Abstract
Background
N-propylparaben (PP), a type of paraben, is commonly used as a preservative or antibacterial agent in daily chemicals, medicine, food, cosmetics, feed, and various industrial preservatives. Although PP promotes the growth of human breast adenocarcinoma (MCF-7) cells by activating the human estrogen receptor (ER), the mechanism responsible for this type of programmed cell proliferation is poorly understood.
Objective
To clarify the effect of PP on cell metabolic function and the potential molecular mechanism of PP induced MCF-7 cell proliferation from a new perspective.
Methods
To use high-resolution mass spectrometry-based metabolomics combined with bioinformatics analysis to analyze the molecular mechanism.
Results
The results illustrated that differential endogenous compounds related to the effects of PP on cell metabolic functions were detected. PP was found to promote glycolysis in MCF-7 cells and enhance the tricarboxylic acid cycle (TCA cycle) in mitochondria, thus improving the energy supply to these tumor cells for metabolic function and promotion of rapid proliferation. Moreover, we found that PP promoted cell proliferation by affecting the mitogen-activated protein kinase (MAPK) signaling pathway of MCF-7 cells.
Conclusion
Our results revealed the molecular mechanism of low concentration PP promoting MCF-7 cell proliferation by activating ER.
Introduction
N-propylparaben (PP), a type of paraben, is characterized by a stable chemical structure with low toxicity, and is non-irritable, resistant to acids and alkalis, and is commonly used as a preservative or antibacterial agent in daily chemicals, medicine, food, cosmetics, feed, and various industrial preservatives. PP displays broad antibacterial activity against molds, yeast, and bacteria, and is a highly effective preservative and fungicide for use in food and medicine.1–3 The concentration range of PP as an antibacterial substance in food, medicine, and cosmetics is typically from 0.01% to 0.20% (w/v). 4 With the increasing use of PP in various fields, the ingestion of PP by humans is becoming an increasingly complex issue, and exposure and absorption levels will likely increase. Thus, the safety of PP has received widespread attention from researchers. PP is a food additive that is generally recognized as safe and can be used without Food and Drug Administration approval. However, in November 2014, the United States Environmental Working Group (EWG) listed it as an ‘unsafe’ food additive and appealed to major food companies in the United States to prevent it from being used in food raw materials and processing. On 8 April 2015, the United States Environmental Working Group, together with several other agencies, filed a petition to the US Food and Drug Administration to prohibit the use of PP in food. Relevant research showed that PP had safety risks, and animal experiments have shown that PP affects late spermatogenesis in mice and reduces the body weight of murine offspring at concentrations of 0.01%, 0.10%, and 1.00% (w/v). Moreover, epididymal sperm count and sperm motility, and testosterone levels have been shown to decrease as the dose of PP increases. 5 PP also reduces superoxide dismutase activity in young Drosophila. 6 Cell experiments found that PP reduced the number of living cells and caused DNA damage in African green monkey kidney Vero cells. 7 The literature has also shown that 10−5 mol/L PP caused the proliferation of human breast adenocarcinoma (MCF-7) cells, mediated by the estrogen receptor (ER). 8 Recent studies have shown that PP has weak estrogen activity, 9 with the doses of exposure leading to harmful effects within the previously considered safe acceptable daily intake range. Therefore, it is necessary to study the mechanism of PP-induced MCF-7 cell proliferation via ER to provide a reference for the reevaluation of PP safety.
Mitochondria exist in most cells except red blood cells, and are the main organelle for oxidative phosphorylation, the tricarboxylic acid cycle and oxidative respiratory chain pathways. Mitochondrial-generated adenosine triphosphate (ATP) provides energy for cell activities, and also participates in many aspects of cell metabolism, such as cell proliferation, and apoptosis.10,11 Mitochondria can also regulate the dynamic balance of calcium. 12 Although the impact of PP on cell energy metabolism has been reported, 13 it has not been characterized how PP affects the levels of cell-related endogenous substances and related metabolic pathways in tumor cells. Additionally, related enzyme activities required by cellular functions and their regulatory genes have not been studied in detail, nor the association between endogenous substances and changes in metabolic pathways. Mitogen-activated protein kinase (MAPK) signaling pathways play important roles in crucial cellular responses, including the processes of cell growth and reproduction, division and death, and changes in various biochemical reaction signals.14,15 Whether the potential mechanism of PP-induced MCF-7 cell proliferation is related to MAPK pathways remains to be studied. The current study used PP at various doses [to simulate the amount of PP exposed to humans in the environment and levels in tissues (2.6 ng/g in tumor tissue)] 16 in cell metabolomics experiments, and determined the effects and molecular mechanisms of PP actions on metabolism and MCF-7 cell proliferation.
