Abstract
The potential of human mesenchymal stem cells (MSCs) for cell therapy has been investigated in numerous immune-mediated conditions; MSCs are considered one of the most promising cellular therapeutics to treat intractable diseases. Recently, approaches to prime MSCs have been investigated, thereby generating cellular products with enhanced potential for a variety of clinical applications. Interferon-gamma (IFN-γ) priming is a current approach used to increase the therapeutic efficacy of MSCs. In this study, we determined the systemic toxicity, tumorigenicity and biodistribution of IFN-γ-primed Wharton’s jelly-derived (WJ)-MSCs in male and female BALB/c-nu/nu mice. There were no deaths or pathologic lesions in the mice treated with 5 × 106 cells/kg IFN-γ-primed MSCs in the repeated dose study. In the tumorigenicity study, one of the subcutaneously treated mice showed bronchioloalveolar adenoma in the lung but tested negative for human-specific anti-mitochondrial antibody, suggesting the spontaneous murine origin of the adenoma. A biodistribution study using real-time quantitative polymerase chain reaction demonstrated the systemic IFN-γ-primed MSC clearance by day 28. Based on the toxicity, biodistribution, and tumorigenicity studies, we concluded that IFN-γ-primed MSCs at 5 × 106 cells/kg do not induce tumor formation and adverse changes.
Introduction
Mesenchymal stem cells (MSCs) can be obtained from various tissues 1 such as umbilical cord blood, adipose tissues, bone marrow, and Wharton’s jelly (WJ). They possess self-renewing capacity, 2 multilineage differentiation potential 3 and immunosuppressive capacity. 4 The therapeutic potential of human MSCs has been reported in numerous immune-mediated conditions in both pre-clinical and clinical studies; MSCs are regarded as one of the most promising cellular therapeutics to treat intractable diseases, including graft-versus-host diseases, 5 Crohn’s disease, 6 and type 1 diabetes. 7 However, MSCs have failed to demonstrate clear efficacy in recent clinical trials 8 owing to their heterogeneity as well as the differences in culture conditions. 9 Recently, priming approaches have been investigated to generate cellular products such as MSCs for enhanced clinical applications.
The Th1 cytokine IFN-γ plays a pivotal role in the immunomodulatory function of MSCs. Several pre-clinical studies have shown the potential of IFN-γ priming in enhancing the therapeutic effects of MSCs.10-13 Improved immunosuppressive effect of IFN-γ primed MSCs led to better outcomes in animal models for graft-versus-host diseases, colitis, and renal fibrosis. 14 Nevertheless, clinical studies using IFN-γ-primed MSCs have not yet been attempted, and it is believed that their safety should be evaluated first.
In this study, we determined the systemic toxicity, tumorigenicity, and biodistribution of IFN-γ-primed WJ-MSCs in BALB/c nude mice.
Materials and Methods
IFN-γ-primed MSCs
IFN-γ-primed MSCs were provided by Samsung Medical Center (Seoul, Republic of Korea). This study was approved by the institutional review board of Samsung Medical Center (IRB No. 2015-10-025), and all samples were obtained with informed consent. WJ-MSCs were isolated and cultivated according to the standard operating procedures of the Good Manufacturing Practice facility at the Samsung Medical Center. At the final stage of the cell manufacturing process, MSCs were primed with 200 IU/mL IFN-γ (Intermax-gamma; LG Life Sciences, Seoul, Republic of Korea) for 24 h, as described previously. 10 To characterize IFN-γ-primed MSCs, antibodies specific for the human CD14, CD34, CD45, CD73, CD90, and CD105 (BD Biosciences, CA, USA) were used for flow cytometry (FACSCalibur; BD Biosciences, San Jose, CA, USA), and the data were analyzed using CELLQUEST software (version 5.2.1., BD Biosciences, San Jose, CA, USA)
Laboratory animal
Both sexes of immunodeficient inbred BALB/c-nu/nu mice (4–5 weeks old) were purchased from Charles River Laboratories (Kanagawa, Japan) and inoculated with IFN-γ-primed MSCs at 6–7 weeks of age after 10 days of acclimation periods. The mice were allocated randomly and maintained under specific pathogen-free conditions and environmental enrichment with cotton pads. 15 Toxicity studies except biodistribution were conducted under Good Laboratory Practice conditions and reviewed and assessed by the Institutional Animal Care and Use Committee of the Korea Institute of Toxicology (Approval number: 17-1-0263). Throughout the IACUC protocols, all efforts were made to minimize the number of animals used and their suffering. Clinical signs and mortality of the animals were observed twice a day. The order of treatments and measurements was recorded. Animal staff alone was aware of the group allocation for blind analysis and performed the behavior observation. The date and time of occurrence and duration of clinical signs including general appearance and behavior changes were recorded using the Pristima System (Xybion Medical System Co., Princeton, NJ, USA). There were no excluded animals and groups for analysis.
