Abstract
Breast cancer is highly prevalent and considered the main challenge to public health among females in Egypt as in other countries. MicroRNA-21 (miR-21) and MEG-2 are noncoding RNA attributed to their aberrant expression in several diseases, including breast cancer. This study aimed to assess the reliability of serum expression levels of miR-21 and MEG-2 in discriminating stages of breast cancer and scrutinize their correlations with the targeted transforming growth factor-beta (TGF-β) expression. One hundred and 30 participants whose ages ranged from 28 to 62 years were included in this study, divided into one hundred breast cancer patients and 30 healthy participants. miR-21 and TGF-β expression levels showed upregulation in patients with BC and elevated miR-21/TGF-β levels consistent with the BC stage. In addition, LncRNA (MEG-2) showed down-regulation in patients with BC. MEG-2 expression levels revealed a gradual decrease consistent with the BC stage. In addition, a negative relationship between the MEG-2 and the miR-21 and TGF-β differential expression was also noticed. This study suggested that miR-21 and MEG-2 can be used as prospective diagnostic biomarkers and emphasized the crucible role of TGF-β as therapeutic targets for BC.
Introduction
Breast cancer is highly prevalent and considered the main challenge to public health among females worldwide. 1 Breast cancer is still the most prevalent disease in Egypt and the Middle East, which has been linked to a lack of awareness of the disease and inadequate medical access to screening and care.2,3
Noncoding RNA molecules with a length of roughly 17–25 nucleotides are known as microRNAs (miRNAs) and are highly conserved. They interact with a specific target messenger RNA (mRNA), which they use to control gene expression at the post-transcriptional stage.4–6 Interestingly, aberrant expression of miRNAs is associated with the molecular pathogenesis of the progression of many diseases.4,7
miR-21 has a crucible role in various cancers and is an oncogenic mediator; located on chromosome 17q23.2. 8 Interestingly, postoperative decreases in circulating miR-21 levels only occurred in cancer patients who had received chemotherapy or potentially curative procedures. 9 Thus, miR-21 could consider a long-term follow-up biomarker in tumor progression.
PTP-MEG2; MEG2, a member of the protein tyrosine phosphatases. 10 Moreover, it acts as a tumor suppressor gene in the progression of various cancer like liver, gastric, and breast cancer. 11
Transforming growth factors-β (TGF-β) is a pluripotent cell regulatory polypeptide that affects cell growth, differentiation, matrix production, and immune function.12,13 TGF-β seems to provide a protection for mammary cells against carcinogenesis by inducing cellular apoptosis and creating a cell microenvironment that inhibits tumor invasion and metastasis. However, aberrant expression of TGF-β in breast cancer, converting its tumor suppressor properties to oncogenic factors, promotes breast cancer and metastasis. 14 Furthermore, according to Dooley and Dijke, 15 TGF-β is a regulator of disease progression via inflammation and fibrosis.
Interestingly, recent studies have demonstrated that noncoding RNAs could participate in the regulation of processes that drive fibrosis which includes; the TGF-β signaling axis. This study sought to evaluate the expression levels of miR-21/MEG-2 and scrutinize their correlations with the targeted TGF-β expression.
Subjects and methods
One hundred and 30 participants whose ages ranged from 28 to 62 years were included in this study from the oncology Department, Faculty of Medicine, Beni-Suef University, and divided into four groups, including 100 breast cancer patients (stage I, stage II, and stage III), we depended on AJCC eighth edition for staging, the following tumor characteristics were noted: tumor size, tumor-node-metastasis (TNM) stage, and estrogen/progesterone status. The other group included 30 healthy volunteers. The study was undertaken as per the Declaration of Helsinki and approved by the Ethics Committee for Research at Beni-Suef University (FMBSUREC/02102022). All participants in the study signed an informed written agreement. Patients undergoing chemotherapy or radiation treatment or who have an acute infection are eligible for exclusion from participation in the current study.
Peripheral blood samples from each participant were collected in separated vacuum tubes then two consecutive centrifugation steps were conducted to remove cellular debris. Sera were stored at −80°C until use for MEG-2 and miR-21 expression assay and Western blot assessment of TGF-β.
MEG-2 and miR-21 expression assay
Primers sequence of all studied genes.

Western blot assay for TGF-β
A Western blotting technique was used to evaluate the TGF-β expression PVDF membrane was used to separate the 20 μg of protein from each sample using SDS polyacrylamide gel electrophoresis, ReadyPrepTM protein extraction kit (total protein) from Bio-Rad Inc (Catalog #163-2086) were used. Membranes were sealed for 1 hour at room temperature with 5% skimmed-milk powder in a buffered Tris-saline solution. Primary antibodies for TGF-β and β-actin were incubated on PVDF membranes for a whole night at 4°C. The secondary antibodies were conjugated to HRP and incubated along with the membranes. With an enhanced chemiluminescence kit from Bio-Rad Inc (Catalog#170-5060) and the imaging program, the intensity of the bands was visualized and evaluated by Odyssey Imaging System (LI-COR Biosciences, Lincoln, NE, USA), according to the method described by Harlow, Lane. 17
Statistical analysis
Results were analyzed using statistical analysis software SPSS 22 software and multiple post-hoc comparison tests (Duncan) used for pairwise comparison. Data were accessible as means ± standard error means (SEM) or percentages when appropriate. Pearson’s correlation analysis assessed the relations between the variables studied.
