Abstract
The current research was performed to evaluate the ameliorative effects of Rhamnetin (RHM) on polystyrene microplastics (PS-MPs)-instigated testicular dysfunction in male albino rats. 48 albino rats were distributed in four groups, i.e., control, PS-MPs treated, PS-MPs + RHM co-treated and RHM only supplemented group. PS-MPs exposure considerably reduced anti-oxidant enzymes i.e., catalase (CAT), glutathione peroxidase (GSR), superoxide dismutase (SOD) and glutathione reductase (GPx) activities. Whereas, reactive oxygen species (ROS) level along with malondialdehyde (MDA) was considerably escalated in PS-MPs treated rats as well as a potential decline was observed in sperm progressive motility. Additionally, a substantial upsurge was noticed in the count of dead sperms, deformity in the tail, mid-piece and head of sperms in PS-MPs treated rats. PS-MPs exposure also decreased steroidogenic enzymes, 17β-hydroxysteroid dehydrogenase (17β-HSD), steroidogenic acute regulatory protein (StAR) and 3β-hydroxysteroid dehydrogenase (3β-HSD) expressions. Moreover, the levels of inflammatory indices i.e., Interleukin-6 (IL-6), Nuclear factor kappa-B (NF-κB), Interleukin-1β (IL-1β), tumour necrosis factor-α (TNF-α) and cyclooxygenase-2 (COX-2) activity were also increased in PS-MPs administrated group. Besides it increased the expression of apoptotic markers (Bax and caspase-3) expression. Whereas, anti-apoptotic marker i.e., Bcl-2 expression was reduced. Moreover, luteinizing hormone (LH), follicle-stimulating hormone (FSH) as well as plasma testosterone levels were also decreased. PS-MPs exposure also led to a substantial histopathological damage in testicular tissues. However, RHM supplementation potentially reduced the damaging effects of PS-MPs in the reproductive tissues of male albino rats. Thus, the current study revealed, RHM possesses potential to prevent PS-MPs-induced testicular damage due to its anti-oxidant anti-apoptotic, anti-inflammatory as well as androgenic properties.

Introduction
Environmental contamination and industrial exposure to numerous toxicants contribute to male infertility. 1 Microplastics (MPs) are among the most emerging environmental toxicants reported in the last few years. 2 Polystyrene microplastics (PS-MPs) are one of the types of MPs3,4 that is most commonly used due to its low cost and outstanding physical properties. 5 PS-MPs are important constituents of disposable cups, toys, paper clips and trays. 2 Human exposure to PS-MPs occurs through drinking water, skin, inhalation and ingestion.6,7 Small size, low density and large relative surface area helps the internalization of PS-MPs into the biota and therefore, they accumulate in the organic food chains. 8 Inappropriate handling and durability are the reasons that cause PS-MPs to become one of the most serious problems of the environment. 9 PS-MPs is a major environmental risk thus, it is named as white pollutant of the environment. 10
Previous literature have shown that PS-MPs exposure led to neurotoxicity, 11 gastrointestinal toxicity, 12 hepatotoxicity, 13 nephrotoxicity, 14 reduced fecundity, abnormal fertilization, embryo dysgenesis 15 and testicular dysfunction. 16 It is reported that PS-MPs exposure induces oxidative stress (OS) as well as inflammation in the testes that results in reduced testosterone level by damaging the leydig cells and decreasing the viable epididyme sperm as well as increasing the rate of sperm deformity in rats. 17 Exposure to PS-MPs can induce histological changes in the testes 18 as well as it may decrease the diameter of seminiferous tubules along with epithelial height. 19
Natural products are the source of most of the active ingredients of medicines. This is widely accepted to be true when applied to drug discovery and more than 80% of drugs are natural products. 20 Flavonoids (plant-based bioactive compounds) have attracted a lot of interest during the last few years due to their diverse pharmacological properties such as anti-oxidant, anti-apoptotic and anti-inflammatory. 21 Flavonoids are used by plants for their defence against plaques, abiotic well as biotic stresses, act as UV filters and for their growth.22,23 They are considered to be safe with little side effects. 24
Rhamnetin (RHM) is one of the important flavonoid that is naturally present in some fruits such as clove, apple, sour cherries and vegetables. 25 RHM displays neuro-protective, 26 anti-tumor, 27 anti-inflammatory, 25 and anti-oxidant properties. 28 By keeping these potential biological activities of RHM under consideration, the recent study was planned to elucidate the potential protective effects of RHM against PS-MPs-instigated reproductive dysfunction in male albino rats.
