Abstract
Recent extensive evidence suggests that ambient fine particulate matter (PM2.5, with an aerodynamic diameter ≤2.5 μm) may be neurotoxic to the brain and cause central nervous system damage, contributing to neurodevelopmental disorders, such as autism spectrum disorders, neurodegenerative diseases, such as Alzheimer's disease and Parkinson's disease, and mental disorders, such as schizophrenia, depression, and bipolar disorder. PM2.5 can enter the brain via various pathways, including the blood–brain barrier, olfactory system, and gut-brain axis, leading to adverse effects on the CNS. Studies in humans and animals have revealed that PM2.5-mediated mechanisms, including neuroinflammation, oxidative stress, systemic inflammation, and gut flora dysbiosis, play a crucial role in CNS damage. Additionally, PM2.5 exposure can induce epigenetic alterations, such as hypomethylation of DNA, which may contribute to the pathogenesis of some CNS damage. Through literature analysis, we suggest that promising therapeutic targets for alleviating PM2.5-induced neurological damage include inhibiting microglia overactivation, regulating gut microbiota with antibiotics, and targeting signaling pathways, such as PKA/CREB/BDNF and WNT/β-catenin. Additionally, several studies have observed an association between PM2.5 exposure and epigenetic changes in neuropsychiatric disorders. This review summarizes and discusses the association between PM2.5 exposure and CNS damage, including the possible mechanisms by which PM2.5 causes neurotoxicity.
Introduction
The problem of air pollution has become a worldwide concern as industrialization has progressed. PM2.5 is one of the most complex and harmful air pollutants, degrading environmental quality and posing a significant risk to human health. Long-term exposure to PM2.5 increases respiratory disease, lung cancer, and cardiovascular disease mortality.1,2 It also exerts pathological effects on the central nervous system (CNS), with the brain as the primary target.
The PM2.5 sources are complex and diverse, including outdoor and indoor sources. Outdoor PM2.5 is primarily derived from heating fuels, industry, and traffic. However, indoor PM2.5 originates from fuel combustion, human activities, equipment operation, cleaning, and cooking. 3 The PM2.5 composition is also determined by its sources, with various chemical components, such as salts of sulfate (and nitrate) and carbons, contributing to its complex composition. PM2.5 can easily adsorb toxic and harmful substances, including polycyclic aromatic hydrocarbons (PAHs), heavy metals, and microorganisms, due to its small particle size, large surface area, and high activity. Carbon components, such as elementary carbon (EC) and organic carbon (OC), have been identified as the primary sources of toxicity for PM2.5, based on various studies.4,5
Recent studies have detected air pollution particles in the placenta, blood, and fetal tissue samples of mothers exposed to air pollution and in human cerebrospinal fluids (CSFs).6,7 The first pathway is blood circulation, where PM2.5 enters the lungs via the respiratory tract, travels via the blood circulation system, and eventually crosses the blood–brain barrier (BBB) to reach the brain.7,8 The second pathway is the olfactory system, where PM2.5 enters the body via the nasal cavity and is transmitted via olfactory neurons to the olfactory bulb, then to the brain via the olfactory cortex.9,10 The third pathway is the gut-brain axis (GBA), where PM2.5 can enter the gastrointestinal tract directly or indirectly, changing the microbiota and intestinal barrier. These changes may lead to abnormalities in the hypothalamus-pituitary-adrenal (HPA) axis and tryptophan metabolism, activating the GBA to affect brain function.11,12
Inflammatory responses and oxidative stress have been proposed as the two primary mechanisms by which PM2.5 damages the CNS, leading to brain toxicity.13,14 Moreover, epigenetic alterations have recently been suggested as one of the major mechanisms of PM2.5-induced brain damage.
15
Numerous epidemiological investigations and animal experiments suggest that PM2.5 may be causally related to neurological or neuropsychiatric disorders, particularly neurodevelopmental disorders, neurodegenerative diseases, and mental illnesses. These diseases, whose causes are unknown and have poor treatment options, significantly diminish the quality of life and pose a substantial challenge to humanity. Therefore, it is crucial to study the potential effects of PM2.5 on the CNS. In this review, we discuss the role of PM2.5 in related CNS disorders, along with the possible underlying mechanisms. Figure 1 illustrates the impact of PM2.5 exposure on the CNS and its potential mechanisms of action.
We indexed relevant reviews and research articles from PubMed database with the following search terms: “PM2.5”, “Neurodevelopmental disorders”, “Autism spectrum disorder”, “Neurodegenerative disorders”, “Alzheimer’s disease”, “ Parkinson’s disease ”, “Mental illness”, “Schizophrenia”, “Depression”, “Bipolar disorders”, and “Air pollution”. A total of 410 articles on PM2.5 and related neurodegenerative and psychiatric diseases published between 2016 and 2023 were selected for reading. The eligible studies were selected by reading literature abstracts, and 196 literatures were finally included.
PM2.5 and neurodevelopmental disorders (NDDs)
NDDs, including autism spectrum disorder (ASD) and attention deficit hyperactivity disorder (ADHD), affect brain development and function. 16 Gestation and the early postnatal period are crucial for brain development and are vulnerable to environmental toxins. Studies have reported that particulate matter exposure during pregnancy can be transmitted to the fetus via uterine and placental functional changes, resulting in detrimental effects on neurodevelopment. 6 Our research group has established an animal model of tracheal drip PM2.5 in pregnant mice and focused on the adverse effects of PM2.5 on the development of offspring, as well as relevant intervention measures. 17 Recent epidemiological studies have revealed an association between PM2.5 exposure and ADHD development.18,19 However, relevant in vivo investigations for the underlying mechanisms are lacking. Epidemiological and animal studies involving populations from diverse geographic regions suggest that PM2.5 may be associated with ASD logy. Therefore, the following section focuses on the neurodevelopmental toxicity of PM2.5 and its possible contribution to ASD.