Materials and methods
Chemicals
PP (purity of 99.8%) was provided by Sigma-Aldrich. Trizol for RNA extraction was purchased from Invitrogen. The One Step RNA PCR Kit was obtained from TaKaRa. Formic acid and ammonium formate were purchased from Sigma-Aldrich. Methanol and acetonitrile (HPLC grade) were provided by Fisher Scientific. Ultra-pure water was prepared by a Millipore-Q pure water system. All other chemical reagents were analytical grade with purity above 99.5%.
Cell experiments
Human breast cancer MCF-7 cells were cultured in Dulbecco’s Modified Eagle Medium (DMEM) containing 1% penicillomycin and 10% fetal bovine serum without phenol red at 5% CO2 and 37°C. After 90% of the cells were infused, the cells were digested with 0.25% trypsin and passaged. Cells in the logarithmic phase were seeded into 48-well plates at 1 × 105 cells/well and cultured overnight in an incubator. After cell adhesion for 24 h, the medium was replaced with DMEM containing different PP concentrations (control and PP concentrations of 1, 10, and 50 μmol/L). Eight multiple wells were used for each group, and the corresponding medium was added and changed every 24 h. After 72 h, the medium was removed, and cells were scraped off for metabolomics and molecular biology analysis.
Detection of cell and culture medium samples by high-resolution mass spectrometry
The cells were ultrasonicated in 200 μL ultrapure water (50% methanol). After centrifugation, 50 μL of the supernatant and culture medium were spiked with 450 μL of precipitant containing the internal standard (methanol:acetonitrile = 1:1), thoroughly mixed for the 60 s, centrifuged at 10,000 g for 10 min, and metabolomics analysis by a QE-Orbitrap high-resolution MS. 17 The chromatographic conditions for detecting mitochondrial related products were as follows: A: water containing 0.1% formic acid; D: acetonitrile; gradient elution protocol: 0–0.5 min, 20% D; 0.5–1.5 min, 20%–40% D; 1.5–2.5 min, 40%–95% D; 2.5–3.5 min, 95% D; 3.5–3.6 min, 95%–20% D, 3.6–5.5 min, 20% D; injection volume: 5 μL; flow rate: 0.3 mL/min; column: Thermo Hypersil Gold C18 3 μm, 2.1 × 100 mm; column temperature: 30°C; autosampler temperature: 4°C. ESI(−): SIM mode; spray voltage: 2800 V; evaporation temperature: 350°C; sheath gas: 40 Arb; auxiliary gas: 10 Arb; capillary temperature: 320°C; S-lens RF: 50; NCE: 20. Tolbutamide (IS): 269.09,654, pyruvate: 87.00,877, lactic acid: 89.02,442, fumaric acid: 115.00,368, succinic acid: 117.01,933, malic acid: 133.01,425, α-ketoglutaric acid: 145.01,425, citric acid: 191.01,973.
Metabolic pathway analysis
Endogenous metabolites were identified, and data were processed following previously reported methods. 17 Differences in endogenous metabolites in cells and culture medium between the control group and PP addition groups were analyzed by MetaboAnalyst 5.0. Endogenous metabolites with a VIP value >1 were identified, and principal component analysis (PCA), partial least squared discriminant (PLS-DA), and topology diagrams were drawn. Pathway analysis at the MetaboAnalyst 5.0 website was then used to analyze the metabolic pathways where PP affected cell physiological function and the culture medium, and the metabolic pathways with an impact value >0.1 were selected as the major pathways. The correlations between cell physiological function and the culture medium were analyzed.
