Abstract
This study was aimed to evaluate the effect of Cymbopogon citratus against carbon tetrachloride (CCl4)-mediated hepatic oxidative damage in rats. Rats were administrated with C. citratus extract (100, 200 and 300 mg/kg b.w.) for 14 days before the challenge of CCl4 (1.2 ml/kg b.w. p.o) on 13th and 14th days. Hepatic damage was evaluated by employing serum biochemical parameters (alanine aminotransferase-ALT, aspartate aminotransferase-AST and lactate dehydrogenase-LDH), malondialdehye (MDA) level, reduced GSH and antioxidant enzymes (catalase: CAT, glutathione peroxidase: GPX, quinone reductase: QR, glutathione S-transferase: GST, glutathione reductase: GR, glucose-6-phosphate dehyrogenase: G6PD). In addition, CCl4-mediated hepatic damage was further evaluated by histopathological examination. However, most of these changes were alleviated by prophylactic treatment of animals with C. citratus dose dependently (p < 0.05). The protection was further evident through decreased histopathological alterations in liver. The results of the present study indicated that the hepatoprotective effect of C. citratus might be ascribable to its antioxidant and free radical scavenging property.
Introduction
Liver damage is a widespread pathology which in most cases involves oxidative stress and is characterized by a progressive evolution from steatosis to chronic hepatitis, fibrosis, cirrhosis and hepatocellular carcinoma. Several xenobiotics are known to cause hepatotoxicity; one among them is carbon tetrachloride (CCl4). 1 CCl4 is metabolized by P450 enzyme system in liver to a highly reactive trichloromethyl radical (CCl3•) that causes lipid peroxidation (LPO) of the cytoplasmic membrane phospholipids, which is considered to be the most important mechanism in the pathogenesis of liver damage induced by CCl4. 2 Hence, it leads to the physiological and morphological changes of hepatocytes and as a consequence leads to liver injury.3,4 ClOO3• can even react with sulfhydryl groups of glutathione (GSH) and protein thiols. In addition, ClOO3• also alters the antioxidant profile of liver including the antioxidant enzymes like superoxide dismutase (SOD), catalase (CAT), glutathione (GSH), glutathione-S-transferase (GST), glutathione peroxidase (GPx), glutathione reductase (GR) and glucose-6-phosphatase dehydrogenase (G6PD). 5
Vaccines, steroids and antiviral drugs, which are commonly used for treating liver diseases, have been found to have side effects and complications to human health, especially when administered chronically or sub-chronically. Therefore, herbal products and traditional medicines with better effectiveness and safe profiles are needed as a substitute for chemical therapeutics. As oxidative stress plays a central role in liver pathologies and their progression, the use of antioxidants has been proposed as therapeutics agents, as well as drug co-adjuvants, to counteract liver damage. A number of studies have shown that the plant extracts having antioxidant activity protect against CCl4-mediated hepatotoxicity by inhibiting LPO and enhancing antioxidant enzyme activity.6–10 Thus, much attention has been focused on the herbal plants and traditional remedies, which have more effectiveness and safe profiles. Many studies have shown that herbal plants constitute high level of antioxidant activity, which protects liver from CCl4 toxicity in rats.6–10
Cymbopogon citratus (Gramineae), local name as lemongrass or serai, is a well-known medicinal plant in Malaysia. It is widely used by local people for treating high fever, stomach, gut problems and headache. It can also act as an antidepressant and as a mood enhancer. C. citratus was investigated by previous researchers having hypoglycemic, hypolipidemic 11 , anxiolytic and sedative effects. 12 Figueirinha et al. 13 reported that C. citratus leaves exhibited antiradical capacity by 2,2-diphenyl-1-picrylhydrazyl (DPPH•) assay and identified tannins, phenolic acids (caffeic and p-coumaric acid derivatives) and flavone glycosides (apigenin and luteolin derivatives) from essential oil-free infusion. Although the studies have been done on C. citratus, the pharmacological effect of the C. citratus stems is still lacking. Therefore in the present study, we examine the antioxidant and hepatoprotective effects of C. citratus against CCl4-induced oxidative damage in rats. We report, herein, the in vivo protective effects of C. citratus against CCl4-induced oxidative damage in rats. Possible mechanisms and its implication will be discussed.
Materials and methods
Chemicals
2,2-diphenyl-2-picrylhydrazyl (DPPH),
Plant samples
Plants were collected from Kebun Rimau Sdn. Bhd., Sabah. Plants were identified and authenticated by Mr. Jonny Gisil (botanist) from Institute for Tropical Biology and Conservation, Universiti Malaysia Sabah. The stem parts of C. citratus were washed with tap water and then oven dried at 35°C to 40°C to obtain the constant weight. The dried plants were then ground to the fine powder by using a blender.