Association between PM2.5 and the risk of ASD
ASD begins in infancy and includes autism and various similar disorders. It is characterized by deficits in social interaction, communication difficulties, limitations in interests or activities, and stereotyped and repetitive behavior patterns. The ASD incidence has increased recently, and its development may be linked to genetic and environmental factors, although the mechanism of action is still unclear. 20 Several epidemiological studies have demonstrated that exposure to PM2.5 increases the risk of ASD.
Several studies have suggested prenatal exposure to PM2.5 is associated with ASD.21–23 However, in a national case-control study of Danish children, reported that exposure to atmospheric pollutants during early infancy, rather than during pregnancy, increases the risk of a child being diagnosed with autism by monitoring and assessing atmospheric pollutant levels in maternal exposure across multiple periods from preconception to infancy. 24 A recent multi-point survey of case-control studies reported a positive association between ASD and PM2.5 exposure in the first year of life (OR = 1.3 [95% CI: 1.0, 1.6] per 1.6 μg/m3 increase in PM2.5). 25 Additionally, a study in Shanghai, China, discovered that PM2.5 exposure (the median levels = 66.2 μg/m3) significantly increased the risk of developing ASD during the first 3 years of life. 26 A meta-analysis also suggests that PM2.5 exposure during pregnancy is associated with an increased risk of ASD in newborns, and postnatal exposure to PM2.5 is also a risk period for the disease. 27 However, several studies have limitations due to multiple confounding factors, such as social class, urban residence, maternal age, and season of conception or birth. For instance, a Canadian population-based birth cohort study discovered no significant association between PM2.5 and ASD. 28 Therefore, the relevance of PM2.5 exposure to ASD and the possible time window of sensitivity remains controversial and requires further exploration.
Mechanisms associated with PM2.5-induced ASD
Oxidative stress and neuroinflammation
Although the mechanisms underlying the neurodevelopmental toxicity of PM2.5 have not been fully elucidated, oxidative stress is currently the most researched issue. Animal models have demonstrated that exposure to specific doses of PM2.5 during pregnancy and lactation causes oxidative stress in the mother, negatively affecting offspring’s the emotional and learning memory abilities. 29 The oxidation product malondialdehyde (MDA) and the antioxidant catalase (CAT) activity are important indicators for evaluating oxidative stress processes. PM2.5 exposure has significantly reduced CAT activity and increased MDA levels in the pregnant rat serum dose-dependently. Open field tests and Morris water maze experiments suggest maternal exposure to PM2.5 may increase anxiety and cognitive impairment in offspring mice. 30 Hypothesized mechanisms associated with environmental particulate matter and ASD include inflammation and microglia activation. 31 Our research group has previously reported that PM2.5 can activate the NLRP3 inflammasome in hippocampal microglia and increase interleukin-18 (IL-18) and interleukin-1β (IL-1β) levels in a dose-dependent manner, leading to hippocampal neuronal damage.32,33
Once PM2.5 enters the brain, it triggers the reactive microglia proliferation, producing inflammatory cytokines, such as interleukin 6 (IL-6) and tumor necrosis factor-α (TNF-α), leading to a cycle of neuroinflammation and oxidative damage and possibly causing brain white matter damage.34,35 Particularly, exposure to high PM2.5 concentrations during gestation resulted in autism-like behavioral deficits in adult mice, including reduced social interaction and increased repetitive behavior. 36 Notably, the autism-like behavioral deficits observed in these animal studies have similar outcomes to those reported in human studies.30,37 Vitamin B, such as folic acid, vitamin B12, and vitamin B6, are essential for neuronal function, and their deficiency has been associated with an increased risk of neurodevelopmental disorders. 38 Therefore, vitamin B supplementation may attenuate autism-like behavior and neurodevelopmental impairment in the hippocampus of mice exposed to PM2.5 during gestation by suppressing inflammation and oxidative stress, exhibiting a preventive effect.39,40 However, the potential mechanisms underlying vitamin B’s amelioration of PM2.5-induced neurodevelopmental impairment in offspring during pregnancy require further elucidation. Vitamin B application is also expected to be a promising therapeutic for ASD.
