Abstract
It has been demonstrated that this sulfasalazine (SF) inhibits the nuclear factor κB (NFκB) pathway, which regulates important genes during inflammation and immune answer. The aim of this work was to evaluate the effects of SF on carbon tetrachloride (CCl4)-induced liver fibrosis. We formed the following experimental groups of rats: controls, damage induced by chronic CCl4 (0.4 g/kg, intraperitoneally, three times a week for 8 weeks) administration and CCl4 + SF (100 mg/kg/day, postoperatively for 8 weeks) administration. We determined the activities of alanine aminotransferase (ALT), γ-glutamyl transpeptidase (γ-GTP), cyclooxygenase (COX)-1 and COX-2, lipid peroxidation, glutathione levels, collagen content, expression of transforming growth factor-β (TGF-β) and nuclear translocation of NFκB. SF was capable to inhibit the ALT and γ-GTP elevated levels induced with the CCl4 administration. SF had antioxidant properties, prevented the lipid peroxidation and the imbalance of reduced and oxidized glutathione produced by CCl4. Importantly, SF blocked the accumulation of collagen in the liver, the expression of TGF-β, the nuclear translocation of NFκB and the activity of COX-2, all induced with the administration of CCl4 in the rat. These results show that SF has strong antifibrotic properties because of its antioxidant properties and its ability to prevent nuclear translocation of NFκB and consequently the expression of TGF-β and the activity of COX-2.
Introduction
Sulfasalazine (SF) is a synthetic drug obtained from the combination of sulfapyridine and 5-aminosalicylic acid, an antibiotic and an anti-inflammatory agent, respectively. This drug is commonly used in the inflammatory diseases of the large intestine and rheumatoid arthritis. 1 It has been demonstrated that SF is a potent inhibitor of nuclear factor κB (NFκB) activation, which explains, at least in part, its pharmacological effects as immunomodulator in chronic inflammation. 2
Hepatic fibrosis results from chronic wound healing response, including inflammation and cellular proliferation producing cytokines and extracellular matrix (ECM) components. The ECM accumulation leads to the formation of nodules followed by architectural distortion and blood flow obstruction of the liver. 3 Hepatic stellate cells (HSCs) are the main producers of ECM during liver fibrosis. 4 HSCs are quiescent in physiological conditions, but they are activated during liver fibrogenesis. When a noxious stimulus is present, Kupffer cells, endothelial cells, monocytes and damaged hepatocytes produce cytokines such as interleukine-1β (IL-1β), tumor necrosis factor-α (TNF-α), platelet-derived growth factor and transforming growth factor-β (TGF-β) to induce HSC transdifferentiation. 5,6 These cytokines and growth factors are regulated by the NFκB, which is activated in response to oxidative stress and signal transduction of IL-1β and TNF-α, among others. 7
Actually, there are experimental models to study the pathophysiology of liver diseases. The carbon tetrachloride (CCl4)-induced liver fibrosis and cirrhosis model is the most used model because of its similarity to human cirrhosis.
8
The progression toward liver injury initiates with the CCl4 biotransformation to produce CCl3 and CCl3OO
Materials and methods
Chemicals
Sulfasalazine, carboxymethylcellulose (CMC), sodium thiosulfate, anthrone, thiobarbituric acid, chloramine-T, p-dimethylaminobenzaldehyde, γ-glutamyl-p-nitroanilide,
Study design
Wistar male rats of weight 90–100 g were used for the study, and the rats were fed Purina chow rat diet ad libitum. Four or five animals were housed per polycarbonate cage under controlled conditions (22 ± 2°C, 50–60% relative humidity and 12-h light–dark cycles).
Liver fibrosis was produced by intraperitoneal administration of CCl4 (0.4 g/kg of body weight) dissolved in mineral oil three times per week for 8 weeks. In order to determine the effects of SF on liver fibrosis, four groups were formed and treated for 8 weeks. Group 1 (n = 8) comprised control animals receiving the vehicle only (mineral oil, 0.25 mL, intraperitoneally); group 2 (n = 15) was administered with CCl4; group 3 (n = 15) was administered with CCl4 + SF (100 mg/kg, postoperatively, daily); group 4 received mineral oil and SF (as mentioned above) only. SF was suspended in CMC (0.7%). All animals were killed under light ether anesthesia.