Preparation of plant extract
Twenty grams of dried powders were extracted with 400 ml of 80% ethanol at 40°C for 4 hr. The material obtained was centrifuged and filtered through Whatman paper No. 1. The filtrate was then rotary evaporated using Rotavapour® and then lyophilized to dryness in vacuo. The lyophilized powder was stored in dark bottle at −80°C until use. 14
Total phenolic content
Total phenolic content was determined by the Folin-Ciocalteau method. 15 The mixture contained 1.5 ml of Folin-Ciocalteau’s reagent (1:10) and 200 μl of plant extract. The mixture was mixed and allowed to stand for 5 min at room temperature. Then, 1.5 ml of sodium carbonate (60 g/l) was added to the mixture. After incubation in dark for 90 min at room temperature, the absorbance was measured at 725 nm against blank using spectrophotometer. Gallic acid was used as a standard for determining the phenol content by the Folin-Ciocalteau method. The results were reported in gallic acids equivalents (GAE) per gram of sample. All the measurements were taken in triplicate.
2, 2-Diphenyl-2-picrylhydrazyl (DPPH) assay
The capacity to scavenge the ‘stable’ free radical DPPH was monitored. 16 Briefly, all the extracts were dissolved in dimethyl sulfoxide (DMSO) at a concentration of 5 mg/ml. Various concentrations of plant extracts (0.3 ml) were mixed with 2.7 ml of ethanol solution containing DPPH radicals (6 × 10−5 mol/L). The mixture was shaken vigorously and left to stand for 60 min in the dark. The reduction of the DPPH radical was determined by measuring the absorption at 517 nm using spectrophotometer. The radical scavenging activity (RSA) was calculated as a percentage of DPPH discoloration using the equation: % RSA = [(A control × A sample)/A control] × 100, where A control is the absorbance of the solution without the extract and A sample is the absorbance of the solution containing extract with different concentration. The extract concentration giving 50% inhibition (EC50) was calculated from the graph of RSA percentage against extract concentration. Ascorbic acid was used as standard and all measurements were taken in triplicate.
Animal experiments
Animal experiment was treated under standard ethical principles as per the university regulations and of the Federal laws for experiment on animals. Male Sprague-Dawley rats, 8–12 weeks and weighing 120 g–150 g were purchased from the Animal house of Health Campus, Universiti Sains, Malaysia. The animals were kept in the animal room under 12-hr dark/light cycle and acclimatized for a week before use. Food and water were available ad libitum. The freeze dried extract was dissolved in distilled water daily prior to oral administration. The animals were divided into five groups, with six rats in each group. CCl4 was dissolved in 1:1 corn oil. Treatment groups were based on previous study with some modification
17
as shown as below: Group I: received saline. Group II: CCl4 (1.2 ml/kg b.w. p.o.) on 13th and 14th days. Group III: C. citratus (100 mg/kg b.w. p.o.) for 14 days + CCl4 (1.2 ml/kg b.w. p.o.) on 13th and 14th days. Group IV: C. citratus (200 mg/kg b.w. p.o) for 14 days + CCl4 (1.2 ml/kg b.w. p.o.) on 13th and 14th days. Group V: C. citratus (300 mg/kg b.w. p.o) for 14 days + CCl4 (1.2 ml/kg b.w. p.o.) on 13th and 14th days.
The effective dose of C. citratus extract was selected according to earlier report. 11 The male rats were used in the study due to the constant metabolism compared to the variation physiology in the female rats. 18 After 24 hr of administration of last dose CCl4, the animals were killed by cervical dislocation. Blood was collected from posterior vena cava using 23-gauge needle into plain tubes. The samples were centrifuged at 3000 rpm for 10 min to obtain the serum. The sera were stored in −80°C freezer until further use. The absolute and relative (organ-to-body weight ratio) weights of the liver were measured.
Determination of serum biochemistry
Enzyme activities of alanine aminotransferase (ALT) and aspartate aminotransferase (AST) were analyzed by Reflotron® system. Lactate dehydrogenase (LDH) activity was measured by monitoring the rate of oxidation of NADH due to reduction of pyruvate to lactate by enzyme. 19 The reaction mixture contained 0.05 ml serum, 2.75 ml phosphate buffer (0.1 M, pH 7.4), 0.1 ml sodium pyruvate (0.01 M) and 0.1 ml NADH (0.002 M). The decrease in absorbance was recorded at 340 nm for 3 min at 30 s interval. The result was expressed as nmol NADH oxidized/min/mg protein using molar extinction coefficient of 6.22 × 10 3 M−1 cm−1.
Preparation of liver post-mitochondrion supernatant (PMS)
Sample livers (10% w/v) were homogenized in phosphate buffer (0.1 M, pH 7.4), and then centrifuged at 3000 rpm for 20 min at 4°C (5804 R, Eppendorf, Germany) to discard nuclei debris. The supernatant obtained was again centrifuged at 10,000 rpm for 30 min at 4°C (5804 R, Eppendorf, Germany). The PMS obtained was used for the measurement of biochemistry parameters. Total protein contents were determined by using bicinchoninic acid and protein assay kit (BCA1). Bovine serum albumin was used as standard.