Dysregulation of the PKA/CREB/BDNF signaling pathway
The PKA/CREB/BDNF signaling pathway has recently been implicated in PM2.5-induced neurodevelopmental toxicity. The cAMP-mediated protein kinase A (PKA) activation is essential for cell signaling. The cAMP response element binding protein (CREB) is a target for PKA that can be activated by PKA phosphorylation at serine 133. CREB, an activated transcription factor, regulates the downstream target gene expression, such as brain-derived neurotrophic factor (BDNF), thereby promoting neuronal development, inhibiting neuronal apoptosis, and enhancing synaptic plasticity.41,42 Synapses provide the structural basis for information transmission between neurons and are essential for cognition and emotion. The synapses formation characterizes the developing brain, and this stage is vulnerable to environmental toxins, 43 such as PM2.5, disrupting synaptic structures, reducing hippocampal synaptic-related proteins (PSD95 and SYP) expressions, and impairing emotional and cognitive development, especially when exposed during the early postnatal period. 44 A recent in vitro study using primary cultured hippocampal neurons as a model reported that exposure to PM2.5 increased neuronal apoptosis in a concentration-dependent manner. When hippocampal neurons cultured for 3 days in vitro (DIV3) were exposed to PM2.5 for 24 h and 96 h, neuronal viability decreased by 18.8% and 32.7% respectively. Transmission electron microscopy revealed damage to synaptic ultrastructure and reduced PKA and CREB phosphorylation and BDNF expression levels at the molecular level. 45 Our in vitro co-culture model of hippocampal neurons and microglia also exhibited significant reductions in SYP and PSD-95 and p-CREB and BDNF under PM2.5 exposure, 32 suggesting that the PKA/CREB/BDNF signaling pathway is involved in PM2.5-induced developmental toxicity in hippocampal neurons. Synaptamide has been discovered to improve synaptic plasticity and cognitive function by specifically activating the PKA/CREB pathway.46,47 Therefore, targeting the PKA/CREB/BDNF signaling pathway may offer a promising therapeutic strategy for ameliorating PM2.5-induced neurodevelopmental damage.
Gut microbiome dysbiosis
The gut microbiome regulates the host brain and body development and an immune system function throughout early human life and childhood.
48
ASD symptomatology is also directly linked to the gut microbiome.
49
Gastrointestinal symptoms are common in autistic children, affecting 46% to 84% of them.
50
Studies have presented that alterations in the gut microbiota and its metabolites can cause ASD-like behavior in rodent models and ASD symptoms in patients.51,52 Additionally, a Jinan, China prospective study demonstrated for the first time a link between PM2.5 and the GBA of the microbiota. The study found changes in neurological outcomes by measuring real-time personal PM2.5 in 76 healthy elderly participants and collecting and analyzing fecal and blood samples, e.g. increases of 19.77% (95% CI: -36.44, 125.69) in anxiety were associated with 10 μg/m3 increase in PM2.5 at the lag 0-72 h. PM2.5 exposure can cause neurological dysfunction by affecting the gut microbiota, tryptophan metabolism and activating GBA by inflammatory factors.
11
Furthermore, environmental factors during the prenatal and neonatal periods can induce dysbiosis and affect microglial maturation and activation, thereby altering brain function and increasing the risk of ASD.
12
However, further research is necessary to elucidate the specific mechanisms by which PM2.5 and gut microbiota combine to cause ASD, particularly in humans. Figure 2 illustrates the ASD pathogenesis associated with exposure to PM2.5.
Epigenetics of PM2.5 and ASD
Epigenetic inheritance is a system of molecular markers that allows two individuals with identical gene sequences to exhibit vastly different traits. Epigenetics may be associated with early environmental and genetic influences on neurodevelopmental risk due to air pollution.53,54 DNA methylation is an important epigenetic marker, and its dysregulation due to genetic or environmental damage can lead to cognitive deficits and behavioral abnormalities. 55 DNA methylation is altered in the ASD brain, suggesting epigenetic dysregulation is a potential mechanism for ASD pathogenesis.56,57 Epidemiological studies also provide strong biological evidence for a correlation between abnormal DNA methylation and PM2.5-related health effects, specifically significant genomic hypomethylation in PM2.5-exposed populations.58,59
Notably, PM2.5 has been shown to interfere with gene-specific DNA methylation and mRNA expression of ASD candidate genes and reduce the synapse-associated gene expression in human neuronal cell models. 60 Many ASD candidate genes contribute to the synapse structure and function. Dendritic spines are small actin-rich protrusions that form the postsynaptic portion of most excitatory synapses and influence synaptic function and plasticity. 61 Individuals with ASD exhibit abnormalities in the number and shape of dendritic spines, resulting in brain dysfunction. Postsynaptic density proteins (PSDs) include cell adhesion molecules, scaffolding proteins, receptors, and cytoskeletal proteins, which underpin synaptic transmission and plasticity. Thus, alterations in these proteins play an important role in ASD pathogenesis. 62
SHANK3 is a highly plausible candidate ASD gene, functioning as a synaptic scaffolding protein predominantly located in the postsynaptic region of excitatory synapses and critical for synapse formation and dendritic spine maturation. 63 Evidence from epidemiological and animal studies suggests that SHANK3 hypermutability and hypermethylation are observed in ASD patients and animal models.64–66 In an in vivo study, neonatal male Sprague-Dawley rats were chosen and exposed to PM2.5 (2 or 20 mg/kg body weight, once a day) by intranasal instillation from postnatal day 8 to 22. It was found that both groups of the exposure rats in the early postnatal period developed an autism-like state, with significantly reduced SHANK3 protein levels in the hippocampus. 67 This reduction may have been caused by PM2.5-induced up-regulation of DNA methylation levels in the SHANK3 promoter. 60 A recent study also confirmed the role of SHANK3 in concentrated ambient PM2.5 exposure-induced autism-like phenotype, providing new ideas for the etiology and prevention of autism. 68
Moreover, studies have identified a significant reduction in Reelin expression in the brains of ASD individuals and a methylation pattern of its promoter.69–71 Reelin is a signaling glycoprotein secreted in the limbic region of the developing cerebral cortex. It is essential in neuronal migration, dendritic spine formation, synaptogenesis, and synaptic plasticity. 72 Exposure to diesel exhaust (DE) during mouse development has reduced Reelin expression, disrupting cortical structures and leading to ASD-like behavior. 73 However, whether PM2.5 can influence ASD progression by regulating the Reelin gene expression remains unknown, and further investigation is needed.