Reverse transcription-polymerase chain reaction (RT-PCR) analysis
TRIzol reagent was added to lyse MCF-7 cells. Total RNA was extracted following the instructions of the TRIzol kit. The design of primers for the following genes are listed in Table S1: Estrogen receptor(ER), Hexokinase 2 (HK2), Pyruvate kinase(PK), pyruvate dehydrogenase (PDH), lactate dehydrogenase (LDH), citrate synthetase (CS), isocitrate dehydrogenase (IDH), succinate dehydrogenase (SDH), malate dehydrogenase (MDH), phospholipase C (PLC), rat sarcoma (RAS), rapidly accelerated fibrosarcoma (RAF), mitogen-activated protein kinase kinase (MEK), extracellular regulated protein kinases (ERK), cellular-myelocytomatosis viral oncogene (C-Myc), sirtuin (silent mating type information regulation 2 homolog) 6 (SIRT6), Cellular Tumor Antigen P53(p53), poly ADP-ribose polymerase 1(PARP1) and β-actin. Real-time quantitative PCR was carried out using an ABI Prism 7300 Sequence Detector (SYBR Premix Ex Taq™) at 95°C for 10 s, 95°C for 30 s, and 60°C for 15 s, with a total of 40 cycles. The mRNA expression differences for ER,HK2, PK, PDH, LDH, CS, IDH, SDH, MDH, PLC, RAS oncogene, RAF, MEK, ERK, C-Myc, SIRT6, p53 and PARP1 between the control and PP groups were analyzed by the 2–∆∆Ct method.
Determination of MCF-7 cell enzyme activities
After culturing MCF-7 cells for 72 h, the culture medium was aspirated, 200 μL 0.1 mM phosphate buffer (pH 7.0) was added to dislodge the cells, and the cell-containing buffer was collected carefully. Cells were disrupted by ultrasonication for 1 min and centrifuged for 10 min at 2000 × g, 4°C. The supernatant was collected for enzyme activity assays. The activities of PDH, SDH, LDH, and MDH were measured according to the manufacturer’s instructions for the respective kits. The protein concentration of the liver homogenate was determined using a BCA protein assay kit. Three parallel samples were taken for each group.
Extraction of MCF-7 cell mitochondria and determination of ATPase activity
After culturing MCF-7 cells for 72 h, the culture medium was aspirated, and 200 μL phosphate buffer (pH 7.0) was added to dislodge the cells. The cell-containing buffer was collected carefully. Cells were disrupted by ultrasonication for 5 min, and mitochondria were obtained by differential centrifugation. 18 The protein concentration of mitochondria was determined using a BCA protein assay kit. ATPase activity in mitochondria isolated from MCF-7 cells was determined using an ATPase assay kit.
Molecular docking
The molecular interactions of PP with human ER were examined by converting both ER and PP into PDBQT format files with Autodock Tools 1.5.619,20 and then performing a molecular docking study using Autodock vina 1.1.2. 21 The structure of human ER (PDB ID: 3ERD; resolution: 2.03 Å) was taken from the RCSB Protein Data Bank database (http://www.rcsb.org). The coordinates of the human ER active pocket were set as: center_x = 5.287, center_y = −0.303, center_z = −5.57: size_x = 15, size_y = 15, and size_z = 15. We set the parameter exhaustiveness to 15 to increase the accuracy of the calculation. All parameters used were the default values unless otherwise stated. Finally, the conformation with the highest score was analyzed by PyMoL.
Statistical analysis
PCA, PLS-DA, and topology were analyzed by MetaboAnalyst 5.0, and the main metabolic pathways of PP that affected cell physiological functions and culture medium with an impact value >0.1 were also analyzed. Data were analyzed by SPSS16.0 software and expressed as mean ± standard deviation (SD).One-way ANOVA was used to compare experimental groups, and Tukey’s test compared the differences between experimental and control groups. * and ** indicated p < .05 and p < .01, respectively.
Results
Effect of PP on human ER
Previous experiments have shown that when p-hydroxybenzoic acid esters contained branched chains, with the increase of the peptide chain, the ability to combine p-hydroxybenzoic acid esters with ER was enhanced continuously, and the relative binding force was essentially identical between various esters and ERα and ERβ.22,23 Figure 1 shows that compared with the control group, mRNA expression levels of ER increased as the PP exposure dose increased. Molecular docking analysis was used to illustrate the correlation between PP and ER. This analysis showed that PP bound tightly in the active pocket of human ER (Figure 2(a)), with an affinity of −6.4 kcal/mol. PP was found in a hydrophobic pocket forming strong hydrophobic interactions (Figure 2(b)). The benzene ring of PP shown in Figure 2(b) forms CH-π, anion-π and cation-π interactions. More importantly, PP forms a hydrogen bond with Glu-353 (Figure 2(b)), the primary non-covalent interaction between PP and human ER. This special binding mode may be responsible for the stable complex formed between the hydroxypropyl ester and human ER. The above results indicated that PP activated the expression of ER in MCF-7 cells, consistent with previous reports.
