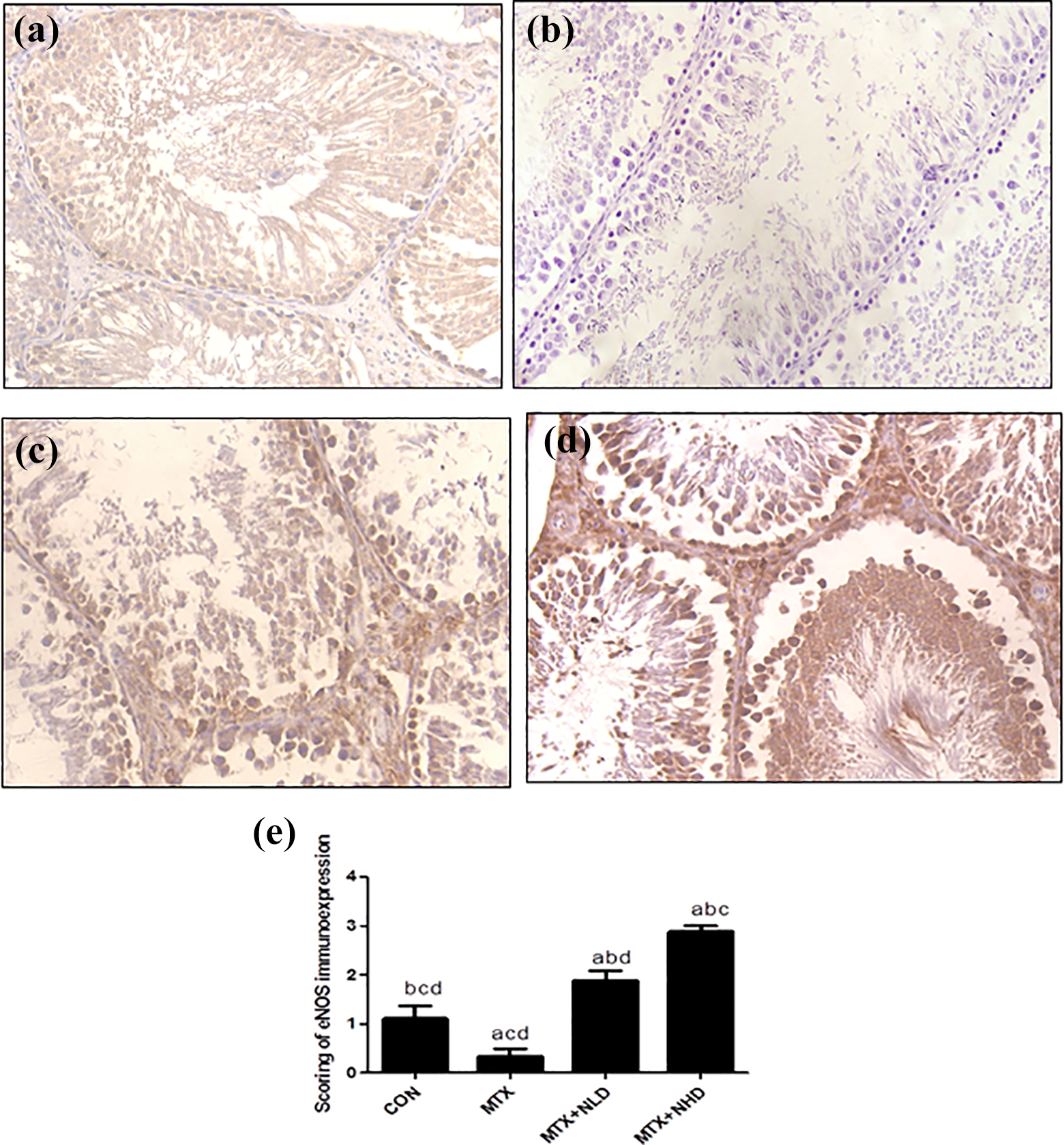
Evaluation of eNOS immunoexpression. Immunohistochemical expression of eNOS in testicular tissue showing: (a) CON group with weak eNOS immunoexpression, (b) MTX group showing negative immunoexpression, (c) MTX + NLD group showing moderate immunoexpression, (d) MTX + NHD group showing strong positive, and (e) semiquantitive analysis of eNOS immunoexpression. Data are expressed as mean ± SEM. a,b,c,dSignificant difference from the CON group, MTX group, MTX + NLD group, and MTX + NHD group, respectively. eNOS: endothelial nitric oxide synthase; CON: control; MTX: methotrexate; NLD: nicorandil low dose; NHD: nicorandil high dose; SEM: standard error of mean.
