Abstract
Acrylamide (ACR) is a chemical intermediate utilized in industry. ACR is also formed during heating of foods containing carbohydrates and amino acids. Therefore, humans are widely exposed to ACR, and ACR neurotoxicity in humans is a significant public health issue attracting wide attention. In this study, we investigated the potential neuroprotective effects of epigallocatechin-3-gallate (EGCG), the most abundant polyphenolic compound in green tea, in PC12 cells treated with ACR. ACR-treated PC12 cells pretreated with various concentrations of EGCG (2.5, 5 and 10 μM) for 24 h had increased viability and acetylcholinesterase activity and reduced apoptosis and necrosis compared to cells exposed to ACR alone. EGCG reduced the expression of bax mRNA, decreased cytochrome c release, reduced intracellular calcium levels, inactivated caspase 3 and increased mitochondrial membrane potential, suggesting that EGCG prevents ACR-induced apoptosis through a mitochondrial-mediated pathway. In addition, EGCG inhibited the formation of reactive oxygen species and lipid peroxidation while enhancing superoxide dismutase activity and glutathione levels, thereby reducing oxidative stress. Our results indicate that pretreatment of PC12 cells with EGCG attenuates ACR-induced apoptosis by reducing oxidative stress. Therefore, drinking green tea may reduce nerve injury induced by ACR.
Keywords
Introduction
Acrylamide (ACR) is an environmental toxin that is a colourless and odourless crystalline substance with low molecular weight. ACR is biodegradable and soluble in water, acetone and ethanol 1 ; ACR can be absorbed via the skin, respiratory and digestive tract and diffuse through various biological membranes. 2,3 ACR is vastly produced for the synthesis of polyacrylamides, 4 which are subsequently utilized in the production of paper, soil conditioning and water treatments. As polyacrylamide flocculants contain residual ACR monomer, drinking water is a source of human exposure to ACR. 5 Consequently, ACR emissions are extensive in the environment, and human exposure is unavoidable, especially for professional workers. Another source of human exposure to ACR is tobacco smoke. 6 In recent years, studies have shown that ACR is formed during the Maillard reaction that occurs during food processing with high temperature, including the production of bread, potato chips and crisps. 4,7,8 Therefore, processed food is a main source of ACR exposure in the general population.
Although polyacrylamides are non-toxic, the ACR monomer is a potent neurotoxin, carcinogen and genotoxin with additional toxic properties. Chronic exposure to ACR induces DNA damage in male mouse germ cells. 9 Experimental animals exposed to ACR have increased rates of tumours of the thyroid gland, central nervous system and uterus. However, reproducibility in humans has been inconsistent to date. 1 In contrast, the neurotoxicity associated with ACR is well documented in exposed populations and animals. 4,10,11 Occupationally exposed workers exhibit characteristic neurotoxic symptoms, including weakness of the legs and numbness of the hands and feet after 3–10 months of exposure. 10 Rat pups treated with ACR exhibit decreased hind limb grip strength and locomotor activity and increased heel splay. 12 The mechanisms of ACR neurotoxicity have not been fully clarified; however, changes in redox status and the induction of apoptosis may be involved. 13,14 ACR and its metabolites (mainly glycidamide) conjugate with reduced glutathione (GSH), resulting in depletion of cellular stores and the overproduction of reactive oxygen species (ROS). ROS activate signalling cascades, leading to apoptosis. 14,15 Apoptosis, or programmed cell death, is an important mechanism that regulates cell proliferation and death required for the health of living systems. 16 Excessive apoptosis can lead to neurodegenerative diseases and other tissue damage.
New strategies for reducing the hazards of ACR have been proposed, including the use of promising new food ingredients. 13 Green tea is a traditional beverage that contains many bioactive constituents, and its health benefits have attracted the attention of many researchers. Polyphenols in green tea are possible neuroprotective agents, among which epigallocatechin-3-gallate (EGCG) is the most abundant with antioxidant, free radical scavenging and anti-apoptotic properties. 15,17 The neuroprotective effects of EGCG have been reported in a number of studies in both cell and animal models. 18,19 EGCG attenuates apoptosis and inflammation to alleviate nerve damage, enhances functional recovery and accelerates nerve regeneration. 20,21
Although the neuroprotective effect of green tea aqueous extract against ACR has been reported, the composition of green tea aqueous extract is complex. 22 Currently, there is a lack of research on the protective mechanism of EGCG against ACR-induced neurotoxicity. Therefore, the aim of this study was to intensively investigate whether EGCG has neuroprotective effects against ACR-induced apoptosis in PC12 cells in vitro.
