Abstract
Background
There are hereditary types of nephroblastoma or Wilms’ tumor associated with exposure of the germ cells of either parent to harmful environmental factors. Some studies have examined the exposure of compounds used pesticides and herbicides as a risk factor for Wilms’ tumor.
Methods
A systematic review and meta-analysis were carried out on case-control studies to establish the potential link between exposure to these organic molecules and Wilms’ tumor occurrence in children rigorously. We examined the monographs on some organo-phosphate insecticides and herbicides issued by the International Association for the Research on Cancer (IARC) under the auspices of the World Health Organization (WHO). PUBMED, SCOPUS, and Google Scholar studies (1960–2021) were identified and systematically reviewed following Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) guidelines. Subgroup analyses were conducted after stratification for occupational versus residential exposure and before birth (prenatal) vs. after birth (postnatal) exposure. In addition, we revised the monographs on chemical compounds issued recently by the IARC/WHO.
Results
Our findings seem to consolidate that parental pesticide exposure during the preconception or pregnancy period is correlated with an increased occurrence risk for Wilms’ tumor. We confirm the validity of the WHO essays on certain organophosphate herbicides and insecticides, which support these compounds, may be highly relevant in future cancer prevention policies.
Conclusion
Parental exposure to pesticides, particularly in household settings, is poorly emphasized in our society. There is a strong association between these organophosphate compounds and pediatric cancer. Public health agencies may need to take stronger action than in the past.

Introduction
Nephroblastoma harboring the eponym of Wilms’ tumor is considered the most common renal tumor of childhood.1,2 This tumor originates from stem cells localized in the mesoderm or nephrogenic rests that give rise to the development of the renal parenchyma. It is classified as an “embryonal” malignant neoplasm.1,3,4 Wilms’ tumor is usually observed in the second infancy (between 2 and 4 years of age). Most cases are typically diagnosed before the age of 5. 5 This tumor may present as unilateral, bilateral, unicentric, or multicentric, and normal or abnormal karyotype. 3 Additionally, it can be combined with various congenital malformations. They include sporadic aniridia, intellectual disability, excessive growth (hypertrophy) of only one side of the body (hemi) or hemihypertrophy, microcephaly, neural tube defect of lower type or spina bifida (“split spine”), trisomy 18 (Edwards) syndrome, and trisomy 21 (Down) syndrome. In addition, familial cases of Wilms’ tumor have been observed and comprise about 1% of all cases. 6
Initially, Wilms’ tumor has been considered to harbor a constant frequency worldwide. 7 However, this tumor occurs with a variation identified at different rates internationally. Incidence variation related to ethnic groups in the same geographic area has also suggested a potential etiologic role of the genetic predisposition.8,9 Wilms’ tumor may occur due to two consecutive genomic (mutational) events that consist of a germinal genetic mutation either with one somatic genetic mutation or, variably, two somatic genetic mutations. 10 This unfolds the critical fact that Wilms’ tumor is dual in origin. It can be presented both in hereditary and somatic types. While genetic conditions of Wilms' tumor are thought to relate to exposures of the germ cells of either parent at preconception period, it is believed that sporadic tumors may be related to vulnerability to carcinogenic molecules during pregnancy.11–14
One of the environmental factors that have been demonstrated to be correlated with Wilms' tumor is pesticide exposure. 15 Although pesticides help protect vegetables and fruit from disease-bearing insects and pests, they are likely to bring about Wilms' tumor among children. Several studies and systematic reviews have investigated the link between childhood cancers and pesticide exposure.4,16–19 Pesticide use is frequently associated with agriculture. Children living on or near treated farmland may primarily be affected. 15 However, in an urban setting, pesticides and herbicides are used for lawn in backyard care as well as private and public gardens. It may not be uncommon that children can potentially get exposed to them. The factors that increase such exposure include excessive or inadequate application and failure to wash pesticide-treated bedding. Children may get exposed to herbicides and pesticides either breathing in settings using such compounds or eating fruit and vegetables exposed to these compounds. 20
Among these studies, the only meta-analysis examined the various risk factors associated with Wilms’ tumor.
21
There remains, however, a lack of a distinct meta-analysis and review of studies that account for the varying methodologies, cohorts (e.g. child exposed, pregnant parent, and working parent exposed), and exposure conditions (e.g. occupational exposure, agricultural exposure, residential exposure) to examine Wilms’ tumor and its association with pesticide use. In addition, since the inclusion of different study designs tackling the same research question may cause some degree of inconsistency in the results,
22
this meta-analysis includes only case-control studies to yield more definitive results. Due to the COVID-19 outbreak, specific preventive measures have been implemented by countries across the globe to control the outbreak.
