Abstract
Background
Ifosfamide (IFO) is a widely used antineoplastic drug with broad-spectrum efficacy against various types of cancer. However, different toxicities associated with IFO has limited its use. This study was to establish the prophylactic effects of betanin, chrysin and ellagic acid against IFO-induced neurotoxicity in rats.
Methods
Animals were randomly divided into eight groups, control, IFO, IFO + betanin, IFO + chrysin, IFO + ellagic acid, betanin, chrysin and ellagic acid groups. Betanin (50 mg/kg, i.p.), chrysin (25 mg/kg, i.p.) and ellagic acid (25 mg/kg, i.p.) were administered to rats once daily for two consecutive days. IFO (500 mg/kg, i.p.) was administered on third day.
Results
Results demonstrated that only ellagic acid markedly decreased the activity of acetylcholinesterase (AChE) and butrylcholinesterase (BChE) compared with IFO alone, while chrysin was only effective on BChE activity. Also, ellagic acid ameliorated IFO-induced lipid peroxidation and glutathione (GSH) depletion, while chrysin only decreased GSH depletion. Histopathological alteration in the IFO-induced brain tissues were decreased especially after administration of ellagic acid. Intraperitoneal pretreatment with betanin, followed by IFO resulted in death of all treated animals. In addition, all mitochondrial toxicity parameters induced by IFO in the rat brain tissue were ameliorated by ellagic acid, chrysin and even betanin.
Conclusion
Taken together, our results demonstrated that especially ellagic acid and to some extent chrysin show a typical neuroprotective effect on IFO-induced acute neurotoxicity through mitochondrial protection and antioxidant properties. Also, the results of our studies showed that pretreatment with betanin followed by IFO was lethal.
Introduction
Ifosfamide (IFO) is an antineoplastic agent used for a variety of hematological and solid organ malignancies. 1 The common side effects of ifosfamide are myelosuppression, urotoxicity and neurotoxicity. 2 The previous studies have reported ifosfamide-induced neurotoxicity with a frequency of 3–19% in children and 10–30% in adults. 3 The first 12 and 146 h after the administration of ifosfamide, the initial symptoms of neurotoxicity are usually manifested and these signs spontaneously removed 72 h after the cessation of ifosfamide. 3 Neurotoxicity induced by ifosfamide can appear in several ways, including agitation, lethargy, hallucinations, confusion, disorientation, seizures and extra-pyramidal signs.4,5 In rare cases, ifosfamide-induced neurotoxicity can progress to irreversible brain damage, coma and death. 6 The exact mechanism of ifosfamide-induced neurotoxicity is unknown. However, it has been suggested that active metabolites of ifosfamide and mitochondrial dysfunction may play a role in ifosfamide-induced neurotoxicity.7,8 Ifosfamide as a prodrug is metabolized in the liver by cytochrome P450 enzymes into the active alkylating agents including 4-hydroxy-ifosfamide, ifosfamide mustard, chloroacetaldehyde and chlorothylamine. 8 The major contributing factors in the development of ifosfamide-induced neurotoxicity are chloroacetaldehyde and chlorothylamine metabolites.8,9 Due to penetration into the blood-brain barrier, the active alkylating agents chloroacetaldehyde and chlorothylamine are thought to be the important neurotoxic agents. 9 It has been reported that chloroacetaldehyde induces its toxicity through alkylation or decrease of ATP via inhibition of mitochondrial oxidative phosphorylation. 10 Additionally, the neurotoxic symptoms induced by ifosfamide are thought to develop due to glutaric acid accumulation. 11 The accumulation of glutaric acid leads to inhibition of mitochondrial respiration. 11 Mitochondrial dysfunction results in an increase in intracellular nicotinamide adenine dinucleotide, inhibiting the oxidation and dehydrogenation of chlorothylamine and chloroacetaldehyde. 9 Mitochondrial dysfunction is associated with increased oxidative stress. 12 Both mitochondrial dysfunction and oxidative stress have been observed in ifosfamide-induced neurotoxicity. 13 Therefore, the use of antioxidant agents may play an important role in reducing the neurotoxicity of ifosfamide without interfering with the therapeutic effect of ifosfamide.