Materials and methods
Materials
Dulbecco’s modified Eagle’s medium (DMEM) and foetal bovine serum were obtained from HyClone (Logan, Utah, USA). In addition, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium (MTT) and ACR (purity >99%) were purchased from Genview (Tallahasses, Florida, USA). EGCG (purity ≥95%) was purchased from Sigma (St. Louis, Missouri, USA). Horse serum was provided by Gibco (Grand Island, New York, USA). Dimethylsulfoxide (DMSO), bicinchoninic acid (BCA) assay kit and intracellular calcium assay kit were purchased from Dingguo Changsheng Biotechnology Co., Ltd. (Beijing, China). Annexin V-FITC fluorescein isothiocyanate apoptosis detection kit and caspase 3 activity assay kit were obtained from Beyotime Institute of Biotechnology (Shanghai, China). Cell apoptosis detection kit (rhodamine 123) was provided by KeyGEN BioTECH (Nanjing, China). RNAprep pure cell/bacteria kit, FastQuant RT kit (with gDNase) and RealMasterMix (SYBR Green) were purchased from Tian-gen (Beijing, China). ROS assay kit, superoxide dismutase (SOD) activity assay kit, malondialdehyde (MDA) assay kit and GSH assay kit were purchased from the Institute of Biological Engineering of Nanjing Jiancheng (Nanjing, China). Rat cytochrome C ELISA enzyme-linked immunosorbent assay kit was provided by R&D Systems (Minneapolis, Minnesota, USA).
Cell culture
PC12 is a cell line derived from a pheochromocytoma of the rat adrenal medulla. According to a previous report, 23 we choose PC12 cells as a model for neurotoxicity studies. PC12 cells were purchased from Dingguo Changsheng Biotechnology Co., Ltd. The cells were routinely cultured in DMEM supplemented with 10% (v/v) horse serum, 5% (v/v) foetal bovine serum, 2 mM glutamine, 100 U/mL penicillin and 100 μg/mL streptomycin in a humidified atmosphere containing 5% CO2 at 37°C.
Cell viability analysis
Cell viability was measured using the MTT assay as previously described. 13,17 Briefly, the cells were seeded in 96-well plates at a density of 1 × 104 cells per well. The cells were pretreated with different concentrations of EGCG (2.5, 5, 10 and 20 μM) for 24 h. ACR was added to each well at a final concentration of 6 mM. After 24 h of incubation, 20 μL of MTT solution was added to the cells at 0.5 mg/mL for 4 h. The solution was then removed, and 200 μL of DMSO was added. The optical density was evaluated at 490 nm using a Microplate Reader (BioTek-Eon, Winooski, Vermont, USA) after dissolution by oscillation for 10 min.
Measurement of acetylcholinesterase activity
Acetylcholinesterase (AChE) activity was measured as described. 24 After freezing and thawing three times, 2 μL of supernatant and 240 µL of the reaction solution containing phosphate buffer saline (PBS) (0.1 M) and 5,5′-dithiobis(2-nitrobenzoic acid) (0.25 mM) were added to 96-well plates. After incubated at 37°C for 5 min, 60 µL of acetylthiocholine iodide (0.075 M) was added. The change in absorbance per minute at 405 nm (ΔA) was measured using a microplate reader.
Annexin V-FITC/PI (propidium iodide) double staining test
Apoptotic and necrotic rates were evaluated by flow cytometry using Annexin V-FITC and PI double staining method. The Annexin V-FITC apoptosis detection kit was used according to the instruction manual. In brief, the cells were seeded in six-well plates. After pretreatment with various concentrations of EGCG (2.5, 5 and 10 μM) for 24 h, cells were incubated with 6 mM ACR. After 24 h, cells were harvested and washed twice in cold PBS and then suspended in 195 µL of Annexin V-FITC binding. Then, 5 µL of Annexin V-FITC was added, and cells were incubated with Annexin V-FITC for 10 min in the dark at room temperature. The cells were then harvested and resuspended in 190 µL of Annexin V-FITC binding, and 10 µL of PI staining solution was added. The cells were placed on ice in the dark for 10 min. The sum of necrotic cells and apoptotic cells was quantitatively measured by flow cytometry (BD LSRFortessa, San Jose, California, USA) within 1 h; 10,000 cells from each sample were counted.
Measurement of mitochondrial membrane potential
To monitor mitochondrial membrane potential (MMP) in PC12 cells, we used the rhodamine 123 assay. 25 Rhodamine 123 is a fluorescent dye that crosses the intact cell membrane and accumulates in mitochondria as a direct function of membrane potential. 26 A cell apoptosis detection kit (rhodamine 123) was used according to the instruction manual. Briefly, cells were seeded in six-well plates and pretreated with various concentrations of EGCG (2.5, 5 and 10 μM) for 24 h. Then, ACR was added at a final concentration of 6 mM and incubated for 24 h. The cells were harvested and suspended in a culture medium at a density of 1 × 106 per millilitre. The cells were then incubated with 10 μg/mL of rhodamine 123 in humidified atmosphere containing 5% CO2 at 37°C for 10 min. After the cells were harvested and washed twice in PBS, the cells were resuspended in the culture medium and continually incubated for 60 min. Fluorescence intensity was measured using flow cytometry.