Single-dose toxicity
Twenty-four male and female mice were randomly assigned to four groups per sex (6 mice/sex/group) treated with vehicle (V.C.), low-dose (1 × 107 cells/kg), middle-dose (5 × 107 cells/kg), and high-dose (1 × 108 cells/kg) of IFN-γ-primed MSCs. The low, middle, and high doses were selected as 10-, 50- and 100-fold higher than the anticipated therapeutic dose in patients, respectively.
16
Fresh medium was injected into the vehicle control group. The mice were inoculated intravenously through the tail vein and observed for 15-days mortality, and clinical symptoms (Figure 1a). Scheme of the animal experiment. Both sexes of BALB/c-nu/nu mice (4–5 weeks old) were administered with IFN-γ-primed MSCs intravenously for (a) the single dose toxicity, (b) repeated dose toxicity, (c) tumorigenicity, and (d) biodistribution studies.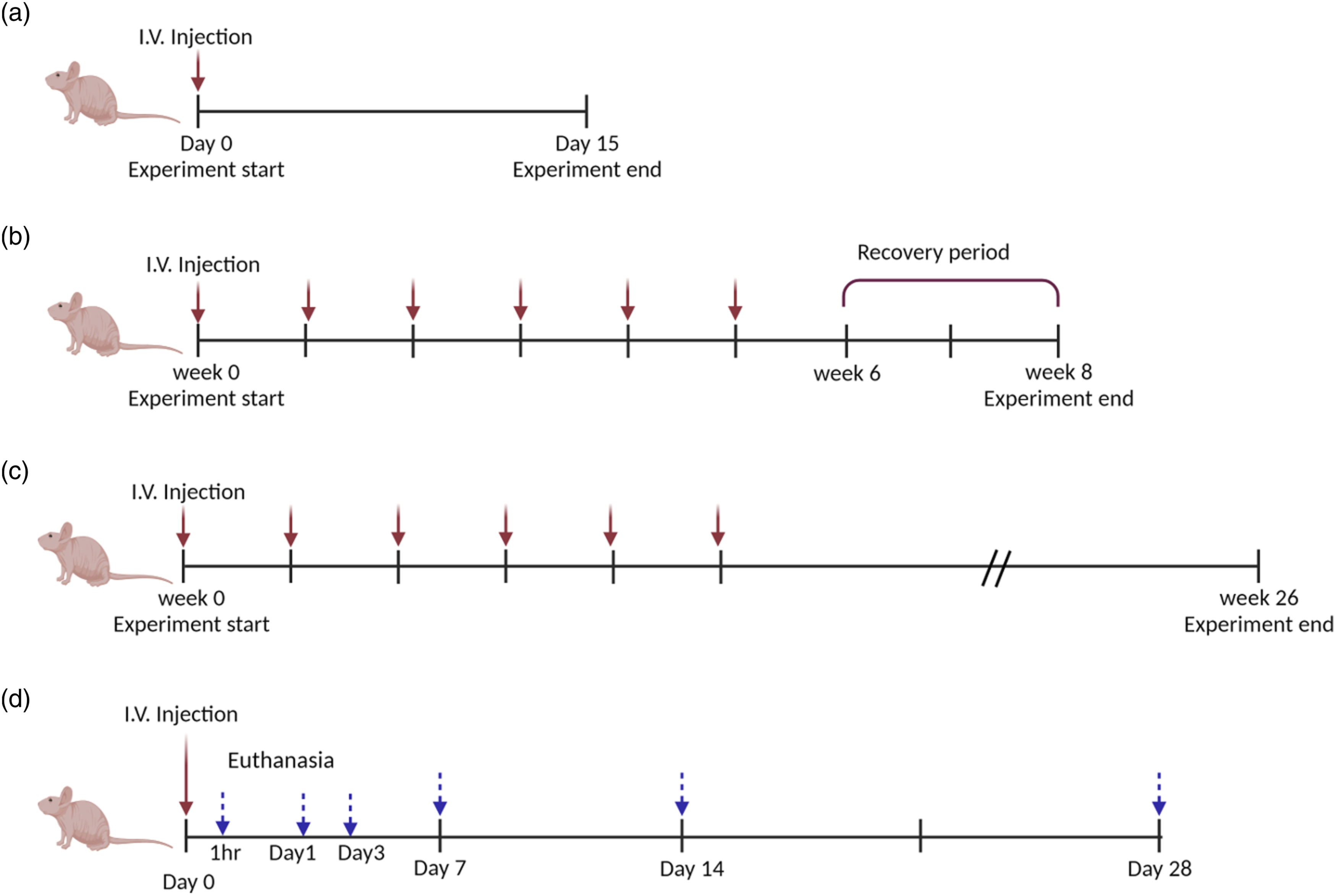
Repeated-dose toxicity
Based on data obtained from the single-dose toxicity study, doses of IFN-γ-primed MSCs were determined to be 0 (vehicle), 5 × 106, 2.5 × 107, and 5 × 107 cells/kg for the repeated dose toxicity study. 50 male and female mice were randomly divided into four groups per sex (10 mice/sex/group) for the 6-weeks intravenous repeated dose toxicity study without a recovery period. Each of five male and female mice independently treated with MSCs at 0 and 5 × 107 cells/kg was assigned for the recovery study, respectively. Vehicle and IFN-γ-primed MSCs were injected intravenously into the tail vein of all mice at 1-week intervals for 6 weeks with or without a 2-weeks recovery period (Figure 1b). 