8
Effects of PP on energy metabolism-related genes and estrogen receptor gene in MCF-7 cells. Docking PP with the human estrogen receptor (ER). (a) PP docking to the active pocket of the ER(overall), (b)PP docking to the active pocket of the ER (details).

Effects of PP on endogenous metabolites and metabolic pathways in 72 h culture medium and MCF-7 cells
Endogenous metabolites were identified, and data were processed using previously reported methods.
17
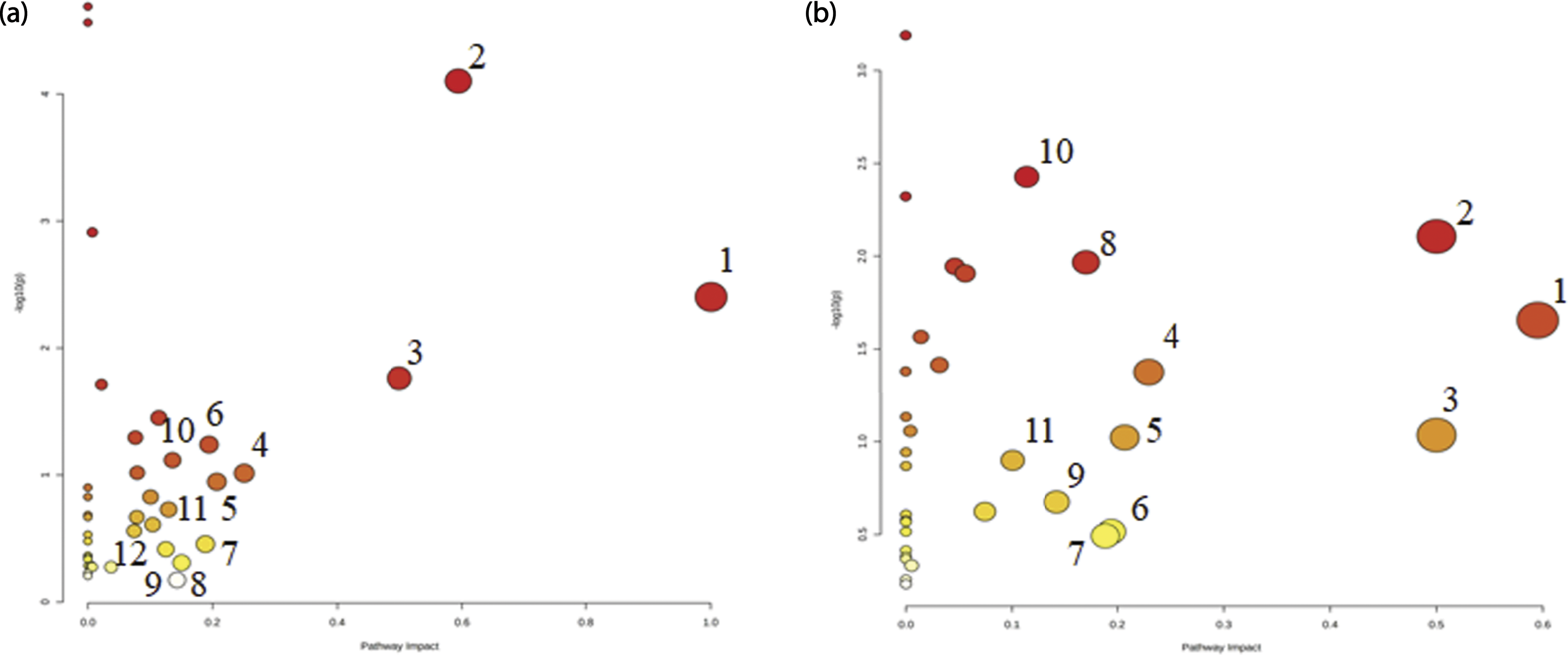
PCA and PLS-DA were performed using MetaboAnalyst 5.0. For 72 h culture medium, the results of PCA (Figure 3(a)) and PLS-DA (Figure 3(b)) showed complete separation of the control group and PP exposure groups. Model quality parameters were: accuracy = 0.90,625, R2 = 0.94,388, and Q2 = 0.64,139. For MCF-7 cells, the results of PCA (Figure 3(c)) and PLS-DA (Figure 3(d)) revealed complete separation of the control group and PP exposure groups. The model quality parameters were: accuracy = 0.875, R2 = 0.96,705, and Q2 = 0.68,781. As shown in Table 1, there were 41 types of endogenous metabolites with VIP >1 in the 72 h culture medium. Table 2 shows 43 endogenous metabolites in MCF-7 cells with VIP >1. As shown in Table 3 and Figure 4 12 and 11 endogenous substance-related metabolic pathways were affected by PP in culture medium and MCF-7 cells, respectively, with an impact value >0.1. These results indicated that PP affected metabolism and the secretion of endogenous substances by MCF-7 cells, yielding significant changes in the content and chemical composition of 72 h culture medium. Amino acid levels showed the greatest changes, consistent with the results of metabolic pathway analysis. PP also affected the energy supply in mitochondria, resulting in changes in the levels of endogenous substances in MCF-7 cells. The effect of PP on metabolic pathways in MCF-7 cells correlated with changes in the 72 h culture medium. Metabolomics analysis of MCF-7 cells cultured in medium in the absence or presence of PP for 72 h. (a) The PCA diagram of 72 h culture medium for the control and PP exposure groups. (b) The PLS-DA diagram of 72 h culture medium for the control and PP exposure groups. (c) The PCA diagram of MCF-7 cells for the control and PP exposure groups. (d) The PLS-DA diagram of MCF-7 cells for the control