Abstract
Except for clinical value, borneol is routinely used in food and cosmetics with seldom safety evaluation. To investigate its hepatoxicity, we exposed 3 dpf (days post fertilization) larval zebrafish to borneol at a gradient of concentrations (200–500 μM) for 3 days. Herein, our results revealed that high doses of borneol (300–500 μM) caused liver size decrease or lateral lobe absence. Borneol also seriously disturbed the hepatic protein metabolism presented with the increased activity of alanine aminotransferase (ALT) and lipid metabolism shown with the increased level of triglycerides (TG) and total cholesterol (TC). The lipid accumulation (oil red staining) was detected as well. Additionally, significant upregulation of genes was detected that related to oxidative stress, lipid anabolism, endoplasmic reticulum stress (ERS), and autophagy. Conversely, the lipid metabolism-related genes were markedly downregulated. Moreover, the changes in the superoxide dismutase activity and the level of glutathione and malondialdehyde raised the likelihood of lipid peroxidation. The outcomes indicated the involvement of oxidative stress, ERS, lipid metabolism, and autophagy in borneol-induced lipid metabolic disorder and hepatic injury. This study will provide a more comprehensive understanding of borneol hepatoxicity and the theoretical basis for the safe use of this compound.
Key contribution
Borneol was firstly evaluated for hepatoxicity using 3 dpf (days post fertilization) larval zebrafish. The outcomes showed hepatic injury and metabolism disorders (protein and lipid), involving oxidative stress, ERS, lipid metabolism, and autophagy pathways.
Introduction
Borneol, a traditional Chinese medicine, is first recorded in the Chinese herbal medicine book Ming Yi Bie Lu and has lots of functions, including heat-clearing, pain relieving, eye-brightening, etc. 1 In modern medical research, borneol has been found to be active in antiinflammation, sedative, and analgesia, in addition to cardiovascular protection and improvement of bioavailability of other medicines.2–4 It has been extensively used in multiple clinical departments, including internal medicine, surgery, gynecology, pediatrics, otolaryngology, etc., especially in the treatment of cardiovascular and cerebrovascular diseases, such as stroke and coronary heart disease.2,5 It is widely used as a vital component in traditional Chinese medicine compounds, and more than 170 have been declared in the Chinese Pharmacopoeia 2020 edition. 6
Otherwise, due to its fragrant property, borneol is commonly added to cosmetics, shampoo, toiletries, and household detergents. And due to its refreshing, anthelmintic, and antiseptic functioning, it is also added to refreshing essential oil and insect repellent or used as a food additive.2,7 The global annual consumption of borneol has been up to 1–10 metric tons. 7 However, the toxicity of this compound was seldomly reported, albeit with a relatively comprehensive illustration of its pharmacological and clinical functions. Therefore, it is urgent to investigate its toxicity and the mechanism of it.8,9
The liver is the largest organ in animals and plays vital roles in both metabolisms (e.g., fat, lipid, drugs, etc.) and detoxification.10,11 Accumulating evidence proved that the metabolic enzymes in the liver are involved in the biotransformation of drugs and toxicants via oxidation, reduction, and hydrolysis.12,13 Therefore, the liver is susceptible to a wide range of diseases. 14 and it is very necessary to perform the assessment of the hepatoxicity of borneol.
Zebrafish (Danio rerio) is a low vertebrate animal from north India and is considered a popular model organism for toxicity evaluation due to its characteristics of embryos, including small size, fast development, transparency, high productivity, etc. 15 Physiologically, in addition to hepatocytes, the zebrafish liver encompasses the same primary cell types (e.g., Kupffer cells, stellate cells, biliary cells, etc.) 16 performing similar functions as their mammalian counterparts 17 and responding with the same mechanism to drugs or toxicants. 18 Otherwise, tissue/organ-specific transgenic zebrafish lines, such as tg(l-fabp:EGFP), make the examination and detection of hepatoxicity more convenient and intuitive. 19
Herein, we aimed to evaluate the hepatoxicity of borneol and investigate the mechanism by analyzing phenotypical changes, lipid metabolism, histological changes, and expression of related genes. Our results will pave the road and provide a theoretical basis for the safe use of borneol routinely and clinically.