10 mice per group were sacrificed at the end of the 6-weeks study period, and the remaining were sacrificed after the recovery period. Singe, repeated dose, and tumorigenicity studies were conducted in compliance with the guidelines of the FDA, WHO, and Korean Ministry of Food and Drug Safety (MFDS).17-19 No Observed Adverse Effect Level (NOAEL) was defined as the highest dose where the effects observed in the treated group do not imply test item-related deaths or adverse pathological lesions.
Tumorigenicity
Sixty male and female mice were divided (12 mice/sex/group) into five groups for the tumorigenicity assessment. For the negative (V.C.) and positive control (P.C.) groups, the mice were treated with one dose of vehicle, and HCT116 cells (human colon cancer cell line; 5 × 108 cells/kg) by subcutaneous administration, respectively. The other two groups were inoculated with one low-dose (2.5 × 108 cells/kg), and high-dose (5 × 108 cells/kg) of IFN-γ-primed MSCs subcutaneously. Another group was treated with 5 × 107 cells/kg of IFN-γ-primed MSCs intravenously once weekly for 6 weeks (Figure 1c). High doses were selected in consideration of the maximum cell number that could be inoculated into mice subcutaneously or intravenously without resulting in death. Mortality, clinical symptoms, and tumor formation were observed for 26 weeks after the first intravenous administration, and gross pathological examination, histopathology, and immunohistochemistry were performed.
Histopathological examination and immunohistochemistry
The lungs (with bronchi), mandibular lymph node, brain, spleen, femur with marrow, heart, uterus with the cervix, kidneys, ovary, liver (with gall bladder), and tail (injection site) were fixed in 10% formalin, and the testes and epididymides were fixed in Bouin’s fixative. Paraffin-embedded sections were used for hematoxylin and eosin (H&E) stain and immunohistochemistry analysis using a human-specific anti-mitochondrial antibody to identify tumor origin (Abcam, UK).