Natural compounds have been shown a spectrum of promising therapeutic effects in shielding humans from disorders such as lipid oxidation, inflammation and cancer. 14 Plant-derived natural compounds have been used as traditional remedies for thousands of years. 15 Recently more studies are being focused on the neuroprotective effects of plant-derived natural compounds. 16 Previous investigations have reported the potential therapeutic effects of natural compounds on brain damage via anti‐inflammation, anti-apoptosis and anti-oxidation activity. 16 These natural compounds have been demonstrated to increase the activation of superoxide dismutase (SOD), catalases (CAT), and the glutathione system. 17 Plant-derived natural compounds have been reported to reduce the malondialdehyde (MDA) level, an index of lipid peroxidation. 17 Several natural compounds have been reported to restore depletion of the mitochondrial membrane potential (MMP) and to diminish the release of cytochrome c. 18 Betanin, ellagic acid and chrysin as plant-derived natural compounds have shown promising effects in reducing brain injury and neurotoxicity. Previous studies have reported neuroprotective effect of betanin, a natural food colorant with powerful antioxidative properties against aluminum chloride (AlCl3) and trimethyltin-induced neurodegeneration via its anti-oxidation activity.19,20 Also, neuroprotective effects of ellagic acid due to its iron chelating, antioxidant and mitochondrial protective effect has been demonstrated by other.21,22 Additionally, chrysin a natural compound found in honey, propolis and plants, suppress neuroinflammation due to its antioxidant and mitochondrial protective effect and possesses potent neuroprotective effects. 23 To our knowledge, the protective role of betanin, ellagic acid and chrysin against ifosfamide-induced neurotoxicity has not been studied. In this study, we investigated the possible protective role of betanin, ellagic acid and chrysin against ifosfamide-induced neurotoxicity in the adult Wistar rats.
Materials and methods
Chemicals
Rhodamine123, 2′,7′-dichlor-fluorescein (DCF), betanin, chrysin, ellagic acid, bovine serum albumin (BSA), hank’s balanced salt solution (HBSS), n-(2-hydroxyethyl) piperazine-n′-(2-ethanesulfonic acid) (HEPES), acetylthiocholine iodide, dimethyl sulfoxide (DMSO), butyrylthiocholine iodide, d-mannitol, rotenone, sucrose, monopotassium phosphate, Ellman’s reagent (5,5′-dithiobis-(2-nitrobenzoic acid) or DTNB), 3-morpholinopropane-1-sulfonic acid (MOPS), 2-amino-2-hydroxymethyl-propane-1,3-diol (TRIS), magnesium chloride, sodium succinate, 4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT), potassium chloride were purchased from Sigma (St Louis, MO, USA). Ifosfamide with purity of about 99% and CAS number 3778-73-2 was purchased from 22 Bahman Training Pharmacy (Ardabil, Iran).
Solutions and buffers
Mitochondrial membrane potential (MMP) buffer contained: 68 mM D-mannitol, 5 mM KH2PO4, 10 mM HEPES, 220 mM sucrose, 2 mM MgCl2, 50 μM EGTA, 2 μM rotenone, 10 mM KCl and 5 mM sodium succinate. Mitochondrial respiration buffer contained: 20 mM MOPS, 0.32 mM sucrose, 0.5 mM MgCl2, 10 mM Tris, 5 mM sodium succinate, 0.1 mM KH2PO4 and 50 mM EGTA, pH adjusted to 7.4. Mitochondrial swelling buffer contained: 2 mmol/L MgCl2, 140 mmol/L KCL, 0.5 mmol/L EGTA, 10 mmol/L NaCl, 20 mmol/L HEPES, 0.5 mmol/L KH2PO4; supplemented 1 mg/mL rotenone, pH adjusted to 7.4. Mitochondrial assay buffer contained: 10 mmol/L NaCl, 0.5 mmol/L KH2PO4, 2 mmol/L MgCl2, 0.5 mmol/L EGTA, 140 mmol/L KCL, 20 mmol/L HEPES; supplemented with 1 mg/mL rotenone and 10 mmol/L succinate, pH adjusted to 7.4. Mitochondrial isolation buffer contained; 0.2 mM EDTA, 75 mM sucrose and 225 mM D-mannitol, pH adjusted to 7.4.
Animals
Male Wistar rats, 6–8 weeks of age (180–220 g), were purchased from Baqiyatallah University of Medical Sciences (Tehran, Iran). The animals were maintained in a clean well-ventilated room at an adjusted temperature (22 ± 2°C) and humidity (55 ± 5%) in polypropylene cages. Before and during the experiment, the rats were allowed free access to standard laboratory diet and water ad libitum. The rats were allowed to acclimatize for 2 weeks prior to experimental procedures. The animal experimental protocol was approved by the Ethics Committee of the Ardabil University of Medical Sciences (Approval No: IR.ARUMS.REC.1399.482).