Material and methods
Chemicals
Both PS-MPs and RHM were obtained from Sigma-Aldrich, (Germany).
Animals
The current study was performed on 48 male albino rats 6–8 weeks old (200–220 g weight). Animals were kept at standard temperature of (22–25°C) with 12 h light/12 h dark period in the animal house of University of Agriculture, Faisalabad (UAF). Rats were given tap water and standard feed. Animals were treated according to the European Union of Animal Care and Experimentation (CEE Council 86/609) approved protocol.
Experimental protocol
Rats were distributed into four groups (n = 12/group). The rats were treated with the following doses: Control group (0.01% DMSO), PS-MPs treated group (0.01 mg/kg. of PS-MPs dissolved in 0.01% DMSO by oral gavage), PS-MPs + RHM treated group (0.01 mg/kg. of PS-MPs and 50 mg/kg. of RHM both dissolved in 0.01% DMSO) and RHM only supplemented group (50 mg/kg. of RHM orally dissolved in 0.01% DMSO). The experiment was carried out for 56 days. At the last day of experiment, rats were anesthetized using 60 mg/kg of ketamine and 6 mg/kg of xylazine, decapitated and blood was stored in sterile tubes. Blood centrifugation was performed for 15 min at 3000 rpm. Plasma was kept at −20°C after separation till analysis. The left testis was kept in 10% formalin solution for histopathological assessment. Whereas, the right testis was stored at −80°C for biochemical assessment. Testicular tissues were homogenized in Na3PO4 buffer at 12,000 rpm for 15 min at 4°C. Finally, obtained supernatant was used to estimate several parameters.
Enzymatic antioxidant status
CAT activity was evaluated according to the protocol of Aebi. 29 Absorbance changes in the mixture were observed at 240 nm and its final values were expressed as unit/mg protein. SOD activity was evaluated by inspecting the change in color intensity (at 560 nm) in accordance with the protocol of Kakkar et al. 30 GPx activity was assessed according to the method of Rotruck et al. 31 Its final values were expressed as unit/mg protein. GSR activity was estimated by computing NADPH disappearance at about 340 nm according to the protocol of Carlberg and Mannervik. 32 The values were displayed as nM NADPH oxidized/min/mg tissue. Whereas, the concentration of ROS was measured according to the protocol of Hayashi et al. 33 as well as the level of MDA was determined using commercial kits.
Sperm analysis
The semen samples were collected from the caudal part of the epididymis. To obtain sperm from the epididymis, the epididymal portion was first minced in 5 mL of physiological saline and then heated for 30 min at 37°C. Using a 400X phase-contrast microscope, sperm progressive motility was observed. 34 Eosin and nigrosin staining was used to determine the viability of the sperm, followed by microscopic analysis. Moreover, epididymal sperm were counted using a hemocytometer. 35 Additionally, Cao et al. methods was employed to measure the structural deformities of the tail, mid-piece and head of sperm. 36
Hypo-osmotic swelling test
The integrity of the morphological sperm plasma membrane was determined using HOS test. In this procedure 20 μL of semen sample was mixed with 180 μL of fructose solution at an osmotic pressure of 80 mOsm/L for about 20 min. The sperm were then stained with eosin and nigrosin following further incubation and mixing. Lastly, 200 sperms with as well as without swollen tails were observed under ×400 magnification. 37
RNA extraction and realtime quantitative reverse transcription-polymerase chain reaction (qRT-PCR)
Primers sequences for the real-time quantitative reverse transcription-polymerase (RT-qPCR).