PM2.5 and neurodegenerative diseases
The World Health Organization (WHO) has predicted that neurodegenerative diseases will be the greatest health problem in the 21st century, with Alzheimer's disease (AD) and Parkinson’s disease (PD) considered the most incurable neurological disorders. 74 These diseases increase dramatically with age, posing a huge medical and public health burden. Although the etiology of most neurodegenerative diseases is unclear, genetic and environmental factors play a crucial role. Extensive research indicates that PM2.5 is an important environmental risk factor for neurodegenerative diseases.75,76 This section discusses the relationship between PM2.5 exposure and AD and PD in more detail, as these diseases represent the most common neurodegenerative disorders. Most evidence points to a potential relationship between them and PM2.5.
PM2.5 and AD
PM2.5 as the independent risk factor for AD
AD is a progressive degenerative nervous system disorder with an insidious onset and is the most prevalent form of dementia. AD is pathologically characterized by amyloid plaques and neurofibrillary tangles in the brain, leading to associated loss of synapses and neurons, cognitive deficits, and eventually dementia. 77 Age is the most important factor influencing AD onset, and as the population ages rapidly, the number of people with AD increases. 78 However, the exact etiology and pathogenesis of AD are still unknown. Age-associated genetic mutations and air pollution are currently known risk factors for AD development, with PM2.5 being an independent risk factor in developing AD.76,79
The Epidemiological Relationship Between PM2.5 and AD.

Mechanisms underlying PM2.5-induced AD
Hyperactivation of microglia
Over the past decade, numerous studies have demonstrated that PM2.5 accelerates the effects of AD by inducing neuroinflammatory processes.87,88 The hallmark of neuroinflammation is immune cell activation, such as microglia and astrocytes. 89 Notably, microglial activation has been identified as a major factor in AD pathogenesis.90,91 Emerging evidence suggests that PM2.5 exposure causes neurotoxicity by activating microglia. 92 Therefore, we hypothesize that PM2.5-induced microglial activation-mediated neuroinflammation plays an important role in AD pathogenesis.
Microglia, the resident macrophages of the CNS, play a crucial role in the brain's immune system. Microglia become activated over a long period during chronic inflammation and subsequently release various inflammatory mediators and neurotoxic substances, such as tumor necrosis factor (TNF), interleukins (IL), and nitric oxide (NO), thereby leading to tau protein deposition and propagation, neuronal loss, and cognitive decline.90,93 Moreover, studies have discovered that microglia activated under PM2.5 conditions release more pro-inflammatory cytokines, such as TNF-α and IL-1β. 94 A recent study using mixed glial and neuronal cultures in vitro from the neonatal rat cerebral cortex revealed that microglia-derived TNF-α can mediate PM in air pollution-related neurodegenerative changes, thereby altering synaptic function and neuronal growth. 95 Another study in an in vitro model of AD demonstrated that PM2.5 exposure exacerbated neuronal damage and inflammation in neuron-microglia co-cultures by increasing IL-1β production. 96 Therefore, PM2.5 may have deleterious effects on AD by causing neuroinflammation through excessive microglia activation. Consequently, microglia-targeted therapy may provide the key to effective AD therapies.
Epigenetics of PM2.5 and AD
The apolipoprotein E gene (APOE) is the strongest genetic risk factor for AD. Several studies have presented that alterations in APOE low DNA methylation may be an important factor contributing to the strong effects of APOE on AD risk.97,98 Moreover, the APOE 4 allele may also play a crucial role in the effects of air pollution in some studies.99,100 A recent study suggests that APOE carriers have a stronger association with PM2.5 and dementia. 101 Clinical and epidemiological studies investigating the long-term effects of PM2.5 exposure suggest that PM2.5 may reduce genomic DNA methylation and trigger neuroinflammation.58,59 Besides nuclear DNA, PM2.5 affects mtDNA methylation, an interesting biomarker for AD. 102 An experimental study has displayed that mtDNA methylation levels in the blood of individuals exposed to PM2.5 were negatively correlated with PM2.5 concentrations. 103 Thus, the effects of PM2.5 exposure on AD progression may be related to DNA hypomethylation, particularly the hypomethylation levels of APOE4 and mtDNA.
Additionally, non-coding RNAs (ncRNAs) play a crucial role in the pathogenesis of many human diseases, including AD, and numerous AD-related ncRNAs have been identified.104,105 However, few studies have examined the relationship between PM2.5-induced AD-like changes and ncRNAs. Recent reports suggest that long non-coding RNAs (lncRNAs) and circular RNAs (circRNAs) may play an important role in PM2.5-mediated inflammatory responses. 106 In an in vivo study investigating AD-like changes in a mouse model of chronic PM2.5 exposure (50 μg/mL), dysregulation of microglia-associated lncRNAs play a key role in cognitive impairment. 107 Additionally, following system exposure to PM2.5 for 3 months in normal mice and in chronic obstructive pulmonary disease (COPD) model mice, circRNAs, such as circBbs9, can exacerbate the PM2.5-induced cellular inflammatory response by activating the NLRP3 inflammasome when overexpressed. 108 These findings suggest that ncRNAs may be involved in PM2.5-induced cognitive impairment and influence AD progression.
Current animal experiments and epidemiological studies have established a correlation between PM2.5 exposure and AD epigenetics. However, the specific epigenetic mechanisms underlying this relationship must be further explored.
