Abstract
Introduction
The toxicity and carcinogenicity of glyphosate have long been debated. Nevertheless, the mortality rate in patients with acute glyphosate-surfactant poisoning varies across different groups.
Methods
Between 2002 and 2020, 109 patients with glyphosate-surfactant poisoning received treatment at Chang Gung Memorial Hospital. Patients were stratified into two subgroups according to their prognosis: good (n = 74) or poor (n = 35). Baseline demographics, psychiatric comorbidities, medical complications, and laboratory data were collected, and mortality data were analyzed.
Results
The patients were 54.1 ± 17.5 years of age and were mostly male (68.8%). Most patients (91.7%) ingested pesticides intentionally, and patients arrived at the hospital within 7.1 ± 12.7 h. Psychiatric comorbidities were prevalent, and the top three comorbidities were mental (71.6%), depressive (48.6%), and adjustment (14.7%) disorder. Patients with poor prognoses were older than those with good prognoses (p = .007). Moreover, patients with poor prognoses had lower Glasgow Coma Scale scores (p < .001) and diastolic blood pressure (p = .008), but higher incidences of upper gastrointestinal bleeding (p < .001), aspiration pneumonia (p < .001), hypotension (p < .001), hyperglycemia (p = .002), acute kidney injury (p < .001), and metabolic acidosis (p < .001) than patients with good prognoses. The mortality rate was 5.5%. A multivariate-logistic-regression model revealed that the Glasgow Coma Scale score was a significant risk factor for poor prognosis (odds ratio 0.653, confidence interval 0.427–0.998; p = .049). However, no risk factors for mortality were identified.
Conclusions
A total of 32.1% of patients with glyphosate-surfactant poisoning had poor prognoses, and 5.5% of patients died despite treatment. The mortality outcome is comparable to that of published reports from other international poison centers.
Introduction
Glyphosate surfactants are nonselective herbicides used extensively for controlling broadleaf weeds and grasses worldwide. It is believed that glyphosate has minimal toxicity because the target enzyme 5-enolpyruvylshikimate 3-phosphate synthase is present in plants and microorganisms, but not in humans. 1 Moreover, the lethal dose of glyphosate in experimental rats is more than 4320 mg per kg body weight when ingested orally and is thought to have low toxicity. 2
The toxicity and carcinogenicity of glyphosate surfactants have long been debated. 3 The International Agency for Research on Cancer announced that glyphosate is a Group 2A carcinogen in 2015. 4 Importantly, the International Agency for Research on Cancer reported strong data indicating that both pure glyphosate and glyphosate surfactants can cause genetic damage. In contrast, the U.S. Environmental Protection Agency argued that there is no evidence linking glyphosate to cancer in humans and that glyphosate use does not pose any health risks. 5 The European Union also stated that under conditions of approval and by following good agricultural practices, glyphosate is considered not to have any harmful effects on human health or unacceptable effects on the environment. 6 Furthermore, Ye et al. 7 suggested that caution is needed in the regulation of glyphosate pesticides because the replacement herbicides could cause more harm to humans.
Literature data on the mortality outcomes of patients with glyphosate-surfactant poisoning.

This observational study investigated the mortality data of Taiwanese patients with glyphosate-surfactant poisoning and compared these data with published studies of glyphosate-surfactant poisoning from other poison centers.
Materials and methods
Patients
Between 2002 and 2020, a total of 109 patients with glyphosate-surfactant poisoning received treatment at Chang Gung Memorial Hospital. The following data for each patient were collected: age, sex, exposure route, time between ingestion and hospital arrival, systemic diseases such as hypertension, diabetes mellitus, stroke, coronary artery disease, chronic obstructive pulmonary disease, chronic kidney disease or malignancy, smoking habits, alcohol consumption, psychiatric comorbidities, clinical manifestations, vital signs, hemograms, biochemistry, arterial blood gas, length of hospital stay, length of intensive care unit stay, respiratory failure, cardiac arrest, and mortality. The designation of exposure routes was based on the patients’ clinical history. Pesticide containers were obtained from family members for confirmation of glyphosate-surfactant ingestion. Baseline demographics, psychiatric comorbidities, medical complications, and laboratory data were collected, and mortality data were analyzed.
Patient groups
Patients were categorized into two subgroups based on their clinical prognosis: good (n = 74) or poor (n = 35). Patients who had a favorable outcome without severe complications were defined as having a good prognosis, whereas those who died or experienced significant complications such as intensive care unit admission, cardiac arrest, acute kidney injury, or respiratory failure were grouped as having a poor prognosis.