Study design
The administration doses of betanin, chrysin, ellagic acid and IFO were chosen according to the previous studies.24–27 Rats were weighed and randomly divided into eight groups, each containing six rats as follows: Group I (Control): Rats received intraperitoneal normal saline for 4 days to serve as control. Group II (IFO): Rats received ifosfamide (500 mg/kg BW, i.p.) the third day to induce neurotoxicity. Group III (IFO + betanin 50): Rats received betanin (25 mg/kg/day; i.p.) for 2 days, with IFO (500 mg/kg BW, i.p) single dose on day 3. Group IV (IFO + chrysin 25): Rats received chrysin (25 mg/kg/day; i.p.) for 2 days, with IFO (500 mg/kg BW, i.p) single dose on day 3. Group V (IFO + ellagic acid 25): Rats received ellagic acid (25 mg/kg/day; i.p.) for 2 days, with IFO (500 mg/kg BW, i.p) single dose on day 3. Group VI (betanin 50): Rats received betanin (25 mg/kg/day; i.p.) for 2 days. Group VII (chrysin 25): Rats received chrysin (25 mg/kg/day; i.p.) for 2 days. Group VIII (ellagic acid 25): Rats received ellagic acid (25 mg/kg/day; i.p.) for 2 days. On the fourth day, 24 h after last administration, the animals were anaesthetized by a combination of xylazine (10 mg/kg) and ketamine (50 mg/kg) and brain tissues were promptly excised, washed in chilled normal saline, and then examined for histopathological, biochemical and mitochondrial analyses.
Histopathological evaluations
Randomly, out of six samples of brain tissue, two samples were selected for histopathological evaluation in each group. The brain tissues were immersed in 10% formalin for fixation. After fixation, the dehydration possess was performed by adding a series of alcohol solutions with different concentration, at the end of steps the tissues were washed with xylene. Then the brain tissues were embedded in paraffin. The brain tissues were sectioned by using the rotary microtome to slices with thickness 5 μm. The obtained slices were placed on a microscopic slide and any additional paraffin was dissolved by passing through Clearene. The staining of sections was performed by hematoxylin and eosin (H & E) stains. Using a light microscope, the slides were observed and their images were captured by the camera fitted with the microscope. 28
Measurement of butrylcholinesterase and acetylcholinesterase enzyme activities
Butrylcholinesterase (BChE) and acetylcholinesterase (AChE) enzymes activities were measured as described by Ellman et al. 29 The reaction solution contained 750 μL distilled water, 50 μL DTNB (0.25 mM), 100 μL sodium phosphate buffer (0.1 M, pH 8.0), 50 μL homogenate, and 50 μL butyrylthiocholine iodide (for butrylcholinesterase) or 50 μL acetylthiocholine iodide (for acetylcholinesterase). The enzyme activities were measured at 412 nm wavelength with a microplate reader (BioTek, USA).
Lipid peroxidation measurement
As a measure of lipid peroxidation in brain tissues, malondialdehyde (MDA) levels were assayed in the form of thiobarbituric acid-reactive substances (TBARS). 30 Briefly, 100 mg brain tissue was mechanically homogenized in presence of 1 mL 0.1% of trichloroacetic acid by a glass homogenizer. Following homogenization, the homogenate of brain tissues was centrifuged at 10,000× g for 10 min at room temperature. The obtained supernatants were transported to new tubes containing 4 mL of 0.5% thiobarbituric acid and 20% trichloroacetic acid, then the tubes were retained in boiling water for 10 min. Following cooling, the absorbance was measured at 535 nm with an ELISA plate reader. Brain tissue proteins were estimated using the Bradford method and the brain tissue MDA content expressed as nmol MDA/mg protein.
Measurement of reduced glutathione and oxidized disulfide
Oxidative stress was measured via determination of the reduced glutathione (GSH) and oxidized disulfide (GSSG) content in brain tissues. Briefly, 100 mg brain tissue was mechanically homogenized with a glass homogenizer and centrifuged at 10,000× g for 10 min at 4°C. Following homogenization, the obtained supernatants were collected. For measurement of GSH, 100 μL supernatant was mixed with 3 mL 500 mM Tris-HCl (pH 8.0) buffer containing 10 mM DTNB and incubated at 25°C for 15 min. The absorbance of the assay mixture was measured at 412 nm. For assessment of GSSG, 100 μL of supernatant was added to 3 mL of reaction solution containing GSH reductase (1 U for each 3 mL reaction solution), 1 mM EDTA, 150 μM NADPH, 3 mM MgCl2, 500 mM Tris-HCl (pH 8.0) buffer and 10 mM DTNB, and incubated at 25°C for 15 min. The absorbance of the assay mixture was measured at 412 nm.