3β-hydroxysteroid dehydrogenase (3β-HSD); 17β-hydroxysteroid dehydrogenase (17β-HSD); Steroidogenic acute regulatory protein (StAR)
Hormonal analysis
The level of LH, FSH, and plasma testosterone was assessed via enzyme-linked immunosorbent assay (ELISA) kits (Los Angeles, CA USA).
Inflammatory markers assessment
The inflammatory indices were measured using commercially available kits. The levels of IL-6, NF-κB, IL-1β, TNF-α and COX-2 activity were measured using a rat ELISA kit (Shanghai YL Biotech Co. Ltd., China). The analyses were carried out using an Elisa Plate Reader as per the manufacturer’s guidelines (BioTek, Winooski, VT, USA).
Histopathology
For the purpose of histopathological assessment testicular tissues were taken from the left testis of the rats. Cold saline solution was used for the cleansing of testes and later on fixing of the tissues was performed in 10% formalin, the tissues were gradually dehydrated by passing through the rising grades (70%, 90% and 100%) of ethanol and embedding was performed in paraffin wax. 820 Spencer rotatory microtome was employed to cut 5 μm thick slices and then stained using hematoxylin-eosin (dissolved in 70% alcohol). At the end, the slides were examined under a light microscope (Nikon, Japan), which was furnished with an automatic microphotographic system. ImageJ2X software was applied to observe the images.
Statistical Analysis
The obtained data was displayed as Mean ± SEM. Data was statistically analysed using one way analysis of variance (ANOVA) followed by Tukey’s test. p < 0.05 was considered as statistically significant.
Results
Effect of PS-MPs and RHM on OS and anti-oxidant enzymes
Mean ± SEM of biochemical markers in the testicles of control, PS-MPs treated, co-treated and RHM supplemented groups.

Values having different superscripts are substantially (p < 0.05) changed from other groups.
Effects of PS-MPs and RHM on sperm progressive motility, dead sperms and abnormalities in the sperm head, mid-piece, and tail
Mean ± SEM of epididymal sperm count, progressive motility, viability, the structural abnormalities of sperm and hypo-osmotic swelled sperm count in control, PS-MPs treated, co-treated and RHM supplemented groups.

Values having different superscripts are substantially (p < 0.05) changed from other groups.
Effect of PS-MPs and RHM on the expression of 3β-HSD, 17β-HSD, and StAR
As displayed in Figure 1, steroidogenic enzymes 17β-HSD, StAR and 3β-HSD expression was substantially (p < 0.05) reduced in PS-MPs administrated rats in contrast to control rats. However, PS-MPs + RHM co-treatment considerably (p < 0.05) upregulated the expression of these enzymes in contrast to PS-MPs administered rat. Whereas, steroidogenic enzymes expression in RHM only administrated group was comparable to control group. Displaying the effects of PS-MPs and RHM on the expression of (a) 3β-HSD, (b) 17β-HSD, and (c) StAR. The bar graphs are composed based on Mean + SEM values. Multiple indexes on bar graphs are displaying meaningful differences at p < 0.05. Polystyrene microplastics (PS-MPs); Rhamnetin (RHM); 3β-hydroxysteroid dehydrogenase (3β-HSD); 17β-hydroxysteroid dehydrogenase (17β-HSD); Standard error of the mean (SEM).
Effect of PS-MPs and RHM on the expression of apoptotic markers
As shown in Figure 2, apoptotic markers Bax and Caspase-3 expression was potentially (p < 0.05) upsurged in PS-MPs treated rats in contrast to the control rats. Whereas, the expression of Bcl-2 was reduced in PS-MPs administrated rats. Nonetheless, the administration of PS-MPs + RHM significantly (p < 0.05) downregulated Bax and Caspase-3 expression and upregulated Bcl-2 expression in co-administrated group and RHM only administrated group showed the values near to the control rats. Representing the effects of PS-MPs and RHM on the expression of (a) Bax, (b) Bcl-2, and (c) Caspase-3. The bar graphs are composed based on Mean + SEM values. Multiple indexes on bar graphs are displaying meaningful differences at p < 0.05. Polystyrene microplastics (PS-MPs); Rhamnetin (RHM); Standard error of the mean (SEM).