To investigate the effect of SF on nuclear accumulation of p65 (which indicates NFκB activation), we formed four more groups as follows: the first group was administered with the vehicle only; the second group received CCl4; the third group received CCl4 + SF; finally, the fourth group was administered with SF only. The vehicle, CCl4 and drugs were administrated only once. These animals were killed 30 min after the administration of mineral oil or CCl4, as applicable.
Animals received humane care according to the institution’s guidelines and the Mexican Official Norm (NOM-062-ZOO-1999) regarding technical specifications for production, care and use of laboratory animals.
Serum enzyme activities
Blood was collected by cardiac puncture and samples were kept on ice until analysis. Samples were centrifuged at 1200g for 15 min to obtain the serum. Then, liver damage was determined by measuring the activities of alanine aminotransferase (ALT) 11 and γ-glutamyl transpeptidase (γ-GTP). 12
Assessment of lipid peroxidation
The extent of lipid peroxidation was determined in liver homogenates by measuring the formation of malondialdehyde (MDA) using the thiobarbituric acid method. 13 Protein was determined according to Bradford method using bovine serum albumin as standard. 14
Reduced and oxidized glutathione determinations in liver
The content of reduced (GSH) and oxidized (GSSG) glutathione was determined in liver. Liver samples were homogenized on ice using a polytron homogenizer. The solution used for the homogenization consisted of 3.75 mL of phosphate-ethylenediaminetetraacetic acid (EDTA) buffer and 1 mL of 25% H3PO4, which was used as a protein precipitant. The total homogenate was centrifuged at 4°C at 17,700g for 30 min to obtain the supernatant for the assay of GSH and GSSG.
Determination of GSH was performed according to Hissin and Hilf. 15 To a 0.1-mL of supernatant, 4.9 mL of the phosphate-EDTA buffer of pH 8.0 was added. The final assay mixture (2.0 mL) contained 100 μL of the diluted tissue supernatant, 1.8 mL of phosphate-EDTA buffer, and 100 μL of the O-phthalaldehyde (1 μg/μL) solution. After thorough mixing and incubation at room temperature for 15 min, the solution was transferred to a quartz cuvette. Fluorescence at 420 nm was determined with the excitation at 350 nm.
In the GSSG assay, 0.5 mL of the original supernatant was incubated at room temperature with 200 μL of 0.04 M N-ethylmaleimide for 30 min to interact with GSH present in the tissue. To this mixture, 4.3 mL of 0.1 N NaOH was added. A 100-μL portion of this mixture was taken for the measuring GSSG, using the above outlined assay for GSH, except that 0.1 N NaOH rather than phosphate-EDTA buffer was employed as a diluent.
Collagen quantification
In order to quantify the collagen content, fresh liver samples (100 mg) were placed in ampules, 2 mL of 6 N HCl was added, and then the samples were sealed and hydrolyzed at 100°C for 24 h. Next, the samples were evaporated at 50°C for 24 h and resuspended in 3 mL of sodium acetate citric buffer, pH 6.0; 0.5 g of activated charcoal was added and the mixture was stirred vigorously and then it was centrifuged at 3000 r/min for 15 min. Chloramine T (1 mL) was added to 1 mL of the supernatant. The mixture was kept for 20 min at room temperature, and the reaction was stopped by the addition of 2 M sodium thiosulfate and 1 N sodium hydroxide. The aqueous layer was transferred into test tubes. The oxidation product from hydroxyproline was converted into a pyrrole by boiling the samples. The pyrrole-containing samples were incubated with the Ehrlich’s reagent for 30 min and then the absorbance was read at 560 nm. Recovery of known amounts of standards was carried out on similar liver samples to provide quantification. 16
Histology
Liver samples were taken from all the animals and fixed with 10% formaldehyde in phosphate-buffered saline for 24 h. Tissue pieces were washed with tap water, dehydrated in alcohol, and embedded in paraffin. Sections of 5 μm thickness were mounted on glass slides covered previously with silane. Masson’s trichromic stain was performed in each slide.
Western blot assays
To carry out Western blot assays, the TriPure reagent (Roche Diagnostics, Indianapolis, Indiana, USA) was used to isolate total protein from liver tissue sample. Fresh tissue was homogenized in 1 mL of TriPure reagent, then 0.2 mL of chloroform was added to the homogenates, and the lower phase was treated with isopropanol to precipitate total protein. Samples were centrifuged at 12,000 r/min for 10 min at 4°C and then three washes were performed with 0.3 M guanidine hydrochloride in 95% ethanol. A final wash was performed with 100% ethanol, the samples were centrifuged as described previously, and the pellet was resuspended in 1% SDS.
