Abstract
Background
We investigated the level of Cysteine-rich 61 (CYR61) in premature ovarian failure as well as its regulatory molecular mechanism in this study.
Methods and Results
Cyclophosphamide (CTX) was used to induce OGCs (rat ovarian granulosa cells) and rats to establish in vivo and in vitro premature ovarian failure models. H&E staining was used to detect the pathological changes of ovarian histopathology. Si-NLRP3 (NOD-like receptor thermal protein domain associated protein 3, NLRP3) and si-CYR61 were transfected into OGCs using lipofectamine 3000. RT-qPCR and western blot were used to detect the expressions of CYR61 in ovarian tissue and OGCs. It showed that the expression of CYR61 was significantly down-regulated in premature ovarian failure model. Cell viability was detected using a Cell Counting Kit-8 (CCK-8) kit. TUNEL (Terminal deoxynucleotidyl transferase-mediated dUTP biotin nick end labeling) staining was used to detect the apoptosis. 5-Ethynyl-2′-deoxyuridine (EdU) and SA-β-gal (senescence-associated β-galactosidase) staining were used to assess the proliferation and senescence. The expression of CYR61 in OGCs and ovarian tissues were detected by immunofluorescence and immunohistochemical staining. Overexpression of CYR61 significantly promoted OGCs proliferation and inhibited pyroptosis and apoptosis. Western blot was used to detect the protein expressions of p53 and p21 in OGCs. Flow cytometry was used to detect the pyroptosis. CYR61 overexpression inhibited the expression of NLRP3 and caspase-1 in CTX-induced OGCs according to western blot results. Moreover, we found that CYR61 overexpression down-regulated the protein expressions of p53 and p21 in CTX-induced OGCs.
Conclusion:
CYR61 inhibited CTX-induced OGCs senescence, and the mechanism may be related to the regulation of caspase-1/NLRP3-induced pyroptosis.
Introduction
It is reported 1 that around two million women suffer from cancer in China every year, and chemotherapy drugs such as cyclophosphamide can improve patients' survival, but also aggravate ovarian granulosa cell damage, which could induce premature ovarian failure (POF). Premature ovarian failure, which usually manifests as ovarian insufficiency, is triggered by a variety of factors, including chemotherapy drug use, metabolic disorders, and estrogen imbalance.2–4 Premature ovarian failure is a serious threat to women’s health and can lead to a variety of diseases including sexual dysfunction, infertility, osteoporosis and cardiovascular disease.5–7 As POF progresses, menopause will occur, leading to premature depletion of the primordial follicular pool of the ovary. With the increased incidence of gynecologic tumors, premature ovarian failure due to chemotherapy is receiving increasing attention. One study reported that the prevalence of premature ovarian failure in women is about 4%, and the current common clinical treatment strategy is hormone replacement therapy. 8 Due to the irreversible pathological mechanism of premature ovarian failure and the accompanying side effects of hormonal therapy, it seems necessary to explore new treatment strategies for premature ovarian failure.
As a CNN (immediate early gene) family molecule, Cysteine-rich 61 (CYR61), also known as CCN1. CYR61 could directly bind to a variety of integrins and regulate cell proliferation, migration and invasion. Previous research has indicated that CYR61 derived from ADSCs (adipose-derived stem cells) promotes CRC (Colorectal cancer) malignant progression by binding to the integrin αVβ5, suggesting that targeting CYR61/αVβ5 is a potential anti-tumor strategy. 9 Sano et al. reported that activation of WNT/β-catenin signaling pathway can promote upregulation of CYR61 expression levels, which may be involved in the malignant development of pancreatic cancer. 10 Chen et al. found that ATF3 (activating transcription factor 3) could upregulate the expression of CYR61 and play a key role as a tumor suppressor in the treatment of hepatocellular carcinoma. 11 CYR61 has been reported to regulate the secretion of IL-6 (interleukin-6) and CRP (C-reactive protein) in the ovarian cancer microenvironment, promoting tumor metastasis and invasion. 12 However, the molecular regulatory mechanism of CYR61 in premature ovarian failure has rarely been reported.