23
The health guidelines released by WHO due to the outbreak of the COVID-19 pandemic due to SARS-CoV-2 have been adopted around the world.
23
One of the standard policies of all nations was to announce a general quarantine following the slogan of ‘stay at home’ to cut the SARS-CoV-2 transmission chain from person to person.24–28 With its advantages over-controlling COVID-19, this policy carries individual and environmental risks from the outdoor environment to homes and indoor environments. People may be exposed to increased disinfectants and pesticides used at home.
4
We propose that ‘stay at home’ may also cause increased exposure to pesticides in addition to disinfectants. Moreover, we investigate the potential role of exposure to occupational pesticides, parental exposure to household pesticides, and children exposure before birth and after birth in Wilms’ tumor using a PRISMA-based25,26 systematic review and meta-analysis including one (case-control) study design.
Material and methods
Study selection method and criteria
A search on PUBMED, SCOPUS, and Google Scholar was performed targeting the literature published between January 1960 to December 2021, spanning over 60 years. At first, an electronic search was done using “(pesticides OR herbicides OR fungicides OR insecticides)” AND “((children OR childhood) and Wilms’ tumor)” AND “(occupation OR occupational)” with different groupings of the words “pesticide(s)”, “child”, “Wilms’ tumor”, and “parental”. We also used the words “children”, “childhood”, “infant”, “newborn”, “preschool child”, “adolescent”, “youth”, “teenager”, “young adult”, “tumors”, “Wilms’ tumor”, “nephroblastoma”, “occupational”, “farmers”, “agriculture”, “horticulture”, “pesticide applicators”, “residential”, “household”.
The following studies were excluded: abstracts, letters, opinions, meta-analysis, case reports, reviews, and studies tackling adults that we predefined as individuals with age more than 24 years or combining children and adults with no separate results.
The tight collaboration between a British and a Canadian institution was key in developing the Newcastle-Ottawa quality assessment scale targeting case-control studies. 29 This scale was applied to each study individually. Previously, we have established that quality assessment of studies included in the meta-analysis is a substantial vital component for an evidence base methodology for observational studies. 30 We considered studies with a NOS score of six and above as investigations harboring a good quality score, while studies with a NOS score of less than six were deemed displaying a poor-quality score. Among various evaluation systems (ad hoc, CONSORT, JADAD, Delphi, and Cochrane Collaboration), we decided for the “ad hoc” method. Baseline features between groups, detection of allotment concealment, and dropout levels were used to evaluate the study quality. All included investigations were judged of satisfactory quality to be elements of this systematic review and meta-analysis.