Determination of lipid peroxidation and GSH
LPO was done following the method of Buege and Aust 20 with some modification, by measuring the rate of production of TBARS (expressed as malondialdehyde equivalents). PMS (1.0 ml) was mixed with trichloroacetic acid (10% w/v) (0.5 ml) and centrifuged at 10,000 rpm for 30 min at 4°C. Supernatant aliquot, 1.0 ml, was mixed with 2.0 ml thiobarbituric acid (0.67% w/v) prepared by dissolving 0.67 g of TBA in warm distilled water. All the tubes were placed in a boiling water bath for a period of 30 min. At the end, the tubes were cooled in an ice bath. The absorbance was measured at 535 nm using spectrophotometer. The result was expressed as the amount of MDA formed/g tissue by using a molar extinction coefficient of 1.56 × 10 5 M−1 cm−1.
Reduced glutathione (GSH) in liver was determined by the method of Jollow et al. 21 An aliquot of 1.0 ml of PMS (10% w/v) was precipitated with 1.0 ml of sulfosalicylic acid (4% w/v). The samples were kept at 4°C for at least 1 hr and then centrifuged at 3000 rpm for 30 min at 4°C. Then, the assay mixture contained 0.2 ml filtered aliquot, 2.6 ml phosphate buffer (0.1 M, pH 7.4) and 0.2 ml DTNB (4 mg/ml of phosphate buffer at 0.1 M and pH 7.4) to make a total volume of 3.0 ml. The yellow color was developed and read immediately at 412 nm with a spectrophotometer. The result was expressed as μmol-reduced GSH/g tissue by using molar extinction coefficient of 13.6 × 10 3 M−1 cm−1.
Catalase (CAT) activity
CAT activity was assayed by the method of Claiborne. 22 CAT catalyzes the conversion of H2O2 to water. The reaction mixture contained 0.01 ml PMS, 0.99 ml phosphate buffer (0.1 M, pH 7.4) and 1 ml H2O2 (0.019 M). The reaction was initiated by the addition of PMS. The decrease of the absorbance was recorded at 240 nm for every 30 s for 3 min. The result was expressed as μmol/min/mg protein by using molar extinction coefficient of 6.4 × 10 3 M−1 cm−1.
Glutathione peroxidase (GPx) activity
GPx activity was measured according to the procedure of Mohandas et al. 23 The reaction solution contained 0.01 ml PMS, 1.58 ml phosphate buffer (0.1 M, pH 7.4), 0.1 ml EDTA (0.5 mM, pH 8.0), 0.1 ml sodium azide (1.0 mM), 0.1 ml reduced glutathione (1.0 mM), 0.1 ml NADPH (0.1 mM) and 0.01 ml H2O2 (30%) in a total volume of 2 ml. The disappearance of NADPH at 340 was recorded for 3 min at 30 s interval. Enzyme activity was calculated as nmol NADPH oxidized/min/mg protein using molar extinction coefficient 6.22 × 10 3 M−1 cm−1.
Glutahione reductase (GR) activity
GR activity was measured according method of Carlberg and Mannervik. 24 The assay consisted 0.025 ml PMS, 1.725 ml phosphate buffer (0.1 M, pH 7.4), 0.1 ml EDTA (0.5 mM), 0.05 ml oxidized glutathione (1.0 mM) and 0.1 ml NADPH (0.1 mM). The disappearance of NADPH at 340 was recorded for 3 min at 30 s interval using spectrophotometer and calculated as nmol NADPH oxidized/min/mg protein using molar extinction coefficient 6.22 × 10 3 M−1 cm−1.
NAD(P)H: quinone oxidoreductase (QR) activity
QR activity was measured according method of Benson et al. 25 as described by Iqbal et al. 26 The reaction solution consisted 0.01 ml PMS, 1.1 ml Tris-HCl buffer (25 mM, pH 7.4), 0.7 ml BSA (1 mg/ml), 0.02 ml Tween-20 (1 % v/v), 0.1 ml FAD (0.15 mM), 0.02 ml NADPH (30 mM) and 0.05 ml 2,6-dichlorophenolindophenol (2.4 mM) in a final volume of 2.0 ml. The disappearance of 2, 6-dichlorophenolindophenol was recorded at 600 nm for 3 min at 30 s interval. The activity was expressed as nmol 2,6-dichlorophenolindophenol reduced/min/mg protein using molar extinction coefficient of 2.1 × 10 4 M−1 cm−1.
Glutathione-S-transferase (GST) activity
GST activity was determined by the method of Habig et al., 27 using 1-chloro 2, 4 dinitrobenzene as a substrate. The reaction mixture contained 0.1 ml PMS, 1.75 ml phosphate buffer (0.1 M, pH 7.4), 0.1 ml reduced glutathione (1.0 mM) and 0.05 ml CDNB (1.0 mM) to a total volume of 2.0 ml. The change of the absorbance was recorded at 340 nm for 3 min at 30 s interval. Enzyme activity was calculated as nmol CDNB conjugate formed/min/mg protein using a molar extinction coefficient of 9.6 × 10 3 M−1 cm−1.