Effect of PS-MPs and RHM on LH, FSH and Plasma testosterone levels
Mean ± SEM of hormonal axis in control, PS-MPs treated, co-treated and RHM supplemented groups.

Values having different superscripts are substantially (p < 0.05) changed from other groups.
Effect of PS-MPs and RHM on inflammatory markers
Mean ± SEM of inflammatory markers in control, PS-MPs treated, co-treated and RHM supplemented groups.
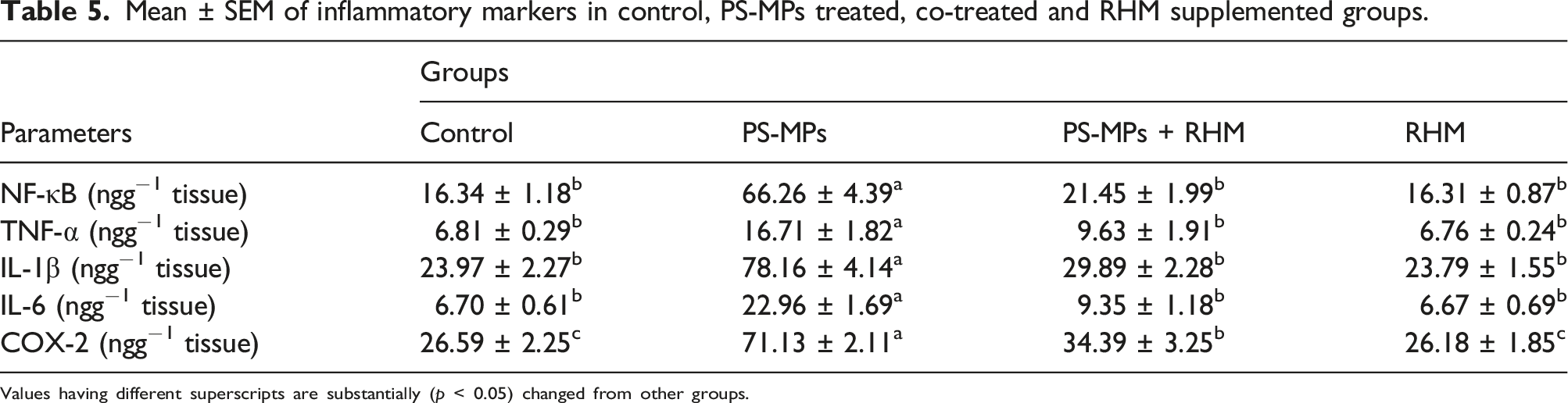
Values having different superscripts are substantially (p < 0.05) changed from other groups.
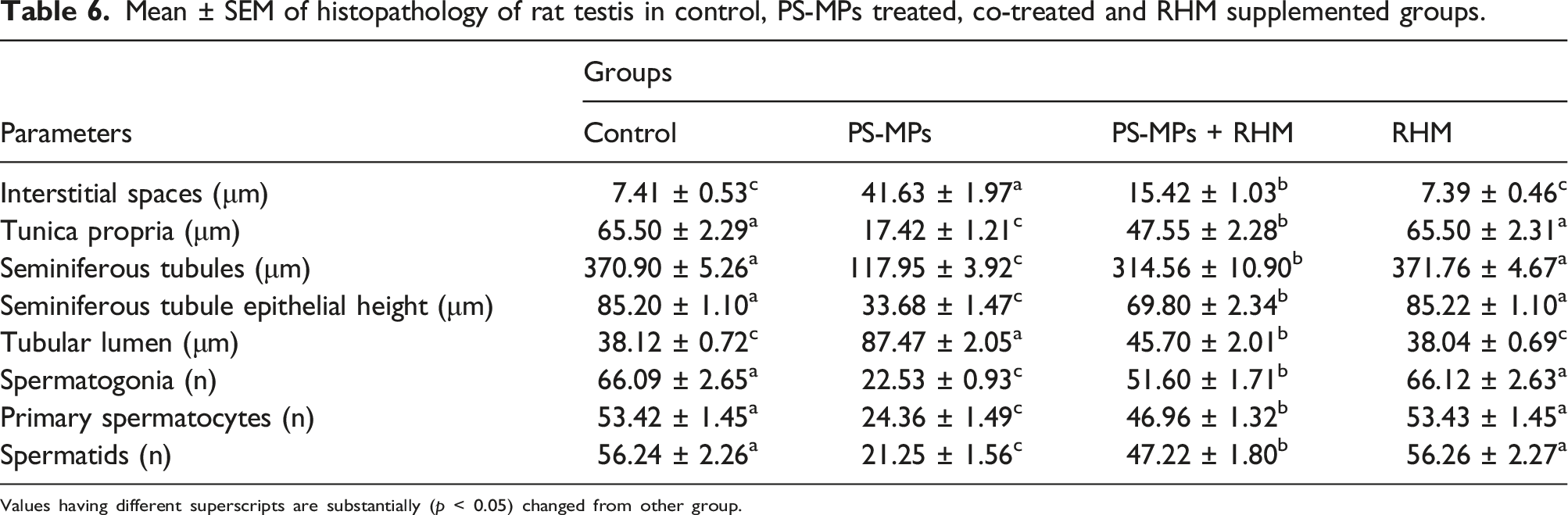
Effect of PS-MPs and RHM on histomorphometry of testicular tissues
As presented in Figure 3, PS-MPs administration considerably (p < 0.05) decreased the diameter of seminiferous tubules as well as seminiferous epithelial height, in addition to tunica propria width. Though, a significant (p < 0.05) escalation was observed in tubular lumen and interstitial spaces in PS-MPs intoxicated rats in contrast to control rats. Co-administration with RHM, considerably (p < 0.05) improved these detrimental morphometric irregularities and raised the diameter of seminiferous tubules + epithelial height along with tunica propria height, besides interstitial spaces as well as tubular lumen were reduced in PS-MPs + RHM treated group in comparison to PS-MPs administrated group. Apart from structural impairment, PS-MPs exposure also noticeably reduced the