Biodistribution
Thirty-five male and female mice were randomly divided into two groups per sex, consisting of 5 mice/time point/sex/group of vehicle control and dosing groups (Figure 1d). Both groups were inoculated once intravenously with fresh media and 5 × 107 cells/kg of IFN-γ-primed MSCs, respectively. The mice were sacrificed at 1 h and 1, 3, 7, 14, and 28 days post-injection. Major organs (lung, liver, spleen, kidney, heart, testis/ovary, epididymis/uterus with cervix, brain, mesenteric lymph node, bone marrow, spinal cord, and injection site (tail)) were harvested, frozen in liquid nitrogen, and homogenized for DNA extraction using a Maxwell® 16 instrument (Promega, Madison, WI, USA). DNA was extracted from the blood on the day of the autopsy. DNA concentration was measured using a SPECTROstar Nano spectrophotometer (BMG Labtech, Offenburg, Germany). Human-specific Alu was amplified from extracted DNA using real-time polymerase chain reaction (RT-PCR). 20 RT-PCR was performed using the QuantStudio 5 Real-Time PCR System and ViiA7 Real-Time PCR System (Thermo Fisher Scientific, MA, USA). The thermal cycle was conducted at 95°C for 2 min, followed by 35 cycles of 95°C for 15 s, 68°C for 30 s, and 72°C for 30 s. This was conducted at 72°C for 20 s, followed by a ramp-up of 1°C per step with a 5-s hold at each step. The validation parameters were examined according to the test guidelines provided by the Korean Ministry of Food and Drug Safety.
Statistical analysis
Statistical analyses for comparing the various dose groups with the vehicle control group were conducted using the Pristima System (Xybion Medical Systems Corporation). Differences in body weight from the control group were examined using Student’s t-test. The values for body weight were calculated as the means ± SD. The variance of homogeneity was also examined using Bartlett’s test. Homogeneous data were analyzed using analysis of variance, and the significance of the inter-group differences was analyzed using Dunnett’s test. Heterogeneous data were analyzed using the Kruskal–Wallis test, and the significance of inter-group differences between the control and treated groups were assessed using Dunn’s rank sum test. Statistical analysis was performed on the results except for death and clinical symptoms. The significance level was p < 0.05.
Results
Characteristics of WJ-derived MSCs with or without IFN-γ priming
MSCs isolated from adult human WJ were morphologically fibroblastic and plastic-adherent in standard culture conditions (Figure 2a). Flow cytometric analysis showed that WJ-MSCs were positive for typical MSC antigens (CD73, CD90, and CD105) but negative for typical hematopoietic antigens (CD14, CD34, and CD45) (Figure 2b),
10
indicating that cultivated cells derived from WJ were defined as MSC according to the minimal criteria for human MSC identification of the International Society for Cellular Therapy.
21
Regardless of IFN-γ priming, the cellular morphology and immunophenotype were identical (data not shown). Characteristics of human WJ-MSCs. (a) Morphological appearance of human MSCs derived from WJ. (b) The representative immunophenotype of WJ-MSC. The expression of surface antigens was plotted against the appropriate IgG isotype controls (gray histogram). MSCs used for the analyses were positive for CD73, CD90, CD105 and negative for CD14, CD34, CD45 (clear histogram).
Single-dose toxicity
Clinical signs of male and female mice after inoculation with IFN-γ primed MSCs.

aintravenous inoculation.
bsubcutaneous inoculation, V.C.: vehicle control, P.C.: positive control, (R): Recovery period.
Repeated-dose toxicity
Histopathology results of male and female mice after inoculation with IFN-γ-primed MSCs.

aintravenous inoculation.
bsubcutaneous inoculation, V.C.: vehicle control, P.C.: positive control, (R): Recovery period. Statistically significant differences (p < 0.05) between vehicle and treatment groups are indicated by asterisks (*).
Incidence of thrombosis at the injection site and emboli in the lung may be considered test item-related, given the dose dependency. IFN-γ-primed MSC-related findings were observed, given 3 male and 1 female death when they were administered with IFN-γ-primed MSC at 5 × 107 cells/kg (Table 1). Moreover, pulmonary embolism/thrombosis was also observed in the groups treated with 2.5 × 107 or 5 × 107 cells/kg of IFN-γ-primed MSC. Therefore, 5 × 106 cells/kg was considered the no adverse effect level in both sexes.