Materials and methods
Chemicals
Borneol (D-Borneol,  , 420247, HPLC ≥ 97%), and tricaine (A5040) were purchased from Sigma-Aldrich. Borneol was dissolved in dimethyl sulfoxide (DMSO spectroscopy grade, D8370, Solarbio, Beijing, China) at a concentration of 600 mM and stored at −20°C. E3 medium (5 mM NaCl, 0.17 mM KCl, 0.4 mM CaCl2, and 0.16 mM MgSO4) was used for borneol dilution.
, 420247, HPLC ≥ 97%), and tricaine (A5040) were purchased from Sigma-Aldrich. Borneol was dissolved in dimethyl sulfoxide (DMSO spectroscopy grade, D8370, Solarbio, Beijing, China) at a concentration of 600 mM and stored at −20°C. E3 medium (5 mM NaCl, 0.17 mM KCl, 0.4 mM CaCl2, and 0.16 mM MgSO4) was used for borneol dilution.
Zebrafish husbandry
Animals used in this study included wide-type AB and tg(l-fabp:EGFP) zebrafish. All of them were provided by Key Laboratory for Drug Screening in Biology Institute, Qilu University of Technology (Shandong Academy of Sciences). Embryos were achieved through natural spawning. Embryos were kept in E3 medium containing 0.1-ppm methylene blue for disinfection. The embryos were kept in a light 14-h/dark 10-h incubator at 28°C before and during the treatment.
All experiments were conducted according to the guidelines of the ethical standards of the committee of the Biology Institute, Qilu University of Technology (Shandong Academy of Sciences) (approval No. SW20210222).
Borneol treatment
The borneol administration was applied in 6-well plates with 30 fish and 5 mL medium per well. Fish were treated with ingredient concentrations of borneol (400, 450, 500, 550, 600, 650, and 700 μM)/vehicle (VHC)/DMSO (0.1%, v/v) for 72 h with daily medium change.
Morphological phenotyping and lethality
At 24 hpe (hours post-exposure), Borneol-/VHC-exposed zebrafish were anesthetized in tricaine 20 and positioned in 0.1% regular agarose G-10 (111,860, Biowest, Shanghai, China) in Milli-Q water for examination using a stereomicroscope (Olympus SZX16, Tokyo, Japan) with cellSens Standard software 2.2. Dead fish were recorded (defining with heart beating) and LC10 and LC50 were calculated by using a dose-mortality curve.
Four borneol concentrations (200, 300, 400, and 500 μM) below LC10 were used for zebrafish intoxication from 3 dpf. And the morphology of embryos (including pericardium, swimming bladder, body shape, and yolk absorption) was examined every 24 h between 24 and 72 hpe using an Olympus SZX16 microscope as described above.
Evaluation of hepatotoxicity
Tg(l-fabp:EGFP) transgenic fish expressing the green fluorescent protein in the liver under liver fatty acid-binding protein ((l-fabp) promotor. 19 From 3 dpf onwards, 30 tg(l-fabp:EGFP) fish were administrated by 200–500 μM borneol in 6-well plates, and the morphology of the liver was examined using a fluorescence microscopy (Olympus SZX16). The liver area and fluorescence intensity were measured by Image pro-plus 5.1.
Alternatively, 3D liver morphology of borneol/DMSO/VHC-treated fish was acquired using ImageXpress Micro Confocal (Molecular Devices, San Jose, CA), and liver volume was analyzed by MetaXpress software 6.5.2351. The photography was performed at 72 hpe and embryos were laterally positioned and maintained in a 96-well plate (1 fish per well) in 0.1% regular agarose G-10 after anesthetizing.