Measurement of intracellular calcium
Intracellular calcium was monitored using the calcium indicator dye FLUO-3AM. 27 The intracellular calcium assay kit was used according to the instruction manual. The cells were treated with EGCG and ACR as described earlier in the methods section of Annexin V-FITC/PI double staining test and MMP measurement. The cells were harvested and suspended in RPMI 1640 culture medium at a density of 1 × 107 cells per millilitre. After adding FLUO-3AM to a final concentration of 5 μM and 1 μL of 25% (w/v) F-127 (molecular probes), the cells were incubated in a humidified atmosphere containing 5% CO2 at 37°C for 1 h. Then, the cells were washed thrice, resuspended in RPMI 1640 and analysed using flow cytometry.
Measurement of cytochrome c release
Cytochrome c was measured using an ELISA kit based on the double sandwich technique. Briefly, cytochrome c antibodies labelled with biotin were added to the samples and then combined with streptavidin-HRP (horseradish peroxidase) at 37°C for 1 h to form an immune complex. The samples were then washed, and chromogenic solutions were added. Absorbance was measured 10 min later at a wavelength of 450 nm to determine colorimetric changes positively correlating with cytochrome c levels.
Measurement of caspase 3 activity
Caspase 3 activity was measured using a colorimetric assay based on the caspase 3 catalysed release of p-nitroaniline (pNA) from acetyl-Asp-Glu-Val-Asp p-nitroanilide (Ac-DEVD-pNA), which has a strong absorption at 405 nm. 28 All procedures complied with the manufacturer’s instructions of the caspase 3 activity assay kit. Briefly, the cells were harvested, washed in cold PBS and then lysed. Supernatants were used to establish a reaction system with the addition of Ac-DEVD-pNA substrate (2 mM). After incubation for 2 h at 37°C, absorbance was measured with a microplate reader at 405 nm. The concentration of protein in samples was determined using the Bradford method, and bovine serum albumin was used as a standard.
Real-time quantitative-polymerase chain reaction (q-PCR) assays
The RNAprep pure cell/bacteria kit was used to isolate total RNA from PC12 cells. Reverse transcription from RNA to cDNA was performed using the FastQuant RT kit (with gDNase). Real-time q-PCR was performed using the iQ5 real-time PCR system (Bio-Rad, Hercules, California, USA), and PCR amplification was performed with cDNA from each example using specific oligonucleotide primers for bax (NCBI reference sequence: NM_017059.2; forward, 5′-CACCAAGAAGCTGAGCGAGT-3′, reverse 5′-CAGTGTCCAGCCCATGATG-3′; 259 bp; from 254 to 512), bcl-2 (NCBI reference sequence: NM_016993.1; forward, 5′-GGTGGTGGAGGAACTCTTCAG-3′, reverse 5′-GTTCCACAAAGGCATCCCAG-3′; 206 bp; from 621 to 826) and β-actin (NCBI reference sequence: NM_031144.3; forward, 5′-CGTGCGTGACATTAAAGAG-3′, reverse 5′-TTGCCGATAGTGATGACCT-3′; 132 bp; from 702 to 833). 29,30 Cycling parameters were defined per the recommendations of the RealMasterMix (SYBR Green) manufacturer. Relative expression in each sample was calculated using the 2−ΔΔCt method.
Measurement of intracellular ROS
To measure intracellular accumulation of ROS in PC12 cells, the fluorescent probe 2,7-dichlorofluorescein diacetate (DCFH-DA) was used as previously described. 13,31 DCFH-DA is a non-fluorescent compound that crosses cell membranes and is hydrolysed to dichlorofluorescin, which is then oxidized to the highly fluorescent dichlorofluorescein (DCF) in the presence of ROS. 13 The cells were processed for measurement of ROS according to the manufacturer’s protocol. Briefly, the cells were incubated with DCFH-DA at a final concentration of 10 μM for 30 min at 37°C. The cells were then harvested and washed twice with PBS. DCF fluorescence positively correlated with intracellular ROS levels as determined by flow cytometry.
Measurement of SOD activity, MDA levels and GSH levels
SOD activity reflects the free radical scavenging ability of cells. For these experiments, a xanthine/xanthine oxidase system was utilized to generate superoxide O2− radicals followed by measurement of SOD-mediated changes in the reduction of nitro blue tetrazolium according to the manufacturer’s instructions. 32 Levels of MDA, the most abundant lipid peroxidation product, were determined using the thiobarbituric acid colorimetric assay according to the manufacturer’s instructions. 33 GSH is an important non-enzymatic antioxidant that can be measured using the 5,5′-dithiobis(2-nitrobenzoic acid) method. The samples were normalized according to protein content, which was determined using the BCA protein assay according to the manufacturer’s instructions.