The isolation of intact and stable nuclei consisted of iso-osmotic lysis procedure obtained as reported by Dyer and Hersog. 17 With this lysis procedure, the nuclear envelope remains intact even during further manipulations of washing, freezing and ultracentrifugation and provides nuclear extract for use in the NFκB assay.
Volumes equivalent to 50 μg of proteins (determined by the Lowry’s method) were transferred onto 12% polyacrylamide gel; separated proteins were transferred onto Inmuno-Blot™ PVDF membrane (BIO-RAD, Hercules, California, USA). Next, blots were blocked with 7% skim milk and 0.05% Tween-20 for 60 min at room temperature and independently incubated overnight at 4°C with specific antibody against TGF-β and p65, respectively (MAB1032 and MAB3026 from Millipore Corp., Billerica, Massachusetts, USA). The following day, membranes were washed and then exposed to a secondary peroxidase-labeled antibody (Zymed, San Francisco, California, USA) in the blocking solution for 1 h at room temperature. Blots were washed and protein was developed using the Western lightning™ Plus-ECL Enhanced Chemiluminescence detection system (NEN Life Sciences Products, Elmer LAS, Inc., Boston, Massachusetts, USA). Blots were stripped and incubated with a monoclonal antibody directed against β-actin, 18 which was used as a control to normalize the expression levels of cytokine protein. The procedure to strip membranes was as follows. First, blots were washed four times with phosphate-saline buffer, pH 7.4 (0.015 M, 0.9% NaCl), then immersed in stripping buffer (2-mercaptoethanol 100 mM, sodium dodecyl sulfate 2% and Tris-HCl 62.5 mM, pH 6.7) for 30 min at 60°C with gentle shaking, membranes were then washed five times with 0.05% Tween-20 in phosphate-saline buffer. Images were digitalized using the BioDoc-It System (UVP, Upland, California, USA) and then analyzed densitometrically using the Lab Works 4.0 Image Acquisition and Analysis software.
Activity of COX
Activity of COX was estimated using a commercial kit (COX Fluorescent Activity Assay Kit, 700200 Cayman Chemical Company, Mexico City, Mexico) and a fluorescent reader following the instructions provided by the manufacturer.
Statistical analysis
Data are expressed as mean ± standard error. Comparisons were carried out by analysis of variance followed by Tukey’s test, as appropriate, using Sigma Stat for Windows 2.0 Version (Systat Software, Inc., San José, California, USA). Differences were considered statistically significant when p < 0.05.
Results
The enzyme activities of serum ALT and γ-GTP serve as the useful tool to evaluate cell death or the increase in plasma membrane permeability. An increase in ALT activity was observed in the CCl4-treated group (Table 1); similarly, an elevation in the γ-GTP activity was found in the same group. These increases are associated with hepatocyte necrosis and cholestatic damage. In the rats, SF significantly prevented the activities of ALT and γ-GTP caused by the CCl4 administration.
Activities of ALT and γ-GTPa

ALT: alanine aminotransferase; γ-GTP: γ-glutamyl transpeptidase; CCl4, carbon tetrachloride; SF: sulfasalazine.
aActivities of ALT and γ-GTP were determined in serum from control rats, rats treated with CCl4 (CCl4), rats treated with CCl4 + SF and rats administered with SF alone. Values represent the mean of experiments performed in duplicate assays ± standard error (n = 6).
bSignificantly different from control, p < 0.05.
cSignificantly different from CCl4 group, p < 0.05.
As mentioned above, the oxidative stress is an important damage mechanism in this experimental model. The measurement of MDA, a product of lipid peroxidation, is useful to assess this process. SF completely inhibited the increase in MDA in the CCl4 group (Figure 1(a)). We determined glutathione levels in the liver to evaluate the redox equilibrium. A statistically significant reduction in the GSH/GSSG ratio can be observed in the CCl4 group when compared with the control group; SF partially but significantly prevented this alteration although caused a decrease in this parameter by itself (Figure 1(b)). There were no differences in the total content of glutathione between the groups (Figure 1(c)).