Results
Demographic characteristics of the studied groups.

Data were expressed as number (%). PR/ER: Progesterone receptor/Estrogen receptor.
The data analysis of the selected biomarkers showed that miR-21 and TGF-β upregulated in BC patients, and the upsurge expression of miR-21 and TGF-β was in the BC stage III patients with remarkable significance from stages I and II (Figure 1). Additionally, MEG-2 was down-regulated in BC patients and the lowest expression pattern of MEG-2 was in the BC stage III patients with noticeable significance from stages I and II (Table 3). Relative expression of TGF-β levels among study participants. The different letters indicate statistical significance different means. G1: healthy control; G2: BC patients with stage I; G2:BC patients with stage II; G3: BC patients with stage III. Expression levels of miR-21,and MEG-2 among study participants. Data were expressed as mean ± SEM; According to the Duncan multiple range test, the different letters indicate statistical significance different means. miR-21: microRNA-21; TGF-β: Transforming growth factors-β.
Pearson correlation coefficient between miR-21, MEG-2, and TGF-β.

miR-21: microRNA-21; TGF-β: Transforming growth factors-β. ** Significant linear correlation at
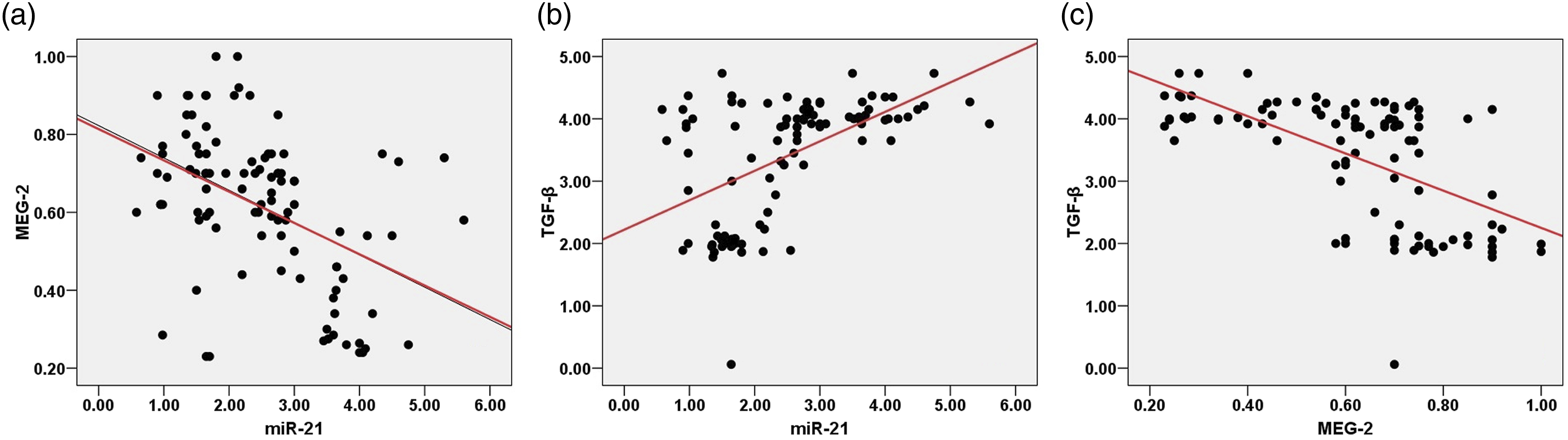
Correlations between (a) miR-21, and MEG-2 (b) miR-21 and TGF-β (c) MEG-2 and TGF-β of the study participants.
Diagnostic performance of serum miR-21 and MEG-2
With an AUC of 0.918, serum miR-21 able to distinguish healthy controls from breast cancer patients in ROC curve analysis, with 95% CI = 0.868 to 0.968, Receiver operating characteristic curves (ROC) for miR-21 and MEG-2 in (a, b) between breast cancer patients and healthy participants; and (c, d) stage III breast cancer from breast cancer patients.
