Abstract
Aluminum phosphide (AlP) poisoning is a serious medical emergency with a high mortality rate. The absence of an exact antidote for AlP poisoning necessitates the quest for alternative treatment options. The study sought to assess the efficacy of adding L-carnitine or medicated paraffin oil to the conventional approach of treatment employed in cases of acute AlP poisoning. We conducted a 1 year, randomized, controlled, parallel-group, single-blind clinical study. 96 individuals with acute AlP poisoning were randomly assigned to one of three groups. The standard AlP therapy was administered to all groups according to the Poison Control Center guidelines at the Ain-Shams University hospitals. All patients underwent a medical history review, clinical examination, and laboratory tests. The outcomes were assessed. The participants in the study groups had mean ages ranging from 25.6 to 26.3 years. The cases analyzed were evenly distributed between genders, with the majority originating from rural areas. The average delay time varied from 2.9 to 4.2 h. All patients in the study reported ingesting AlP during suicide attempts. 12 hours after admission, many clinical and biochemical data improved in both intervention groups including cytochrome c oxidase, caspase-3, caspase-9, catalase, and superoxide dismutase. The intervention groups required significantly less mechanical ventilation and had a lower mortality rate than the control group. Decontamination with paraffin oil could be advantageous for reducing the severity of AlP poisoning, improving prognosis, and lowering the mortality rate.
Keywords
Introduction
Aluminum phosphide (AlP) is a fumigant used to ensure the safety of stored grains. 1 In many developing nations, including Egypt, Iran, and India, it is the leading chemical agent used in suicide by poisoning, with a high mortality rate.2,3 AlP is available in tablet form and emits a highly toxic phosphine (PH3) gas when exposed to moisture and an acidic environment. 2 Its low cost, ease of access, and high toxicity, combined with the lack of definitive treatment or antidote for acutely poisoned patients, resulted in significant public health problems. 3 Various studies indicate that even 500 mg of this compound is lethal to humans, with mortality rates as high as 70%–100%. 4
Phosphine induces mitochondrial dysfunction by inhibiting the cytochrome c oxidase (Complex IV) activity and disrupting the Complex I and Complex II chains. Ultimately, this inhibits adenosine triphosphate (ATP) formation. 5 In addition, it has been reported that PH3 causes oxidative stress by inhibiting antioxidant enzymes such as catalase (CAT). Furthermore, it releases reactive oxygen species (ROS), resulting in cellular apoptosis and damage to cell membranes and nucleic acids, as indicated by elevated active caspase-3 and caspase-9 levels. 6 Patients typically die due to cardiovascular failure, refractory shock, severe acidemia, fulminant hepatic failure, and/or acute respiratory distress syndrome. 7
In almost all cases, the current standard treatment for acute AlP poisoning is supportive care until the toxin has been eliminated by the lungs and kidneys. 7 Even in intensive care units (ICUs), the mortality rate associated with AlP poisoning is extremely high, exceeding 60% in hospitals with advanced life-support equipment and a high level of specialization. 3 Many AlP-poisoned patients deteriorate despite receiving adequate supportive care and no known antidote exists. It is one of the leading causes of poisoning deaths worldwide.3,8,9
N-acetyl cysteine (NAC) is an innovative thiol compound that functions as a precursor to L-cysteine and reduced glutathione (GSH). Moreover, it interacts with ROS, including OH and H2O2. 10 Several studies found that NAC reduced the length of hospitalization, as well as the frequency of intubation and mechanical ventilation, in patients with phosphide poisoning, but there are few human studies demonstrating a significant mortality benefit.11,12
Acetyl-L-Carnitine, also known as “ALCAR,” is the most potent form of L-carnitine. ALCAR is considered relatively safe and free of serious side effects for most patients. 13 Human studies showed a rare survival after administration of ALCAR to a patient who developed severe myocarditis with shock, atrial fibrillation and global hypokinesia of both ventricles after being acutely intoxicated with AlP. Despite supportive measures, including vasopressor support, the patient’s blood pressure could not be restored. 14
Several studies on the proper method of gastric decontamination in acute AlP poisoning have produced conflicting results. However, it was hypothesized that AlP tablets could maintain their integrity in a lipid-rich environment with reduced PH3 liberation. 15 Consequently, with positive results, vegetable oils have recently been recommended for gastric decontamination following acute AlP poisoning. 16 Paraffin is a mineral oil used as a safe laxative lubricant for the treatment of chronic constipation in children. 17 In addition, it does not cause the common side effects of osmotic laxatives, such as abdominal cramps, flatulence, diarrhea, and electrolyte disturbances. 18
Surprisingly, the use of ALCAR and paraffin oil has not been adequately investigated. To introduce a more promising therapy for these patients, the authors of this study compared the mortality rate, length of intensive care unit or hospital stay, and various other measured parameters for patients with acute AlP poisoning. In addition, we investigated potential mechanisms of AlP toxicity, as well as clinical and laboratory findings that could be used to predict the clinical outcome of AlP-poisoned patients.