Oil red O staining
20 embryos in each group were fixed in paraformaldehyde (PFA, in PBS) at 4°C for 24 h. Following washing using phosphate-buffered saline (PBS, B1139-066, BIOEXPLORER®, Colorado, USA) twice, the embryos were dehydrated in 25%, 50%, 75%, and 100% propanediol (in PBS) sequentially. The dehydrated embryos were stained in 1% oil red O solution (DH222-1, Ding Guo Chang Sheng, Beijing, China) for 4 h. The hydration was formed in propanediol at the opposite concentration order. Before re-fixation using PFA, the embryos were washed three times. The staining was examined using a stereomicroscope SZX16.
Histological analysis of zebrafish liver
Secondary to fixation in PFA, 10 embryos in each group were embedded using paraffin, sectioned, and stained with Hematoxylin and Eosin Staining (H&E, G1003, Servicebio, Wuhan, China). The examination of liver histological change was formed using an Olympus FSX100 inverted microscope equipped with FSX-BSW imaging software (Olympus FSX100, Tokyo, Japan).
Activity measurement of liver-related enzymes
At 72 hpe, 35 zebrafish in each group were collected using 1.5 mL Eppendorf tubes and washed three times with PBS before draining and weighing. The homogenization of samples was applied using Bullet BlenderTM 24 (Next Advance, New York, USA) in NaCl 0.9% with a ratio of 1:9 (zebrafish: NaCl 0.9%, w/v). Total protein was quantified using BCA Protein Assay Kit (P0012, Beyotime, Shanghai, China). The activity of alanine aminotransferase (ALT, C009-2-1, Nanjing Jiancheng, Nanjing, China), aspartate aminotransferase (AST, C010-2-2, Nanjing Jiancheng, Nanjing, China), glutathione (GSH, BC1175, Solarbio, Beijing, China), and superoxide dismutase (SOD, BC0170, Solarbio, Beijing, China) were performed according to the manufacturer’s protocols. Meanwhile, the quantification of malondialdehyde (MDA, BC0020, Solarbio, Beijing, China), triglycerides (TG, A110-1-1, Nanjing Jiancheng, Nanjing, China), and total cholesterol (TC, A111-1-1, Nanjing Jiancheng, Nanjing, China) were conducted according to the manufacturer’s protocols as well.
Qualification of gene expression
At 72 hpe, 25 embryos were collected for RNA extraction using Fastpure tissue Total RNA Isolation Kit (RC112-01, Vazyme, Nanjing, China). 1 μg RNA/sample was reverse transcribed with SPAPKscriptⅡRT Plus Kit (Sparkjade, Shangdong, China). qPCR was performed with AceQ® GPCR SYBR Green Master Mix (Q711-03, Vazyme, Nanjing, China) and the housekeeping gene was β-actin. Primers for all genes are listed in Supplementary Table S1.
Statistical analysis
The statistical analysis of all the data was performed with One-way ANOVA followed by Sidak’s multiple comparisons test using Graphpad prism 7.0. Results are presented as mean ± SEM of three independent experiments performed in triplicate.
Results
Borneol caused death and phenotypical changes in zebrafish
As the results showed, the death of zebrafish embryos was observed in borneol solution, excluding vehicle control (VHC), DMSO, and 400-μM groups. And the death rates were 4.53%, 7.86%, 61.2%, 68.83%, and 100% in 450-, 500-, 550-, 600-, 650-, and 700-μM groups separately (Figure 1(a)). The “Concentration - Death rate” curve was calculated using Graphpad prism 7.0 (Figure 1(b)), LC10 and LC50 were 500 μM and 556.92 μM separately. Therefore, zebrafish were treated with 200, 300, 400, and 500 μM borneol for further experiments. Compared to VHC, zebrafish exhibited no phenotypical changes at the concentrations of 200–500 μM at 24 hpe. While lack of swimming bladder was observed in 300-, 400, 500-μM groups at 48 hpe and curved bodies in 500-μM group at 72 hpe (Figure 1(c)). Mortality and phenotypes of embryos treated with borneol. Embryos were administrated with 200–500 μM borneol (or VHC/DMSO) from 72 hpf for 3 days. (a) The cumulative mortality of zebrafish at 24, 48 and 72 hpe. (b) The “Concentration - Death rate” curve of zebrafish. (c) The morphology of larval zebrafish at 24, 48 and 72 hpe. Swimming bladder loss was donated with red asterisk; spinal curvature was donated with black arrowhead, n ≥ 30. *p < 0.05, **p < 0.01 vs the VHC group. a: The data were obtained from three independent experiments.