(a) Liver lipid peroxidation determined as MDA content; (b) reduced/oxidized ratio (GSH/GSSG); and (c) total glutathione (GSH + GSSG) determined in livers from control rats, rats treated with CCl4, rats treated with CCl4 + SF and rats administered with SF alone. Each bar represents the mean value of experiments performed in duplicate assays ± standard error (n = 6). a: Significantly different from control, p < 0.05; b: significantly different from CCl4, p < 0.05. MDA: malondialdehyde; SF: sulfasalazine; GSH: reduced glutathione; GSSG: oxidized glutathione; CCl4, carbon tetrachloride.
The collagen content is the most important parameter used to evaluate liver fibrosis because it is the main component of the connective tissue accumulated in this disease. Herein, we observed a significant increase in this protein in the group treated with CCl4 during 8 weeks; SF prevented collagen deposition in the liver (Figure 2(a)). In order to corroborate the biochemical findings of collagen content, we decided to perform the Masson’s trichromic stain of liver slices. The normal parenchyma of the liver is shown in Figure 2(a), panel A; the CCl4 group had altered parenchyma architecture and severe necrosis, due to the accumulation of collagen fibers (Figure 2(b), panel B); SF maintained the normal architecture, since the liver slices of the group treated with CCl4 + SF showed less necrotic areas and lower collagen fibers compared with the CCl4-treated group (Figure 2(b), panel C). This histological approach is in agreement with the enzymatic activity of ALT (Table 1) and the collagen content in the liver (Figure 2(a)).
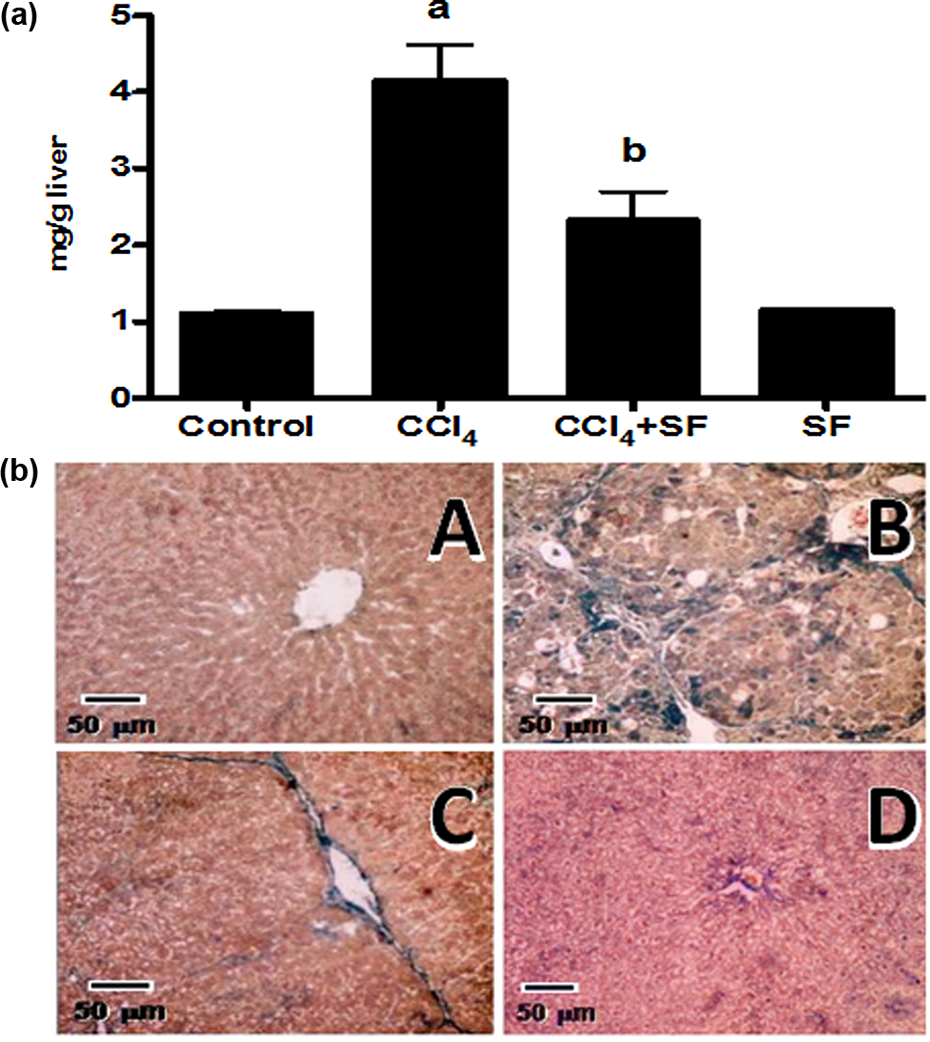
Liver fibrosis determined as collagen content and trichromic stain of I liver slices. (a) Collagen content determined in livers from control rats, rats treated with CCl4, rats treated with CCl4 + SF and rats administered with SF alone. Each bar represents the mean value of experiments performed in duplicate assays ± standard error (n = 6). a: Significantly different from control, p < 0.05; b: significantly different from CCl4, p < 0.05. (b) Representative Mallory’s trichromic stain of liver sections obtained from rats treated with vehicle (panel A), CCl4 (panel B), CCl4 + SF (panel C) and rats administered with SF alone (panel D). Collagen can be visualized as blue color. SF: sulfasalazine; CCl4, carbon tetrachloride.