Patients and methods
Study design, setting, and ethical considerations
This study was a randomized, controlled; parallel-group clinical trial (RCT) that enrolled AlP-poisoned patients admitted to the Poison Control Center of Ain-Shams University hospitals (PCC-ASUH), Egypt, in 2020. The protocol was approved by the PCC-ASUH and the Institutional Review Board of the Faculty of Medicine at Ain-Shams University [IRB No. FMASU (R 66/2019)]. This study was registered with clinicaltrials.gov (NCT04509258). Patients or their legal guardians (if the patient was unable to participate in the consent process) provided informed consent, and data were collected using a standardized checklist and following the declaration of Helsinki. 19
Eligibility criteria
Inclusion criteria
The current study included all adult patients of both sex with a history of acute intoxication with AlP who presented to the PCC-ASUH within the first 6 h after acute exposure and agreed to participate in the trial between January 2020 and December 2020. Diagnosis was established on history of exposure to AlP tablets with reliable identification of the compound based on the container brought by the patient or the patient relatives, in addition to suggestive clinical manifestations following a single exposure to AlP tablets.
Exclusion criteria
1. Patients younger than 18 and older than 65years of age. 2. Pregnant and lactating females. 3. Patients who presented with a delay time greater than 6 h after AlP exposure. 4. Patients who received any treatment for the AlP intoxication before admission to PCC-ASUH. 5. Co-ingestion or exposure to other toxins. 6. Patients with a history of cardiac, renal, or hepatic diseases. 7. Patients with an ALCAR or paraffin oil hypersensitivity.
Methods
The sequentially numbered opaque sealed envelopes method was used to randomly assign patients to one of the three groups. 20 The allocation sequence was generated using a variable block size and permuted block randomization technique. In this study, a single-blind methodology was utilized. 21
Group I (NAC group)
Thirty-two patients received standard and routine treatment for acute AlP poisoning upon admission in accordance with PCC-ASUH guidelines as follows: • Care for the airway, the respiratory system, and the circulatory system. • In the event of hypotension (SBP <90 mmHg): A central venous line was inserted to administer intravenous fluids guided by central venous pressure measurement, steroids (hydrocortisone 100 mg qid), and vasopressors (norepinephrine) to maintain SBP >90 mmHg. • The metabolic acidosis was corrected by intravenous sodium bicarbonate (NaHCO3) administration. • Magnesium sulfate 1g as an initial dose, followed by 1g every 3 hours, then 1–1.5 g every 6 h for 24 h was administered. • Aspiration of gastric content through a nasogastric tube was used to perform gastric decontamination whenever possible, followed by administration of normal saline 0.9% mixed with NaHCO3 solution 8.4% (50 mL NaHCO3 added to each 500 mL saline) if the delay time was less than 2 h. Gastric lavage was repeated until the aspirated gastric contents were clear.
16
• NAC was administered in three doses of 300 mg/kg diluted in Dextrose 5% in water (D5W) over 21 h: 150 mg/kg IV, mixed in 200 mL of D5W and infused over 1 h, followed by 50 mg/kg IV, mixed in 500mL of D5W and infused over 4 h, and finally, 100 mg/kg IV, mixed in 1000 mL of D5W and infused over 16 h.
22
• Hemodialysis was considered in cases with severe persistent acidosis or pulmonary edema unresponsive to diuretics. • If acute lung injury or acute respiratory distress syndrome developed, mechanical ventilation was provided. • Antiemetics in the form of intravenous metoclopramide 10 mg qid or intravenous ondansetron 0.15 mg/kg tid.