Borneol caused liver injury
A transgenic fish line tg(l-fabp:EGFP) was used to investigate the effects of borneol on the liver of larval zebrafish. Borneol exhibited hepatotoxicity with respect to the decrease of liver size in all groups, excluding the 200-μM group (Figure 2(a) and (b)), albeit with no variation in fluorescence intensity (Figure 2(c)). Effects of borneol on hepatic morphology of zebrafish. Embryos were administrated with 200–500 μM borneol (or VHC/DMSO) from 72 hpf for 3 days. Borneol resulted in the loss of lateral lobes. (a) 2-dimensional whole-mount images of liver. (b) Fluorescence area and (c) fluorescent intensity of liver measured in (a). (d) 3-dimensional whole-mount images of liver. (e) Liver volume measured in (d). Lateral lobes are donated with yellow triangles; Ventral lobes are indicated with red arrowheads. *p < 0.05, **p < 0.01 vs the VHC group.
Meanwhile, the hepatotoxicity of borneol was detected in three-dimensional images of the liver (Figure 2(d)). Zebrafish liver is composed of ventral and lateral lobes. 16 VHC/DMSO-treated fish exhibited well-organized and flat-shaped ventral and lateral lobes. Conversely, borneol-administrated ones exhibited dose-independent length reduction of ventral lobes and lateral lobes loss at high concentrations, especially 400 and 500 μM. Significantly, the volume of the liver was reduced by borneol-elicitation in 300-, 400- and 500-μM groups (Figure 2(e)).
Borneol disturbed lipid metabolism
As shown in Figure 3(a) and (b), borneol caused a dose-dependent yolk absorption delay, especially at high concentrations (400 and 500 μM). The effects of borneol on lipid metabolism of zebrafish. Embryos were administrated with 200–500 μM borneol (or VHC/DMSO) from 72 hpf for 3 days. (a) Yolk sac (outlined with red dotted lines) absorption analysis. (b) Qualification of yolk sac area in (a). (c) Lipid accumulation analysis in the liver (oil red O staining), outlined with white dotted lines. (d) Quantification of lipid accumulation in (c), n ≥ 20. *p < 0.05, **p < 0.01 vs the VHC group.
We also performed Oil red O staining to investigate the effects of borneol on lipid deposition in larval zebrafish. Compared to VHC/DMSO-treated fish, borneol-treated ones presented a significant increase of darkness in the liver region (Figure 3(c) and (d)) which indicated the possible hepatic steatosis caused by lipid accumulation.
Borneol caused the pathological change in liver
To confirm these results, we histologically examined the damage in the liver elicited by borneol at 72 hpe. Sagittal sections through the liver tissue of VHC-treated animals showed clear texture, and cells with clear cytoplasm and nuclei were uniformly and compactly arranged. Conversely, borneol-treated larvae presented less-stained cytoplasm and extracellular matrix, lipid droplet accumulation, blood vessel expansion, and blood cell retention, especially in 400- and 500-μM treated ones (Figure 4). Effect of borneol on hepatic histo-architecture (H&E staining). Embryos were administrated with 200–500 μM borneol (or VHC/DMSO) from 72 hpf for 3 days. Borneol caused liver injury shown with blood vessel dilation (donated with red triangles) and lipid droplets (donated with yellow triangles), (n ≥ 10).