PM2.5 and PD
Association between PM2.5 and the Risk for PD
PD is the second most prevalent neurodegenerative disease after AD. It is characterized by the loss of dopaminergic neurons in the substantia nigra and the formation of Lewy bodies composed of α-synuclein.109,110 When approximately 80% of dopaminergic neurons are lost, patients may experience clinical symptoms such as motor retardation and non-motor symptoms such as depression and cognitive impairment. Despite being unknown, the exact cause of PD is attributed to several factors, including aging, genetic, and environmental factors that increase the risk of PD. 111 Air pollution, particularly PM2.5, is a contributing factor to PD development. Several epidemiological studies have investigated the possible association between PM2.5 and PD.
Exposure to air pollutants, particularly PM2.5 (mean annual concentration = 9.8 µg/m3), was related to an increased risk for PD in a population-based cohort study conducted in Ontario, Canada. 112 Furthermore, short-term exposure to PM2.5 increased the risk of PD hospitalization in a case-crossover analysis. 113 Two independent studies in New York State have also demonstrated that long-term exposure to PM2.5 may lead to clinical deterioration in PD. The findings support the idea that the adverse effects of PM2.5 on PD may vary depending on particle composition.76,114 The above epidemiological studies have revealed that short- and long-term exposure to PM2.5 significantly increases the risk for PD. However, a recent meta-analysis reported no statistically significant association between long-term PM2.5 exposure and PD incidence. 115 The relationship between PM2.5 exposure and PD development should not be ignored, despite the epidemiological limitations, such as the small number of articles selected and geographical differences in this meta-analysis.
Mechanisms associated with PM2.5-induced PD
Mitochondrial dysfunction
Accumulating evidence suggests that PM2.5-induced mitochondrial dysfunction and oxidative stress may be potential biological mechanisms underlying the deterioration of PD. Previous studies have reported that PM2.5 exposure may induce ultrastructural damage to mitochondria and myelin sheaths, increase apoptosis-related protein expressions, such as Caspase-3 and Caspase-9, and impair neurobehavior. 116 Mitochondrial dysfunction has also been associated with PD, leading to oxidative damage and neuronal death. 117 Mitochondrial dysfunction promotes reactive oxygen species (ROS) production, causing α-synuclein aggregation or reduced α-synuclein degradation. Increased alpha-synuclein levels lead to mitochondrial damage and dysfunction, creating a positive feedback loop.118,119 A recent experimental study, using PM2.5 with rotenone treated PC4 cells and C57BL/6 J mice as the in vitro and in vivo models respectively, has revealed that PM2.5 exacerbates PD development both in vitro and in vivo by inducing mitochondrial dysfunction and oxidative stress. 120 This confirms that mitochondrial dysfunction may play a crucial role in PD development caused by PM2.5.
Systemic inflammation
Systemic inflammation also plays an important role in PD pathogenesis. PM2.5 particles can reach the fine bronchi and alveolar spaces via the nose due to their small size, causing lung and systemic inflammation via the circulatory system.121,122 Notably, the systemic inflammation observed in PD patients is believed to be conducive to CNS neuroinflammation.
123
PM2.5 promotes cytokine secretions, such as TNF-α and IL-1β, in the serum. These inflammatory mediators can enter the brain via the damaged BBB, activating CNS immune cells and causing downstream effects, such as neuronal damage.124,125 Animal models prove that systemic inflammation promotes neuroinflammation, leading to dopaminergic neuron loss and alpha-synuclein accumulation, particularly the formation of Lewy body-like inclusions.
126
In conclusion, PM2.5 causes neuroinflammation and induces a systemic inflammatory response that indirectly produces neurotoxicity and increases the risk of PD. Figure 3 presents the pathogenesis and clinical manifestations of PD associated with exposure to PM2.5.
PM2.5 and mental disorders
“An era of mental illnesses is upon us. " The Global Burden of Disease (GBD) 2019 report highlights that mental disorders are among the top 10 leading causes of global burden. 127 Mental illnesses are a group of neurological disorders that manifest with varying degrees of impairment in cognitive, emotional, volitional, and behavioral mental activities, including schizophrenia, depression, and bipolar disorder, among others. 128 Biological, psychological, and social-environmental factors cause mental illnesses. New evidence from human and animal studies suggests that sustained exposure to PM2.5, affecting the CNS via various cellular, molecular, inflammatory, and oxidative stress pathways, increases the risk of mental illnesses.129–131 However, research into the underlying mechanisms remains incomplete. Considering the increasing burden of mental disorders in today's highly polluted world, these findings will increase awareness of vulnerable populations and support the urgent need to reduce exposure to air pollutants.
PM2.5 and schizophrenia
Association between PM2.5 and schizophrenia
Schizophrenia (SZ) is a severe and chronic psychiatric disorder characterized by the dissonance between mental activity and the environment. It typically begins in the middle-aged and young adult stages and is marked by thinking, perception, emotion, and behavior disorders. Although the potential etiology of SZ is unknown, there is agreement that both genetic and environmental factors contribute to somatic vulnerability.132,133 Urban exposure to environmental toxins and pollutants has been identified as a reliable risk factor for SZ and other psychiatric disorders. Recent evidence indicates that exposure to air pollutants is associated with an increased risk for SZ.134,135 PM2.5 play a key role in these epidemiological studies on SZ.