number of multiple stages of germ cells such as, spermatogonia, primary and spermatids in comparison to control group. While, the co-administration of rats with PS-MPs + RHM resulted in considerable (p < 0.05) rise in the count of all germ cells in contrast to PS-MPs administrated rats. The group treated only with RHM, showed regular count of germ cells and efficient spermatogenesis as in the control rats (Table 6). Micro-images of the adult albino rat testes (H&E, 400X): (a) Control group displaying normal germinal epithelium including different stages of germ cells and the tapered luminal area containing spermatozoa; (b) PS-MPs-intoxicated group showing sloughing of the epithelial layer, vacant lumen, and degenerated area of IS; (c) PS-MPs + RHM co-treated group showing restoration in epithelial part, and TL filled with ST as well as retrieved the deteriorated IS; (d) RHM supplemented group displaying compacted seminiferous tubules with less IS and luminal part filled with all stages of spermatogenic cells thus improved spermatogenesis. PS-MPs: Polystyrene microplastics; RHM: Rhamnetin; IS: Interstitial spaces; TL: Tubular lumen; EH: Epithelial height; SG: Spermatogonia; PS: Primary Spermatocytes; ST: Spermatids. Mean ± SEM of histopathology of rat testis in control, PS-MPs treated, co-treated and RHM supplemented groups.
Values having different superscripts are substantially (p < 0.05) changed from other group.