SOD activity and MDA and GSH levels in PC12 cells were all determined with assay kits. Estimation of all these parameters was determined according to the instruction manuals of the kits based on colorimetric methods.
Statistical analysis
Data were analysed and expressed as the mean ± SE (standard error) of at least three experiments. Statistical analyses were performed using SPSS 16.0 software. Statistical significance was analysed by one-way analysis of variance. The level of differences among groups was analysed by least significant digit (LSD). A p value of <0.05 was considered statistically significant.
Results
Effect of EGCG on cell viability
Cell viability was determined in PC12 cells exposed to ACR using the MTT assay as described in the methods section. Treatment with different concentrations of ACR (2, 4, 6, 8 and 10 mM) for 24 h in PC12 cells resulted in a dose-dependent decrease in cell viability. The median inhibition concentration (IC50) for ACR was approximately 6 mM (Figure 1(a)). In contrast to ACR, PC12 cells exposed to varying concentrations of EGCG (2.5, 5, 10 and 20 μM) for 24 h showed no significant decrease in cell viability (p > 0.05; Figure 1(b)).

Effect of EGCG on viability of PC12 cells exposed to ACR: (a) effect of ACR on viability of PC12 cells. (b) Effect of EGCG on viability of PC12 cells. (c) Effect of EGCG on ACR-induced reduction in the viability of PC12 cells. Data represented as (percentage of control) mean ± SE of three separate experiments. *p < 0.05 compared to control cells. # p < 0.05 compared to ACR-treated cells. EGCG: epigallocatechin-3-gallate; ACR: acrylamide.
To determine the effect of EGCG on ACR-induced toxicity, we treated PC12 cells with EGCG (2.5, 5, 10 and 20 µM) for 24 h prior to exposing them to ACR (6 mM). Pretreatment with EGCG for 24 h attenuated ACR-induced cytotoxicity in a concentration-dependent manner; the effects were significant at 5 and 10 μM EGCG (p < 0.05). Pretreatment with 2.5 μM EGCG had no significant effect, and the effect observed with 20 μM EGCG was similar to 10 μM EGCG (Figure 1(c)). Therefore, 2.5, 5 and 10 μM of EGCG were used in subsequent experiments to attenuate the effects of 6 mM ACR in PC12 cells.
Effect of EGCG on AChE activity
As illustrated in Figure 2, 6 mM ACR alone significantly decreased AChE activity, whereas pretreatment with 5 and 10 μM EGCG significantly increased AChE activity in ACR-exposed cells. Pretreatment with 2.5 μM EGCG resulted in no obvious effects on AChE activity. The cells treated with EGCG alone also showed slightly increased AChE activity compared with the cells without ACR exposure.

Effect of EGCG on AChE activity of PC12 cells exposed to ACR data represented as (percentage of control) mean ± SE of three separate experiments. *p < 0.05 compared to control cells. # p < 0.05 compared to ACR-treated cells. EGCG: epigallocatechin-3-gallate; AChE: acetylcholinesterase; ACR: acrylamide.
Effects of EGCG on apoptosis
Annexin V-FITC/PI assay
Treatment with ACR (6 mM) alone significantly increased the percentage of apoptotic cells and necrotic cells compared to control cells. In addition, 10 μM EGCG alone did not obviously affect the apoptosis and necrosis of PC12 cells in this model. Pretreatment with 5 μM and 10 μM EGCG significantly decreased the percentage of ACR-induced apoptotic and necrotic cells; pretreatment with 2.5 μM EGCG had no obvious effects (Figure 3(a) and (b)).

Effect of EGCG on ACR-induced apoptosis and necrosis of PC12 cells: (a) flow cytometry scatter diagram obtained via Annexin V-FITC and PI tests of each sample. (b) Column bar graph of the percentage of apoptotic and necrosis cells. In a flow cytometry scatter diagram, cells in Q3 that were Annexin V (−) and PI (−) were living cells, those in Q4 that were Annexin V (+) and PI (−) were early apoptotic cells, and those in Q2 that were Annexin V (+) and PI (+) were late apoptotic cells or necrotic cells. Data represented as mean ± SE of three separate experiments. *p < 0.05 compared to control cells. # p < 0.05 compared to ACR-treated cells. EGCG: epigallocatechin-3-gallate; ACR: acrylamide.
MMP assay
Disruption of MMP is a key step in the mitochondrial apoptotic pathway and is considered an important indicator of cell health or injury. MMP was evaluated using the rhodamine 123 assay to compare mitochondrial function between treatments. ACR significantly reduced MMP after 24-h exposure compared with control cells. The ACR-treated cells pretreated with 5 μM and 10 μM EGCG had significantly increased MMP compared to the cells treated with ACR alone; the cells pretreated with 2.5 μM EGCG had no significant effects (Figure 4(a) and (b)).
