Abstract
Background:
Cyclophosphamide (CP) causes premature ovarian failure (POF) due to ovarian toxicity. The toxicity mechanism is attributed to oxidative stress, inflammation, and apoptosis. We assessed whether quercetin and rosuvastatin could promote ovarian protection against CP ovotoxicity.
Methods:
A total of 80 female BALB/c mice were randomly assigned; 10 mice into each of eight groups. Group 1 (control), group 2 (EH), group 3 (CP), group 4 (QH), group 5 (QL), group 6 (RH), group 7 (RL), and group 8 (COM).
Results:
Quercetin and rosuvastatin groups (4:8) showed signs of restored ovarian function in the form of a significant, dose-dependent increase in primordial follicles number, serum anti-Mullerian hormone level, and ovarian tissue glutathione level (p < 0.05) versus group 3, and a significant, dose-dependent decrease in atretic follicles number and ovarian tissue level of malondialdehyde (p < 0.05) versus group 3. Immunohistochemistry analysis demonstrated a lower expression of caspase and nuclear factor-kappa B of groups (4:8) versus group 3, although quercetin and rosuvastatin showed a nonsignificant reduction in tumor volume.
Conclusions:
We demonstrated the protective effect of quercetin and rosuvastatin against ovarian toxicity and POF induced by CP without compromising its antitumor effect.
Introduction
Premature ovarian failure (POF) is a heterogeneous disorder, which could be defined as hyper gonadotropic hypoestrogenism in women below 40 years. In this case, patients typically complain of oligomenorrhea, amenorrhea, or infertility. Additionally, patients can suffer from long-term consequences of hypoestrogenism, including osteoporosis, cardiovascular diseases, and psychic disorders. 1 The condition affects one per 10,000 women by the age of 20 years, and one per 100 by the age of 40 years. 2
The underlying causative agent of POF could include genetic, autoimmune, metabolic, infectious, and iatrogenic causes. 3 The most predictable cause of POF is iatrogenic one which occurs during cancer chemotherapy and radiotherapy. The incidence of chemotherapy-induced POF is continuously rising and reached approximately 70–100% of patients who undergo chemotherapy. 4 Cyclophosphamide (CP) is one of the most widely used chemotherapeutic drugs that are involved in the treatment of breast cancer and is the most extensively studied agent. 5,6 Moreover, it is an alkylating agent that causes double-strand breaks in DNA resulting in DNA damage, deleterious toxic consequences for the organ cells including the heart, liver, and kidney, and has severe toxic effect on the ovary. 7,8 Its cytotoxicity is also due to generation of reactive oxygen species with subsequent lipid peroxidation, mitochondrial damage leading to activation of the caspase family and apoptosis, 9,10 and inflammation by different inflammatory mediators. 11
One of the most common side effects of CP in a female patient is POF, which accounts for 42.4% in 2000. 12 It induces disruption of follicular reserve with depletion of dominant primordial follicles and accelerated follicular atresia through induction of follicular apoptosis that resulted in ovarian dysfunction and a reduction of anti-Mullerian hormone (AMH) level. 13
Quercetin is one of the flavonoids abundantly distributed in fruits, vegetables, olive oil, red wine, and tea. 14 Recently, it showed cardioprotective, anti-inflammatory, antiproliferative, and anticancer activities due to its potent antioxidant properties. 15,16 Quercetin has an important therapeutic role in the prevention and treatment of different chronic diseases, including cardiovascular, neurodegenerative diseases, and cancer. 17,18 It has a protective effect in reperfusion ischemic tissue damage, hypertension, and atherosclerosis by inhibition of LDL oxidation. 19 In addition, it has an antiapoptotic effect 20 and it has been reported to repair DNA damage through epigenetic modification by inhibition DNA acetylation. 21 Moreover, it has been showed that quercetin can reduce CP-induced cardiotoxicity and genotoxicity in mice. 22
Rosuvastatin is one of 3-hydroxy-3-methyl-glutaryl-coenzyme A (HMG-CoA) reductase inhibitors which are widely used to lower blood cholesterol level in hypercholesterolemic patient. Rosuvastatin was found to be significantly more potent inhibitor of hepatocyte sterol synthesis than any of the statins currently available. 23 It decreases the relative risk of heart attack and stroke in patients even without hyperlipidemia. 24 In addition, it exhibits a pleiotropic activity independent of HMG-CoA reductase inhibition, which includes antioxidant, antiapoptotic, and anti-inflammatory effects. 25 Moreover, it induces epigenetic modifications of DNA. These modifications include inhibition of histone and DNA methylation and deacetylation of histone and these changes thought to protect cells against DNA injury and against alteration of tumor suppressor genes. 26
Therefore, the present study is designed to investigate the possible protective effect of quercetin and rosuvastatin each alone or in combination on CP-induced POF without compromising the antitumor effect of CP in Ehrlich tumor-bearing mice.