In the present study, we mainly established POF model to analyze the expression level of CYR61 in vivo and in vitro. We also explored the effects of CYR61 on the cell proliferation and aging in CTX-induced OGCs, and we found that CYR61 may alleviate CTX-induced premature ovarian failure by regulating pyroptosis.
Materials and methods
Cell culture and transfection
Ovarian granulosa cells were extracted according to the previously reported method. 13 Briefly, 8-week-old rats were anesthetized with sodium pentobarbital and ovarian tissues were isolated under aseptic conditions. Follicular cells were then isolated under a microscope. The obtained cells were washed with PBS and cultured in medium containing 10% fetal bovine serum, 10 ng/mL growth hormone and 15 ng/mL estradiol in an incubator under appropriate conditions. Following the instructions, 5 μM si-NC (negative control) or si-CYR61 was transfected into OGCs cells using Lipofectamine 3000. After 48 h transfection, RT-qPCR analysis was performed to confirm the success of transfection.
Animals and premature ovarian failure model
Eight-week-old female SD rats (n = 12, SPF, specific pathogen free) were obtained from Beijing HFK Bioscience Co., LTD. The rats were kept in animal houses with the right temperature, humidity and light cycle, and had free access to food and water. The rats were randomly divided into control and model groups. The rats in the model group were given intraperitoneal injection of 50 mg/kg cyclophosphamide, and the rats in the control group were given an equal volume of saline. After one week of feeding, the rats were anesthetized and executed by intraperitoneal injection of an overdose of 1% sodium pentobarbital solution. All animal care and experimental procedures were approved by the Animal Care and Use Committee of the Institute of Radiation Medicine, Chinese Academy of Medical Sciences (Approval No. IRM-DWLL-20022119)
H&E (hematoxylin and eosin) staining
H&E staining was performed according to the reported method, 14 and 4-μm sections were made by fixing ovarian tissue with 4% paraformaldehyde for 24 h and paraffin embedding. Then stained with hematoxylin and eosin, respectively. Finally, the tissue morphology was observed under a light microscope and randomly photographed and recorded.
5-Ethynyl-2′-deoxyuridine staining
As previously reported, 15 the treated cells were stained with the EdU kit, following the manufacturer’s instructions. Nuclei were marked with DAPI staining. The fields of view were then randomly selected for observation under a fluorescent microscope and photographed. The EdU-positive cells and DAPI-positive cells were counted by Image J software. The ratio of EdU-positive cells/DAPI-positive cells ratio was calculated to evaluate the EdU-positive rate.
Cell Counting Kit-8 kit assay
The cell activity was determined by following the CCK-8 kit instructions. Briefly, cells were cultured in 96-well plates for 24 h and subsequently transfected with NC, si-CYR61, or pcDNA3.1-CYR61. After transfection, 10 μL CCK-8 was added and incubated at 37°C for 3 h. Finally, the absorbance was detected at 450 nm by microplate reader.
RT-qPCR analysis
TRIzol reagent was used to extract total RNA from ovarian tissues and OGCs cells. RT-qPCR experiments were performed using SYBR Premix Dimer Eraser (Perfect Real Time) assay kits on LightCycler 480 (Roche) as previously reported. 16 GAPDH was used as an internal reference and 2−ΔΔCt method was used to calculate the relative expression levels of target genes.
Western blotting assay
Protein was extracted from the cells and tissue using RIPA lysate, and protein concentration was determined by BCA kit. The protein samples were diluted to the same concentration with the loading buffer and stored in a −80°C refrigerator until used. As previously reported, 17 protein samples were separated by sodium dodecyl sulfate polyAcrylamide gel electrophoresis (SDS-PAGE), transferred to polyvinylidene fluoride (PVDF) membranes, and incubated with primary and secondary antibodies. Finally, the exposure was developed in the dark using enhanced chemiluminescence (ECL) reagent. The relative expression levels of the proteins were evaluated by measuring the gray values.