Glucose-6-phosphate dehydrogenase (G6PD) activity
G6PD activity was measured by the method of Zaheer et al. 28 The total volume of 3.0 ml consisted of 0.1 ml PMS, 0.5 ml Tris-HCl buffer (0.2 M, pH 7.6), 0.05 ml NADP (0.1 mM), 0.05 ml glucose-6-phosphatase (0.8 mM), 0.25 ml MgCl2 (8 mM) and 2.05 ml distilled water. The change of the absorbance was recorded at 340 nm for 3 min at 30 s interval. The activity was expressed as nmol NADP reduced/min/mg protein using molar extinction coefficient of 6.22 × 10 3 M−1 cm−1.
γ-Glutamyl transferase (γ-GGT) activity
γ-GGT activity was measured according method of Orlowski and Meister. 29 PMS, 0.5 ml, was mixed with 1 ml of substrate mixture consisting of glycyl glycine (40 mM), magnesium chloride (11 mM) and L-γ-glutamyl-p-nitroanilide (4 mM) in Tris-HCl buffer (185 mM, pH 8.25). The mixture was allowed to stand for 10 min at 37°C in water bath. Then, 1 ml of TCA (25% w/v) was added into each tube and subjected to centrifugation at 3000 rpm. The absorbance was measured at 405 nm by spectrophotometer. The activity was expressed as nmol p-nitroanaline released/min/mg protein.
Histopathological examination
A portion of the liver was cut and fixed in 10% (w/v) phosphate buffered formaldehyde solution. After embedding in paraffin wax, thin sections of 4 μm thickness of liver tissue were cut and stained with haematoxylin–eosin (H&E), using standard techniques. The thin sections of liver were made into permanent slides and mounted. The slides were examined under microscope with photographic facility and photomicrographs were taken.
Statistical analysis
The results were represented as mean ± SD. All statistical comparison was made by means of one-way analysis of variance (ANOVA). The homogeneity of the variance was tested by Levene’s test. The data were analyzed by using computer program SPSS (Release 17.0, SPSS). A p-value less than 0.05 were considered to be significant different. The EC50 were determined using non-linear regression analysis of the dose–response curve by using Graph Pad prism 5.
Results
Total phenolic content
Ethanol extract of C. citratus showed 30.74 ± 1.13 mg GAE/ g extract of phenolic content.
Effect of C. citratus on DPPH radical scavenging
As shown in Figure 1 , C. citratus scavenged 13% of the activity and ascorbic acid scavenged 96% at 300 μg/ml. The EC50 were calculated. EC50 of C. citratus and ascorbic acid were 994.77 ± 12.57 μg/ml and 10.06 ± 1.42 μg/ml, respectively.
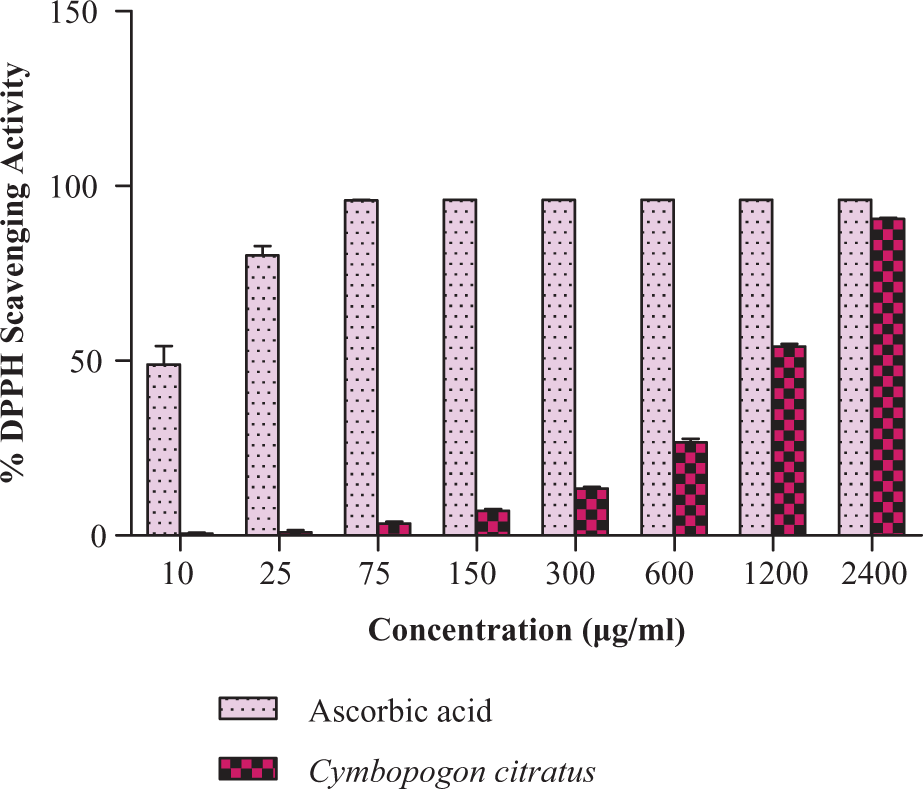
The DPPH free radical scavenging activity of C. citratus at different concentrations (μg/ml). Each value represents the mean ± SD of three independent experiments. Experimental conditions are described in materials and methods section.