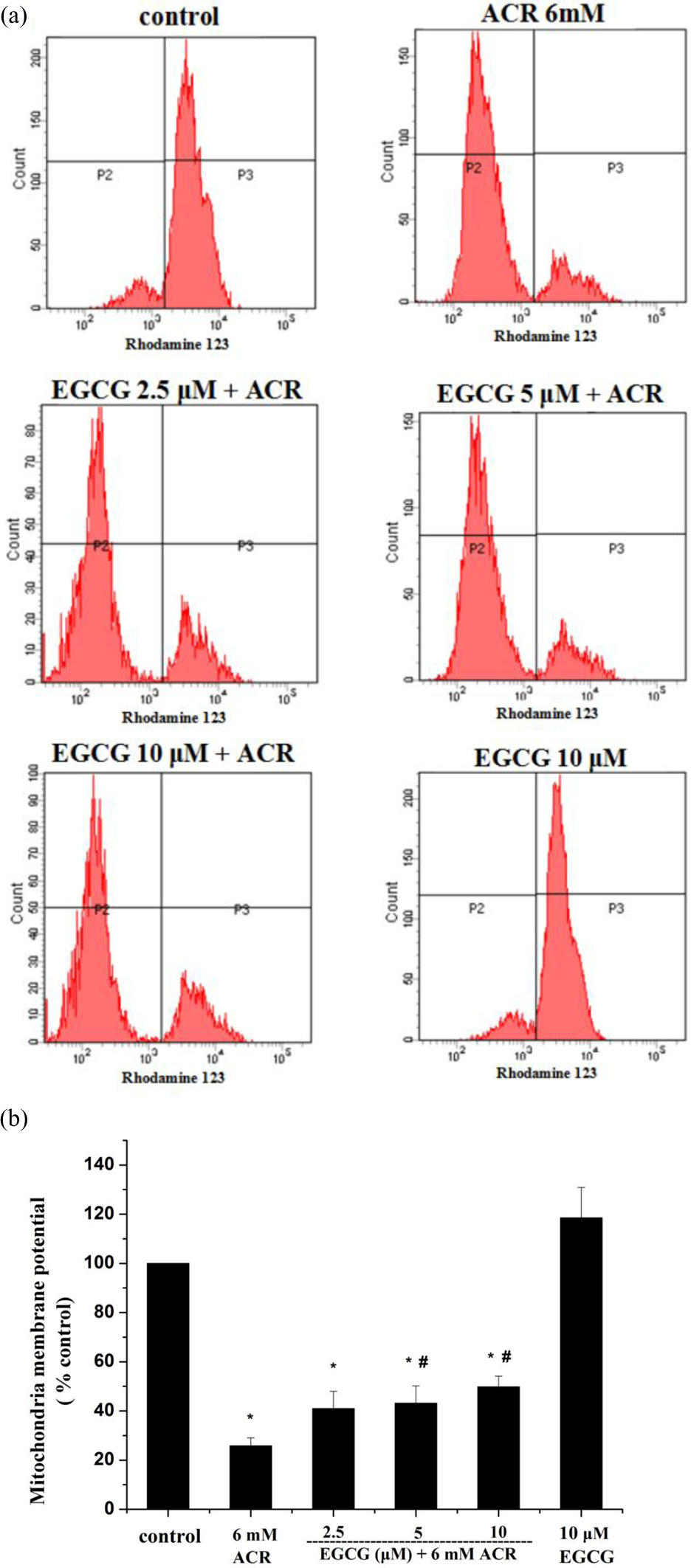
Effect of EGCG on the MMP in ACR-treated PC12 cells: (a) flow cytometry histograms of each sample. (b) Column bar graph of mean cell florescence for rhodamine 123. Data represented as (percentage of control) mean ± SE of three separate experiments. *p < 0.05 compared to control cells. # p < 0.05 compared to ACR-treated cells. EGCG: epigallocatechin-3-gallate; MMP: mitochondrial membrane potential; ACR: acrylamide.
Intracellular calcium assay
The apoptotic process can be triggered by an increase in intracellular calcium in numerous cell types. Sustained elevations in intracellular calcium disrupt mitochondrial function, which subsequently damages membranes, organelles and chromatin, and eventually causes cell death. 34 Intracellular calcium levels were significantly increased in PC12 cells exposed to ACR. In contrast, no significant changes were observed in intracellular calcium levels in cells treated with EGCG alone compared to control cells. The ACR-treated cells pretreated with 5 μM and 10 μM EGCG had significantly decreased intracellular calcium levels compared to the cells exposed to ACR alone; pretreatment with 2.5 μM EGCG did not significantly affect intracellular calcium levels (Figure 5(a) and (b)).