The next step involved the verification of the reference lists of the published papers to detect any additional studies. The quest was limited to investigations issued in the English language in the open access and subscription-based (non-open access) scientific literature. Finally, all titles and abstracts were assessed to ascertain the suitability of the publications. Figure 1 offers details of the inclusion and exclusion criteria showing the steps to decide on the articles for the study. Figure 1 is adapted from a flow diagram, which has essentially been proposed as standard by the PRISMA statements.25,26 Steps followed in selecting articles for the study.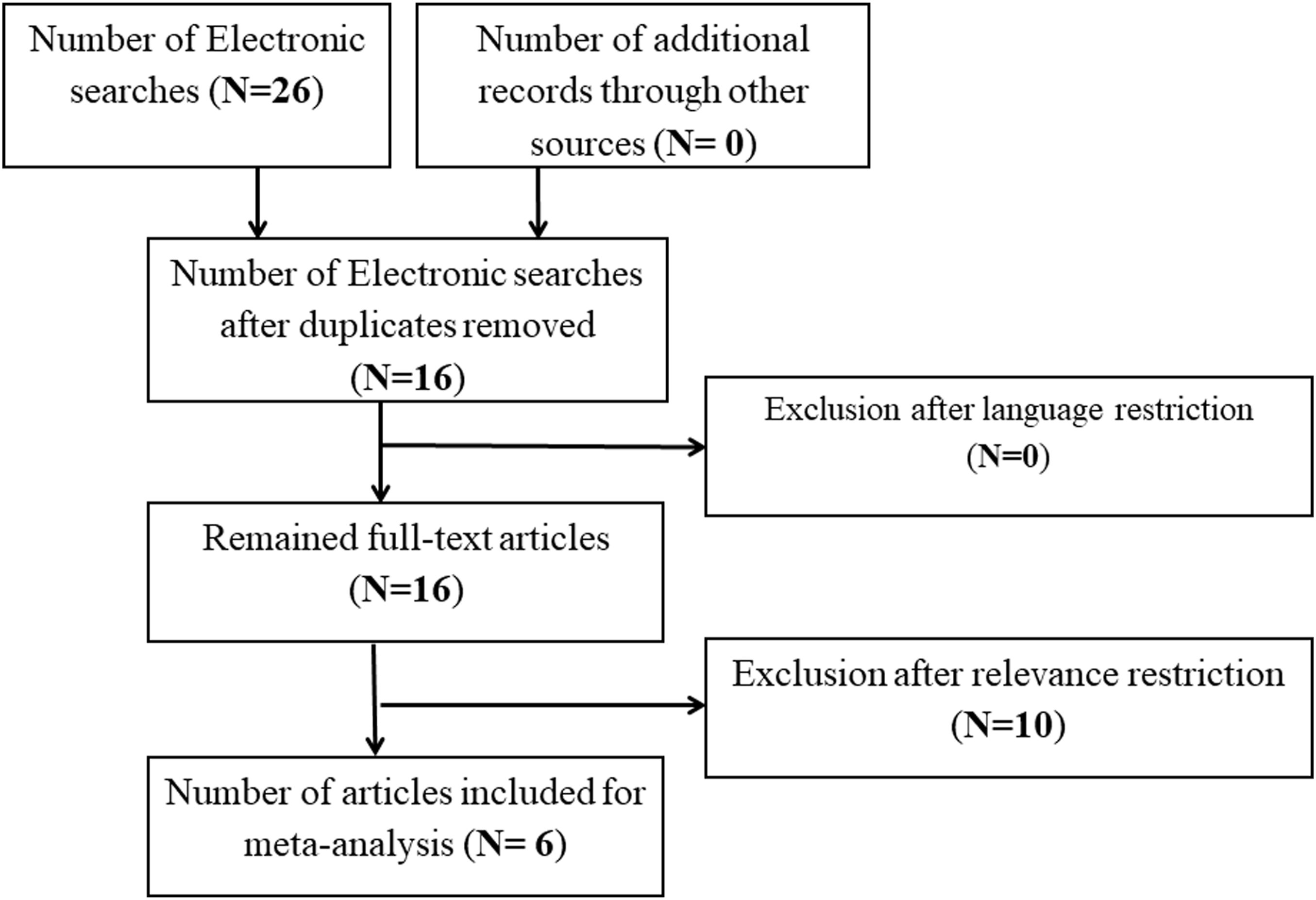
After the studies have been selected, all three authors went through the abstracts and studies to peruse the study design, site of the study, number of individuals participant to the study, source of chemical exposure, the timing of chemical exposure, and the study outcome. The authors then assembled a list of variables thought to play a critical role in clarifying the relationship between parental exposure to herbicides and pesticides and pediatric neoplasm.31,32 Table 1 shows studies that were selected for the meta-analysis. We considered a study suitable to be the part of our meta-analysis if it: (1) Mentioned only children and young adults (up to 24 years) exposed to herbicides and pesticides from parental occupational exposure (e.g. farmers, non-farmers agricultural workers, pesticide applicators, greenhouse workers), (2) Debated only the link between Wilms’ tumor and organophosphate compounds and not as a part of various studies, and (3) Included case-control investigations. Characteristics of studies included in the meta-analysis.
Case-control studies were included in the meta-analysis in analogy to another study targeting neuroblastoma and pesticide exposure published in this journal, 33 and pediatric brain tumor and pesticide exposure published in another journal. 34 The following study designs were excluded: cohort studies, reviews, abstracts, opinions, case reports, letters, meta-analysis, and pool analysis. Finally, we revised 114 monographs of the IARC/WHO collections, which are available on the internet. We concentrated on insecticides and pesticides and organophosphate insecticides and pesticides.
Statistical methods
Relevant data obtained from the appropriate articles were then enrolled into the Comprehensive Meta-Analysis (CMA) software. 35 These variables included specifically the sample size, odds ratio (OR), and the confidence intervals (CI) (lower and upper limit). In a few instances, we performed preliminary calculations, as the studies directly provided not all relevant variables.