With an AUC of 0.787, miR-21 discriminated patients with stage 3 breast cancer from breast cancer patients with 95% confidence interval = 0.672 –0.901,
Discussion
In the present study, miR-21, MEG-2, and TGF-β expression levels were assessed as potential diagnostic biomarkers of breast cancer. To our knowledge it is the first investigation to reveal the association of miR-21, MEG-2, and TGF-β levels with each other in breast cancer patients. Herein, our findings showed that patients with breast cancer had significantly higher levels of miR-21 and TGF-β expression as well as a substantial decrease in MEG-2 expression. Accordingly, previous studies have reported remarkable elevation in the expression levels of TGF-β in patients with breast cancer.18,19
Recent studies pointed out that the role of sustained TGF-β expression in breast cancer is mainly associated with the advanced stages of the disease.20,21 Our results revealed that TGF-β elevated gradually among BC stages. Here, the TGF-β elevation in expression level is related to the tumor progression and its association with oncogenic activity. TGF-β upregulation accelerates tumor growth and metastasis while preventing immune surveillance.21–23
Herein, the present investigation set out the expression patterns of miR-21 and MEG-2 as bioindicators for breast cancer. According to our findings, miR-21 and MEG-2 expression levels in patients with BC are considerably distinguishable from healthy controls. Our results are consistence with Han et al., 24 and Wang et al., 25 who reported that miR-21 expression levels were elevated in BC patients and considered miR-21 a crucial biomarker for breast cancer diagnosis and a promising strategy in breast cancer therapy. Additionally, miR-21 expression levels of breast cancer patients tended to elevate gradually among BC stages, and the expression levels of miR-21 were associated with the BC stages. Therefore, miR-21 might be a viable biomarker for detecting breast cancer and predicting its prognostications. Previous studies on breast cancer showed that miR-21 expression is elevated and consistent with our results about the oncogenic effects of miR-21 on breast cancer.9,26
Along with the upregulation of miR-21 and TGF-β expression, MEG-2 expression downregulation was observed in BC patients as previously described. 27 It is hypothesized that the MEG-2/TGF-β axis plays a crucial role in controlling genes involved in the progression of cancer and metastasis. Additionally, our results were consistent with Liu et al., 11 who found that MEG2 is downregulated in human gastric cancer. Furthermore, according to Ying et al., 10 MEG2 blocks the AKT pathway, hich in turn prevents hepatocellular carcinoma from proliferating and metastasizing.
Additionally, MEG-2 expression levels of breast cancer patients tended to decrease gradually among BC stages, and the expression levels of MEG-2 were associated with the TNM stage in breast cancer, indicated the tumor suppressive role of MEG-2 in BC. Previous studies were consistent with our results about the tumor suppressive effects of MEG-2 on cancer.10,28–30
To explore the correlation underlying the signaling efficacy of TGF-β and its modulatory effect on the miR-21/MEG-2 expression axis. Herein, Pearson’s statistical analysis revealed a noticeable negative correlation between the signaling of MEG-2 and the differential expression of miR-21 and TGF-β. Moreover, remarkable positive association between the signaling of TGF-β and the expression level of miR-21 among study participants. Our results were consistent with Dai et al. 31 demonstrated that miR-21 up-regulation in breast cancer tissue and its expression level were strongly correlated with higher TGF-1, mediating the effects of TGF-1 for cell invasion and chemoresistance by directly down-regulating phosphatase and tensin homolog (PTEN). As stated by Bao et al., 29 miR-21 expression plays a crucible role in regulating MEG-2 expression in colorectal cancer (CRC), which may represent a potential target for CRC diagnosis and management. Additionally, Liu et al. 11 stated that the MEG-2 expression level was down-regulated in gastric cancer and was suppressed by miR-181a-5p.
The miR-21 diagnostic performance was constructed utilizing ROC curve analysis, which had good sensitivity and specificity performance in discrimination of patients with breast cancer from study participants. In concordance with our results, Han et al., 24 Papadaki et al., 32 and Ali et al., 33 have identified miR-21 expression as a discriminator biomarker between breast cancer and healthy controls, providing a potential diagnostic candidate. As far as we know, this is the first investigation pointed to the correlation of TGF-β with the miR-21/MEG-2 expression. However, The small sample size can be considered the main limitation of this study due to this study having no financial support and not being funded. Therefore, further large-scale studies and clinical trials are needed to clarify the relationship between miR-21, MEG-2, and TGF-β with the therapeutic effect in clinical trials.
Conclusion
The study revealed that miR-21 and MEG-2 can be used as prospective diagnostic biomarkers and emphasized the significant correlation of TGF-β with miR-21 and MEG-2 expression in breast cancer.
Footnotes
Author contributions
Ekram Desouky, Saeed Mohamed and Nabil Hasona contributed to the study’s conception and design. Material preparation, data collection and analysis were performed by Nabil Hasona, Saeed Mohamed, and Amal Khaliefa. The first draft of the manuscript was written by Nabil Hasona, Ekram Desouky and Amal Khaliefa. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
This study was conducted in compliance with the Declaration of Helsinki, and approved by the Faculty of Medicine’s Ethics Committee at Beni-Suef University.
Informed consent
Patient consent for publication was covered by the informed consent document.
Data availability
All data generated or analysed during this study are included in this published article.