Inclusion and exclusion criteria
All patients with glyphosate-surfactant poisoning treated at our Poison Center were included. Patients were excluded from this research if they co-ingested other pesticides or if the exposure route was not oral.
Clinical diagnosis
The diagnosis of glyphosate-surfactant poisoning was based on the pesticide exposure history, clinical manifestations, physical examination results, and laboratory findings. As blood and urine glyphosate tests were not available, a meticulous clinical history was obtained, which included inquiring the patient and families about the herbicide information and requesting the container for verification.
Clinical management
Patients were treated with gastric lavage using 2 L of normal saline, followed by activated charcoal administration given by a nasogastric tube. Gastric lavage was indicated if the patient arrived at the hospital 1 hour after pesticide intake. The contraindications for gastric lavage involve deficits in airway protective responses, swallowing of a corrosive acid/alkali substance, or the hazard of alimentary tract hemorrhage due to a primary disease. The purpose of activated charcoal administration was to prevent further gastrointestinal absorption. Since there is a lack of antidotes for patients with glyphosate-surfactant poisoning, clinical management is largely supportive of symptoms.
Statistical analysis
Comparisons between the good and poor prognosis groups were performed via t test for quantitative variables and chi-square tests for categorical variables. Univariate binary logistic regression analysis was carried out to examine the clinical and laboratory variables that might be linked with poor prognosis or mortality. To avoid the influence of confounders, a multivariate regression analysis was carried out to investigate the significant variables during the univariate analysis. A p value of less than 0.05 was considered statistically significant. The analysis was performed with IBM SPSS Statistics for Windows, version 25.0. (Armonk, New York, United States, 2017).
Results
Baseline characteristics of patients with glyphosate-surfactant poisoning stratified into two subgroups according to their prognosis: good or poor (n = 109).

Note: **p < .01.
Psychiatric comorbidities of patients with glyphosate-surfactant poisoning stratified into two subgroups according to their prognosis: good or poor (n = 109).

Note: *p < .05.
Clinical findings of patients with glyphosate-surfactant poisoning stratified into two subgroups according to their prognosis: good or poor (n = 109).
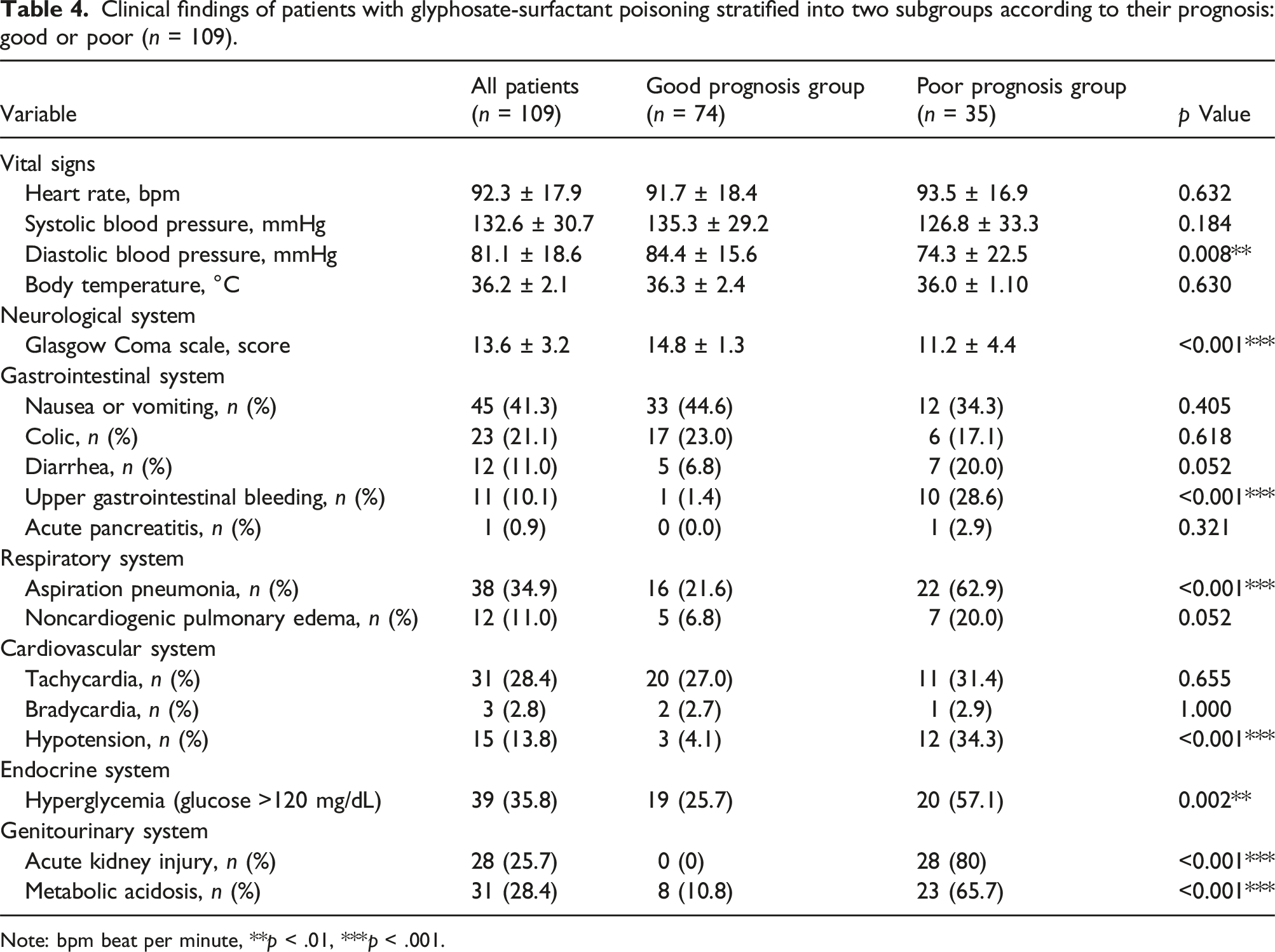