Effect of EGCG on the ACR-induced increase in intracellular calcium of PC12 cells: (a) flow cytometry histograms of each sample. (b) Column bar graph of mean cell florescence for FLUO-3AM. Data represented as (percentage of control) mean ± SE of three separate experiments. *p < 0.05 compared to control cells. # p < 0.05 compared to ACR-treated cell. EGCG: epigallocatechin-3-gallate; ACR: acrylamide.
Cytochrome c assay
Release of cytochrome c from the mitochondrial inner membrane into the cytoplasm is critical for apoptotic cell death in many cell types. 35 Cytochrome c release was significantly increased in PC12 cells exposed to ACR compared to control cells. No significant changes in cytochrome c release were observed in cells treated with EGCG alone. The ACR-treated cells pretreated with 5 μM and 10 μM EGCG had a significant decrease in the release of cytochrome c compared to the cells exposed to ACR alone; pretreatment with 2.5 μM EGCG did not significantly affect cytochrome c release (Figure 6). The decrease in cytochrome c release from the ACR-treated cells pretreated with EGCG confirms the inhibitory effects of EGCG against ACR-induced apoptosis.

Effect of EGCG on the release of cytochrome c induced by ACR in PC12 cells. Data represented as (percentage of control) mean ± SE of three separate experiments. *p < 0.05 compared to control cells. # p < 0.05 compared to ACR-treated cells. EGCG: epigallocatechin-3-gallate; ACR: acrylamide.
Caspase 3 activity assay
Caspase 3 activity plays a regulatory role in apoptosis and can be activated during the early stages. Treatment with ACR alone significantly increased caspase 3 activity in PC12 cells compared with control cells. The ACR-treated cells pretreated with 5 μM and 10 μM EGCG had a significant decrease in caspase 3 activity compared to the cells exposed to ACR alone; pretreatment with 2.5 μM EGCG did not significantly affect caspase 3 activity (Figure 7).
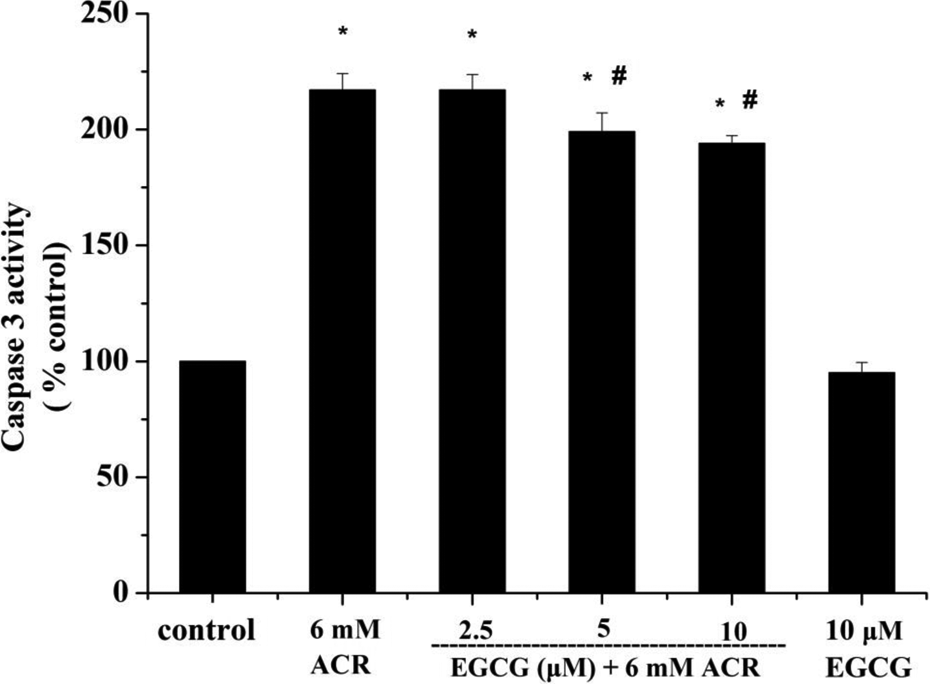
Effect of EGCG on the caspase 3 activity induced by ACR in PC12 cells. Data represented as (percentage of control) mean ± SE of three separate experiments. *p < 0.05 compared to control cells. # p < 0.05 compared to ACR-treated cells. EGCG: epigallocatechin-3-gallate; ACR: acrylamide.