Tumorigenicity
All mice administered with HCT116 cells showed tumor-related clinical signs including hard skin and palpable mass, thereby establishing the tumorigenicity study in BALB/c-nu/nu mice (Table 1). No body weight alterations were observed in the tumorgenicity phase of this study, whereas the body weight of male and female mice significantly decreased 11 and 25 post-inoculation of HCT116 cells. The tumor-forming rates were 100%, indicating the tumorigenicity protocol with the proper positive control group.
22
Out of 12 male mice treated with IFN-γ-primed MSCs via subcutaneous administration, 4 mice did not survive with the low dose, while 5 mice did not survive with the high dose during the study period of 6 months. It was not considered test item-related tumor or toxicity because the death occurred due to skin trauma in the male mice and no deaths were observed in the female mice. In the female group treated with a low dose of IFN-γ-primed MSC by subcutaneous administration, bronchioloalveolar adenoma in the lung was observed in one female mouse (Table 2). In Figure 3, the human mitochondrial markers were detected in bronchioloalveolar adenoma of the mice treated with HCT116 cells, but not 2.5 × 108 cells/kg IFN-γ-primed MSC. This result implies the tumor-derived spontaneously from the mouse sources rather than human MSC (Figure 3). One male mouse inoculated with IFN-γ-primed MSC at 5 × 107 cells/kg by intravenous administration died. Although further study is needed to clarify if pathogenic lesions of the respiratory system are related to IFN-γ-primed MSC administration, there was no evidence of tumor formation related to the high dose of IFN-γ-primed MSC administration. Therefore, in this tumorigenicity study, 5 × 108 cells/kg and 5 × 107 cells/kg of IFN-γ-primed MSC could be considered the no adverse effect level in both sexes when they were administered via subcutaneous and intravenous injections, respectively (Table 2, Figure 3). Histopathologic changes and immunohistochemical analysis of a human mitochondrial marker. Compared with the mice treated with (a) vehicle, (b) embolus with mineralization was observed in the lung and (c) injection site of mice treated with IFN-γ-primed MSCs subcutaneously (2.5 × 108cells/kg). Immunohistochemistry staining with a human-specific anti-mitochondrial antibody was performed for the lung of mice treated with (d) vehicle, (e) HCT116 cells, and (f) IFN-γ-primed MSCs (2.5 × 108cells/kg) intravenously. The test-item inoculated mouse presented no positive reaction after probing with a human-specific antibody. Bar = 200 μm.
Biodistribution
The average amount of human-specific Alu in organs of the mice treated with IFN-γ primed MSCs.

V.C.: vehicle control, UND: under the lower limit of quantification.
Discussion
IFN-γ-primed MSCs are a potential ex vivo strategy for cell therapy because IFN-γ is known to enhance the immunosuppressive properties of MSCs by directly or indirectly modulating T cell responses by inducing MSC inhibitory factors.11,23,24 Such ex vivo manipulation of MSCs may be more effective than untreated MSCs; however, ex vivo expansion of MSCs induced tumorigenic transformation25,26 and long-term culture may affect their proliferative capacity, differentiation potential, and trophic activity.27-29 Although large-scale ex vivo expansion and preservation processes of the cells are required for clinical application, the comprehensive safety analysis of IFN-γ-pretreated and expanded MSCs has not been fully explored in preclinical models. 30 To address these issues, preclinical safety, and toxicological studies have been conducted in animal models.
In single- and repeated-dose toxicity studies, clinical signs including severe irregular respiration, subdued behavior, and prone position were observed. MSCs may be trapped in the lungs as emboli after intravenous inoculation in rodent models.31-33 because these are large cells with an estimated size of 30 μm (ranging from 16 μm to 53 μm) relative to the diameter of capillary in the mouse lung. Temporal lung emboli due to MSCs with large diameters may not cause death or clinical signs. Similar to the partial recovery of emboli in the lung with bronchi (Table 2), regeneration in the bronchiole region was observed 2 weeks post-WJ-MSCs administration in the previous study. 34 Therefore, respiratory symptoms may not be correlated with histopathological observations. In comparison, hematopoietic stem cells of mice have a much smaller diameter, ranging from 4 μm to 12 μm.35,36 MSCs demonstrated the highest interaction with lung endothelium through integrin ligand vascular cell adhesion molecule (VCAM)-1, and obstructive events such as embolism during lung passage are expected after intravenous inoculation of MSCs. 37 The lung emboli are consistent with the observation that the biodistribution of MSCs was primarily to the lung (Table 3).