The main mechanism of action of SF is the NFκB inhibition, and because this protein plays a role in inflammatory responses, we determined its translocation to the nucleus. CCl4 induces NFκB translocation compared with the control group; importantly, SF prevented this effect (Figure 3).

SF blockade of nuclear translocation of NFκB (p65). Western blot of nuclear extracts was used to determine the presence of p65 into the nucleus in samples of liver tissue from control rats, rats treated with CCl4, rats treated with CCl4 + SF and rats administered with SF alone. 3-Actin was used as an internal control. Signal intensities were determined by densitometric analysis of treated blots and values calculated as the ratio of p65/β-actin. Each bar represents the mean value of experiments performed in duplicate assays ± standard error (n = 6). a: Significantly different from control, p < 0.05; b: significantly different from CCl4, p < 0.05. SF: sulfasalazine; CCl4, carbon tetrachloride; NFκB: nuclear factor κB.
TGF-β is an important cytokine in the progression toward liver fibrosis. We evaluated the effect of SF on TGF-β expression; as it was expected, CCl4 induced the TGF-β expression and SF inhibited it significantly (Figure 4).

SF blockade of TGF-β protein expression in samples of liver tissue determined by Western blot analysis from control rats, rats treated with CCl4, rats treated with CCl4 + SF and rats administered with SF alone. β-Actin was used as an internal control. Signal intensities were determined by densitometric analysis of treated blots and values calculated as the ratio of TGF-β/β-actin. Each bar represents the mean value of experiments performed in duplicate assays ± standard error (n = 6). a: Significantly different from control, p < 0.05; b: significantly different from CCl4, p < 0.05. SF: sulfasalazine; CCl4, carbon tetrachloride; TGF-β: transforming growth factor-β.
The chronic inflammation plays an important role in the establishment of liver diseases. The importance of COX in the liver fibrosis has been demonstrated. Figure 5 shows the activity of both the isoforms of this enzyme determined using a commercial kit. The activity of COX-2 increased in the rats treated with CCl4 for 8 weeks, while the COX-1 activity was unaltered; SF prevented the increase in COX-2 activity.

Activities of COX-1 and COX-2 determined with a commercial kit in livers from control rats, rats treated with CCl4, rats treated with CCl4 + SF and rats administered with SF alone. Each bar represents the mean value of experiments performed in duplicate assays ± standard error (n = 6). a: Significantly different from control, p < 0.05; b: significantly different from CCl4, p < 0.05. SF: sulfasalazine; CCl4, carbon tetrachloride; COX: cyclooxygenase.
Discussion
Liver fibrosis and cirrhosis are related to the accumulation of ECM components, leading to hepatic insufficiency. 19 Hepatic chronic diseases are the results of recurrent liver injuries leading to an imbalance between fibrogenesis and fibrolysis. Kupffer cells are the main source of cytokines involved in the signals that lead to the production of scar tissue by the activation of HSCs. 7 SF inhibits the nuclear translocation of NFκB, a protein that regulates the transcription of several target genes including IL-1β, TNF-α and COX-2, which have an important role in the establishment of liver injury. 2,9 Thus, we evaluated the effects of SF in the experimental model of liver fibrosis induced by the administration of CCl4. We demonstrated the ability of SF to prevent oxidative stress, inflammation and hepatic fibrosis induced by CCl4 treatment in the rat.