Bax and bcl-2 mRNA expression
Relative bax mRNA expression significantly increased in ACR-treated PC12 cells. In contrast, bcl-2 mRNA expression significantly decreased in PC12 cells treated with ACR. In ACR-treated cells pretreated with 5 μM EGCG, a significant decrease in the relative expression of bax mRNA was observed compared to cells treated with ACR alone. The relative expression of bcl-2 mRNA did not significantly change with EGCG pretreatment. The relative expression of bax and bcl-2 mRNA was not significantly changed in cells treated with 5 μM EGCG (Figure 8(a)). The bcl-2/bax ratio is also considered as an indicator that reflects the apoptosis status of cells. We also analysed the relative expression ratio of bcl-2/bax mRNA in each group. The relative expression ratio of bcl-2/bax mRNA in cells treated with ACR alone was significantly decreased compared to the control group. Compared to the ACR-treated cells, the relative expression ratio of bcl-2/bax mRNA in cells pretreated with EGCG significantly increased (Figure 8(b)).

Effect of EGCG on the relative expression of bax mRNA and bcl-2 mRNA of PC12 cells exposed to ACR. (a) The relative expression of bax mRNA and bcl-2 mRNA of PC12 cells. (b) The relative expression ratio of bcl-2/bax mRNA in PC12 cells. Data represented as mean ± SE of three separate experiments. *p < 0.05 compared to control cells. # p < 0.05 compared to ACR-treated cells. EGCG: epigallocatechin-3-gallate; ACR: acrylamide.
Effects of EGCG on oxidative stress
Common approaches for evaluating oxidative stress include monitoring ROS production and ROS-induced modifications of cellular constituents (i.e. MDA, the production of lipid peroxides in cells). 36 In this study, we measured ROS, MDA, SOD and GSH.
A significant increase in ROS and MDA was observed in PC12 cells exposed to ACR (p < 0.05). In contrast, the cells pretreated with EGCG had significantly decreased levels of ROS and MDA compared to the cells exposed to ACR alone; no significant changes were observed in ROS and MDA of cells treated with EGCG alone compared to the control cells (Figure 9(a) to (c)). To compare the antioxidant capacity of cells, we measured SOD activity and GSH content. In cells treated with ACR alone, SOD activity and GSH levels were significantly decreased compared to the control cells. In contrast, the ACR-treated cells pretreated with EGCG exhibited significantly increased SOD activity and GSH levels compared with cells treated with ACR alone (Figure 9(c)). These results suggest that ACR increases oxidative stress in PC12 cells as indicated by increased ROS and MDA along with decreased GSH content and SOD activity; pretreatment with EGCG reduced ACR-induced oxidative stress in PC12 cells.

Effect of EGCG on ACR-induced oxidative stress in PC12 cells: (a) and (b) ROS production ((a) flow cytometry histograms of each sample and (b) column bar graph of median cell florescence for DCF). (c) SOD activity and MDA and GSH levels. Data represented as mean ± SE of three separate experiments. *p < 0.05 compared to control cells. # p < 0.05 compared to ACR-treated cells. EGCG: epigallocatechin-3-gallate; ACR: acrylamide; ROS: reactive oxygen species; DCF: dichlorofluorescein; SOD; superoxide dismutase, MDA: malondialdehyde; GSH: glutathione.
Discussion
In this study, we explored the neuroprotective effects of EGCG on PC12 cells exposed to ACR by measuring markers of apoptosis with positive results.
EGCG increased the survival rate of PC12 cells exposed to ACR and increased AChE activity, which may play an important role in the mechanisms underlying the neuroprotective effects of EGCG. Decreased viability in ACR-treated PC12 cells has been previously reported 13,37 ; however, there is a lack of information available regarding the effects of ACR on AChE in neurons. Recently, it was shown that ACR decreased AChE activity in the brains of mice by mechanisms that likely involve oxidative stress. 38 AChE catalyses the breakdown of acetylcholine, which is important in a variety of brain functions, such as neurotransmission, reward, learning, memory formation and neuronal development. 39 Inhibition of AChE by ACR has significant implications for brain functions. Furthermore, in the present study, we observed the protective effect of EGCG against ACR-induced nerve damage, which has significant implications for the study of public health issue.
Here, we performed a series of assays to investigate the role of apoptosis in the neuroprotective effects of EGCG against ACR. Externalized phosphatidylserine on the surface of nucleated cells is an important marker of apoptosis. To determine phosphatidylserine content, we measured annexin binding by flow cytometry. 40 Mitochondria are vital organelles for the generation of cellular energy and play an important role in the intrinsic apoptotic pathway. 41 Mitochondrial dysfunction leads to the loss of MMP and release of calcium and cytochrome c from the mitochondria into the cytoplasm, eventually causing cell death and apoptosis 42,43 (Figure 10). Caspase 3 is a key protease that is activated during the early stages of apoptosis and mediates cell death 44 ; therefore, we evaluated mitochondrial functions and caspase 3 activity. Our study showed that ACR increases the percentage of necrotic and apoptotic cells, intracellular calcium levels, release of cytochrome c and caspase 3 activity while decreasing MMP. These data suggest that ACR induces apoptosis in PC12 cells, which is consistent with the previous report. 13 EGCG decreased the percentage of necrotic and apoptotic cells and maintained mitochondrial dysfunction via increasing MMP and decreasing intracellular calcium and cytochrome levels and caspase 3 activity in this study, suggesting anti-apoptotic effects attributable to EGCG.