Mitochondria isolation
Mitochondria were extracted from the midbrain regions of the rats using the mechanical lysis and differential centrifugation. Fresh tissue samples were homogenized in mitochondrial isolation buffer containing 75 mM sucrose, 0.2 mM EDTA and 225 mM D-mannitol (pH adjusted to 7.4 with 2 mM NaOH) with a glass homogenizer on ice bath and centrifuged at 1000× g for 10 min at 4°C to isolate the nuclear fraction. The obtained pellet containing unbroken cells and nuclei were removed and the obtained supernatants were centrifuged at 12,000× g for 10 min at 4°C to sediment mitochondria. The protein content of each sample was determined using the Bradford protein assay. For each test, 100 μg proteins of rat brain isolated mitochondria were used. 31
Measurement of succinate dehydrogenase activity
The mitochondrial function was defined as the ability of the mitochondrial succinate dehydrogenase to metabolically reduce the MTT to a purple formazan dye. Rat brain isolated mitochondria were incubated in mitochondrial assay buffer containing 10 mmol/L NaCl, 0.5 mmol/L KH2PO4, 2 mmol/L MgCl2, 0.5 mmol/L EGTA, 140 mmol/L KCL, 20 mmol/L HEPES; supplemented with 1 mg/mL rotenone, 10 mmol/L succinate and 0.4% MTT in 96-well plates at 37°C for 30 min. To dissolve the formazan crystals, 100 μL of DMSO was added into each well and the absorbance was measured using an ELISA reader at 570 nm. The inhibition rate was calculated using the formula: Inhibition rate = (A570 of untreated group – A570 of treated group)/(A570 of untreated group) × 100%. 32
Measurement of mitochondrial membrane swelling
Mitochondrial membrane swelling was estimated by changes in light scattering at 540 nm. The decrease in light absorbance at 540 nm is indicator of mitochondrial membrane swelling. Briefly, rat brain isolated mitochondria were incubated in a 96 well plate with total volume of 100 μL/well (100 μg/well), in mitochondrial swelling buffer (2 mmol/L MgCl2, 140 mmol/L KCL, 0.5 mmol/L EGTA, 10 mmol/L NaCl, 20 mmol/L HEPES, 0.5 mmol/L KH2PO4; supplemented 1 mg/mL rotenone, pH adjusted to 7.4). Then, after 10 min, the absorbance was measured at 540 nm in a microplate reader (BioTek, USA). 33
Measurement of mitochondrial ROS formation
A fluorescent dye 2′,7′-dichlorofluorescein diacetate (H2DCFDA) was used to measure the mitochondrial ROS formation. This dye is not fluorescent, but it can emit fluorescence when it is oxidized in presence reactive oxygen species. Therefore, measurement of fluorescence intensity of DCFH can be used indirectly for the rate of mitochondrial ROS formation. The isolated rat brain mitochondria were incubated into 96-well plates at a density of 100 μg proteins per well in mitochondrial respiration buffer containing 20 mM MOPS, 0.32 mM sucrose, 0.5 mM MgCl2, 10 mM Tris, 5 mM sodium succinate, 0.1 mM KH2PO4, 50 mM EGTA and 10 μM H2DCF at 37°C for 15 min. Following the incubation, the mitochondrial respiration buffer was replaced with fresh mitochondrial respiration buffer without H2DCF and after 15 min, the isolated mitochondria were immediately analyzed using flow cytometry (Cyflow Space-Partec, Germany). The fluorescence intensity of DCFH was detected on the FL1 channel and 2 × 104 particles were collected for analysis. The mean of fluorescence intensities was compared between groups. 34
Mitochondrial membrane potential collapse measurement
A cationic, green-fluorescent dye rhodamine 123 was used to measure the mitochondrial membrane potential collapse. Rhodamine 123 is widely used as a fluorescent probe for the detection of the mitochondrial membrane potential (ΔΨm). In isolated mitochondria, this cationic dye distributes electrophoretically into the mitochondrial matrix and energization induces a red shift and extensive quenching of the fluorescence intensity of rhodamine 123, so that dye accumulation could be suggested as a sensitive and specific probe of mitochondrial membrane potential. 35 The isolated rat brain mitochondria were incubated into 96-well plates at a density of 100 μg proteins per well in MMP buffer containing 68 mM D-mannitol, 5 mM KH2PO4, 10 mM HEPES, 220 mM sucrose, 2 mM MgCl2, 50 μM EGTA, 2 μM rotenone, 10 mM KCl, 5 mM sodium succinate and 5 μM rhodamine 123 at 37°C for 15 min. Following the incubation, the MMP buffer was replaced with fresh MMP buffer without rhodamine 123 and after 15 min, the isolated mitochondria were immediately analyzed using flow cytometry (Cyflow Space-Partec, Germany). The fluorescence intensity of rhodamine 123 was detected on the FL1 channel and 2 × 104 particles were collected for analysis. The mean of fluorescence intensities was compared between groups. 36
Statistical analysis
The data were shown as mean ± standard error of the mean (SEM). Comparison between the groups was performed using one-way analysis of variance, followed by Tukey’s post hoc test using GraphPad Prism 9.0 (GraphPad Prism 9; GraphPad Software, San Diego, USA). p < 0.05 was considered as statistically significant. The flow cytometry data was analyzed by FlowJo software.