Effects of C. citratus on liver index
There was no significant change of the body weight and liver index of rats (Data not shown).
Effects of C. citratus on CCl4-induced hepatotoxicity
As shown in Table 1 , during the administration of CCl4 there were significant (p < 0.05) increased levels of ALT (838.17 ± 65.09 IU/I), AST (4191.67 ± 707.18 IU/I) and LDH (26.83 ±0.30) compare to control group ALT (38.12 ± 4.87), AST (193.00 ± 38.28) and LDH (19.17 ± 0.19). In contrast, the pretreatment of C. citratus was significant (p > 0.05) and decreased the elevation of ALT, AST and LDH. This result showed that C. citratus extract could inhibit the elevated ALT, AST and LDH in rats intoxicated with CCl4.
Effects of C. citratus on CCl4-induced alterations in serum biochemistry a
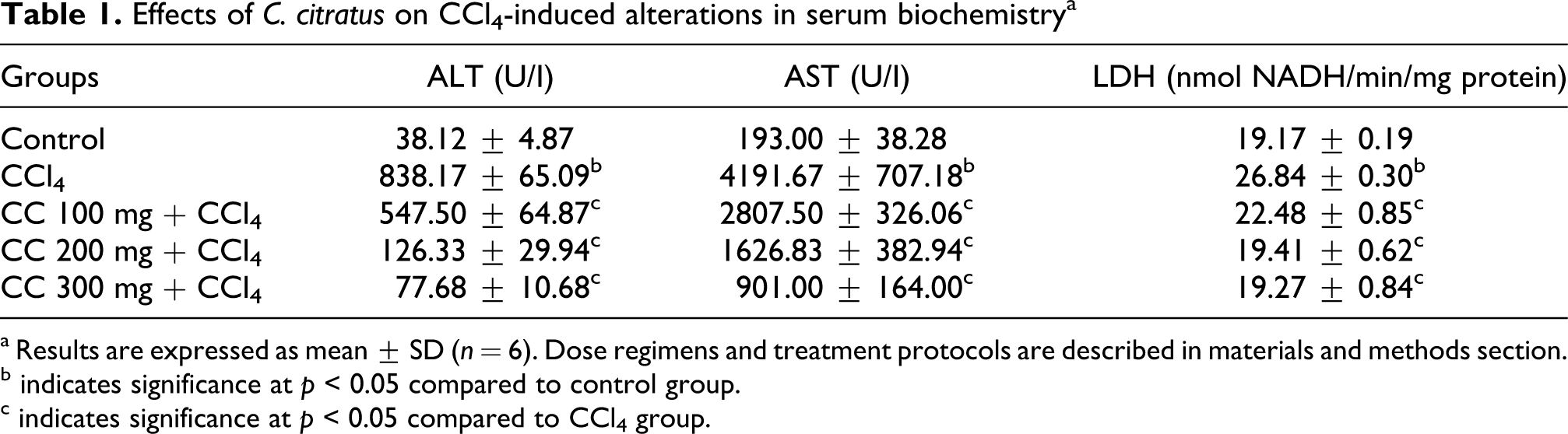
a Results are expressed as mean ± SD (n = 6). Dose regimens and treatment protocols are described in materials and methods section.
b indicates significance at p < 0.05 compared to control group.
c indicates significance at p < 0.05 compared to CCl4 group.
Effects of C. citratus on lipid peroxidation and GSH
The effects of C. citratus on MDA and GSH are shown in Table 2 . The level of hepatic MDA significantly (p > 0.05) increased in CCl4 intoxificated rats by 51% (33.55 ± 0.67 nmol/g tissue) compared to control group (22.23 ± 0.71 nmol/g tissue). Interestingly, the pretreatment of C. citratus extract offsets the level of hepatic MDA significantly (p < 0.05) by 24%, 30% and 44% in a dose-dependent manner. Although the administration of CCl4 alone significantly (p < 0.05) diminished the GSH in liver (36%, 6.29 ± 0.17 μmol/g tissue). The pretreatment of C. citratus extract protected the depletion of GSH level by increasing 31%, 36% and 48% in a dose-dependent manner.