The pooled values of the pertinent variables were computed using an inverse variance weighting method. All the studies’ results included in the software followed the same measurement scale.35,36 The 95% CI around the pooled effect size was gained from the pooled variance. We decided for the fixed effects model to combine effect sizes inside each study, as we expected random variations due to sampling errors. We preferred a random-effects model. We used it to merge effect sizes across various studies since we accepted more between-study variability as stated previously. 36
We went further to address the issues of heterogeneity. We applied I-square (I2) index to determine the extent of heterogeneity.36,37 The I2 stands for the fraction of total variation across investigations due to heterogeneity rather than pure chance. I2 was computed as follows:
In the above equation, Q is Cochran’s heterogeneity statistic and df the degree of freedom.38,39 It has been determined that I2 values vary from 0% and 100%, while values lower than zero are turned into zero. If I2 amount to 0%, it indicates no observed heterogeneity. On the contrary, a larger value shows increasing heterogeneity.31,32
The next step involved the perusal of the selected studies for publication bias. Thus, we charted the estimate of the natural logarithm (Ln or Loge) of OR versus the standard error (SE). Natural logarithms are logarithms to the base of the constant e, an irrational and transcendental number. Finally, funnel plots of asymmetry were calculated using Egger’s regression asymmetry test and Begg’s modified funnel plot.31,40,41
Results
Four categories defined by exposure were identified, namely (1) pre-partum or before birth, (2) post-partum or after birth, (3) occupational, and (4) residential exposures, to compare the association of Wilms' tumor with pesticide exposure. In addition, we ran a meta-analysis. This task included only the case-control studies as stated above. Our results showed significant proof against the null hypothesis that Wilms’ tumor occurrence was similar between exposed (“treatment”) and control groups (OR = 2.1; CI = 1.2–3.6; p-value = .006). Moreover, from the forest plots in Figure 2(a), the I2 statistic reveals that the rate of between-study heterogeneity attributable to variability in the proper treatment effect amounted to 57.86%. Forest plots displaying a meta-analysis of the effect of pesticides on Wilms' tumor in children during mother’s Pregnancy/Preconception (A = Before birth; B = After Birth; C = Occupational Exposure; D = Residential Exposure). The size of each box indicates the relative weight of each study in the meta-analysis, while the horizontal bars reflect the 95% confidence interval.