Results
Effects of betanin, chrysin and ellagic acid on 24-h survival rate of IFO-induced neurotoxicity in rats
Compared with the control group and the IFO group, the rats in IFO + chrysin group, IFO + ellagic acid group, chrysin group, ellagic group and betanin group, the 24-h survival rate was 100% and no deaths were observed. In the group exposed to betanin and IFO, the 24-h survival rate was zero and all animals were dead. Lower (25 mg/kg) and higher (100 mg/kg) doses of betanin were tested in the presence of IFO and again the 24-h survival rate was zero. No deaths were observed in the groups exposed to betanin or IFO alone. Due to the importance of the results obtained from the simultaneous exposure of betanin and IFO, the groups of control, IFO, IFO + betanin and betanin were repeated and the same results were observed again (n = 12). The frequency of death in betanin + ifosfamide group was 12, but it was zero for the other three groups (control, IFO and betanin). These results suggest that simultaneous exposure of betanin and IFO led to death in treated rats, however, further investigation is needed to find the cause of death.
Effects of betanin, chrysin and ellagic acid on histopathological changes induced by IFO in rat brain
The histopathological examination of the control group (normal saline) illustrated normal neurons with prominent nuclei in a fibrillary background, and the same picture was seen in groups treated with betanin, chrysin and ellagic acid. IFO exposed group showed numerous degenerating neurons that became visibly dark and shrunken. As well as dilated blood vessels, neuroinflammation and demyelination were also observed in IFO group. The histopathological analysis of IFO + betanin group and IFO + chrysin group indicated dilated blood vessels, demyelination and some degenerating neurons with moderate neuroinflammation in comparison to IFO group. The analysis of IFO + ellagic acid group in comparison to IFO group revealed mild degeneration, minimal neuroinflammation with only a few dilated blood vessels. Histological examination showed that the neuroprotective effects of ellagic acid against IFO-induced neurotoxicity is more than chrysin and betaine (Figure 1). The severity of pathological lesions was graded semiquantitatively as follows: score − was considered normal, score + mild, score ++ moderate, and score +++ severe and summarized in Table 1. Histopathological findings of brain selected of control group, IFO + betanin group, IFO + chrysin group, IFO + ellagic acid group, betanin group, chrysin group and ellagic acid group. Neuroinflammation, dilated blood vessels and demyelination (arrow). Chemical structure of ellagic acid, ifosfamide, chrysin and betanin. Histopathological semi-quantitative scoring showing the effects of ifosfamide with/without betanin, ellagic acid and chrysin pretreatment on severity of histopathologic lesions in ifosfamide-treated experimental animals. Note: Scores represent values obtained from tissue sections of six animals of each group. Scores –, normal; + mild; ++, moderate; +++, severe levels.

Effect of betanin, chrysin and ellagic acid on increase of BChE and AChE activity induced by IFO
Since there are studies showing that IFO-induced neurotoxicity is associated with alterations in AChE and BChE activity, we investigated whether betanin, chrysin and ellagic acid restores BChE and AChE activity induced by IFO. Figure 2(a) shows the effect of betanin, chrysin and ellagic acid on the activity of AChE with and without IFO. Statistical analysis of data also demonstrated a significant difference between IFO group and IFO + ellagic acid, suggesting that only the ellagic acid treatment prevented the increase in AChE activity induced by IFO. Also, statistical analysis did not show a significant difference between IFO group and IFO + betanin group on the activity of AChE. Also, Figure 2(b) shows the effects of betanin, chrysin and ellagic acid on the activity of BChE with and without IFO. Statistical analysis showed a significant difference between IFO group with IFO + chrysin group and IFO + ellagic acid group, suggesting that chrysin and ellagic acid treatment prevents the increase in BChE activity induced by IFO. Statistical analysis of the activity of acetylcholinesterase (a) and butrylcholinesterase (b), is represented as mean ± standard error of the mean (SEM) of five observations (n = 5); where symbols *, ** and *** denote a particular group compared with the control group as p < 0.05, p < 0.01 and p < 0.001 respectively, Symbols #, ## and ### signifies the comparison of a particular group with IFO group as p < 0.05, p < 0.01 and p < 0.001 respectively. Also, symbol “ns” expresses non-significant in comparison to control group (results were analyzed with one‐way ANOVA followed by post hoc Tukey test). IFO, Ifosfamide.
Effect of betanin, chrysin and ellagic acid on IFO-induced oxidative stress in rat brain tissue
Oxidative stress markers (GSH, GSSG and MDA levels) were selected to examine the bona fide encephalopathy of IFO, and the results are shown in Figure 3. Findings revealed a significant increase in brain tissue GSSG and MDA levels as well as the decrease in tissue GSH level in IFO group in comparison to the control group (normal saline), indicating IFO-induced oxidative stress and toxicity. While statistical analysis showed that group treated with ellagic acid in all three oxidative stress markers recovered IFO-induced oxidative stress. In addition, chrysin was also able to increase the amount of glutathione in comparison to the IFO group, but did not affect on MDA levels. Also, statistical analysis showed that group treated with betanin in all three oxidative stress markers did not recover IFO-induced oxidative stress. Statistical analysis of oxidative stress parameters including; GSH (a), GSSG (b) and MDA (c), is represented as mean ± standard error of the mean (SEM) of five observations (n = 5); where symbols *, ** and *** denote a particular group compared with the control group as p < 0.05, p < 0.01 and p < 0.001 respectively, Symbols #, ## and ### signifies the comparison of a particular group with IFO group as p < 0.05, p < 0.01 and p < 0.001 respectively. Also, symbol “ns” expresses non-significant in comparison to control group (results were analyzed with one‐way ANOVA followed by post hoc Tukey test). GSH, Reduced Glutathione; GSSG, Oxidized Disulfide; MDA; Malondialdehyde; IFO, Ifosfamide.