Group II (NAC + Car group)
This group consisted of 32 patients. They received a continuous IV infusion of nine ampoules (9 gm) of L-carnitine added to 500 mL of 0.9% normal saline until improvement or death. 14 In addition to the acetyl L-carnitine doses, the patients also received the standard and routine treatment for acute AlP poisoning.
Group III (NAC + Par group)
Thirty-two patients participated in this group and received the standard treatment for AlP poisoning described above, with the exception that gastric lavage was performed by aspiration of gastric content through a nasogastric tube whenever possible, followed by the administration of 250 mL liquid paraffin oil to be left in the stomach or orally administered in patients who refused insertion of a nasogastric tube. 23
All patients included in the study were subjected to a comprehensive history, including age, gender, place of residence, special habits, and history of medical or psychiatric comorbidities. Intoxication data included the manner of poisoning (whether intentional or accidental), number of AlP tablets consumed, route of poison intake, prehospital management, and time between AlP exposure and arrival at the PCC-ASUH (delay time).
Upon admission, an immediate clinical examination was performed, including vital signs (pulse, blood pressure, respiratory rate, and temperature). The Glasgow Coma Scale (GCS) was used to evaluate the patient’s level of consciousness. Immediate electrocardiography (ECG) was performed upon admission and was repeated 12 h later. Before starting any treatment, arterial and venous blood samples were collected for blood gas analysis and assessment of the baseline routine laboratory investigations, including random blood sugar (RBS), serum electrolytes (Na and K), hepatic profile [alanine aminotransferase (ALT), aspartate transaminase (AST), and total bilirubin], complete blood count, blood urea nitrogen (BUN), and serum creatinine to determine the estimated glomerular filtration rate (eGFR).
24
Colorimetric kits (Bio-diagnostic; Giza, Egypt) were used to measure some oxidative stress biomarkers, including catalase (CAT) and superoxide dismutase (SOD) activities. MyBioSource enzyme-linked immunosorbent assay (ELISA) kit (Vancouver, British Columbia) was used to measure Complex IV (cytochrome c oxidase) activity and active caspase-3 level, while the Cusabio ELISA kit (Houston, USA) was used to estimate active caspase-9 level. After 12 h, another blood sample was taken to reevaluate the oxidative stress biomarkers.
Evaluation of the severity of acute intoxication
The new Simplified Acute Physiology Score II (SAPS II) was used to assess the severity of acute intoxication in patients within the first 24 h after admission, and the mean predicted mortality rate was compared across all studied groups. The SAPS II calculator generates a score between 0 and 163 in integer points with the involving of several parameters according to Le Gall and Lemeshow. 25
The Sequential Organ Failure Assessment (SOFA) score was another ICU scoring system used to assess a patient’s organ function or rate of failure. 26 All study participants were prospectively observed until hospital discharge or death.
Study outcomes
At the conclusion of this study, the primary outcome was in-hospital mortality; secondary outcomes included the need for intubation and mechanical ventilation, the need for vasopressors, the duration of ICU admission, and the total length of hospital stay.
Statistical analysis
Package of Statistics for Social Sciences SPSS Statistics for Windows, version 26 by IBM Corp. (Armonk, New York, United States) was used to tabulate and analyze the data. The three groups were compared using ANOVA tests while student T-test was used to compare two groups. The Kaplan-Meier method was used to analyze survival data, and the log-rank test was used to compare treatment groups. Subsequently, the Cox proportional hazard model was used to calculate the hazard ratios (HR) and 95% confidence intervals. Continuous variables that were found to be significantly related to mortality were converted to binary values, and the cut-point was determined using receiver operating curve (ROC) analysis to determine the area under the curve (AUC, c-statistic) and the highest predictive value. The data were presented as the mean and standard deviation and differences were considered significant at p < 0.05.
Results
In the present study, we sought to determine the effect of L-carnitine and liquid paraffin on the survival rate of acutely poisoned AlP patients. As depicted in the flowchart, a total of 110 patients were admitted to the PCC-ASUH, 14 patients were excluded, and the remaining 96 patients were divided into three groups of 32 patients each (Figure 1). A flow chart showing the steps of the current study, total number of patients,period of follow up and different assessed parameters.
Baseline demographic and toxicological characteristics of the three treatment groups on admission (n = 96).

ais statistically significant when p < 0.05.