Immunofluorescence staining
As previously reported, 18 OGCs cells were fixed using 4% paraformaldehyde for 15 min 0.1% Trizon X-100 was used to break the membrane, followed by 5% BSA closure. CYR61 primary antibody was incubated overnight for 4°C. The nuclei were labeled with DAPI. Finally, images were observed under a confocal microscope and randomly selected fields of view were photographed.
Flow cytometry
As previously reported, 19 pyroptosis was determined using the FAM-FLICA caspase-1 assay kit. Briefly, the cells were stained with FAM-FLICA and PI for 30 min at room temperature. The fluorescence intensity of the samples was subsequently measured by flow cytometry. The positive rate of pyroptosis was calculated according to the percentage of double staining.
Terminal deoxynucleotidyl transferase-mediated dUTP biotin nick end labeling staining
After transfection, the rat OGCs cells were continued to be cultured for 24 h and TUNEL assay was performed according to the kit instructions. Briefly, OGCs cells were fixed with 4% paraformaldehyde, TUNEL solution stained, and DAPI stained for cell nuclei. 20 Finally, the TUNEL positive rate was observed and analyzed under confocal microscope.
SA-β-gal staining
The OGCs cells were collected and fixed with 4% paraformaldehyde for 5 min. After incubation for 16 h in the dark with the addition of SA-β-gal solution according to the instructions. 21 The field of view was randomly selected under the microscope for observation and photographed.
Statistical analysis
The experimental results were expressed as mean ± SD, and SPSS 22.0 software package was used for statistical analysis. For comparison between two or more groups, differences between groups were analyzed by Student’s t-test or ANOVA. A p value of less than 0.05 indicates statistical significance.
Results
Effects of cyclophosphamide on rat OGCs and changes of CYR61 expression
First, H&E staining was performed to assess morphological changes in the ovaries. As shown in Figure 1(a), we showed many mature and healthy follicles were present in the ovaries of the control group, whereas many atretic follicles were observed in the POF group. As shown in Figure 1(b) and (c), we found the levels of CYR61 in POF group were reduced significantly compared with control group. Besides, immunohistochemical staining results (Figure 1(d)) also showed the same trend. Cyclophosphamide causes injured in OGCs and a decreased expression of CYR61. (a) Representative H&E staining images, magnified 100 times below. (b) RT-qPCR was applied to measure the mRNA level of CYR61 in different groups. (c) Western blotting assay was performed to detect the protein expressions of CYR61 in different groups. (d) The representative images of immunohistochemical staining for CYR61. The data was expressed as mean ± SD, n = 3. *p < 0.05, **p < 0.01, versus control group.