Effect of betanin, chrysin and ellagic acid on mitochondrial complex II activity in presence IFO in rat brain tissue
We investigated mitochondrial complex II activity in rat brain isolated mitochondria. Statistical analysis showed that mitochondrial complex II activity remarkably reduced in IFO treated group compared with control group. Statistical analysis in IFO + betanin group, IFO + chrysin group and IFO + ellagic acid group in comparison to IFO group showed that mitochondrial complex II activity was remarkably enhanced. Also, statistical analysis showed than betanin, chrysin and ellagic acid alone in comparison to control group did not affect mitochondrial complex II activity in rat brain isolated mitochondria (Figure 4(a)). Statistical analysis of mitochondrial complex II activity (a) and mitochondrial membrane swelling (b), is represented as mean ± standard error of the mean (SEM) of five observations (n = 5); where symbols *, ** and *** denote a particular group compared with the control group as p < 0.05, p < 0.01 and p < 0.001 respectively, Symbols #, ## and ### signifies the comparison of a particular group with IFO group as p < 0.05, p < 0.01 and p < 0.001 respectively. Also, symbol “ns” expresses non-significant in comparison to control group (results were analyzed with one‐way ANOVA followed by post hoc Tukey test). SDH, Succinate Dehydrogenase; IFO, Ifosfamide.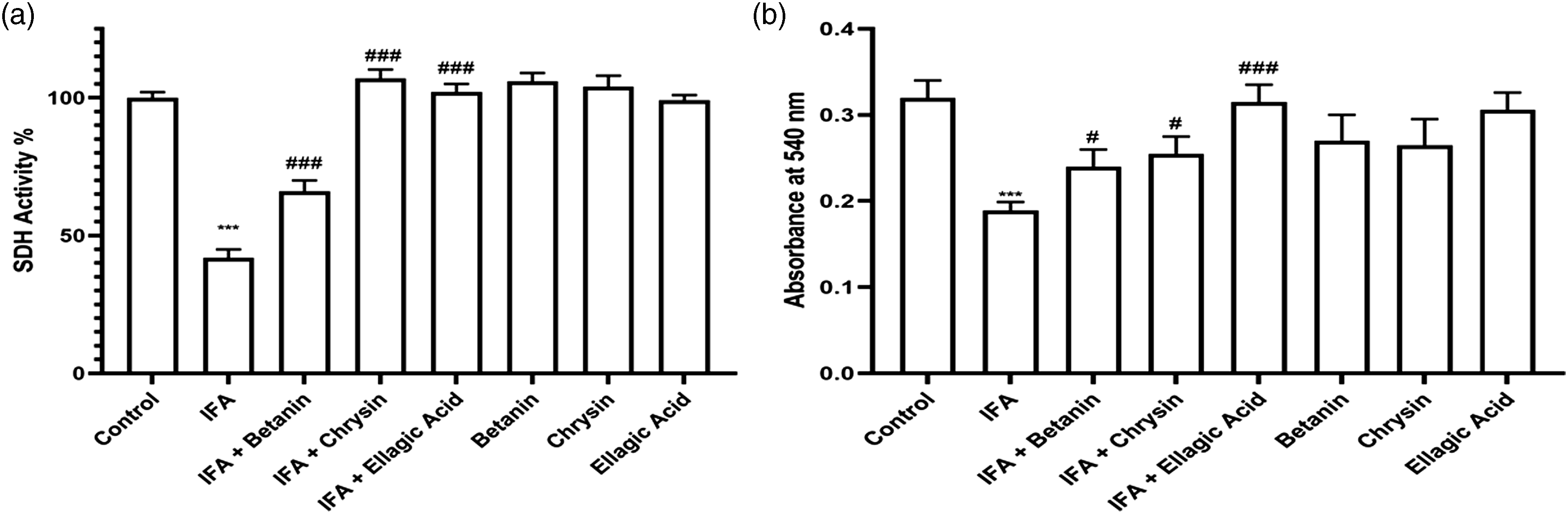
Effect of betanin, chrysin and ellagic acid on mitochondrial membrane swelling in presence IFO in rat brain tissue
Measurements of mitochondrial membrane swelling are shown in Figure 4(b). Four animals from each group were randomly selected for the measurement of mitochondrial swelling. It was observed that mitochondrial suspension absorbance at 540 nm from IFO group (0.19 ± 0.02) showed a significant decrease compared with control group (0.32 ± 0.03) which can be restored by betanin (0.23 ± 0.02), chrysin (0.24 ± 0.02) and ellagic acid (0.29 ± 0.02) pretreatment. Also, statistical analysis showed than betanin, chrysin and ellagic acid alone in comparison to control group did not change mitochondrial suspension absorbance at 540 nm.