The two independent time-series study results have revealed that the risk of hospital admission for SZ patients increases with PM2.5 concentrations.136,137 Another study in a coastal city in China discovered a positive association between PM2.5 exposure and hospital readmission in SZ patients, with a stronger association in men and young adults (<45 years). 138 Furthermore, a case report including 1193 patients with SZ stated that ambient PM2.5 concentrations were associated with deterioration in SZ. 139 In this study, the stratified analysis by age group showed that the ORs associated with PM2.5 concentration increased substantially for patients over 65 years of age, and significant increases were observed at a single lag of 6 days. A prospective study reported a statistically significant correlation between environmental PM2.5 and an increased risk of recurrence in SZ patients. 140 However, despite these findings, a few studies have reported a less consistent association between PM2.5 exposure and SZ risk.141,142 In contrast, data on the association between the diagnosis or severity of schizophrenia and PM2.5 are insufficient; therefore, we hypothesize a positive association between PM2.5 and schizophrenia.
Mechanisms associated with PM2.5-induced SZ
Oxidative stress
Emerging research suggests that oxidative stress is associated with SZ pathophysiology, and deficiencies in antioxidant capacity increase the risk of SZ. 143 This prompts us to consider the role of PM2.5-induced increased oxidative stress in the risk of SZ. Total Antioxidant Capacity (T-AOC, also known as TAS) is the total antioxidant level consisting of various antioxidant substances and antioxidant enzymes, 144 which may play a role in this association. People living under conditions with a high concentration of PM2.5 have a 1.85-fold decrease in T-AOC compared to those living under relatively low concentration, reducing the body’s antioxidant capacity.145,146 A study investigating the correlation between thirty-two DSM-IV SZ paranoid patients and their T-AOC levels also reported a negative correlation between TAS levels and symptom severity in SZ patients. 147 A study employing a mediated effects model indicated that PM2.5 exposure might increase the risk of SZ recurrence and reduce the function of the antioxidant system, particularly T-AOC levels. 148 Thus, oxidative stress may mediate the association between PM2.5 exposure and SZ recurrence, with T-AOC possibly playing a key role.
Neuroinflammation and nasal inflammation
A prospective study conducted in the UK suggests that biological and psychological factors, such as neuroinflammation and social stress, may be relevant mechanisms explaining the increased risk of psychosis due to air pollution. 149 Animal model studies have revealed that PM2.5 exposure increases inflammatory cytokine levels, such as TNF-α and IL-1β, and activates microglia, promoting neuroinflammation. 88 Additionally, PM2.5 can enter the CNS via the olfactory neuron pathway and induce inflammation in the olfactory neuroepithelium, with brain effects.150,151 Olfactory disturbances, associated with cognitive abnormalities and negative symptoms in SZ patients, frequently precede neurological and psychiatric manifestations.152–154 According to a recent inducible olfactory inflammation (IOI) mouse model study, air pollutants, including PM2.5, cause local inflammation in the nasal cavity, affecting brain function and leading to SZ-related neurobehavioral consequences. 155 Therefore, current evidence supports the intriguing hypothesis that PM2.5 may induce SZ via neuroinflammatory mechanisms and nasal inflammation, leading to pathological effects on the CNS.
Dysregulation of the WNT/β-catenin pathway
Dysregulation of the WNT/β-catenin pathway may be associated with neuroinflammation in SZ.156,157 The WNT/β-catenin signaling pathway is highly conserved and tightly controlled, regulating embryonic development, cell proliferation and differentiation, epithelial-mesenchymal transition (EMT), and metabolic engagement. 158 Down-regulated WNT/β-catenin signaling is necessary and sufficient for microglia activation, 159 and glycogen synthase kinase-3 beta (GSK-3β) is a major inhibitor of this pathway. 160 Several studies have presented dysregulation of WNT/β-catenin pathway-related protein levels in the hippocampus of mouse SZ models, manifesting primarily as decreased β-catenin and increased GSK-3β., WNT gene expression is also impaired in SZ patients. 161 Some findings suggest that disrupting the WNT/β-catenin pathway may initiate the neurotoxic process in SZ patients.162,163 PM2.5 is associated with the WNT/β-catenin pathway activation.164,165 A study using healthy 7-week-old SPF SD rats and their offspring as an in vivo model showed that early-life exposure to PM2.5 can disrupt the synaptic plasticity and reduce the β-catenin and p-GSK-3β protein levels in the WNT/β-catenin pathway of the hippocampus in offspring rats. 166 These results suggest that PM2.5 may play a pivotal role in the SZ pathophysiology by disrupting the WNT/β-catenin pathway, and GSK-3β may be a potential therapeutic target for SZ.
PM2.5 and mood disorders
Mood disorders are a group of mental illnesses characterized by a simultaneous decline in mood, energy, and motivation, including depression, bipolar disorder, and anxiety spectrum disorders.167,168 The complex etiology of mood disorders is due to biological, psychological, and social factors. Human and animal investigations suggest that PM2.5 exposure can negatively affect mood and increase the risk of mental disorders. 169 However, the specific mechanisms underlying depression and bipolar disorder involving PM2.5 remain unexplored. Therefore, the following sections focus on the possible contributions of PM2.5 to these two diseases.
PM2.5 and depression
Association between PM2.5 and the Risk of Depression
Depression, also known as the “common cold” of psychiatry, is the most common mental disorder, characterized by core symptoms such as persistent depressed mood and decreased interest. If left untreated, depression can worsen and eventually develop into major depressive disorder. Depression etiology is unknown, but it is widely accepted that biological, psychological, and social environmental factors play a role in its pathogenesis. Genome-wide association studies (GWAS) have revealed a moderate genetic correlation between major depressive disorder and SZ, suggesting a potential common etiology or mechanism between both. 170 In the previous section, we identified a positive association between PM2.5 and SZ, and increasing scientific evidence supports the correlation between PM2.5 and depression.