Borneol caused the abnormal level of ALT, AST, oxidoreductase, and lipid metabolism-related enzymes
At 72 hpe, we also assessed the borneol-elicited hepatic injury by measuring the level of alanine aminotransferase (ALT), aspartate aminotransferase (AST), superoxide dismutase (SOD), glutathione (GSH), malondialdehyde (MDA), triglycerides (TG) and cholesterol (TC) with homogenate of whole larvae. The activity of aminotransferase (e.g., ALT and AST) and the level of metabolites (e.g., TG, and TC) are both well-determined hallmarks of liver injury.21,22 ALT level was significantly increased by borneol albeit with a normal level of AST (Figure 5(a) and (b)). Significantly, the level of TG and TC, both well-identified central indicators of hepatic lipid metabolism,21,23 were raised after borneol intoxication (Figure 5(c) and (d)). Additionally, hallmarks of lipid peroxidization, the level of SOD exhibited a dose-dependent decline (Figure 5(e)) while GSH level showed a slight increase in lower-dose groups (e.g., 200–400 μM) and a marked decrease in high-dose ones (e.g., 500 μM) (Figure 5(f)). Intriguingly, the level of MDA was significantly alleviated by borneol induction (Figure 5(g)). Analysis of metabolic hallmarks at 72 hpe. Embryos were administrated with 200–500 μM borneol (or VHC/DMSO) from 72 hpf for 3 days. (a) The activity measurement of alanine aminotransferase (ALT) and (b) aspartate aminotransferase (AST). Qualification of the level of triglycerides (TG) (c) and total cholesterol (TC) (d). The activity measurement of superoxide dismutase (SOD) (e) and glutathione (GSH) (f), and level of malondialdehyde (MDA) (g). *p < 0.05, **p < 0.01 vs the VHC group.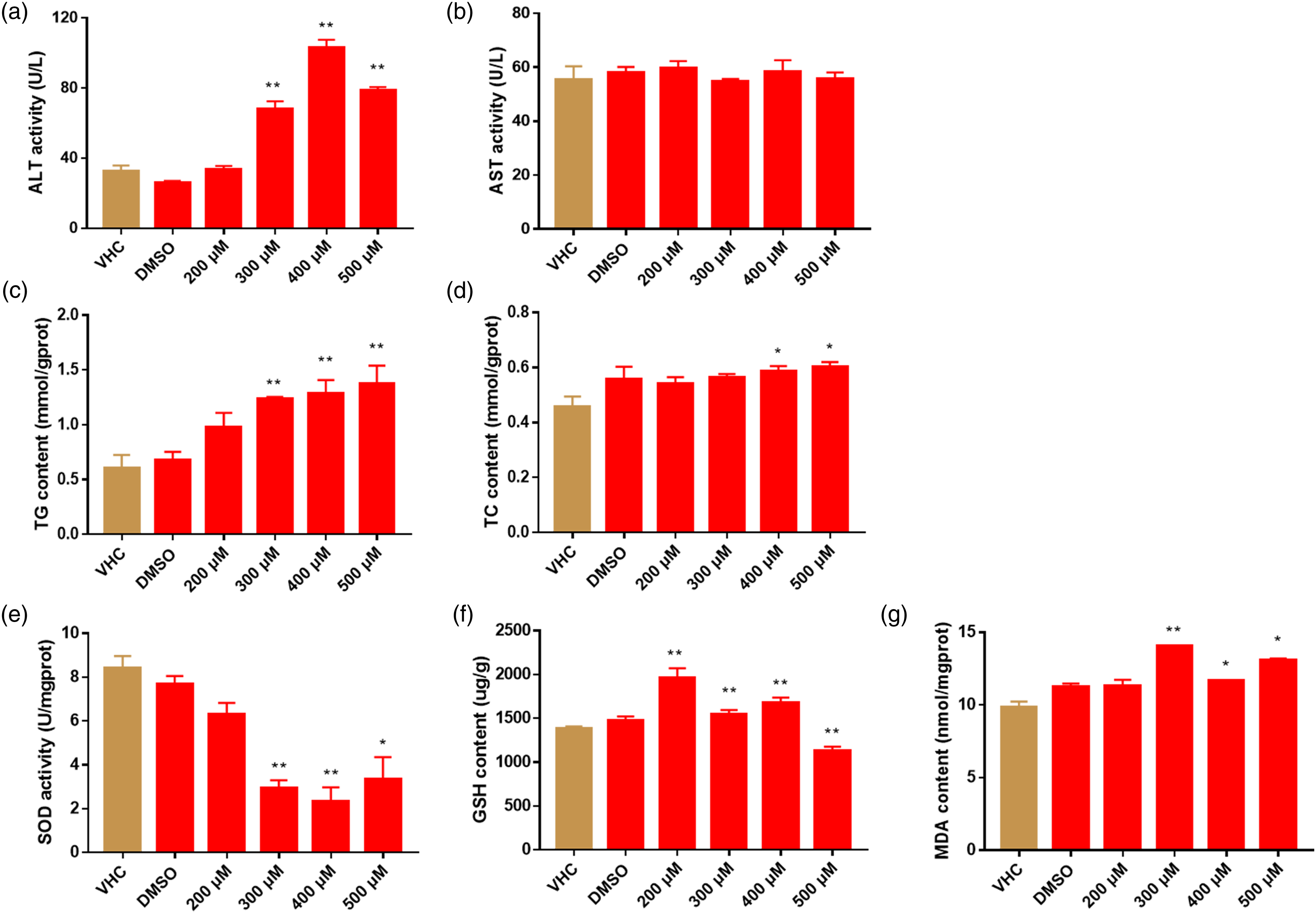
Borneol caused abnormal gene expression