As mentioned in the Results section, oxidative stress was determined by the measurement of lipid peroxidation (levels of MDA) and the ratio GSH/GSSG and total content of glutathione (GSH + GSSG). Lipid peroxidation is an indicative of cell membrane damage produced by free radicals and detergent action of bile salts. 20 Glutathione is a tripeptide that protects the organism from free radicals and reacts with toxic substances to eliminate them. 21 Both these markers showed the antioxidant properties of SF. According to our results, it has been reported that SF and its metabolites, 5-aminosalicylic acid and sulfapyridine, act as scavengers of free radicals. 22 Linares et al. 23 found an important effect of SF on the liver and kidney of rats treated with SF; they reported alterations in the oxidative markers and attribute the liver and kidney toxicity to this finding. In this regard, we found an imbalance in the GSH/GSSH ratio produced by SF itself; although there is no alteration in the total content of glutathione and the lipid peroxidation in the same rats. This result is probably due to the fact that we used a lower dose and despite the GSH/GSSG imbalance, it was not sufficient to induce a real damage to the liver.
HSCs undergo a transdifferentiation process, acquiring proinflammatory and fibrogenic abilities mainly upon stimulation by TGF-β. 19 TGF-β signaling induces nuclear translocation of co-Smads and, consequently, gene transcriptions, such as collagen type I. 24 In fact, in this study, SF was capable of preventing TGF-β expression induced by CCl4 and then inhibiting the production and accumulation of collagen in the liver, biochemical and histologically evaluated, suggesting lower HSCs activation.
COX is a protein that plays an important role in the inflammatory process; it synthesizes prostaglandins, the principal mediators of inflammation. 25 Ramirez-Yañez et al. demonstrated a direct correlation between PGE2, an enzymatic product of COX, and TGF-β levels in vitro and in vivo. 26 It has been shown that COX-1 maintains its expression without a change in healthy and damaged liver but in liver diseases COX-2 expression increase both in humans and experimental animals, compared with normal conditions. 27 –29 Herein, we demonstrated the ability of SF to prevent CCl4-induced inflammation caused due to COX-2 activity. This finding could explain the effect observed on TGF-β expression and collagen deposition in the liver. It is important to point out that COX activity produces reactive oxygen species 30 and because SF prevented its activity, it might be an alternative mechanism to inhibit the redox imbalance.
NFκB belongs to a family of transcription factors that importantly regulate inflammation. The canonical pathway is triggered by proinflammatory cytokines such as TNF-α and IL-1β; this signal leads to p65:p50 nuclear accumulation and activation of gene transcription. When the canonical pathway is activated, IκB is phosphorylated by the IkB kinase (IKK) and degraded via ubiquitin-26S proteasome, leading to the nuclear translocation of NFκB and its transcriptional activity. 30 Wahl et al. demonstrated that SF is a specific inhibitor of NFκB; this drug, but not its metabolites 5-aminosalicylic acid and sulfapyridine, was capable of preventing IκB phosphorylation. 2 We showed the ability of SF to block the p65 nuclear accumulation in the liver induced with the administration of CCl4 to the rats. This effect is very important since the expression of COX-2 is induced through the NFκB pathway, 31 thus explaining our finding of activity of COX-2 inhibition by SF.
In this study, we showed the beneficial effects of SF on the CCl4-induced liver fibrosis. In summary, SF prevented oxidative stress, inflammation, nuclear translocation of NFκB and finally collagen deposition in the liver. Due to the properties of SF, in this experimental model, the hepatic markers of necrotic and cholestatic damage were normal in the rats that were treated with SF.
This study shows the antifibrotic properties of SF in the CCl4-induced liver fibrosis model of the rat. This experimental model is mostly used to study promising beneficial drugs in the treatment of the liver fibrosis. However, distinct models of liver diseases available to evaluate the possible helpful effect of SF. So, it is necessary to carry out the study of this drug in more experimental models in order to understand its properties in liver diseases caused by different etiologic agents and at different disease phase. Then, if SF shows positive properties, it should be considered to carry out clinical probes.
Footnotes
Acknowledgements
The authors thank Mr Benjamín Salinas Hernández, Mr Ramón Hernández and QFB Silvia Galindo for their excellent technical assistance.
Declaration of Conflict of Interest
E.C. was a fellow of Conacyt. All other authors declared no conflicts of interest.