The tumorigenic potential of any cell therapy is a major concern in its clinical application, especially as the potency to form teratomas of pluripotent stem cells was reported in immunocompromised animals.38,39 Furthermore, the inherent genetic instability of human embryonic stem cells, induced pluripotent stem cells, and adult stem cells have been reported.40-43 MSCs could be transformed during the expansion process ex vivo due to chromosomal abnormalities, increased levels of telomerase activity, and c-Myc expression.44,45 Therefore, regulations, such as US Food and Drug Administration and European Medicines Agency, recommend evaluating their tumorigenicity by transplantation for immunodeficient animals.46,47 In the tumorigenicity study, no tumor formation-related findings were observed in body weight, clinical signs, gross findings, or macro- and microscopic examination. Only one female mouse in the subcutaneous low-dose group showed bronchiolo-alveolar adenoma in the lung (Table 2). Tumor formation due to inoculation of human cells can be detected by immunostaining with a human-specific antibody; however, no human mitochondrial antigen expression was observed in the bronchioloalveolar adenoma, indicating that this tumor did not originate from MSCs. Compared with inbred BALB/c-nu/nu mice, severe immunodeficient mice such as SCID, NOD/SCID/IL2rγnull strain have been used for yearlong tests with high sensitivity for tumorigenicity studies of engineered cell therapeutics, recently. Our results are consistent with the previous study, which reported no tumorigenicity after inoculating BALB/c-nude mice with umbilical cord blood-derived MSCs. 48 Thäte et al. 49 also reported that no MSC-induced tumors in immunodeficient NSG mice after repeat-dose I.V. injections at 4 × 107 cells/kg after a follow-up of 6 months, which was similar to data in this study.
In this study, MSCs administered intravenously were mostly distributed in the lungs, which is consistent with previous reports.37,50
In a previous study, none or extremely low amounts of Alu DNA were observed in the lung 7 days after MSCs administration. No Alu DNA was detected in most tissues at the 4 weeks post-administration. 49 These results were consistent with those found with respect to Alu DNA detection in the lung up to 3 days post-inoculation in this study. Moreover, there was no Alu DNA detection in all tested tissues except the injection site (tail) 7 days post-administration.
Although in vivo toxicity and biodistribution of IFN-γ-primed MSCs were observed in a previous study, 51 there was a paucity of safety data with different doses, administration routes, and study periods. These IFN-γ-primed MSCs were approved for clinical trials (Approval No. 20210021802) by the Korean Ministry of Food and Drug Safety in 2021 (MFDS). Therefore, this work may provide insights into the safety of cell therapy using primed MSC in preclinical studies, possibly, helping to guide treatment decisions in clinical studies.
We concluded that 5 × 106 cells/kg of IFN-γ-primed MSC do not induce tumor formation and adverse toxicity in mice. These preclinical studies would provide fundamental data for the safety of candidate cell therapy using IFN-γ-primed MSCs for human intractable diseases.
Footnotes
Acknowledgements
The authors would like to thank the members of the Korea Institute of Toxicology for their technical support according to Good Laboratory Practice guidelines and the department of Pediatrics, Samsung Medical Center for sample collection.
Author Contributions
Sang-Jin Park and Dae Seong Kim carried out the design and coordinated the study, and prepared the manuscript. Myeongjin Choi, Kang-Hyun Han, Ji-Seok Han, provided assistance in the design of the study and contribute to the result analysis and preparation of the manuscript. Kyoung-Sik Moon and Keon Hee Yoo directed the study and provided feedback and critical comments for this manuscript. All authors read the manuscript and agreed to its publication.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Health and Welfare, Republic of Korea [grant numbers HI15C2963]; and the Korea Institute of Toxicology, Republic of Korea [grant number 1711159828].
Ethical approval
All experimental protocols were approved by the Institutional Animal Care and Use Committee (IACUC) of the Animal and Plant Quarantine Agency of Korea.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