Materials and methods
Animals
Strain: BALB/c mice weighting 20–25 g and aged 8–12 weeks, obtained from Tanta University Animal House, were used in this study.
Housing: The animals were housed in groups (total numbers, n = 80 female mice for this experiments) in polyethylene cages and had free access to food and water ad libitum 1 week prior to the experiment. The study protocol has been approved by the research ethics committee at the Faculty of Medicine, Tanta University (AL Gharbia, Egypt).
Drugs
CP (Endoxan vial 200 mg) white powder was purchased from Baxter Healthcare Company, Hong Kong. Quercetin yellow powder was purchased from Sigma-Aldrich, St. Louis, Missouri, USA. A white powder of calcium salt of rosuvastatin was obtained from SAJA Pharmaceutical Company, Saudi Arabia, as a gift.
Ehrlich tumor
The Ehrlich ascitic tumor, obtained from a spontaneous mammary adenocarcinoma of mice, was maintained in ascitic form (obtained from National Cancer Institute, Cairo, Egypt). The ascitic fluid was collected by intraperitoneal (IP) puncture using a sterile syringe. Ascitic tumor cell counts were done in a Neubauer hemocytometer (Sigma-Aldrich). The cells were found to be more than 99% viable by the trypan blue dye exclusion method. 1 × 106 cells in 0.5 ml saline were injected SC in the left thigh of used female mice. The day of tumor implantation was assigned as day 0. 27
The experiment study
Accustomed mice were allocated randomly to eight different groups, and each had 10 mice.
Group (control): (no CP or Ehrlich tumor); mice received 0.5 ml sterile saline IP per day for 10 days.
Group (EH): mice were injected with Ehrlich tumor cells SC by 1 × 106 cells in the left thigh at day 0.
Group (CP): mice were injected with Ehrlich tumor cells SC in the left thigh. Each mouse received 90 mg/kg of CP single IP injection on the eighth day of the experiment; these served as the untreated POF group.
Group (QH): received high dose of quercetin IP in a daily dose of 100 mg/kg for 10 days starting the first day after Ehrlich injection and CP was injected IP on the eighth day; these served as the QH-treated POF group.
Group (QL): received low dose of quercetin IP in a daily dose of 20 mg/kg for 10 days starting the first day after Ehrlich injection and CP was injected IP on the eighth day; these served as the QL-treated POF group.
Group (RH): received high dose of rosuvastatin IP in a daily dose of 3 mg/kg for 10 days starting the first day after Ehrlich injection and CP was injected IP on the eighth day; these served as the RH-treated POF group.
Group (RL): received low dose of rosuvastatin IP in a daily dose of 0.3 mg/kg for 10 days starting the first day after Ehrlich injection and CP was injected IP on the eighth day; these served as the RL-treated POF group.
Group (COM): received combination of a low dose of quercetin and rosuvastatin IP in a daily dose of 20mg/kg and 0.3mg/kg respectively for 10 days starting the first day after Ehrlich injection and CP was injected IP on the eighth day; these served as the QH-treated POF group.
After the 10th day from the beginning of treatment and according to estrous cycle, mice were anesthetized by ether and euthanized, and blood samples were collected under anesthesia immediately by a cardiac puncture for AMH level detection. Ovarian tissues and Ehrlich mass were excised and washed by phosphate-buffered saline.
Biochemical assays
Tissues were homogenized in 150 mM KCl (pH 7.4) at a 1:10 (w/v) dilution of the whole homogenate. The homogenates were centrifuged at 18,000 rpm × g and 4°C for 30 min to determine the malondialdehyde (MDA) (nmol/g tissue) and reduced glutathione (GSH) level (mg/g tissue) (catalog number MD 2529, GR 2511), respectively Biodiagnostic company according to the method of Ohkawa et al. and Beutler, 28,29 respectively.
AMH assays
AMH was quantitatively estimated in mouse serum samples using enzyme-linked immunosorbent assay kits according to the method of Cate et al. (1986) 30 (catalog number: BYEK2651; Chongqing Biospes Company, Chongqing, China). The values were expressed as ng/ml.