Effects of C. citratus on CCl4-induced in hepatic lipid peroxidation and reduced glutathione a
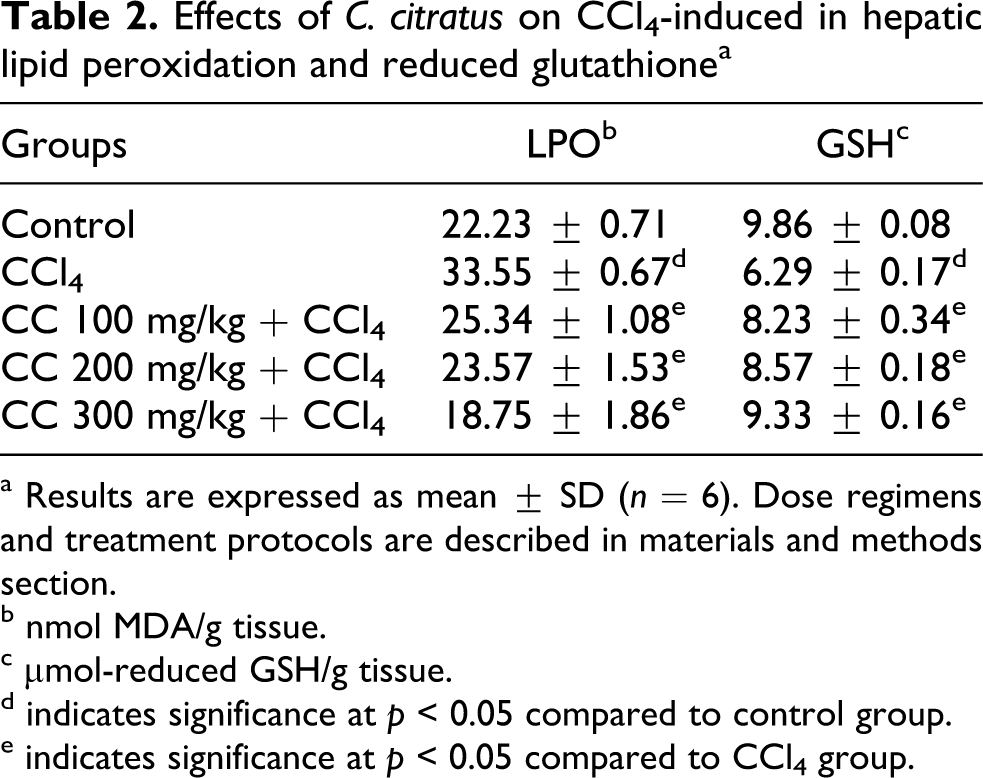
a Results are expressed as mean ± SD (n = 6). Dose regimens and treatment protocols are described in materials and methods section.
b nmol MDA/g tissue.
c μmol-reduced GSH/g tissue.
d indicates significance at p < 0.05 compared to control group.
e indicates significance at p < 0.05 compared to CCl4 group.
Effects of C. citratus on antioxidant enzymes
The activities of CAT, GPx, GR, QR, GST and G6PD are shown in Table 3 . Administration of CCl4 caused alteration of the antioxidant enzymes in liver. Significantly (p < 0.05) lower hepatic CAT (30%, 1.52 ± 0.06), GPx (37%, 214.77 ± 6.78), GR (40%, 59.10 ± 7.05), QR (49%, 43.35 ± 3.98), GST (13%, 97.83 ± 1.87) and G6PD (20%, 10.77 ± 0.19 mg protein) compared to control groups (2.17 ± 0.08, 338.66 ± 43.32, 98.32 ± 13.61, 85.26 ± 1.83, 112.47 ± 3.09 and 13.52 ± 0.23 mg protein) were observed in the CCl4-induced hepatotoxicity. Interestingly, the pretreatment of C. citratus extract was significant and increased the level of antioxidant enzymes in a dose-dependent manner. Pretreatment of C. citratus extract significance increased the GR and QR by 80% and 29% at 100 mg/kg b.w. Meanwhile, the antioxidant enzymes (CAT, GPx, GR, QR, GST and G6PD) were significant and increased at 200 mg/kg b.w. and 300 mg/kg b.w. The γ-GGT was not significantly altered in acute CCl4-induced hapatotoxicity (Data not shown).
Effects of C. citratus on CCl4-induced in hepatic antioxidant enzymes a

Results are expressed as mean ± SD (n = 6). Dose regimens and treatment protocols are described in materials and methods section.
a indicates significance at p < 0.05 compared to control group.
b indicates significance at p < 0.05 compared to CCl4 group.
c μmol H2O2/min/mg protein.
d nmol NADPH oxidized/min/mg protein.
e nmol 2,6- DCP reduced/min/mg protein.
f nmol CDNB conjugate/min/mg protein.
g nmol NADP reduced/min/mg protein.
h nmol p-nitroanaline released/min/mg protein.
Effects of C. citratus on histopathological examination
Histopathological examination of the liver revealed normal cellular architecture with distinct hepatic cells, central vein and sinusoidal spaces as shown in Figure 2A . In contrast, the CCl4-treated group showed the degenerative changes such as fatty change, necrotic cells, cellular hypertrophy, sinusoidal dilatation, hemorrhage and inflammatory cell infiltration (Figure 2B). The pretreatment of AP extract before CCl4 challenge was remarked ameliorated in a dose-dependent manner with the parallel results of serum aminotransferase activity and hepatic oxidative stress profile.