The second subgroup study addressed the effect of pesticide exposure just after birth. Although only two studies were included, it is worth pointing out that one of the researches, i.e. Schuez et al. (2001), 38 involved three relevant case-control experiments, which were summarized to obtain study-specific odds ratios. The meta-analysis results showed the odds ratio of 0.9 (CI = 0.7–1.2; p-value = .611). Following Figure 2(b), the null hypothesis that the exposure effect was similar between exposure and control at birth should not be rejected. There is a seeming absence of between-study heterogeneity, as attested by an insignificant Q-value and null value of I 2 .
Another exposure category we considered was occupational exposure, whether it is more prone to be associated with Wilms' tumor. Figure 2(c) indicates that the summary effect size for “occupational” exposure was not significant. We arrived at the OR of 1.6 (CI = 0.7–3.8; p-value = .249). Hence, pesticides due to “occupational” exposure were not connected with Wilms’ tumor. The significant Q-value points to the presence of heterogeneity. The I 2 statistic revealed a high percentage of between-study heterogeneity (77.17%).
The last aspect that we explored through our studies examined whether exposure through home/residence played an important role in Wilms' tumor etiology. Four studies were involved in the analysis. We found an OR of 1.4 (CI = 1.2–1.7; p-value < .0001). Pesticides or herbicides in a household setting was highly correlated with the occurrence of Wilms' tumor, as it is further illustrated in the forest plot (Figure 2(d)). The Q-value (p = .486) indicates the lack of heterogeneity (I 2 = 0.00).
Testing for publication bias
Visual inspection
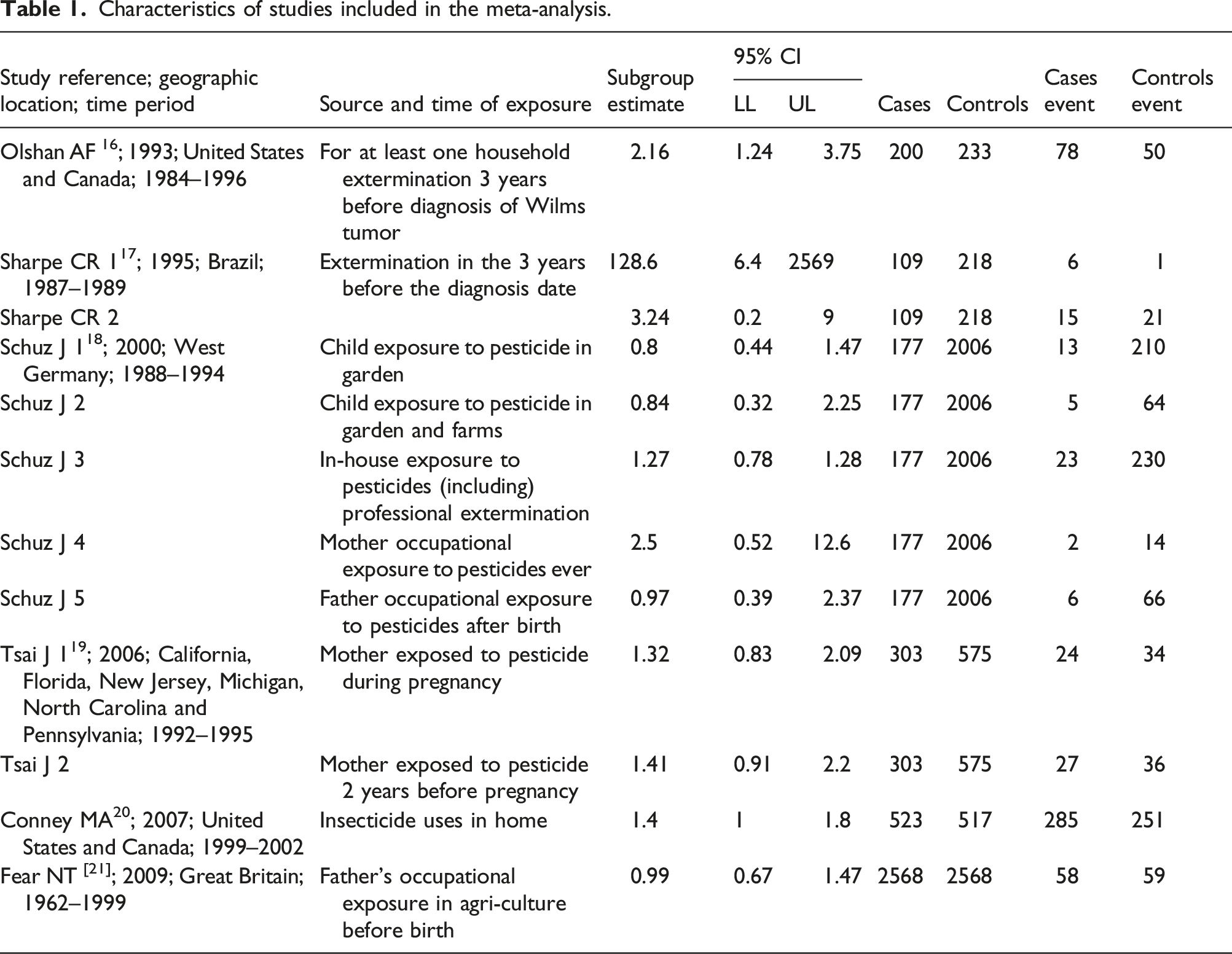
The four subcategories defined by exposure were inspected for publication bias using funnel plots in Figure 3 (A through D). The before-birth exposure case’s funnel plot (Figure 3(a)) looks asymmetrical, which eventually indicates a publication bias. It may also reflect either the low quality of trials causing far too high treatment effect estimates or less methodological rigor in the quality of some investigations.
42