Discussion
The recent study was intended to evaluate the attenuative effects of RHM against PS-MPs-instigated testicular dysfunction in adult male albino rats. PS-MPs exposure can deteriorate the quality as well as the quantity of sperms in adult individuals. 39 Mammalian exposure to PS-MPs occurs through inhalation and ingestion. Since PS-MPs are ubiquitous in the environment, exposure to them is almost inevitable. 6 Human exposure to PS-MPs via aquatic food chain is well known. Moreover, PS-MPs have also been reported in the beer, honey and salt. 40 The natural plant derivative flavonoid RHM has numerous therapeutical properties and it is an important component of various foods. 21 RHM has been reported as an antioxidant compound that can potentially scavenge the free radicals to reduce OS. 41 Moreover, RHM displays anti-inflammatory and anti-tumar activities.25,27
PS-MPs decreased antioxidant enzymes (CAT, GPx, SOD and GSR) activity in PS-MPs-intoxicated rats, while upsurged MDA as well as ROS level. Antioxidant enzymes are the 1st line of defence, which lowers ROS generation and protect the biological components (proteins, DNA and lipids) from damage. 42 The antioxidant defence system in semen comprises of CAT, GPx and SOD. 43 CAT is regarded as the key antioxidant enzyme that is involved in H2O2 catabolism. 44 SOD converts superoxide radicals into H2O2 and O2. 42 GSR retains the concentration of GSH that helps to maintain the continuous action of GPx. 45 GSH aids in the conversion of peroxides into less harmful hydroxyl compounds and effectively prevents the formation of ROS. MDA is a harmful end product of lipid peroxidation and its quantity can indicate the damage induced by lipid peroxidation and ROS in an indirect way. 46 Antioxidant enzymes are generally involved in neutralization of ROS. When ROS are produced in excess, they overwhelm the body’s antioxidants defence system and prompts the production of OS. 47 Other than endogenous antioxidant, these may also be administrated from different plants to counter OS. 48 Co-administration with RHM alleviated the harmful effects of PS-MPs by decreasing OS in the testes. The current research demonstrated that the co-administration with RHM improved CAT, GPx, SOD and GSR activity; however, MDA and ROS levels were considerably decreased. The mitigative effects of RHM on these enzymes might be attributed to its anti-oxidant potential. 41 Moreover, RHM has flavan nucleus in its structure which consist of two benzene rings attached with oxygen containing pyran ring that is likely to contribute to its curative functions. 49
The number of sperms, progressive motility and viability of sperm were reduced in PS-MPs administrated group in contrast to control group. Sperm number, progressive motility as well as viability are the indications of normal spermatogenesis and male reproductive health. 50 Male gametes are sensitive to ROS. The exposure to PS-MPs induced OS in the testicular tissues. 17 OS can influence membrane fluidity as well as sperm motility. 51 As stated earlier, the presence of excessive polyunsaturated fatty acids (PUFAs) makes the spermatozoa vulnerable to OS. 52 PUFAs are necessary for sperm activity and membrane integrity. 53 However, excessive production of ROS disturbs the viscosity and permeability of spermatozoa’s membranes, resulting in peroxidation of PUFAs. 54 OS also reduces the synthesis of ATP by affecting the mitochondria of sperm cells directly, which affects flagellar activity and prompts sperm immobility as well as apoptotic death. 55 However, RHM treatment improved the state of these spermatogenic damages.
Steroidogenic enzymes expression was investigated to ascertain the mechanism underlying the decreased testosterone level after the treatment of PS-MPs. PS-MPs decreased steroidogenic enzymes 17β-HSD, StAR and 3β-HSD expression. 56 Testicular steroidogenesis, as proposed by Raucci et al. (2014) is a critical step in the synthesis of testosterone and it is arbitrated by steroidogenic enzymes and proteins.57,58 17β-HSD, StAR and 3β-HSD play a significant role in the production of steroid hormones. 59 StAR is a rate-limiting steroidogenic protein that regulates cholesterol translocation in the membranes of mitochondria to enhance testosterone synthesis. 60 Although, conversion of this cholesterol to testosterone is catalyzed by steroidogenic enzymes 3β-HSD and 17β-HSD.61,62 Conversely, RHM treatment to PS-MPs-induced rats resulted in a significant upsurge in 17β-HSD, StAR and 3β-HSD expression.