Histopathological changes in rat livers (H & E). Dose regimens and treatment protocols are described in materials and methods section. (a) Control liver; (b) rat liver intoxicated with CCl4; (c) rat liver intoxicated with CCl4 and pretreated with C. citratus (100 mg/kg b.w.); (d) rat liver intoxicated with CCl4 and pretreated with C. citratus (200 mg/kg b.w.); (e) rat liver intoxicated with CCl4 and pretreated with C. citratus (300 mg/kg b.w.). Magnification ×100.
Discussion
Phenolic compounds are the major antioxidant constituents in the natural products and contribute in treating various pathological diseases. The higher content of polyphenols is correlated with the higher antioxidant activities. A great number of medicinal plants worldwide showed a perceptible content of phenolic content, strong antioxidant activity and powerful scavenger activity against free radicals.14,30–33 DPPH scavenging activity is a common in vitro test to evaluate the scavenger activity of the sample. DPPH radical is a stable-free radical. Antioxidant present in the plant reduces the DPPH to a yellow colored compound, diphenylpicrylhydrazine, and the extent of reducing will depend on the hydrogen-donating ability of the antioxidants. 34 Current study showed that ethanol extract of C. citratus contains a perceptible amount of phenolic contents and DPPH-free RSA.
Liver disease is considered a major health problem worldwide which in most cases involves oxidative stress damage. Hepatic oxidative stress is progressive from hepatic steatosis to chronic hepatitis, fibrosis, cirrhosis and hepatocellular carcinoma. 5 The modern drugs and vaccines used by modern medicine are proved to have side effects and complications to human health. In view of this, researchers are concerned to investigate the medicinal plants with more safety profile and effectiveness is needed to protect or cure the liver disease. Medicinal plants are being used by traditional ways to treat liver disease since last decade. However the pharmacological and biological studies, especially animal model, have not been fully investigated. Thus, present study was conducted to evaluate the efficacy of ethanol extract of C. citratus extract against CCl4-induced hepatotoxicity in rats.
CCl4 is a potent chemical hepatoxin and widely used as hepatotoxicity model. 35 It is commonly used for the screening protective effects of plants against hepatic dysfunction or hepatic oxidative stress damage.9,10,36 Study showed that administration of CCl4 causes acute hepatocellular injury with centrilobular necrosis and steatosis. 37 CCl4 is metabolized by P450 system such as cytochrome (CYP)2E1, CYP2B2 or CYP2B2 and possible CYP3A to highly reactive metabolite, trichloromethyl radical (CCl3•) and trichlromethyl peroxyl radical (CCl3OO•) in the liver. CCl3• can attach to nucleic acid, protein and lipid cells and initiate cellular impairment such as lipid metabolism.3,4 CCl3OO• attacks the polyunsaturated fatty acids by initiating the LPO. Thus, it affects the permeability of the cellular membrane and causes loss of cellular calcium ions and consequence cell damage. 3 The cellular damage of the hepatic cell attributes to increase the leakage of ALT and AST in to bloodstream. 4 The ALT and AST is a major and sensitive hepatic damage marker. 38 The elevated ALT and AST denotes acute liver damage and inflammatory hepatocellular disorders. Hepatic damage induced by CCL4 administration is observed by evaluating the increasing level of serum ALT, AST and LDH. Our results showed that ALT and AST were significantly elevated in the CCl4-intoxificated rats. This reflects that acute liver damage occurs, which is consistent with the findings from other investigators.5–7 It denotes the hepatic damage after the administration of CCl4. Interestingly, the pretreatment of ethanol extract of C. citratus decreased the level ALT, AST and LDH significantly in a dose-dependent manner. Furthermore, these findings were confirmed by histopathological examination. This result suggested that pretreatment of ethanol extract of C. citratus could inhibit the CCl4-induced hepatic oxidative damage by maintaining the permeability of the cellular membrane as well as repair of hepatic tissue damage caused by CCl4 and inhibiting the leakage of ALT, AST and LDH into the bloodstream.
The increase of MDA is an indicator of liver tissue damage 39 because MDA is an end product formed during the peroxidation of membrane polyunsaturated fatty acids. 40 When O2 tension is high, CCl3•radicals formed from the metabolism of CCl4 by P450 system react rapidly with O2 and form more reactive CCl3OO•. The CCl3OO• radical abstracts the hydrogen atom from polyunsaturated fatty acids and initiates the LPO. 3 This means that LPO is elevated when the level of free radical is raised. In the present study, MDA was significantly increased in CCl4-administration group. This result was supported by the previous studies and indicates the increase of LPO by evaluating the elevation of MDA.9,10,41,42 In contrast, the pretreatment of ethanol extract of C. citratus before CCl4 challenge offsets the level of MDA significantly in a dose-dependent manner. Meanwhile, GSH is important for the detoxification of reactive toxic metabolites in cells and the necrosis is initiated when the GSH is depleted. 43 GSH was significantly decreased in the CCl4-induced toxicity group. This result was correlated to the elevated level of MDA in the CCl4-administration rats. In contrast, the GSH level was elevated when pretreatment of C. citratus extract was given before the CCl4 challenge. It strongly suggests that ethanol extract of C. citratus could protect the liver from CCl4 oxidative stress damage.