The expression of CYR61 in cyclophosphamide induced OGCs in vitro
In vitro experiments, CCK-8 kit results (Figure 2(a)) showed the cell viability of OGCs cells in CTX group was significantly decreased in a dose-dependent manner. EdU staining results (Figure 2(b)) showed that EdU positive rate of OGCs cells decreased significantly in CTX group. According to SA-β-gal staining results (Figure 2(C)), we found that compared to control and DMSO group, SA-β-gal positive cells were increased significantly in CTX group, and the difference was significant. As shown in Fig. 2DE, it indicated that the mRNA and protein expressions of CYR61 in CTX group were significantly lower than those in the control group. Immunofluorescence staining results (Figure 2(e)) showed the same trend. It suggested that the expression of CYR61 was down-regulated in cyclophosphamide induced-OGCs. The expression of CYR61 in cyclophosphamide induced OGCs in vitro. (a) CCK-8 kit was applied to detect the cell viability. (b) EdU staining was used to detect the proliferation. (c) SA-β-gal staining was used to detect the senescence. (d) RT-qPCR was performed to detect the mRNA expressions of CYR61 in different groups. (e) Western blot was performed to detect the protein expression of CYR61. (f) The representative images of immunohistochemical staining for CYR61. *p < 0.05, **p < 0.01, versus DMSO group.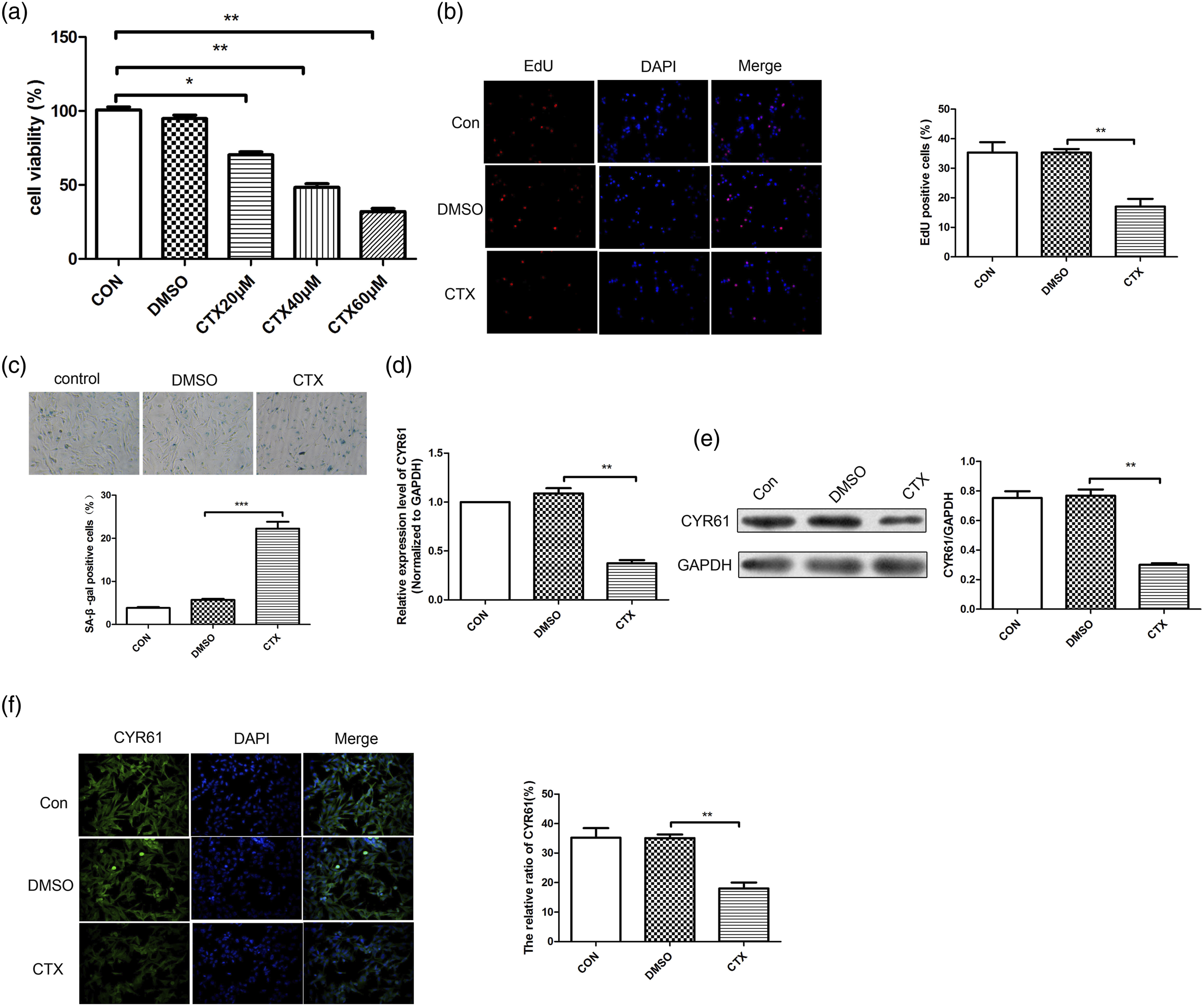
Cysteine-rich 61 regulated cyclophosphamide induced proliferation inhibition and aging in OGCs
Next, we further investigated the effect of CYR61 on proliferation inhibition of CTX-induced OGCs cells. Western blot results (Figure 3(a)) confirmed the success of transfection. CCK-8 kit (Figure 3(b)) revealed compared with CTX + NC group, the cell viability in CTX + pcDNA3.1-CYR61 group was increased significantly, while the cell viability in CTX + si-CYR61 group was reduced obviously. EdU staining results (Figure 3(c)) showed the same trend. SA-β-gal staining (Figure 3(d)) displayed that compared with the CTX + NC group, SA-β-gal positive rate was significantly lower in the CTX + pcDNA3.1-CYR61 group. However, SA-β-gal positive rate was significantly increased in the CTX + si-CYR61 group. Besides, western blot results (Figure 3(e)) revealed that the expression of p53 and p21 in CTX + pcDNA3.1-CYR61 group was significantly less than that in CTX + NC group, while the expressions of p53 and p21 group were increased significantly. Taken together, we suggested that CYR61 attenuated cyclophosphamide-induced proliferation inhibition in OGCs. CYR61 regulated cyclophosphamide induced proliferation inhibition and aging in OGCs. (a) Western blot was performed to detect the CYR61 proteins in different groups. (b) CCK-8 was applied to detect the cell viability. (c) EdU staining was used to detect the proliferation. (d) SA-β-gal staining was used to detect the senescence. (e) Western blotting was used to detect the expressions of p21 and p53. *p < 0.05, **p < 0.01, versus CTX + vector group.
Up-regulation of CYR61 significantly reversed CTX-induced pyrocytosis in OGCs
Then, pyroapoptosis was detected by flow cytometry. It (Figure 4(a)) showed that compared with DMSO group, the pyrocytosis in CTX + pcDNA3.1 group was increased significantly. While the pyrocytosis in CTX + pcDNA3.1-CYR61 group was reduced significantly. TUNEL staining results (Figure 4(b)) showed that the apoptosis was increased significantly in CTX + pcDNA3.1 group compared with DMSO group. Compared with CTX + pcDNA3.1 group, the apoptosis was reduced significantly in CTX + pcDNA3.1-CYR61 group. As shown in Figure 4(c), we found the expressions of caspase-1, IL-1β, GSDMD (gasdermin D) and NLRP3 in CTX + pcDNA3.1 group were increased significantly compared with DMSO group. While compared with CTX + pcDNA3.1 group, the expressions of caspase-1, IL-1β, GSDMD and NLRP3 in CTX + pcDNA3.1-CYR61 group were reduced significantly. Besides, we used immunofluorescence staining to detect the positive rate of caspase-1 in different groups. It (Figure 4(d)) showed that compared with DMSO group, the positive rate of caspase-1 in CTX + pcDNA3.1 group was increased significantly. While the positive rate of caspase-1 in CTX + pcDNA3.1-CYR61 group was reduced obviously. Up-regulation of CYR61 significantly reversed CTX-induced pyrocytosis in OGCs. (a) Pyroptosis was detected using Flow cytometry. (b) TUNEL staining was performed to detect the apoptosis. (c) The expressions of caspase-1, GSDMD, IL-1β and NLRP3 were detected using western blot. (d) Positive rate of caspase-1 in different groups were detected by immunofluorescence staining. *p < 0.05, **p < 0.01, versus DMSO group; #p < 0.05, ##p < 0.01, versus CTX + pcDNA3.1 group.