The asymmetrical look of the funnel plotting foreshadows publication bias, experimental issues and/or methodological gaps. The residential exposure case’s funnel plot (Figure 3(d)) appears asymmetrical. It includes studies of average to moderately high SEs (i.e. low precision) and high OR, hence with the possibility of publication bias. Additional testing will be necessary. Funnel plot of standard error showing the effect of pesticides on Wilms' tumor in children during mother’s Pregnancy/Preconception (A = Before birth; B = After Birth; C = Occupational Exposure; D = Residential Exposure).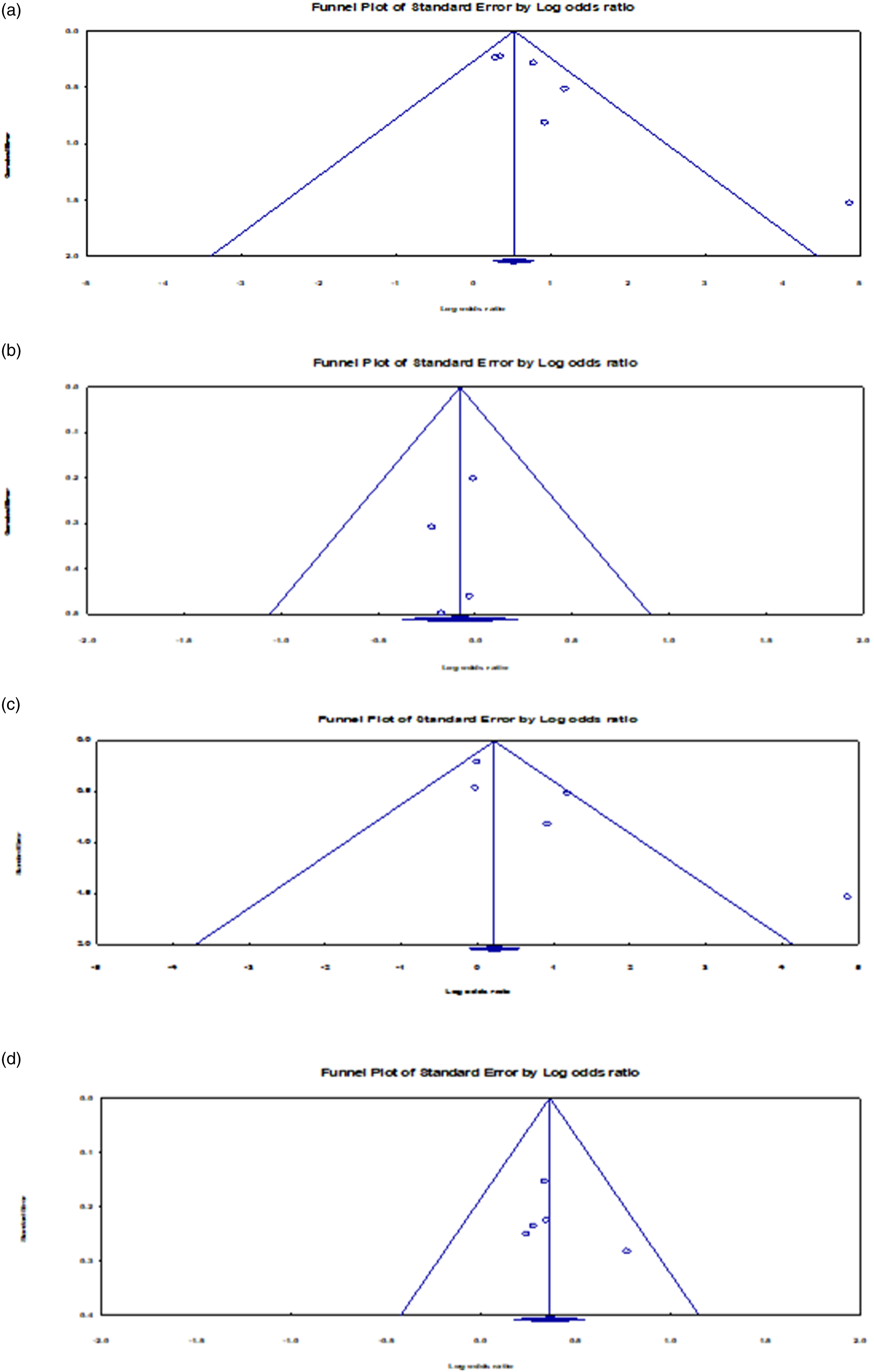
The funnel plots for the after-birth exposure case (Figure 3(b)) and the occupational exposure case (Figure 3(c)) show summary log odds ratios that require attention. For the former, the summary effect fell below the point of no effect (i.e. log OR = 0 or OR = 1). For the latter, although the summary effect fell above the threshold of no effect, it was nonetheless insignificant. No further test of the publication bias for these two categories was initiated.
Test for publication bias
We statistically tested and assessed the impact of publication bias, considering the case-control experiments within each pesticide exposure category. Under the before birth exposure case, the Egger test revealed the presence of a bias. The intercept (B0) was 2.582, 95% CI (0.532, 4.633), the 1-tailed p-value was .013. We observed a net difference of 0.448 between the imputed point estimate and the original combined point estimate. Figure 4 shows both point estimates. In considering the random-effects model, the point estimate and, of course, 95% CI for the blended studies was 2.006 (CI: 1.231, 3.271). The imputed point estimate instead amounted to 1.558 (0.917, 2.648) using the Duval and Tweedie’s Trim and Fill technique (Figure 4(a)).43,44 Thus, the adjusted point suggested a much lower risk than the original estimate. The difference between both estimates (ORs of 2.01 vs 1.56) suggests a bias of likely modest impact. That is, the inclusion of all meaningful studies might shift the effect size only modestly, without altering the major finding that exposure before birth to pesticides is a risk factor to Wilms’ tumor. Funnel plot with imputed studies (A = Before birth exposure; B = Residential exposure).
Under residential exposure case, the Egger test of the intercept revealed to be insignificant: B0 = 1.259, 95% CI (−4.122, 6.640), 1-tailed p-value = .255. We observed no difference between the imputed point estimate and the original combined point estimate, as shown in Figure 4(b) with both point estimates. In fact, the Duval and Tweedie’s Trim and Fill data suggest that, irrespective of the model taken (fixed effects vs. random effects), the point estimate for the merged studies revealed a value of 1.439, 95% CI (1.192, 1.736) would remain unchanged.