In the mean time, cellular antioxidants are also important for protection against oxidative stress. ROS, such as superoxide anions, and H2O2 are produced during the normal cellular metabolism and well regulated by homeostasis. The intracellular concentration of ROS is raised when the equilibrium balance of both production or removal of free radicals by various antioxidants is hampered. CAT and GPx catalyze the decomposition of harmful H2O2 to water. GSH is oxidized to GSSG in the catalyzation of GPx. GSSG formed is then reduced by GR to GSH. GST, the primary phase II enzyme involved in detoxification 5 , catalyzes the conjugation of GSH with xenobiotics and their reactive metabolites to produce water-soluble compounds. 18 QR is a flavoprotein which utilizes NADH or NADPH as a reducing cofactor and detoxifies the reactive quinone to less toxic hydroquinones as described by Iqbal et al. 26 The interaction between semiquinone and oxygen molecules is prevented because of the reducing process of QR and thus bypasses the production of semiquinone. Furthermore, endogenous catechol estrogen quinones formed generates O2•− and thus covalently binds to DNA-forming depurinating adducts which leads to cancer induction. 39 G6PD is a membrane bound enzyme that eliminates the lipid peroxides and toxic oxygen radicals. It is a key enzyme which catalyses the first steps of pentose phosphate metabolic pathway. 44 The damage to cell membrane, caused by LPO, leads to the decrease of G6PD. 45 Previous experiments reported that the CCl4 administration decreased the level of antioxidant enzyme, such as CAT, GPx, GST, GR, QR and G6PD.5–10,46 Our findings showed that the levels of antioxidant enzymes (CAT, GPx, GST, GR, QR and G6PD) were reduced in the CCl4-administration group. Interestingly, the pretreatment of ethanol extract of C. citratus before CCl4 administration significantly elevated the antioxidant enzymes. This result suggested that the pretreatment of C. citratus extract could inhibit the reducing activity of antioxidant enzyme and protect from CCl4-induced oxidative stress.
Histopathological study showed that CCl4 induced a marked massive fatty change, necrosis of hepatocytes, sinusoidal dilatation, inflammatory infiltration of neutrophils, lymphocytes and kupffer cells, which was supported by previous study.47,48 This finding is correlated to the elevation of serum aminotransferase, LDH and hepatic oxidative stress markers which denote the progression of acute liver damage by the intoxification of CCl4. However, the pretreated rats with ethanol extract of C. citratus before administration of CCl4 revealed the lesser or mild lesion in the liver. A similar evident was observed from serum aminotransferase, LDH and hepatic oxidative stress marker.
In conclusion, it suggested that the alteration of biochemical profile of CCl4-induced hepatotoxicity is reversed towards normalization by ethanol extract of C. citratus. Ethanol extract of C. citratus protects the integrity of plasma membrane by reducing the leakage of serum aminotransferase and LDH. At the same time, it also increased the regenerative and reparative capacity of liver by normalizing the hepatic oxidative stress marker profile. Apart from biochemical profile, the histological examination revealed lesser or mild liver damage caused by CCl4. On the basis of our results, it is hypothesized that the efficacy of hepatoprotective effects of ethanol extract of C. citratus may be largely related to the phenolic content and free RSA by stabilizing the membrane and maintaining the normal functional of hepatocytes. We conclude that the ethanol extract of C. citratus could be used as a therapeutic agent to protect the liver from oxidative stress damage. Prior to considering the therapeutic agent of C. citratus, the mechanism of action, comprehension investigation of pharmacokinetic and bioavailability of its bioactive constituents are much essential and needed.
Footnotes
Acknowledgements
This study was done under FRG166-SP-2008 from Ministry of Higher Education, Government of Malaysia. The authors thank the Director of Biotechnology Research Institute, Prof. Datin, Dr. Ann Anton for her kind support and facilities in the study. They acknowledge Kebun Rimau Sdn. Bhd for the providing the plant. They also acknowledge Prof. Dr. Amran Ahmed, School of Science and Technology, UMS for the suggestion and advice on the statistical analysis and Dr. Mie Mie Sein, School of Medicine, UMS for the analysis and interpretation of histology. The authors would also like to express special thanks to Dr. Khoo Yong Pheng for the animal care, suggestion and helpful discussion.
This research work was supported by research grant from Ministry of Higher Education, Government of Malaysia (Project No.: FRG0166-SP-2008).