We also performed qPCR to compare the gene expression level, in borneol and VHC/DMSO treated animals, of common molecular biomarkers of oxidative stress, lipid anabolism, endoplasmic reticulum stress (ERS), and autophagy (Figure 6). Effects of borneol on gene expression at 72 hpe. (a) Expression of metabolism-related genes. (b) Expression of oxidative stress-related genes. (c) Expression of endoplasmic reticulum stress related genes. (d) Expression of lipid metabolism-related genes. (e) Expression of autophagy-related genes. Gene expression level was quantified relative to reference gene β-actin expression by ∆∆Cq method. Results for each gene are normalized to VHC administrated fish and are shown with mean ± SEM of three independent experiments performed in triplicate. Statistical analysis was performed using one-way ANOVA followed by Sidak’s multiple comparisons test. *p < 0.05, **p < 0.01 vs the VHC group.
Known as hallmarks of hepatotoxicity,24,25 compared with the VHC/DMSO group, si:ch211-127i16.2 were markedly downregulated while cyp2e1, man2b2, and g6pca.1 were significantly upregulated (Figure 6(a)). Alternatively, borneol significantly upregulated the expressions of oxidative stress-related factors (e.g., cat, nrf2, nqo-1, ho-1, gstp2, and gsto1), lipid anabolism (e.g., srebf2 and fasn), ERS-related factors (e.g., chop, hspa5, perk, atf6, ern1, bip, and atf4a) and autophagy-related genes (e.g., beclin, lc3, atg3, and atg5). Nevertheless, the expression of other oxidative stress-related factors (e.g., cat, nrf2, nqo-1, ho-1, gstp2, and gsto1) and lipid anabolism (e.g., srebf2 and fasn) were significantly decreased.
Discussion
Borneol has been used for more than one thousand years in Chinese medicine, as a preservatives, adsorbent, and perfume in food or cosmetics.1,7 However, its toxicity was rarely elaborated. Therefore, we systematically illustrated the possibility of hepatoxicity of this compound and explored its mechanism that would probably provide guidelines for its routine and clinical application.