Baseline vital signs, clinical data, and electrocardiographic findings for the three investigated groups (n = 96).

MAP: Mean arterial pressure, GIT symptoms: Gastrointestinal symptoms, ECG (1): Electrocardiography performed immediately after admission, ECG (2): Electrocardiography performed 12 h after the administration of each group’s dose.
* is statistically significant when p < 0.05.
Laboratory findings in the three treatment groups on admission (n = 96).
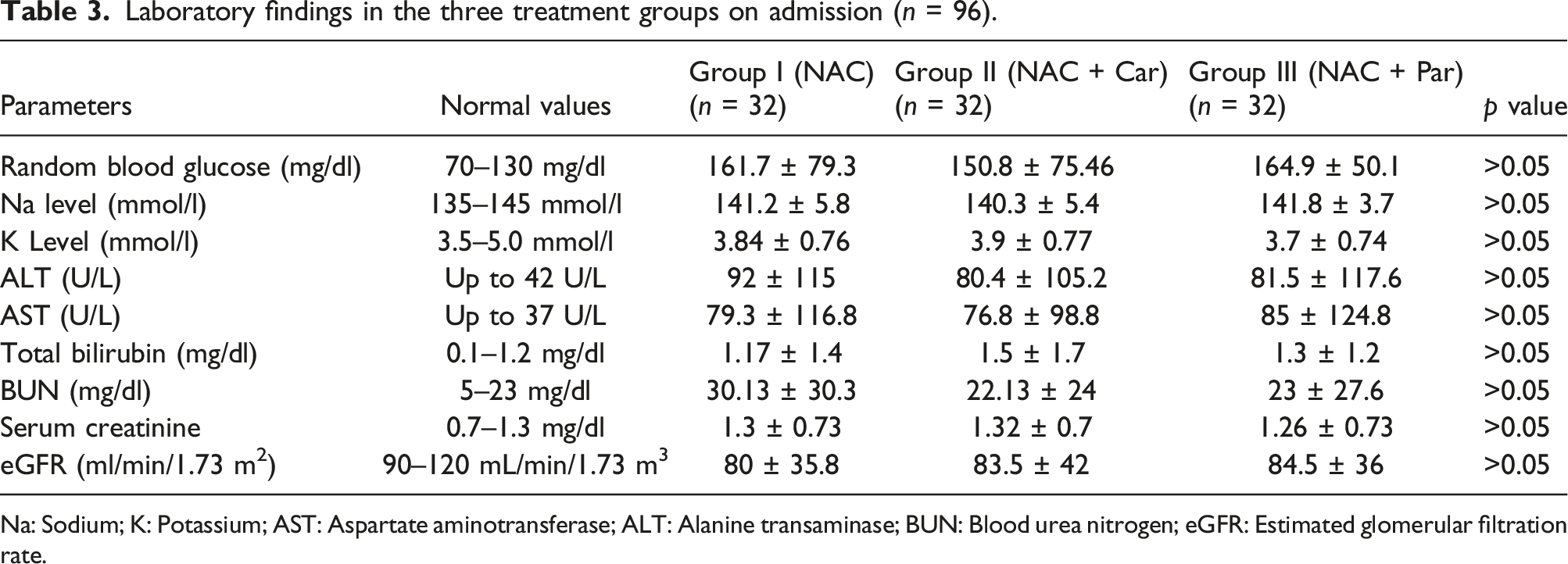
Na: Sodium; K: Potassium; AST: Aspartate aminotransferase; ALT: Alanine transaminase; BUN: Blood urea nitrogen; eGFR: Estimated glomerular filtration rate.
The arterial blood gas analysis parameters for the three investigated groups (n = 96).

PH: Potential of hydrogen; PCO2: Carbon dioxide partial pressure; SaO2: Oxygen saturation; HCO3: Bicarbonate; CL−: Chloride ion. The data were expressed as mean ± SD, and,
*is statistically significant when p < 0.05.
Evaluation of cytochrome c oxidase, caspase-3, caspase-9, catalase, and superoxide dismutase levels in the three treatment groups on admission and after 12 h of receiving the treatment (n = 96).

SOD: Superoxide dismutase.
*is statistically significant from the same group before treatment.
#is significantly different from the remaining groups when p < 0.05.