and PP exposure groups. The average change of main metabolites(VIP value >1) in medium contributing for discrimination of control and PP-treated group in PLS-DA models (Mean ± SD) (VIP >1). Arrow indicates significantly up-regulated or down-regulated metabolites in the PP different dose groups compared with the control. The average change of main metabolites(VIP value >1) in cell contributing for discrimination of control and PP-treated group in PLS-DA models (Mean ± SD) (VIP >1). Arrow indicates significantly up-regulated or down-regulated metabolites in the PP different dose groups compared with the control. Results for medium and cell pathway analysis with MetaboAnalyst5.0 Effects of PP on the main metabolic pathways in the 72 h culture medium and MCF-7 cells. (a) Metabolic pathway map of 72 h culture medium for the PP exposure groups. (b) Metabolic pathway map of MCF-7 cells for the PP exposure groups.



Correlation and hierarchical cluster analysis of endogenous substances in MCF-7 cells
The correlation analysis of differentially expressed endogenous substances in MCF-7 cells is shown in Figure 5(a), where the horizontal and vertical axes both represent variable information. Figure 5(b) shows the heat map obtained by hierarchical clustering analysis, where the horizontal and vertical axes represent sample and variable information, respectively. Figure 5(b) illustrates the content change trend of 43 differential endogenous substances in different groups. These results demonstrated changes in contents of endogenous substances in MCF-7 cells after 72 h exposure to PP. Correlation analysis and hierarchical cluster analysis. (a) The correlation of differential endogenous substances in MCF-7 cells for the control and PP exposure groups was analyzed by hierarchical clustering. (b) The correlation of differential endogenous substances in MCF-7 cells for the control and PP exposure groups was examined by a heat map.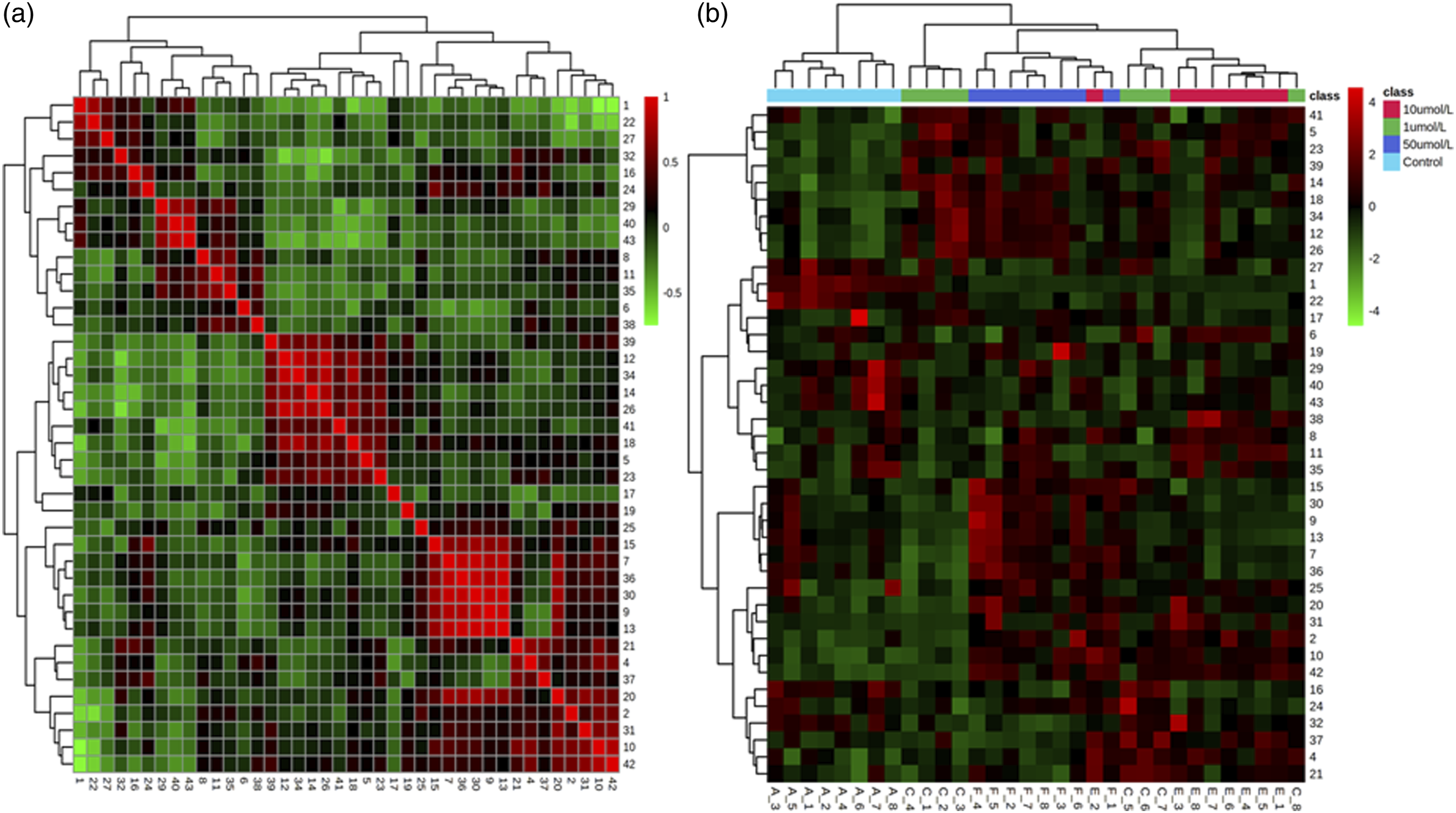
Effect of PP on glucose metabolism in MCF-7 cells
The average change of metabolites for TCA cycle in cell (VIP <1).

Effect of PP on enzyme activities in MCF-7 cell.

Data are the mean ± SEM. (n = 3). Statistical analysis were performed by one-way Anova with Tukey’s test for comparisons between groups. *p < .05, **p < .01.