The Epidemiological Relationship Between PM2.5 and Depression.
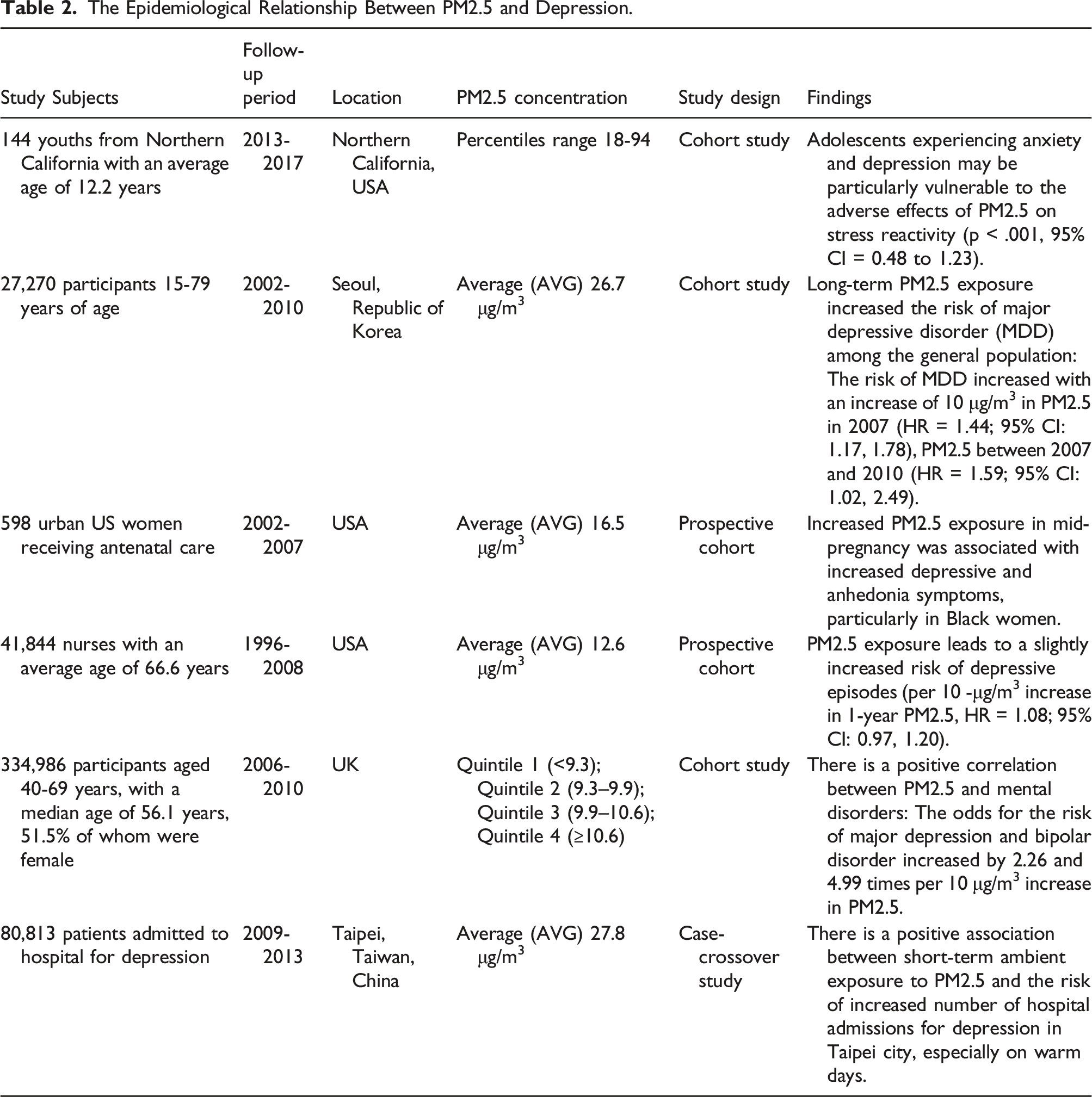
Mechanisms associated with PM2.5-induced depression
Dysregulation of the Nrf2/NLRP3 signaling pathway
Nrf2/NLRP3 signaling pathway, regulating inflammation, may play a critical role in PM2.5-induced depression. 177 PM2.5 exposure can activate Nrf2, an important transcription factor that regulates intracellular defense against oxidative stress, thereby enhancing resistance to oxidative stress. 178 However, Nrf2 deficiency promotes PM2.5-induced inflammatory responses via up-regulation of astrocyte activation and nerve damage. 179 Regarding the interaction between oxidative stress and inflammation, the key pro-inflammatory effects also must be mediated by NLRP3 inflammatory vesicle activation. The NACHT, LRR, and PYD domains-containing protein 3 (NLRP3) inflammasome is a multimeric protein that mediates caspase-1 activation and secretion of the pro-inflammatory cytokine IL-1β/IL-18 in response to microbial infection and cellular injury. 180 The NLRP3 inflammasome has been reported to be a central mediator of depression induced by the immune inflammatory response. 181 PM2.5 induces an inflammatory response via NLRP3 inflammasome pathway activation, which requires Nrf2.182,183 Therefore, we hypothesize that PM2.5 exposure can mediate inflammation and oxidative stress via the Nrf2/NLRP3 signaling pathway, leading to depression.