Semiquantitive analysis of eNOS immunoexpression
As shown in Figure 2(e), there was a significant decrease of immunoexpression in the MTX group in comparison with the CON group. Pretreatment with NIC led to significant increase in eNOS immunoexpression compared to the MTX group. The increase in eNOS immunoexpression is more significant in NHD than NLD.
Evaluation of p53 level using Western blotting
There is a significant increase of p53 level in the MTX group in comparison with the CON group. However, it significantly decreased on pretreatment with NIC as compared to the MTX group (Figure 3).

P53 level determined by Western blotting. There is a significant increase in p53 testicular tissue level in the MTX group in comparison with the CON group. However, p53 level significantly decreased in the MTX + NLD group and the MTX + NHD group compared to the MTX group. Data are expressed as mean ± SEM. a,b,c,dSignificant difference from the CON group, MTX group, MTX + NLD group, and MTX + NHD group, respectively. CON: control; MTX: methotrexate; NLD: nicorandil low dose; NHD: nicorandil high dose; SEM: standard error of mean.
Discussion
In our study, the protective effect of NIC against MTX-induced testicular injury was evaluated. The results revealed that NIC could ameliorate such damage and NHD (10 mg/kg/day) was more protective than NLD (3 mg/kg/day). This was detected through evaluation of testicular weight, serum testosterone, TAC, MDA, GSH, TNF-α, IL-1β, p53, eNOS immunoexpression, P-gp, and histopathological changes.
Many drugs used in cancer chemotherapy result in toxic adverse effects in different organs. 21 Testicular germinal epithelium have a high mitotic activity. Therefore, it is especially susceptible to toxicities by cytotoxic drugs. 22 It is known that chemotherapeutic agents not only affect tumor cells but also affect healthy tissues resulting in undesirable consequences, and their adverse effects on the male reproductive system can lead to infertility and sterility. 4
MTX causes an increase in free radicals and reduces the effectiveness of the antioxidant enzyme system causing the cells to become more susceptible to ROS and eventual cellular damage. Increased oxidative stress in testes causes abnormal sperm and infertility. It is one of the major causes of male infertility. 23 GSH is considered to be one of the most important enzymes involved in removal of ROS and free radicals in the male reproductive system. 4
Oxidative stress activates several intracellular signaling pathways leading to the upregulation of proinflammatory cytokine synthesis such as TNF-α and IL-1β. The severe inflammatory reaction could be attributed to the accumulation of inflammatory lymphocytes, neutrophils, and macrophages, which have been implicated in the destruction of connective tissue through the release of proteolytic enzymes and the further release of free radicals. 7 The same was found in the current study which showed a significant increase in TNF-α and IL-1β and decrease in both GSH and TAC in the MTX group.
MTX-induced oxidative stress is followed by membrane lipid peroxidation. MDA is a highly toxic molecule which is the best indicator of oxidative stress and tissue damage as it causes degradation of unsaturated fatty acids in the cell membrane. 24 Previous studies have reported that MTX led to a significant increase in MDA levels in the testis, liver, and kidneys. 25 Our study also showed an increase in the MDA levels in the testicular tissue following MTX administration.