The changes of compound concentration in cell under the influence of different doses of PP.

Data are the mean ± SEM. (n = 8). Statistical analysis were performed by one-way Anova with Tukey’s test for comparisons between groups. *p < .05, **p < .01.
Effect of PP on mitochondrial energy metabolism in MCF-7 cells
Table 3 shows that PP affected the TCA cycle, indicating that PP affects mitochondrial energy metabolism in MCF-7 cells. Our study revealed that, compared with the control group, CS mRNA expression levels (Figure 1) and citric acid content (Tables 2 and 6) were significantly changed in cells exposed to PP. However, they were inconsistent with the increase in the mRNA expression level of CS and citric acid content, and this observation remains to be further analyzed. It has been reported that the accumulation of the dehydrogenation product [nicotinamide adenine dinucleotide (NADH)] of isocitrate markedly inhibited the activity of IDH. 27 Our research showed that, compared with the control group, the content of NADH and IDH mRNA expression levels were significantly changed with increasing PP exposure dose (Table 4 and Figure 1), indirectly suggesting that PP can enhance the activity of IDH. Previous research reported that citric acid can participate directly in energy generation and enzymatic reactions of the body, and this metabolite can be oxidized completely to generate ATP in the TCA cycle, with a high energy utilization rate. 28 Therefore, it is likely that PP can promote the use of citric acid in the TCA cycle, explaining the decrease in citric acid levels in the PP-exposed group. Compared with the control group, the levels of endogenous metabolites related to the TCA cycle (α-ketoglutamic acid, fumaric acid, and succinic acid, shown in Tables 2 and 6) and mRNA expression and enzyme activity levels of regulatory enzymes (SDH and MDH, shown in Figure 1 and Table 5) had changed significantly with increasing PP exposure dose. Previous reports showed that SDH and MDH were two key enzymes involved in the TCA cycle, and SDH was a marker enzyme that reflects mitochondrial function, with its activity used as an important indicator for activity of the TCA cycle. 29 The current results showed that PP promoted the TCA cycle of mitochondria in MCF-7 cells, to provide sufficient energy for the growth of MCF-7 cells.