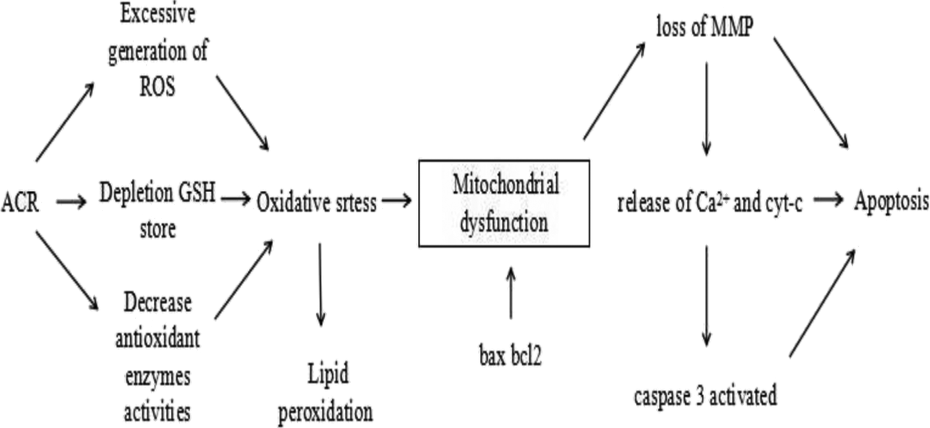
A schematic of the timeline of the different physiological changes in the cells exposed to ACR. ACR induces oxidative stress that causes mitochondrial dysfunction and finally leads to apoptosis. ACR: acrylamide.
In mammals, the mitochondrial apoptosis pathway is controlled by the bcl-2 family of proteins, which are divided into pro- and anti-apoptotic proteins based on structure and function (Figure 10). Change in the balance between pro-apoptotic bax mRNA and anti-apoptotic bcl-2 mRNA leads to apoptosis. 45,46 In the present study, treatment with ACR alone significantly decreased the relative expression ratio of bcl-2/bax mRNA, resulting in apoptosis. However, pretreatment with EGCG significantly increased the relative expression ratio of bcl-2/bax mRNA in PC12 cells exposed to ACR (p < 0.05). These data suggest that EGCG attenuates the pro-apoptotic effects of ACR in PC12 cells.
Oxidative stress or an imbalance between pro-oxidants and antioxidants, resulting from increased ROS or reduced antioxidant scavenging ability, is closely linked to apoptosis. 31,43 Mitochondria are a major source of intracellular ROS; however, excessive generation of ROS affects the mitochondrial function, leading to the loss of MMP, cytochrome c release, intracellular calcium overload and ultimately apoptosis 43,47 (Figure 10). ACR induces oxidative stress primarily by depleting cellular GSH stores. In addition, the results of this study show that ACR increased ROS and MDA production while decreasing antioxidant SOD activity and GSH levels. These data confirmed the induction of oxidative stress by ACR. The antioxidant properties of EGCG are attributed to two triphenolic groups within its structure. EGCG prevents oxidative stress against ACR, including decreased ROS and lipid peroxidation and increased GSH levels and SOD activity in PC12 cells in this study.
The neuroprotective effects of EGCG are attributed to its antioxidant and anti-apoptotic properties in both cell and animal models. 15,17 EGCG prevents mitochondrial damage against hydrogen peroxide in PC12 cells. 23 EGCG provides a neuroprotective role against α-amino-3-hydroxy-5-methyl-4-isoxazolo and subarachnoid haemorrhage by attenuating ROS production. 48,49 Our study showed that EGCG protects PC12 cells against ACR-induced oxidative stress and apoptosis, which also confirms that antioxidant and anti-apoptotic properties are the neuroprotective mechanisms of EGCG.
In summary, the results obtained in the present study indicate that ACR induces PC12 cell damage, including decreased viability and AChE activity and increased apoptosis and oxidative stress, and EGCG attenuates ACR-induced neurotoxicity in PC12 cells. The mechanism of EGCG against ACR includes maintaining mitochondrial function and regulating the expression of bax and bcl-2 mRNA and redox state. Thus, the protective effects of EGCG are partly due to suppression of apoptosis and prevention of oxidative stress, suggesting that drinking green tea may reduce nerve damage induced by ACR.
Footnotes
Author Contribution
YH and DT are co-first authors and they have contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the grants of National Natural Science Foundation of China (31570687).