Zebrafish is highly conservative to humans on the level of anatomy, physiology, and molecular26,27 and used for the evaluation of drug‐induced liver injury. 21 From 72 hpf onward, the hepatic organogenesis is completed, and the liver is perfused with blood and functional. 28 Thus, we initiate the treatment at 72 hpf. Borneol exposure caused a decrease in hepatic size, abnormal lipid metabolism, histologic alterations, increased levels of ALT, TG, TC, and MDA, and decreased GSH and SOD. These results implied the prominent hepatotoxicity of borneol.
It has been reported that si:ch211-127i16.2, man2b2, and g6pca.1 were all involved in various biological processes of metabolism and associated with hepatoxicity.24,25 Similar to cyp2e1, si:ch211-127i16.2 encodes a drug-metabolizing enzyme, a member of the P450 family. G-6-pase (glucose-6-phosphatase) and Man2b2 (mannosidase, alpha, class 2B, member 2) were encoded by g6pca.1 and man2b2 genes respectively, play an important role in the glucose metabolism pathway. 29 In this study, the expression of cyp2e1, man2b2, and g6pca.1 was significantly increased in the borneol-administrated groups suggesting the hepatic injury, albeit with the opposite response of si:ch211-127i16.2 gene.
Since oxidative stress is closely involved in liver injury, 30 we investigated the effects of borneol on related enzyme activity and gene expression using zebrafish. GSH is a non-enzymatic antioxidant that plays an important role in the cellular antioxidant response and the maintenance of redox balance. 31 MDA is the final product of lipid peroxidation, and elevated MDA levels reflect the increase of lipid peroxidation in the body.32,33 Thus, the excessive accumulation of GSH and MDA is considered as a sign of oxidative stress.34,35 Meanwhile, SOD and CAT are the first-line defense against oxidant-caused damage and play an important role in alleviating injury from oxygen-free radicals. 36 In our research, borneol inhibited SOD activity and resulted in the deposition of MDA albeit with the decrease in GSH. This indicated the borneol-induced oxidative stress, especially at high concentrations. Intriguingly, borneol only upregulated the expression of sod in lower-concentration groups which was probably due to the overwhelming oxidation in higher-concentration ones. As an antioxidant regulator, 37 nrf2 plays a vital role in drug-induced oxidative stress 38 and normally remains as heterodimers by binding to keap1. The oxidative stress can lead to the nuclear translocation of nrf2 and activates the expression of downstream antioxidant genes to exert antioxidant effects, including nqo-1 and ho-1. 39 Jia 31 has proved that the expression of nrf2, nqo-1, and ho-1 in zebrafish are significantly increased due to isoniazid-induced oxidative stress. Consistently, our data indicated the borneol-induced oxidative stress with respect of the upregulation of nrf2, nqo-1, and ho-1 and decreased keap1 level. Moreover, glutathione-S-transferase (GST), mainly produced in the liver, has dual functions of detoxification and antioxidant. 40 Gstp2 and gsto1 are members of the GST family, 41 in this study, we found borneol increased the expression of gstp2 and gsto1 which demonstrated the oxidative stress in the process of hepatic injury as well.
Accumulating evidence demonstrates that endoplasmic reticulum stress (ERS) is strongly related to oxidative stress. 42 Therefore, we examined the expression of ERS-related genes. As a hallmark of ERS, 43 the expression of chop was highly upregulated. Hspa5, the homologue of grp78 in zebrafish, is also accepted as a central regulator of ER homeostasis and apoptosis. 44 Under the stimulation, it disintegrates the binding with perk, atf6, and ire1 and alleviates their expression. 45 Additionally, bip is the first identified protein in the molecular chaperone and its activation can reduce the production and accumulation of unfolded or misfolded proteins. 46 The RT-PCR result showed that ERS was involved in borneol-induced hepatic injury with respect of upregulated ERS-related genes, such as chop, hspa5, perk, atf6, ern1, bip, and atf4a.