Effects of PP on Na+-K+-ATPase and Ca2+-Mg2+-ATPase activities in mitochondria.

Data are the mean ± SEM. (n = 3). Statistical analysis were performed by one-way Anova with Tukey’s test for comparisons between groups. *p < .05, **p < .01.
Effects of PP on the MAPK signaling pathway in MCF-7 cells
The mRNA expression levels of RAS, RAF, ERK2, MEK1 and c-Myc were significantly changed in cells exposed to PP compared with control cells (Figure 6), indicating that PP may promote tumor cell proliferation by activating the cRAF-MEK1 complex. It was previously reported that c-Myc regulates aerobic glycolysis and enhances glycolysis by directly activating the LDH gene and all glycolysis genes. c-Myc also activates genes regulating mitochondrial biosynthesis to increase the activity of the TCA cycle,31–34 which is consistent with our previous research. The literature showed that when oxidative stress caused DNA damage, SIRT6 can enhance the activity of polyadenyldiphosphate ribosylase of PARP1 by modifying the protein of PARP1 with monoadenosine diphosphate ribosylation, and then participate in the repair of DNA double-strand break.
35
The results showed that compared with the control group, the mRNA expression level of genes SIRT6 and PARP1 showed a significant upward trend. The reason may be that the cellular immune mechanism wanted to inhibit the growth of tumor cells by increasing the expression level of tumor suppressor genes, which also confirmed and explained the results and reasons that PP could promote the growth of MCF-7 tumor cells from the reverse side. Compared with the control group, the mRNA expression level of tumor suppressor gene p53 did not decrease significantly, and the reason needed further study. The above research results indicated that PP affected the MAPK signaling pathway of MCF-7 cells, providing further evidence to explain how PP promoted the growth of MCF-7 tumor cells. Effects of PP on the MAPK signaling pathway-related genes in MCF-7 cells.
Discussion
People are increasingly exposed to more types of chemicals in contemporary daily life, introducing new risks to health. The preservative PP is used widely in food, medicine, agriculture, and other industries to protect products against degradation by bacteria and fungi. However, studies on PP have shown that it can significantly lower sperm numbers in rats and interfere with estrogen secretion in women, thus affecting fertility, and create other problems. The literature has also shown that 10−5 mol/L PP can cause the proliferation of MCF-7 cells, mediated by the ER. 8 Our current research used RT-PCR technology and molecular docking analysis to show that PP can activate ER, and to study the molecular mechanism of low dose PP actions in the promotion of tumor cell growth.
Metabolomics is a powerful methodology to study the physiological and pathological states of an organism and the biochemical effects of exogenous substances. Metabolomics focuses on changes in concentrations of metabolites with molecular weights <1000 Da. These small-molecule metabolites are downstream products of genes and proteins, and result from the combined action of genes and the environment. Their abundances reflect the state (phenotype) of the cell or organism. Considering the complexity of diseases, metabolomics has significant advantages and potential in disease diagnosis and treatment. Changes in endogenous metabolites can also be used to study mechanisms of toxicity and identify potential biomarkers of toxicity. By applying metabolomic study, we found that many endogenous substances were differentially expressed in cells exposed to PP compared with control cells, including amino acids and small molecule chemicals involved in energy metabolism. Further analysis revealed that the affected metabolic pathways were related to glycolysis and the TCA cycle, indicating that PP exposure had an impact on the energy metabolism of MCF-7 cells. Metabolomics provided the direction to further study the molecular mechanism of PP-induced growth of MCF-7 cells.
The current metabolomics study found that PP exposure affected the glycolytic metabolic pathway of MCF-7 cells. Tumor cells exhibit an abnormal mode of energy metabolism that is better able to meet energy needs and provide material for cellular growth, helping them to interact with the surrounding environment, and promoting metastasis. Glycolysis plays an important role in the coordination between tumor cells and the surrounding environment and is a key beneficial metabolic pathway for tumor cells.