Caspase-3 levels were significantly lower in patients of the paraffin group after receiving treatment (2.5 ± 0.8) when compared to the same group before treatment (3.9 ± 1.3) or to its levels in patients of the supportive group after treatment (3.5 ± 1.36) (Table 5).
Patients treated with carnitine and paraffin had significantly lower levels of caspase-9 (8.2 ± 2.8, 8.9 ± 1.7) than the same groups before treatment (12.1 ± 2.8, 13.3 ± 3.6).
Moreover, both carnitine and paraffin-treated patients exhibited significantly higher levels of catalase (149.7 ± 21.47, 144.4 ± 19.73) than the same patients prior to treatment (103.3 ± 12, 102.9 ± 10.6) and significantly higher levels than the supportive group patients following treatment (106.1 ± 17.1). In addition, the superoxide dismutase levels of the carnitine- and paraffin-treated groups decreased significantly after treatment (34.58 ± 7.2, 31.8 ± 7.8) when compared to their admission levels (47.54 ± 9.6, 44.9 ± 7.7) and to their level in the supportive treatment group (42 ± 10.3) (Table 5).
Glasgow coma scale, SOFA score, SAPS II score, and mortality rate (percent) for the three tested groups of AlP-poisoned patients who received treatment (n = 96).

SOFA: Sequential organ failure assessment; SAPS: Simplified acute physiology score II.
Comparison of primary and secondary outcomes in the studied groups (n = 96).

ICU: Intensive care unit; MV: Mechanical ventilation. Data were expressed as mean ± SD.
* is statistically different when p < 0.05.
The effectiveness of each treatment was evaluated using Kaplan–Meier survival curves for the three groups of patients. The median survival time had not yet been reached in the carnitine- and paraffin-treated groups (NAC + Car and NAC + Par), whereas it was 30 h (interquartile range [IQR] 20.8–39.2 h) in the NAC-treated group (NAC) (Figure 2). Both intervention groups had a significantly higher mean survival time than the control (NAC) treated group, as shown in Table 8. The Kaplan–Meier survival curves according to the received treatment in the three groups. Mean survival time in the studied groups with acute AlP poisoning (n = 96).

A subsequent ROC analysis was constructed to analyze different evaluated scores and biomarkers to identify the optimal cut-point for regression analysis. GCS had an AUC of 0.96 ± 0.029 with an optimal cut-point of six points with a sensitivity of one and specificity of 0.9 to predict survival. Although the SOFA score had an AUC of 0.94 ± 0.024 with an optimal cut-point of 7 with a sensitivity of one and specificity of 0.889 to predict mortality, the SAPSII score had an AUC of 0.97 ± 0.015 with a cut-point of 52, a sensitivity of 0.81, and a specificity of 0.96 to predict mortality. Thus, these results indicated that the estimated scores are highly predictive in acutely AlP-poisoned patients (Figure 3(a) and (b)). The receiver operating curves (ROC) of different measured scores and biomarkers in predicting survival or mortality. (a); ROC in predicting survival for GCS, (b); ROC for SOFA and SAPS II in predicting mortality. (c); ROC for cytochrome oxidase-c (1) and catalase (CAT 1) in predicting survival. (d); ROC for caspase-3, caspase-9, superoxide dismutase in predicting mortality [Casp-3 (1), Casp 9 (1), SOD (1)]. Cytochrome oxidase c (1), CAT (1), Casp-3(1), Casp-9(1), and SOD (1) are parameters measured upon admission.
For the estimated biomarkers, the catalase enzyme had an AUC of 0.6, showing that it was a poor indicator. However, the cytochrome-c oxidase enzyme had an AUC of 0.82 with a cut-point of 69.5, sensitivity of 0.78, and a specificity of 0.76, indicating that it can be a good predictor for patients’ survival. Additionally, caspase-3 and caspase-9 were poor mortality indicators as they had an AUC of 0.64 and 0.61 respectively. As shown in Figure 3(c) and (d), superoxide dismutase was a fair indicator of mortality with an AUC of 0.78, a cut-point of 51.4, a sensitivity of 0.62, and a specificity of 0.92. For logistic regression analysis, no parameters with poor predictive ability, as demonstrated by AUC analysis, were selected.
Unadjusted cox proportional hazard models predicting survival time for patients according to each estimated biomarker (n = 96).