PS-MPs exposure augmented Bax and Caspase-3 expression, whereas it downregulated Bcl-2 expression. Proteins of Bcl-2 and Caspase families are particularly important in apoptosis. 63 Bcl-2 is an anti-apoptotic protein that safeguards cells from dying as a result of apoptosis. 17 Bax, on the other hand, is a pro-apoptotic protein that acts as an antagonist and accelerates cell death. 17 These proteins alter mitochondrial membrane permeability, allowing cytochrome c to leak into the cytoplasm and activate caspase 3. 64 Caspase-3 is a member of the cysteine proteases family that is responsible for splitting cellular proteins and causing structural changes, resulting in apoptotic cell death. 7 Caspases-3 overexpression is a critical event in the regulation of the mitochondrial apoptotic pathway. 65 PS-MPs exposure induced apoptosis by increasing the expression of apoptotic proteins Bax as well as Caspase-3 and decreasing the expression of anti-apoptotic protein Bcl-2. 17 However, administration of RHM to PS-MPs treated rats resulted in a substantial decline in Bax and Caspase-3 expression as well as a substantial upsurge in Bcl-2 expression.
In the present study, LH, FSH and plasma testosterone levels in PS-MPs treated rats were substantially (p < 0.05) reduced in contrast to control group. Spermatogenesis is regulated by appropriate ratio of LH, plasma testosterone and FSH. 66 Testosterone production is an important biomarker of male reproductive health. 67 LH and FSH both are released by hypothalamus to regulate spermatogenesis. 68 LH causes Leydig cells to synthesize testosterone, while FSH promotes Sertoli cell multiplication. 69 Our results are further endorsed by the study of Jin et al. (2021) who reported that the level of testosterone was decreased following the exposure to PS-MPs. 70 The results of our research showed that the supplementation of RHM recovered the hormonal levels and eventually reinstated the process of spermatogenesis. Our results are further endorsed by the findings of Agrawal (2011) who reported that flavonoids can act as regulator on hormones i.e., thyroid hormones androgen, and estrogen. 71
The administration of PS-MPs increased the level of inflammatory markers such as, IL-6, NF-κB, IL-1β, TNF-α and COX-2 activity in PS-MPs administrated group in contrast to the control. The production of pro-inflammatory cytokines such as IL-6, NF-κB, IL-1β, TNF-α and COX-2 is associated with acute inflammatory reactions as well as other ROS linked illnesses. IL-6, TNF-α, IL-1β production is stimulated as a result of NF-κB activation, which contributes to acute impairment. 72 COX-2 is another important inflammatory marker that plays a prime role in inflammation. 73 However, the administration of RHM to PS-MPs-intoxicated group resulted in a significant decrease in IL-6, NF-κB, IL-1β, TNF-α level and COX-2 activity. This activity may be attributed to anti-inflammatory nature of RHM.
PS-MPs treatment considerably augmented the interstitial spaces and diameter of the tubular lumen. Furthermore, it also decreased the epithelial height along with the diameter of seminiferous tubules. PS-MPs substantially (p < 0.05) decreased the count of different germ cells. Previous studies have shown that reduced epithelial height is linked with reduced testosterone level, which also affects the germ cells count. 19 The disruption of the blood-testis barrier, followed by exposure to PS-MPs, may be responsible for the histopathological changes. 70 However, co-administration of RHM with PS-MPs significantly increased the count of germs cells and restored the testicular impairments due to its androgenic, antioxidant, anti-inflammatory as well as anti-apoptotic activities.
Conclusion
In conclusion, the results of our study suggested that RHM showed excellent attenuative effects against PS-MPs-instigated testicular damage. RHM administration considerably recovered anti-oxidant enzymes activities, ROS and MDA levels, regulated the level of inflammatory indices, steroidogenic enzymes as well as apoptotic markers expression and histological architecture. Taken together, it can be concluded that RHM may hold some clinical prospects in future to cure testicular dysfunctions in animals and humans.
Footnotes
Author’s contribution
AH and MUI conceived the idea and designed the study. AH and MUI performed the experiments. HA helped in statistical analysis. AH and MUI wrote the manuscript. All authors have read and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The study was approved by the Institutional Biosafety/Bioethics Committee (IBC) of the University of Agriculture, Faisalabad, in compliance with the (CEE Council 86/609) protocol.
Data availability statement
The datasets used/analyzed in this study are available from the corresponding author on reasonable request.