Down-regulation of CYR61 promotes CTX-induced pyrocytosis in OGCs
Similarly, pyroapoptosis was detected by flow cytometry. It (Figure 5(a)) showed compared with DMSO group, the pyrocytosis in CTX + NC group was increased significantly. While the pyrocytosis in CTX + si-CYR61 group was increased significantly. TUNEL staining results (Figure 5(b)) showed that the apoptosis was increased significantly in CTX + NC group compared with DMSO group. Compared with CTX + NC group, the apoptosis was increased significantly in CTX + si-CYR61 group. As shown in Figure 5(c), we found the levels of caspase-1, IL-1β, GSDMD and NLRP3 in CTX + NC group were increased significantly compared with DMSO group. While compared with CTX + NC group, the expressions of caspase-1, IL-1β, GSDMD and NLRP3 in CTX + pcDNA3.1-CYR61 group were increased significantly. Besides, we used immunofluorescence staining to detect the positive rate of caspase-1 in different groups. It (Figure 5(d)) showed the positive rate of caspase-1 in CTX + NC group was increased significantly compared with DMSO group. While the positive rate of caspase-1 in CTX + si-CYR61 group was increased obviously. In summary, down-regulation of CYR61 significantly promotes CTX-induced pyrocytosis in OGCs. Down-regulation of CYR61 promotes CTX-induced pyrocytosis in OGCs. (a) Flow cytometry was used to detect the pyroptosis. (b) TUNEL was performed to detect apoptosis. (c) Western blot was used to detect the expressions of caspase-1, GSDMD, IL-1β and NLRP3 (d) Positive rate of caspase-1 in different groups were detected by immunofluorescence staining. *p < 0.05, **p vs DMSO group; #p < 0.05, versus CTX + NC group.
Si-NLRP3 or caspase-1 inhibitor represses CTX-induced OGCs proliferation inhibition and aging
Furthermore, the effect of si-NLRP3 or caspase-1 inhibitor on CTX-induced proliferation of OGCs was investigated to investigate whether CTX -induced OGCs is associated with pyrocytosis. As shown in Figure 6(a), we found that the expression of NLRP3 was reduced significantly in si-NLRP3 group compared with NC group. Compared with control group, the expression of caspase-1 was reduced significantly in VX765 group. In addition, the positive rate of caspase-1 was detected by immunofluorescence staining. It (Figure 6(b)) revealed that compared with CTX + vector group, the positive rate of caspase-1 in CTX + si-NLRP3 and CTX + VX-765 group reduced significantly. The level of pyrocytosis measured by flow cytometry (Figure 6(c)) showed a same trend. EdU staining results (Figure 6(d)) showed that compared with CTX + vector group, the proliferation in CTX + si-NLRP3 group and CTX + VX-765 group was increased significantly. Reports have confirmed that p53 and p21 are key signaling molecules in the aging process. As shown in Figure 6(e), it showed that compared with CTX + NC group, the SA-β-gal positive rate were reduced significantly both in CTX + si-NLRP3 and CTX + VX-765 group. Western blot results (Figure 6(f)) revealed compared with CTX + vector group, the expressions of p53 and p21 were reduced significantly in CTX + si-NLRP3 and CTX + VX765 group. In conclusion, it suggested that si-NLRP3 or caspase-1 inhibitor (VX765) represses CTX-induced OGCs proliferation inhibition and aging. Si-NLRP3 or caspase-1 inhibitor represses CTX-induced OGCs proliferation inhibition and aging. (a) Western blot was used to detect the protein expressions of NLRP3 and caspase-1. (b) Representative immunofluorescence image for detection of caspase-1. (c) Flow cytometry detection of pyroptosis. (d) Representative EdU staining images. (e) SA-β-gal staining was used to detect the senescence. (f) Western blot was performed to detect the protein expressions of p21 and p53 in different groups. *p < 0.05, **p < 0.01, versus NC/CTX + vector group.
Discussion
Cyclophosphamide is widely used in the treatment of gynecologic tumors, but its alteration causing premature ovarian failure has also attracted clinical attention. Premature ovarian failure is a reproductive disorder accompanied by reduced estrogen levels and is the leading cause of infertility in women of childbearing age under 40 years. 22 In the study, we used a cyclophosphamide-induced premature ovarian failure model to investigate the expression level of CYR61 and its potential molecular regulatory mechanisms in premature ovarian failure. We found that CYR61 was down-regulated in premature ovarian failure rats and that overexpression of CYR61 significantly slowed cyclophosphamide-induced proliferation inhibition and pyroptosisof ovarian granulosa cells.