Since the liver is the main organ for lipid metabolism, hepatic steatosis is considered an indicator of abnormal lipid metabolism. 47 Our results further demonstrated the borneol-induced steatosis shown as decreased yolk area, lipid accumulation, vacuolated hepatocytes, and increased levels of TG and TC. Meanwhile, we examed the expression of related genes. Sterol regulatory element binding factor 1 (SREBF1) and sterol regulatory element binding factor 2 (SREBF2) are central players which participate in lipid metabolism and cholesterol synthesis respectively.48,49 Additionally, fatty acid synthase (FASN) is a master enzyme for fatty acid biosynthesis and fat formation, and its expression can be regulated by its upstream SREBF1. A high expression level of FASN indicates excessive lipid accumulation. 50 Consistently, as the regulators of lipid synthesis and metabolism, PPAR-γ is associated with lipogenic enzyme expression, while PPAR-α is involved in the activity of enzymes for fatty acid β-oxidation, such as carnitine palmitoyltransferase gene 1(CPT-1), peroxisome fatty acyl-COA oxidase 1(ACOX-1)), etc.51,52 This manuscript exhibited a significantly upregulated expression of srebf2 and fasn and downregulated ppar-γ, ppar-α, cpt-1, mgst, and acox1, which implied the increased hepatic lipogenesis, decreased lipolysis, and steatosis.
Autophagy can be activated by liver injury to prevent excessive inflammatory response and hepatocyte apoptosis and promotes liver homeostasis.53,54 Conversely, autophagy also evokes hepatocyte death through beclin1, an upstream factor, 55 and some downstream factors (e.g., atg3, atg5, and atg7) that are crucial for autophagic membrane elongation. 56 In autophagy, the microtubule-associated protein lc3 is accepted as a hallmark of autophagy tonoplast. The overexpression of lc3 indicates excessive autophagy which caused the formation of autophagosomes or inhibition of downstream degradation. 57 In this manuscript, we found borneol significantly upregulated the expression of beclin1, lc3, atg3, and atg5 and resulted in autophagy in zebrafish.
Collectively, our results showed borneol caused the liver injury of zebrafish. The mechanism was speculated that borneol induced hepatotoxicity through oxidative stress and endoplasmic reticulum stress followed by lipid metabolism disorder and autophagy dysfunction. This manuscript probably provided the theoretical basis for the safe and rational use of borneol.
Supplemental Material
Supplemental Material - A mechanistic investigation about hepatoxic effects of borneol using zebrafish
Supplemental Material for A mechanistic investigation about hepatoxic effects of borneol using zebrafish by L Liu, Y Yang, F Yang, Y Lin, K Liu, X Wang and Y Zhang in Human & Experimental Toxicology.
Footnotes
Author contributions
Conceptualization, Formal analysis, Methodology, Validation, Funding acquisition: L.L.; Investigation, Validation, Software, Formal analysis, Writing-original draft: Y.Y.; Validation: F.Y.; Formal analysis, Methodology: Y. L.; Project administration, Funding acquisition: K.L.; Formal analysis, Methodology, Validation, Visualization, Writing-original draft, Writing-review & editing, Funding acquisition: X.W.; Conceptualization, Methodology, Formal analysis, Writing-review & editing, Visualization, Project administration, Funding acquisition: Y.Z.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Shandong Provincial Natural Science Foundation (ZR2020YQ60), the Taishan Scholars Program, Jinan Talent Project for University (2021GXRC047), the Science and Technology Project of Changzhou City (CE20215028), Funding of the Key Project at Central Government Level (YDZX2021023), Science, Education and Industry Integration Innovation Pilot Project of Qilu University of Technology (Shandong Academy of Sciences) (2022PYI016).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