Effect of betanin, chrysin and ellagic acid on IFO-induced mitochondrial ROS formation in rat brain tissue
The effects of betanin, chrysin and ellagic acid on IFO-induced ROS production in rat brain isolated mitochondria were investigated using H2DCF by flow cytometry. DCF fluorescence intensity was significantly increased in IFO treated group in comparison to control group. Mitochondrial ROS levels significantly decreased in IFO + betanin group, IFO + chrysin group and IFO + ellagic acid group in comparison to IFO group. Also, statistical analysis showed than betanin, chrysin and ellagic acid alone in comparison to control group did not increase mitochondrial ROS formation in rat brain isolated mitochondria (Figure 5). Statistical analysis of ROS formation in isolated mitochondria is represented as mean ± standard error of the mean (SEM) of five observations (n = 5); where symbols *, ** and *** denote a particular group compared with the control group as p < 0.05, p < 0.01 and p < 0.001 respectively, Symbols #, ## and ### signifies the comparison of a particular group with IFO group as p < 0.05, p < 0.01 and p < 0.001 respectively (results were analyzed with one‐way ANOVA followed by post hoc Tukey test). DCF, 2′,7′-Dichlorofuorescin; IFO, Ifosfamide.
Effect of betanin, chrysin and ellagic acid on IFO-MMP induced in brain tissue
Mitochondrial membrane potential is a sensitive index of the mitochondrial inner membrane status was measured by Rhodamine 123 redistribution as a fluorescent cationic dye. As shown in Figure 6, MMP was remarkably increased in the mitochondria isolated from rat brain of IFO treated group compared with control group. Four days after injection of betanin, chrysin and ellagic acid alone we did not any significant alterations compared with control group. But in IFO + betanin group, IFO + chrysin group and IFO + ellagic acid group in comparison to IFO group we observed a significant decrease in MMP (the fluorescence intensity of rhodamine 123 is reduced). Statistical analysis of mitochondrial membrane potential collapse in rat brain isolated mitochondria is represented as mean ± standard error of the mean (SEM) of five observations (n = 5); where symbols *, ** and *** denote a particular group compared with the control group as p < 0.05, p < 0.01 and p < 0.001 respectively, Symbols #, ## and ### signifies the comparison of a particular group with IFO group as p < 0.05, p < 0.01 and p < 0.001 respectively (results were analyzed with one‐way ANOVA followed by post hoc Tukey test). IFO, Ifosfamide.
Discussion
The aim of the current study was to investigate the effect of betanin, chrysin and ellagic acid on ifosfamide-induced oxidative stress, mitochondrial toxicity and histopathological changes. The results suggest that ellagic acid and partly chrysin treatment may have therapeutic potential for ifosfamide-induced neurotoxicity through mitochondrial protection and antioxidant properties in rat brain. Also, interestingly we observed that intraperitoneal pretreatment with betanin followed by IFO is lethal in male Wistar rats.
It has been reported that the pharmacological and toxicological effects of IFO as a prodrug are related to its bioactivation through the cytochrome P450 (CYP) system in the liver. 37 The activation of ifosfamide to the therapeutically active metabolite, 4-hydroxyifosfamide, is catalyzed by the hepatic CYP isoforms CYP2B6 and CYP3A4. 37 . Although ifosfamide requires CYP2B6 and CYP3A4 for metabolism and bioactivation, CYP3A5 is mainly responsible for the transformation of ifosfamide into active metabolite through autoinduction. 37 The formed 4-hydroxyifosfamide is unstable and quickly converts to aldoifosfamide, or is oxidized into 4-keto-4-hydroxyifosfamide by alcohol dehydrogenase. 37 These two active metabolites passively diffuse out of hepatocytes, circulate in the blood, and then passively enter other cells and cellular organelles such mitochondria. 37 Aldoifosfamide may be detoxified to the inactive metabolite carboxyifosfamide or the therapeutically active metabolite cytotoxic nitrogen mustards (isophosphoramide mustard) and highly electrophilic byproduct acrolein (AC). 37 Acrolein as a toxic substance has neurotoxic effect and can induce mitochondrial dysfunction and oxidative stress. 38 Also, chloroacetaldehyde as one of the metabolites of ifosfamide, can cross the blood-brain barrier and induce the oxidative stress in the brain. 39 The therapeutic and prophylactic treatment of Methylene Blue (MB) in ifosfamide-induced neurotoxicity is due to the inhibition of chloroacetaldehyde formation. 40 Chloroacetaldehyde inhibits mitochondrial complex I, suggesting that this active metabolite is responsible for the nephrotoxicity induced by ifosfamide. 41 Mitochondrial dysfunction and oxidative stress are one of the major mechanisms causing neurotoxicity. The generation of ROS mainly occurs in mitochondria that play a prominent role in the induction of oxidative stress in the neurons. 42 The homeostasis of ROS in the neurons is maintained by enzymatic and non-enzymic antioxidants like superoxide dismutase, catalase and glutathione. 43 Our results showed that ifosfamide caused lipid peroxidation, GSH depletion, mitochondrial dysfunction, mitochondrial swelling, MMP collapse and mitochondrial ROS formation. A recent study has reported that ifosfamide treatment causes a significant decrease in cellular viability, which is accompanied by ROS generation, GSH depletion, lipid peroxidation and mitochondrial and lysosomal damages in isolated rat neurons. 44 The findings of this study are consistent with our results in the current study.