CYR61 is highly expressed in a variety of cancers and promotes cell proliferation, invasion, and metastasis.23–25 IGF1 has been reported to upregulate CYR61 expression, activate the/PI3K/Akt signaling pathway, and promote breast cancer cell proliferation and invasio. 26 Studies have reported that CYR61 was highly expressed in both placental vascular endothelial cells and stromal cells of pregnant women. 27 Moreover, CYR61 could mediate VEGF, wnt and other signaling proteins, which are involved in regulating ovarian granulosa cell proliferation, migration and angiogenesis. 28 Kipkeew et al. suggested that CYR61 is a key regulator of proliferation and invasion control in extravillous trophoblasts. 29 In the study, we found that the expression of CYR61 was significantly downregulated in the POF model, while overexpression of CYR61 significantly alleviate the effects of CTX-induced cell injury in OGCs. We confirmed that CYR61 is closely related to the cell proliferation of CTX-induced OGCs.
In addition, it has been found that CYR61 could act as a direct target molecule of wnt signaling pathway and participate in regulating the development of muscle aging. 30 Jun et al. reported that CYR61 can activate the RAC1/NOX1 signaling axis and promote fibroblast senescence. 31 Similarly, in combination with SA-β-gal staining results, we found that overexpression of CYR61 significantly attenuated CTX-induced ovarian granulosa cell senescence. It is well known that the oncogene p53 plays a key role in promoting the aging process by upregulating the expression of p21. 32 We found that CYR61 significantly down-regulated CTX-induced expression levels of p53 and p21 genes and proteins. To address the regulatory mechanism of CYR61 in aging, we found that overexpression of CYR61 attenuated CTX-induced inhibition of ovarian proliferation and the effects of cellular senescence.
In addition, it has been reported that premature ovarian failure is accompanied by a chronic inflammatory response and that pyroptosis is an inflammatory response-mediated cellular necrosis process with complex endogenous molecular mechanisms. 33 The inflammatory vesicle NLRP3 could act as a regulator to promote caspase-1-dependent IL-1β, IL-18 and cleavage of GSDMD. 34 Critically, upregulation of inflammatory cytokine expression can accelerate cellular senescence. Our study also suggested that CYR61 significantly inhibited NLRP3- and caspase-1-mediated pyroptosis. Combined with the previous findings, we hypothesized that CYR61 attenuated CTX-induced cellular senescence, which may be closely associated with its inhibition of pyroptosis.
In summary, we found significant downregulation in both ovarian tissue and granulosa cells in the POF model. Overexpression of CYR61 significantly inhibited CTX-induced granulosa cell senescence, and the mechanism may be related to the regulation of caspase-1/NLRP3-induced pyroptosis. Our results suggested that CYR61 may serve as a potential drug target for the treatment of POF.
Footnotes
Acknowledgement
We are grateful to all participates for their contributions for the present study.
Authors contributions
Hongxia Xu, Xiumin Bao, Junya Yang, Hanxin Kong, Yan Li, Zhiwei Sun. D. X.: Writing, Conceptualization, Methodology, Software. X. B.: Data curation, Writing, Original draft preparation. J. Y.: Visualization, Investigation, Software. H. K.: Methodology. Y. L.: Data curation. Z. S.: Reviewing and Editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the project of Medical Leading Talents Fund of Yunnan Province (L-2017016).
Ethical Approval
All animal care and experimental procedures were approved by the Animal Care and Use Committee of the Institute of Radiation Medicine, Chinese Academy of Medical Sciences (Approval No. IRM-DWLL-20022119).
Availability of data and materials
All data in the article will be made available by the authors, without undue reservation.