MTX accelerates the apoptotic process, DNA damage, and uncontrolled germ cell apoptosis which could interfere with normal spermatogenesis and eventually cause infertility. MTX could reduce the seminiferous tubule length and epithelium thickness, decrease the number and mobility of sperms, and reduce the level of serum testosterone. 25 –27 In our study, MTX administration revealed testicular injury characterized by degeneration in seminiferous tubule and germ cells apoptosis with high level of an essential apoptotic marker (p53) in testicular tissue.
The blood–testis barrier contains a number of active membrane transporters which acts as efflux pumps such as P-gp that accepts MTX as a substrate. It is responsible for testicular protection and highly expressed in Sertoli cells, interstitial cells, spermatids, Leydig cells, and peritubular cells. 28,29 Our study revealed the important role of P-gp in controlling MTX testicular injury. MTX group showed a significant decrease in testicular P-gp level associated with accumulation of MTX in testicular tissue and marked injury of the testes. However, pretreatment with NIC showed marked increase in P-gp level leading to decreased MTX accumulation and protecting testes from MTX harmful effect. These results were in agreement with previous studies. 2,29
It has been stated that nanomolar concentrations of NO have anti-inflammatory and protective capacities that are mediated via the attenuation of nuclear factor kappa B (NF-κB). 30 eNOS is one of NO synthases and plays a functional role in spermatogenesis. 31 This encouraged us to examine the effect of NIC as NO donor against MTX-induced testicular toxicity. NIC increased endothelium-derived NO production by activation of eNOS, inhibited endothelial cell death, and exerted anti-inflammatory and antioxidative effects. 32
Additionally, it stimulates cytoplasmic guanylate cyclase causing an increase in the cellular levels of cyclic guanosine monophosphate, a reduction in cytosolic calcium, and consequently relaxation of vascular smooth muscle. 32 The increase in eNOS activity may contribute to the protective effect of NIC against MTX-induced testicular toxicity. The current findings revealed that MTX triggered decreased immunoexpression of eNOS in testicular tissue. However, pretreatment with NIC increased eNOS immunoexpression.
NIC, a nitrite-like drug, has been widely used as an ATP-sensitive potassium-channel (KATP) opener and a potent stimulant of guanylylcyclase and used for treatment of angina. 33 It was reported to have a significant protection in different rat models; myocardial ischemia reperfusion injury and renal toxicities. 34,35
In the current model, pretreatment with NIC preserved the testicular weights and increased the serum testosterone levels. These effects were correlated with the amelioration of biochemical and histological alterations caused by MTX. Prophylactic use of NIC in our model prevented MTX-induced ROS production in testicular tissues manifested by a significant increase in testicular level of GSH and TAC. This is in consistence with the findings of Aizawa et al. and El-Kashef. 36,37
Additionally, NIC produced anti-inflammatory action proven via a significant decrease of TNF-α compared to the MTX group and also decreased lipid peroxidation manifested by a significant decrease in testicular level of MDA. These findings are in agreement with previous studies. 12,38
Wang et al. suggested that NIC ameliorates hypoxia-induced apoptosis via unregulated eNOS expression, which in turn inhibits the NF-κB pathway and the mitochondrial apoptotic pathway. 39 Findings of our experiment observed that pretreatment with NIC upregulates the decreased protein expression of eNOS compared with the MTX group. Moreover, NIC decreased p53 testicular tissue level explaining its antiapoptotic action. These findings were consistent with He et al. 40
Conclusion
In conclusion, results of the present study revealed that NIC is a potentially beneficial protective agent against MTX-induced testicular damage via increased eNOS immunoexpression, antiapoptotic, anti-inflammatory, and antioxidant properties. Moreover, NIC high dose (10 mg/kg/day) is more protective than low dose (3 mg/kg/day). Further studies are recommended to evaluate this protective effect in human patients.
Footnotes
Author contributions
MMMR, YHA, and SS selected the point, performed the experimental part and statistical analysis, and revised the manuscript. HMK wrote and revised the manuscript, and sent it for publication. ME performed and wrote the part of histopathology, immunohistochemistry, and revised the manuscript. AMAB wrote the part of Western blotting and revised the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