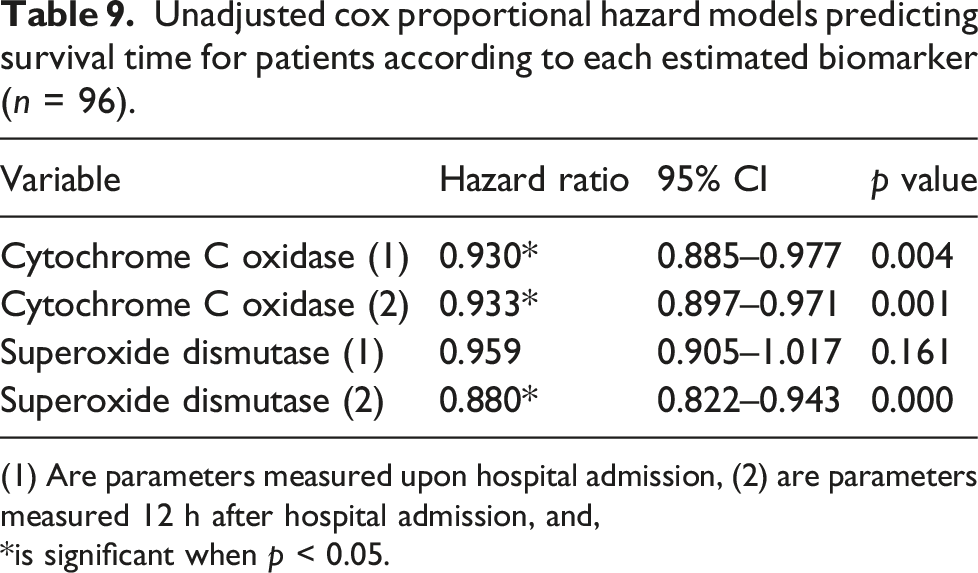
(1) Are parameters measured upon hospital admission, (2) are parameters measured 12 h after hospital admission, and,
*is significant when p < 0.05.
Discussion
AlP is a fumigant with an estimated 30%–100% mortality rate in cases of acute poisoning. 3 Cardiovascular toxicity, which can lead to cardiogenic shock and severe acidosis, is responsible for the majority of clinical symptoms. 27 According to previous reports, the lethal dose for a 70 kg subject is between 150 and 500 mg. 28
All acutely AlP-poisoned patients admitted to the ICU of the Poison Control Center of Ain-shams University Hospitals who met the inclusion criteria were included in the current study. Patients were randomly divided into three groups to determine the efficacy of adding any additional treatment to the poison center’s supportive care. The first group received only supportive treatment, the second group received supportive treatment plus acetyl L-carnitine, and the third group received supportive treatment plus medicated paraffin oil.
Our findings revealed that most patients were between the ages of 25 and 27 years old, there were no significant differences between the sexes within each group, and that the majority of patients did not have any special habits, such as smoking or drug abuse. All included patients ingested fewer than two tablets and arrived at the hospital between 2 and 5 h after ingestion. A negligible minority of patients were affected by psychological disorders. Most hospitalized patients presented with various GIT symptoms, including nausea, vomiting, and abdominal pain. These findings were consistent with previous reports which stated that most AlP-poisoned patients were between the ages of 20 and 30.29,30 In addition, Helal et al. 31 reported that patients with AlP poisoning experienced gastrointestinal symptoms.
Patients in the three investigated groups exhibited a low MAP and tachycardia that could be a reflex to hypotension or due to cardiotoxicity, a slightly low temperature, and an elevated respiratory rate. Hypotension and tachycardia have been documented in several prior studies.32,33
Our current findings importantly revealed that the three groups showed elevated random blood glucose levels that was reported as a cause of several organ damages and consequently lead to death in previous studies which considered hyperglycemia as a mortality predictor.33,34
Several studies advocated the estimation of arterial blood gas analysis for AlP-poisoned patients, as the majority of admitted patients had metabolic acidosis in addition to cardiomyopathy.23,31 Patients in all three groups exhibited metabolic acidosis and low oxygen saturation, as reported by multiple studies. The reported metabolic acidosis may have resulted from the accumulation of lactic acid due to the inhibition of oxidative phosphorylation, as previously reported. 35 This was consistent with our study, as the addition of carnitine or paraffin was accompanied by the higher reported levels of cytochrome-c, catalase, and lower levels of SOD in both groups, indicating a better prognosis. Moreover, the group that received supportive treatment had the lowest levels of PH and HCO3, the highest lactate level and anion gap, as previously reported, indicating the worst prognosis.