36
Therefore, we analyzed the regulatory enzymes and intermediate metabolites in the glycolytic pathway using RT-PCR technology, enzyme activity detection and mass spectrometry quantitative detection methods. It was found that PP exposure enhanced the glycolytic and pyruvate reduction reactions in MCF-7 cells (summarized in Figure 7), consistent with the results of metabolic pathway prediction. Only endogenous pyruvate can enter the TCA cycle, and our research showed that the cellular pyruvate content decreased after PP exposure, indicating that mitochondria in MCF-7 cells used pyruvate to generate acetyl CoA as the material basis for the TCA cycle. The impact of PP on mitochondrial energy metabolism became the focus of our research, to further determine the molecular mechanism of low-dose PP-induction of tumor cell growth. Effects of PP on the energy metabolism and the MAPK signaling pathway in MCF-7 cells.
Mitochondria are the main energy metabolism organelles of the body and the integrator of key metabolic pathways in the body, including oxidative phosphorylation, fatty acid oxidation, and TCA cycle pathways. Among the key metabolic pathways of mitochondria, the TCA cycle is a key pathway for mitochondrial energy production. Insufficient energy supply of mitochondria interferes with the normal function of cells, leading to dysfunction of organs and tissues with high energy demand, such as heart, muscle and brain, leading to human diseases. 37 The current results showed that acetyl CoA generated from pyruvate enters the TCA cycle under the action of CS (Figure 7), indirectly explaining the decrease in pyruvate content. The mRNA expression levels of key regulatory enzymes that promote the TCA cycle were significantly increased in PP exposed cells, likely promoting the generation of mitochondrial ATP energy. AMP is formed after the release of energy from ATP and ADP in the body and can combine with phosphate groups to form ADP and ATP, and when ATP production is less than consumption, the concentration of AMP increases. 38 The results of this study were just the opposite. The PP-exposed cells exhibited enhanced activity of ATPases (Table 7), likely to increase the utilization rate of ATP. These findings reveal that the PP exposure resulted in sufficient ATP for MCF-7 cell growth.
Changes to signaling pathways in tumor cells are closely associated with cell proliferation and apoptosis. 39 The MAPK signaling pathway plays an important physiological role in eukaryotes,40,41 which also primarily includes the p38, JNK, and ERK pathways that can be activated or inhibited to participate in cell proliferation, differentiation, and apoptosis.42,43 In general, after activation of RAF in the MAPK pathway, RAF binds to MEK to form the cRAF-MEK1 complex through the C-terminus. This complex gives rise to active MEK1 and promotes tumor cell proliferation. Because the MAPK pathway plays an important role in tumor cell proliferation, we used RT-PCR technology to analyze the genes related to this pathway. The present findings showed that PP can promote tumor cell proliferation by promoting the MAPK signaling pathway. Exposure to PP affected glycolysis and the TCA cycle via the regulation of c-Myc (Figure 7), confirming the above research results from the side.
In summary, on the basis of the metabolomics analysis of high-resolution MS and bioinformatics analysis, we investigated the effect of PP on cellular metabolic functions and the underlying molecular mechanism of PP actions from a new perspective. Metabolomics detected differential expression of endogenous substances associated with the effects of PP on cellular metabolic functions. Molecular biology and other technology combined with changes in the levels of endogenous substances. This study revealed that PP promoted the glycolysis process of MCF-7 cells, and also enhanced the TCA cycle of mitochondria, likely to provide sufficient energy requirements for the metabolic function and enhanced promotion of MCF-7 cell proliferation. Additionally, PP promoted cell proliferation by affecting the MAPK pathway of MCF-7 cells. Overall, these results demonstrated the molecular mechanism of low concentration PP-induction of MCF-7 cell proliferation via the ER. A limitation of this research was the in vitro exposure of MCF-7 tumor cells, and human exposure to PP is mainly via oral intake or skin absorption. Further research is needed in the in vivo setting, such as on animal tumor models, to study the effects of PP on metabolic function and the potential new mechanism of tumor proliferation in vivo.
Supplemental Material
Supplemental Material - Discovery of the mechanism of n-propylparaben-promoting the proliferation of human breast adenocarcinoma cells by activating human estrogen receptors via metabolomics analysis
Supplemental Material for Discovery of the mechanism of n-propylparaben-promoting the proliferation of human breast adenocarcinoma cells by activating human estrogen receptors via metabolomics analysis by Yunxia Chen, Chan Zhao, Jun Zheng, Ning Su and Hainan Ji in Human & Experimental Toxicology
Footnotes
Acknowledgements
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the Basic Research Fund for Central Public Interest Institutes (No.2020JK035), The State Administration for Market Regulation Special project for improving chemical quality and safety detection capability (No.2013810-22-004).
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