Hippocampal inflammation
The mood disorders caused by PM2.5-induced hippocampal inflammation have attracted significant attention. Animal models have demonstrated that PM2.5 exposure up-regulates cytokines, such as IL-1β, IL-6, TNF-α, and chemokines, in the hippocampus. 184 Elevated inflammatory cytokine levels in the hippocampus can lead to estrogen deficiency and depression-like behavior in rodents. 185 Additionally, abnormal feedback mechanisms between mood-sensitive glucocorticoids (GC) and hippocampal glucocorticoid receptors (GR) play an important role.169,186 The hypothalamic-pituitary-adrenal (HPA) axis is the primary stress response system responsible for regulating GC. A hyperactive HPA axis and hypercortisolism are also important biological features of depression. The GR in the hippocampus inhibits HPA axis activity and subsequent GC production via negative feedback under normal physiological conditions.187,188 In a study in the mouse model, it was found that PM2.5 exposure reduces the GR expression level in the hippocampus, leading to decreased negative feedback to the HPA axis and increased GC secretion. The human test in this study also showed an increase in plasma cortisol secretion after exposure to severe air pollution. This activates the inflammatory response in the hippocampus and ultimately leads to marked depression-like behavior. 169 Notably, the GC concentrations in the saliva samples of human subjects suffering from depression exhibited the same trend as that observed in animal models, supporting this hypothesis.189,190
PM2.5 and bipolar disorder
Bipolar disorder (BD) is characterized by alternating episodes of mania or hypomania and depression. BD has a peak onset between 15 and 19 years and is one of the leading causes of disability in young people. The causes of BD are unknown, but genetic, neurobiochemical, and psychosocial factors play a role in its development. BD has a high heritability rate (approximately 70%) and is strongly associated with schizophrenia and depression.191,192 Meanwhile, the impact of environmental factors on these disorders is receiving widespread attention. Several pieces of evidence now assess the association between air pollution and BD.
A cross-sectional study conducted in Milan, Italy, reported that short-term exposure to air pollution was associated with the severity of manic episodes in hospitalized bipolar disorder patients. 193 Furthermore, a systematic review and meta-analysis presented a statistically significant correlation between PM2.5 and multiple adverse mental health outcomes, including bipolar disorder. 194 The risk of BD increased 4.99 times 174 for every 10 µg/m3 increase in PM2.5. 174 Despite the positive results of current research on the association between PM2.5 and BD, there is still a scarcity of research, and the understanding of the underlying mechanisms remains incomplete. It is well-known that increasing urban greenery, coverage helps reduce PM2.5 concentrations. 195 Exposure to larger and closer green spaces may decrease the risk for BD. 196 Therefore, a comprehensive understanding of the association between PM2.5 and BD and the relevant intervention strategies should receive more attention.
Conclusions and Future Perspectives
In summary, air pollution is a significant environmental health concern. PM2.5, one of the most critical components of air pollutants, is highly linked with developing CNS disorders. PM2.5 exposure can adversely affect the CNS via several pathways, including the BBB, olfactory system, and GBA. The primary adverse outcomes associated with PM2.5 exposure are ASD, AD, PD, SZ, depression, and BD. However, some studies did not discover a significant association, most epidemiological studies have reported a positive association between PM2.5 exposure and these diseases. Animal and clinical studies have provided new evidence that short- or long-term exposure to PM2.5 may cause neurotoxicity via mechanisms, such as neuroinflammation, oxidative stress, systemic inflammation, and intestinal flora dysbiosis. Promising therapeutic targets for alleviating PM2.5-induced neurological damage include inhibiting microglia overactivation, regulating gut microbiota with antibiotics, and targeting signaling pathways, such as PKA/CREB/BDNF and WNT/β-catenin. Additionally, several studies have observed an association between PM2.5 exposure and epigenetic changes in neuropsychiatric disorders.
In this review, we provide comprehensive information regarding the role of PM2.5 in neurodevelopment and neurological disorders. Through the meticulous collection of extensive epidemiological data and evidence from both in vivo and in vitro studies, we elucidate the associations and potential mechanisms of action linking PM2.5 exposure to a variety of neurological disorders. These elucidated mechanisms, operating at both the cellular and molecular levels, significantly contribute to the existing theory of PM2.5 neurotoxicity, while also highlighting potential targets for future interventions and treatments of PM2.5-mediated diseases. Nonetheless, it is important to acknowledge the limitations of our study and address several crucial issues to further enhance our understanding of the effects of PM2.5 on the brain. For instance, human epidemiological studies possess inherent limitations, such as the identification of critical 'windows of susceptibility' for PM2.5 exposure, gender differences in potential vulnerability, and possible gene-environment interactions. Furthermore, the complexity of environmental PM2.5 underscores the incomplete nature of our current understanding of the underlying mechanisms, necessitating further exploratory research.
Given the increasing burden of CNS diseases in our polluted world, further targeted and effective interventions are needed to mitigate its detrimental effects on human health. For instance, advocating for the promotion of energy-efficient and environmentally friendly lifestyles and consumption patterns can significantly contribute to environmental preservation. Furthermore, optimizing policies aimed at reducing the permissible levels of PM2.5 emissions holds great potential in effectively preventing and controlling PM2.5 pollution. By implementing these measures, we can collectively work towards mitigating the impact of PM2.5 pollution and safeguarding public health.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Shandong Province (No.ZR2021MH031) and Weifang Medical University Scientific Research Innovation Program Doctoral foundation (No.2021BKQ)>.