Discussion
The above results show a link between parental exposure to pesticides/herbicides and Wilms’ tumor, in the event of exposure before birth. However, after-birth such exposure does not seem to be significantly associated with the onset of this pediatric renal tumor. Additionally, parental exposure to pesticides/herbicides in household settings may need closer attention in the future. These results presented across subgroups of case-control experiments are robust across studies after computing the relevant summary effects. With regard to the poor association with Wilms’ tumor regarding pesticides due to occupational exposure, a reduced number of investigations may be reported as responsible for this difference in results. Therefore, we should pledge to have more investigative studies in the future to elucidate the role of pesticide exposure due to occupational exposure in the etiology of Wilms’ tumor worldwide across both low-/medium-income countries and high-income countries, despite some scientists support the controversial thesis that additional studies could not contribute to a definitive statement regarding this pediatric renal tumor due to limited data regarding exposure. 45
There are very few explicitly done studies about Wilms' tumor risk related to pesticide exposure. One possible reason is the lack of research in this area. Pediatric renal tumors are rare, but not an insignificant portion of pediatric neoplasms being strongly associated with abnormal blastogenetic processes.3,46 It has been suggested that the risk of developing Wilms' tumor increases with childhood insecticide exposures. 16 In addition, parental pesticide/herbicide exposures before birth (pre-partum) increase the risk of developing Wilms’ tumors.16,17 Another important point is the limited expertise on pediatric renal tumors comparing with other more studied pediatric tumors (e.g. leukemia, neuroblastoma).
In perusing the literature, we found that another meta-analysis that investigated the perinatal and environmental risk factors for Wilms’ tumor, reported an increased risk of Wilms' tumor associated with maternal pesticide exposure (OR = 1.37 CI = 1.09, 1.73). However, some evidence of publication bias was observed and needs to be emphasized. A link appears to exist between pesticide exposure and the etiology of this tumor. 4 Maternal contact to pesticides has been revealed to be strongly associated with this pediatric small round blue cell tumor. Since the maternal versus paternal exposure subgroup has already been discussed, we aimed to study the residential versus occupational and before-birth versus after-birth sub-groups. Our meta-analysis studies identified that pesticide exposure during the preconception and pregnancy period might contribute to Wilms' tumor development. A recent French population-based study also exposed an association between Wilms’ tumor and maternal exposure to chemicals during pregnancy confirming our results. 47
Similarly, our study did not support the positive association between parental occupational contact to pesticides and augmented Wilms’ tumor risk. This data contrasts with the meta-analyses performed in the past, which elucidated that Wilms’ tumor has been linked with occupational contact of both parents to pesticides and/or herbicides. 17 We consider that it is important to educate parents to take proper precautions while using pesticides or insecticides during pregnancy and preconception.
Even though the specificity of pesticides in the etiology of pediatric cancer has been postulated, the complex chemical nature of pesticide formulations, variation in the source, and the timing of exposure makes it difficult to understand its role in the process of Wilms’ tumor development fully. In addition, the limited data information about pesticides can be a problem as there are only rare studies that distinguished between herbicides, fungicides, insecticides, or other types of pesticides. They are not always mutually exclusive categories. It is conceivable that differences in the chemical composition of different compounds, application methodologies, differences in timing as well as the source of exposure could cause the different studies to differ in the final results. 18 Also, carcinogenicity could result from various specifically immunotoxin-related molecules or deliberately mutagenic properties of pesticides, impairing various biological processes. Furthermore, to complicate the pediatric tumorigenesis, neoplasms in childhood have been regarded as multi-step diseases with possibly multiple biological pathways. 20
It should be noted that for some of the above indicated reasons and the following arguments, the findings of our study should be interpreted cautiously and contain some limitations. Firstly, meticulous consideration is required while examining deductions of case-control studies because of the chance of methodological shortcomings. For example, contact misclassification, insufficient size of samples, biases in selecting controls, and confounding factors are some of the well-known limitations associated with case-control studies. On the other hand, it can also be challenging to find many investigations on this topic, because Wilms’ tumor is the most common tumor of the pediatric kidney, but it always remains a rare pediatric neoplasm. Furthermore, some unpredictability in case and control participation rates may constitute another limitation. Since Wilms’ tumor is a relatively rare entity, the studies are usually small. Hence, more extensive size studies are recommended, which may help assess the real differences between cases and controls and evaluate potential confounders and effect modifiers.48,49
It is to note that observation of funnel plots shows asymmetry throughout some of the sub-groups, which may show publication bias. Considering our data, large-scale and concerted actions among countries worldwide to augment awareness of nephroblastoma risk and pesticide/herbicides exposure are worthwhile.