Note: bpm beat per minute, **p < .01, ***p < .001.
Laboratory findings of patients with glyphosate-surfactant poisoning stratified into two subgroups according to their prognosis: good or poor (n = 109).

Note: *p < .05, **p < .01, ***p < .001.
Outcomes of patients with glyphosate-surfactant poisoning intoxication stratified into two subgroups according to their prognosis: good or poor (n = 109).

Note: **p < .01, ***p < .001.
Prediction of poor prognosis via logistic regression analysis.

Note: *p < .05, **p < .01, ***p < .001.
Discussion
This study contributes to the literature by demonstrating that acute, large-dose exposure to glyphosate-surfactant herbicides could cause significant mortality and morbidity. Among the 109 patients, 35 (32.1%) had poor prognoses, and the overall mortality rate was 5.5%. As shown in Table 1, the mortality data are comparable to published data from international poison centers, for which the mortality rates ranged between 1.8% and 46.0%.8–25
The analytical data indicate that lower Glasgow Coma Scale scores in individuals exposed to glyphosate surfactants may be a predictor of a poor prognosis. Other investigators also suggested that glyphosate exposure can lead to a loss of consciousness and lower Glasgow Coma Scale scores15,22,25 and that the pesticide may also induce seizures. 15 Several types of neurologic damage caused by glyphosate have been reported in previous studies. Animal studies have shown that exposure to glyphosate can result in changes in gene regulation and neurotransmitter levels in the cerebrum, leading to neurotoxicity in the forebrain and midbrain regions of zebrafish. 26 An in vitro study revealed that treatment with high concentrations of glyphosate can negatively affect the integrity of the blood–brain barrier, leading to alterations in neuron metabolic processes and glucose uptake by cerebral vascular endothelial cells, resulting in neurological damage. 27 Glyphosate also affects serotonin, dopamine, and norepinephrine concentrations in the cerebrum and modifies monoamine neurotransmitters in a regional- and dose-associated way, according to a laboratory study. 28 Studies have suggested that glyphosate has a potent toxic effect on cerebral transmission and causes oxidative stress, inflammation, and mitochondrial malfunction. These biological activities can eventually result in neuronal mortality through mechanisms such as autophagy, necrosis, or apoptosis and trigger behavioral symptoms in addition to motor neuron disease. 29 These findings may help explain the observed changes in Glasgow Coma Scale scores in this study.