The present study revealed that patients receiving only supportive care had the lowest GCS scores, indicating severe brain damage. Low GCS was also observed in previous studies, and it was attributed to the downregulation of cytochrome-c oxidase enzyme and oxidative phosphorylation by phosphine gas, causing multi-organ damage.31,36 This strongly supported our findings as the lowest GCS was accompanied by the highest SOFA score. Sharif and colleagues 37 reported that a SOFA score above 4.5 was a significant predictor of unfavorable outcomes.
The hospital and ICU stay for patients in the current study ranged from one to 2 days, the same as in the study by Abd Elghany et al. 29 AlP poisoning may result in ECG changes and arrhythmia. In addition, patients may require the administration of vasopressors or mechanical ventilation support. The current reports demonstrated that the addition of paraffin to the standard supportive treatment reduces the need for mechanical ventilation. Moreover, the administration of L-carnitine or paraffin reduced the ECG changes for the patients significantly although there were no significant differences in the need for vasopressors among the three groups.
In the present investigation, the group receiving supportive care had the highest in-hospital mortality rate (62.5%). In nearly every AlP poisoning study, high mortality rates were observed. 9,32,33 Importantly, the addition of carnitine decreased the mortality rate to 37.5%, while the paraffin-treated group had the lowest mortality rate at 31.25%. The promising results of the paraffin-treated group may be attributable to the significantly increased levels of cytochrome-c and catalase with decreased caspase-3, caspase-9, and SOD levels after 12 h of admission. 38
The Kaplan–Meier curve of the current results revealed that 50% survival had not been reached in the supportive group, indicating the highest mortality rate among the investigated group, whereas the mean survival time in hour for the carnitine- and paraffin-treated groups were nearly identical, indicating a higher survival rate among these groups. Several studies supported these findings by demonstrating the beneficial effect of using different types of oils in gastric decontamination.39,40 In addition, Darwish et al. 23 reported that adding paraffin oil to the treatment protocol of acute AlP poisoning improved patients’ outcomes.
After testing different used scores and parameters as a good survival protector, the current study highlighted that the GCS, SOFA, and SAPS II are strong survival predictors in acutely AlP-poisoned patients, which is strongly supported by previous studies. However, our study revealed that certain biomarkers, such as cytochrome-c oxidase with a cutoff point of 69.5 and superoxide dismutase with a cutoff point of 51.4, can also predict the survival rate of poisoned patients. To our knowledge, this was the first time that these biomarkers were evaluated as survival predictors for AlP-poisoned patients. As previously reported, cytochrome-c oxidase is a strong marker for mitochondrial damage and cell death in kidney injury and liver injury.41,42 In addition, SOD was considered a novel biomarker for measuring the severity of renal diseases. 43 He et al. revealed that serum levels of SOD can be a potential predictor of the prognosis of hepatitis E virus-induced liver failure. 44 The significance of the HR of serum levels of cytochrome-c oxidase upon admission, after 12 h, and superoxide dismutase only after 12 h of admission was also confirmed by a Cox proportional hazard model.
Conclusion
The addition of L-carnitine or medicated paraffin oil to the standard supportive treatment for acutely AlP-poisoned patients is beneficial, although the medicated paraffin has a more potent effect than L-carnitine. Both are expected to improve the prognosis of acutely AlP-poisoned patients through modulating the antioxidant and anti-apoptotic pathways; however, medicated paraffin has a superior action through elevating cytochrome-c oxidase levels and inhibiting caspase-3 levels which has not been achieved by adding L-Carnitine.
In acutely AlP-poisoned patients, severity scoring systems such as GCS, SOFA, and SAPS II had the potential to be a useful tool for predicting mortality. Furthermore, combining these scores with other biomarkers, such as cytochrome-c oxidase and superoxide dismutase, may aid in stratifying the severity and mortality risk associated with AlP poisoning. Therefore, the inclusion of these tools into decision-making strategies could facilitate the early detection of patients’ prognoses and enhance clinical outcomes.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.