The natural products such as pure compounds derived from herbs, herbal formula, crude extracts and fractions rich in bioactive components have been shown to prevent and ameliorate drug/chemical toxicity. 45 Animal and clinical studies have shown that some natural products derived from herbs can reduce radiotherapy and chemotherapy-induced neurotoxicity, hepatotoxicity cardiotoxicity, nephrotoxicity, gastrointestinal toxicity and hematopoietic system injury. 45 As mentioned above our results demonstrated that ifosfamide enhances histopatological changes, disruption of cholinergic neurons, oxidative stress and mitochodrial dysfunction. Oxidative stress and mitochondria dysfunction plays an important role in causing the ifosfamide-induced neurotoxicity. Administration of ellagic acid and partly chrysin prior to ifofamide caused a significant improvement in the histopatological changes, disruption of cholinergic neurons, oxidative stress and mitochodrial dysfunction, clearly demonstrating the antioxidant, mitochondrial protection and neuroprotective effect of ellagic acid. Also, betanin, ellagic acid and chrysin treatment prior to ifosfamide administration significantly recoved the mitochondrial functions, suggesting the mitochondrial protective effect of these naural compounds. In several studies, ellagic acid and chrysin have been widely reported to exert potent antioxidant activity and free radical scavenging by restoring the activity of cellular antioxidant enzymes and scavenging ROS. There are many studies that suggest ellagaic acid, chrysin and betanin play an important role in protecting mitochondrial function during exposure to neurotoxic agents.25,27,46 Firdaus et el., 47 have reported that ellagic acid attenuates arsenic induced neuro-inflammation and mitochondrial dysfunction associated apoptosis in hippocampus of Wistar rats. Also, ellagic acid has been able to reduce ROS and the permeability of the mitochondrial membrane potential induced by acrolein as a toxic metabolite of ifosfamide, through its antioxidant properties. 48 The findings of the study of Ardah et al., 49 demonstrated that ellagic acid protects against MPTP-induced Parkinson’s disease (PD) and the observed neuroprotective effects are attributed to its potent anti-inflammatory and antioxidant properties. It has been reported that chrysin increases antioxidant capacity, as well as regulates inflammation and apoptosis in rat brain in presence of isoniazid. 27
Our observations showed that the simultaneous presence of betanine and ifosfamide caused death in exposed animals. Symptoms such as anorexia, inactivity, lethargy, gastritis, hepatomegaly and organ adhesions were observed in IFO + Betanin. However, in the groups that received betanin and ifosfamide alone, there were no obvious symptoms and death during study. The deadly betanin-ifosfamide combination is related to; (1) a narrow therapeutic window of ifosfamide, thus any alteration in its plasma concentrations can lead to toxic manifestations; (2) ifosfamide is prodrug whose efficacy depends on effective biotransformation by cytochrome P450 enzymes, which may be inhibited or induced by betanin; and (3) alteration in ifosfamide excretion pathways by betanin, may be another reason. We observed that ifosfamid-induced neurotoxicity was not augmented in the presence of betanin, and mitochondrial toxicity parameters were significantly reduced, which could indicate that mitochondrial damage and neurotoxicity are not effective in causing this deadly betanin-ifosfamide intervention.
Overall, the findings of this study showed that ellagic acid is more effective than chrysin and can reduce the neurotoxicity induced by ifosfamide and is recommended for further investigation in clinical trials. Also, due to the observation of death in the simultaneous presence of betanin and ifosfamide, it is suggested that further studies be done on the causes of this mortality.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Ardabil University of Medical Sciences, Deputy of Research with ethics code IR. ARUMS.REC.1399.482.