A total of 28 (25.7%) patients experienced acute kidney injury after glyphosate-surfactant poisoning. Renal complications have been reported in other studies 19, 25, 30. In this study, 11 patients were found to have both acute kidney injury and hypotension when they visited the emergency room; therefore, a prerenal form with reduced blood flow to the kidney was considered. However, reports have indicated the direct kidney toxicity of the surfactant used in this herbicide. 30 In a laboratory study, 31 Ma et al. reported that glyphosate exposure modified the transcription of immunoglobulin M, complement C3, and lysozyme in the kidneys of common carp and caused renal damage, such as vacuolization of the kidney parenchyma and intumescence of the kidney tubules. In a clinical study, Mohamed et al. suggested that elevated urinary levels of cytochrome C and interleukin-18 after glyphosate-surfactant self-poisoning could help in the diagnosis of acute kidney injury and confirm glyphosate-surfactant-induced renal apoptosis and mitochondrial toxicity. 23 Kimura et al. published a clinical case of acute kidney injury after glyphosate and surfactant ingestion. A biopsy of the patient’s kidneys revealed damage to the tubular epithelium, specifically in the proximal tubules. Pathology revealed vacuolization of the proximal tubular epithelium, which was noted throughout the biopsied sample.30,32 Therefore, the mitochondrial damage caused by glyphosate could be associated with acute kidney injury, and the combination of glyphosate and surfactants could damage vascular endothelial cells and produce circulatory insufficiency.
In this study, 38 out of 109 patients (34.9%) experienced aspiration pneumonia, and 12 patients (11.0%) had non-cardiogenic pulmonary edema. Significant laryngeal injury was noted after glyphosate intoxication and was strongly correlated with aspiration pneumonitis and significantly longer hospital stays. 11 Another study showed that after the ingestion of glyphosate, various degrees of corrosive esophageal injury from 1 to 2b were noted. 19 This study revealed that 10.1% of patients had upper gastrointestinal bleeding. The pain related to corrosive injury may contribute to swallowing difficulty and choking, and some patients with upper gastrointestinal bleeding also experience coffee ground vomiting, all of which can lead to aspiration pneumonia. Additionally, gastric lavage with activated charcoal in the emergency room may also be a potential contributor. 33 Previous studies have reported that pulmonary edema is an uncommon complication of glyphosate poisoning. 34 However, in our study, 12 patients (11.0%) experienced non-cardiogenic pulmonary edema. Overall, 16.5% of patients experienced respiratory failure necessitating ventilatory support. Several studies have shown that glyphosate can induce inflammation in pulmonary and airway tissues, triggering increased levels of eosinophils, neutrophils, and asthma-associated cytokines (interleukin-5, interleukin-10, interleukin-13, interleukin-33, and thymic stromal lymphopoietin), which cause airway tightening.35,36 One study suggested that lung and kidney toxicity are the main contributing factors to the mortality of patients who are poisoned by glyphosate. 13 Pulmonary toxicity, in particular, appears to be a critical factor according to the above studies.
The majority of patients intentionally ingested glyphosate-surfactant herbicides (91.7%, Table 2). Psychiatric comorbidities were prevalent (Table 3), and the top three comorbidities were mental disorders (71.6%), depressive disorders (48.6%), and adjustment disorders (14.7%). Studies have shown that glyphosate-surfactant poisoning is always associated with suicide attempts in Asia.9,13,17,19,20,22 To date, no studies have explored the correlation between glyphosate poisoning and underlying psychiatric illnesses. This is the first study to analyze the psychiatric comorbidities of glyphosate-surfactant-treated patients to identify the underlying psychiatric patterns. A previous systematic review 37 revealed that a high proportion of people who self-harm has a psychiatric disorder, highlighting the need to include appropriate psychological and/or pharmaceutical interventions in their post-incident care.
The limitations of this study include its retrospective nature, small sample size, lack of data on urine/blood glyphosate levels, lack of data on surfactants, and short follow-up duration. However, further studies are needed.
Conclusions
A total of 32.1% of patients with glyphosate-surfactant poisoning had poor prognoses, and 5.5% of these patients died despite intensive treatment. A prompt diagnosis of glyphosate-surfactant poisoning and an immediate institution of a treatment protocol are necessary. Moreover, the mortality outcome is comparable to that of published reports from other international poison centers.
Footnotes
Author contributions
K.H.L. data collection and manuscript writing; S.S.C., C.Y.T., H.Y.C., W.C.L., P.Y.K., K.F.T., J.C.Y. data analysis; I.K.W. manuscript editing; T.H.Y. study supervision, patient care and management. All the authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from Chang Gung Memorial Hospital (CORPG3N0391 and CORPG3K0195).
Ethical statement
Data Availability Statement
The datasets used or analyzed in the study are available from the corresponding author upon reasonable request.