Most recently, IARC/WHO experts from several countries have already met in Lyon, France, to assess the carcinogenicity of organophosphate pesticides. These included mainly diazinon, glyphosate, malathion, parathion, and tetrachlorvinphos.50–56 It has been stated that it would be crucial to focus on parental exposure to pesticides, herbicides, insecticides, and other household chemicals and pediatric cancer while emphasizing evidence-based, proactive measures. They may include the wearing of chemical-resistant gloves during pesticide application and, even the application of chemicals for household cleaning. Due to one co-author’s (CMS) participation in IARC/WHO monographs, this meta-analysis has taken a relatively more extensive view than previous ones. Previously, we examined in depth the effects of exposure to pesticides on neuroblastoma and pediatric brain cancer under different time windows of exposure from early human development stages (e.g. before birth, during pregnancy, and after birth) and in socio-economic setting attributes (e.g. professional and residential exposures).33,34 The IARC Monographs recognize human cancer’s preventable causes (e.g. pharmaceuticals, complex mixtures, single chemicals, physical, and biological agents). In the volume 112 of the monograph series, there are in-depth evaluations of the carcinogenicity of some organophosphate insecticides and herbicides, including diazinon, glyphosate, malathion, parathion, and tetrachlorvinphos.57–61
Awareness and education programs to limit the pollution of irrigation fields and the contamination of agricultural products have been increasing in the last few years. 62 They target the adverse effects of chemical compound exposure on health and how it may be beneficial to minimize the human and animal exposure, mainly in the time of repeated drought and climate change Warmer temperatures increase evaporation, reduce surface water, and dry out soils and vegetation. Thus, the environment can be heavily loaded with chemicals, now more than a few decades or centuries ago. As the pandemic of SARS-CoV2 persists with variants and sub-variants, it will influence the behavior of humans in the future. It should be emphasized that a stay-at-home lifestyle may trigger less infections, but it will enhance the risk of exposure to household chemicals even more. Currently and in the nearest future, all attempts should be maximized to control children, men, and women’s exposure to chemical compounds. Furthermore, householders should consider that dosage, timing, and length of exposure are critical to the capability of a pesticide to cause detrimental effects, as satisfactorily indicated by other studies.21–23
In conclusion, Wilms' tumor is less likely to develop when pesticide exposure occurs after birth. On the other hand, contact to pesticides or herbicides during pre-conception or pregnancy is more probable to increase the risk of this pediatric small round blue cell tumor. Pesticide exposure in household/residential settings seems to contribute to Wilms’ tumor etiology. Additional investigations with an extensive sample size are required to conclude more confidently, probably involving low-/middle-income and high-income countries. This may be considered important in the post-pandemic era. In our opinion, there is some compelling evidence to robustly educate parents and/or guardians more regarding the appropriate use of chemical compounds and take necessary precautions to minimize the potential risks associated with their application.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. CMS receives royalties from Springer Nature and NOVA Publishers, but he devotes them to pediatric charities. CMS receives consulting fees from Health and Medical Research Fund (HMRF), Food and Health Bureau, Government Secretariat, The Government of the Hong Kong Special Administrative Region, The People’s Republic of China. The authors are responsible for the material and the text of this article. Affiliated institutions and grant societies had no role in study design, data collection, analysis, decision to publish, or manuscript preparation. The authors have no other conflicts of interest to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research has been funded by the generosity of the Children’s Hospital of Eastern Ontario, Ottawa, Ontario, and the Stollery Children’s Hospital Foundation and supporters of the Lois Hole Hospital for Women through the Women and Children’s Health Research Institute (WCHRI, Grant ID #: 2096), Hubei Province Natural Science Funding for Hubei University of Technology (100-Talent Grant for Recruitment Program of Foreign Experts Total Funding: Digital PCR and NGS-based diagnosis for infection and oncology, 2017–2022), Österreichische Krebshilfe Tyrol (Krebsgesellschaft Tirol, Austrian Tyrolean Cancer Research Institute, 2007 and 2009 - “DMBTI and cholangiocellular carcinomas” and “Hsp70 and HSPBP1 in carcinomas of the pancreas”), Austrian Research Fund (Fonds zur Förderung der wissenschaftlichen Forschung, FWF, Grant ID L313-B13), Canadian Foundation for Women’s Health (“Early Fetal Heart-RES0000928”), Cancer Research Society (von Willebrand factor gene expression in cancer cells), Canadian Institutes of Health Research (Omega-3 Fatty Acids for Treatment of Intestinal Failure Associated Liver Disease: A Translational Research Study, 2011–2014, CIHR 232514), and the Saudi Cultural Bureau, Ottawa, Canada. The funders had no role in study design, data collection, analysis, decision to publish, or manuscript preparation.