Histopathological examinations of the ovarian tissue
For light microscopic evaluation, ovarian tissues were fixed in 10% formalin and embedded in paraffin wax. Paraffin wax-embedded specimens were cut into 5 μm thick sections, mounted on slides, and stained with hematoxylin and eosin. Each section was viewed at power (100× magnification). The primordial follicle number was counted in every fifth section by the same observer to minimize the interobserver error according to Kishk and Ali. 31 To reach a value representative of the total number of primordial follicles in the ovary, their numbers were then multiplied by five. The same was done for atretic follicles counting.
Caspase immunohistochemistry examinations of the ovarian tissues
Immunohistochemical expression of caspase in the ovary was done using rabbit polyclonal caspase-3 antibody purchased from Lab Vision Corporation, UK. Caspase staining was scored according to Buchwalow and Böcker (2010) 32 as follows: under 1% of the total cell number are positive scored as 0, 2–10% of the total cell number are positive scored as 1, 11–50% of the total cell number are positive scored as 2, 51–90% of the total cell number are positive scored as 3, and over 90% of the total cell number are positive scored as 4.
NF-κ B immunohistochemistry examinations of the ovarian tissues
Immunohistochemical expression of nuclear factor-kappa B (NF-κB) in the ovary was done using rabbit polyclonal antibody (ready-to-use for immunohistochemical staining) purchased from Thermo Fisher Scientific Anatomical Pathology, Cheshire, UK. NF-κB staining was scored according to Weichert et al. 33 regarding to the number of stained cells (cytoplasmic as well as nuclear). No cells stained scored (0), less than 10% of cells stained scored (1), 10 - 50% of cells stained scored (2), 50 - 80% of cells stained scored (3), or more than 80% of cells stained scored (4).
Estimation of the Ehrlich solid tumor volume
After tumor mass was dissected, each mass was measured with the help of vernier caliper (major and minor tumor axis of the tumor). Tumor volume of each animal was calculated using the following formula 34
where A is the minor tumor axis, B is the major tumor axis, and π equals 3.14.
Histopathological examinations of the Ehrlich tumor tissues
For light microscopic evaluation, Ehrlich tumor tissues were fixed in 10% formalin and embedded in paraffin wax. Paraffin wax-embedded specimens were cut into 5 μm thick sections, mounted on slides, and stained with hematoxylin and eosin. Each section was viewed at low power (50× magnification). Histopathological analysis of tumor sections was focused on the extension of the necrotic and proliferative area.
Statistical analysis
All values are presented as means ± standard error. Differences were considered significant at p < 0.05. The GraphPad Prism, version 5, for Windows was used for the statistical analyses. The biochemical values were analyzed using one-way ANOVA and post hoc Tukey’s honestly significant difference test. Immunohistochemical results were compared using Kruskal–Wallis variance analysis. When differences among the groups were detected, group means were compared using the Mann–Whitney U test.
Results
Amelioration of oxidative stress by both quercetin and rosuvastatin therapy
CP group revealed a significant increase in MDA level as compared to the normal control group. QH, RH, RL, and COM groups revealed a significant decrease in MDA level as compared to the CP group, but QL group revealed a nonsignificant decrease in MDA level as compared to the CP group. QH, QL, RH, and RL groups revealed a significant decrease in MDA level as compared to the COM group. CP group revealed a significant decrease in GSH level as compared to the normal control group. QH, RH, and COM groups revealed a significant increase in GSH level as compared to the CP group, but QL and RL groups revealed a nonsignificant increase in GSH level as compared to the CP group. QH, QL, RH, and RL groups revealed a significant increase in GSH level as compared to the COM group (Figure 1).

The biochemical assay in different groups: (a) MDA and (b) GSH. Data were represented as mean ± standard deviation 35 (n = 10). ns: nonsignificant (p > 0.05); #: significant (p < 0.05) as compared with the control group; *: significant (p < 0.05) as compared with the CP group; &: significant (p < 0.05) as compared with the COM group; MDA: malondialdehyde; GSH: glutathione; CP: cyclophosphamide.
Restoration of AMH levels by quercetin and rosuvastatin therapy
CP group revealed a significant decrease in AMH level as compared to the normal control group. QH, QL, RH, RL, and COM groups revealed a significant increase in AMH level as compared to the CP group. QH, QL, RH, and RL groups revealed a significant increase in AMH level as compared to the COM group to reach near-normal serum values (Figure 2).
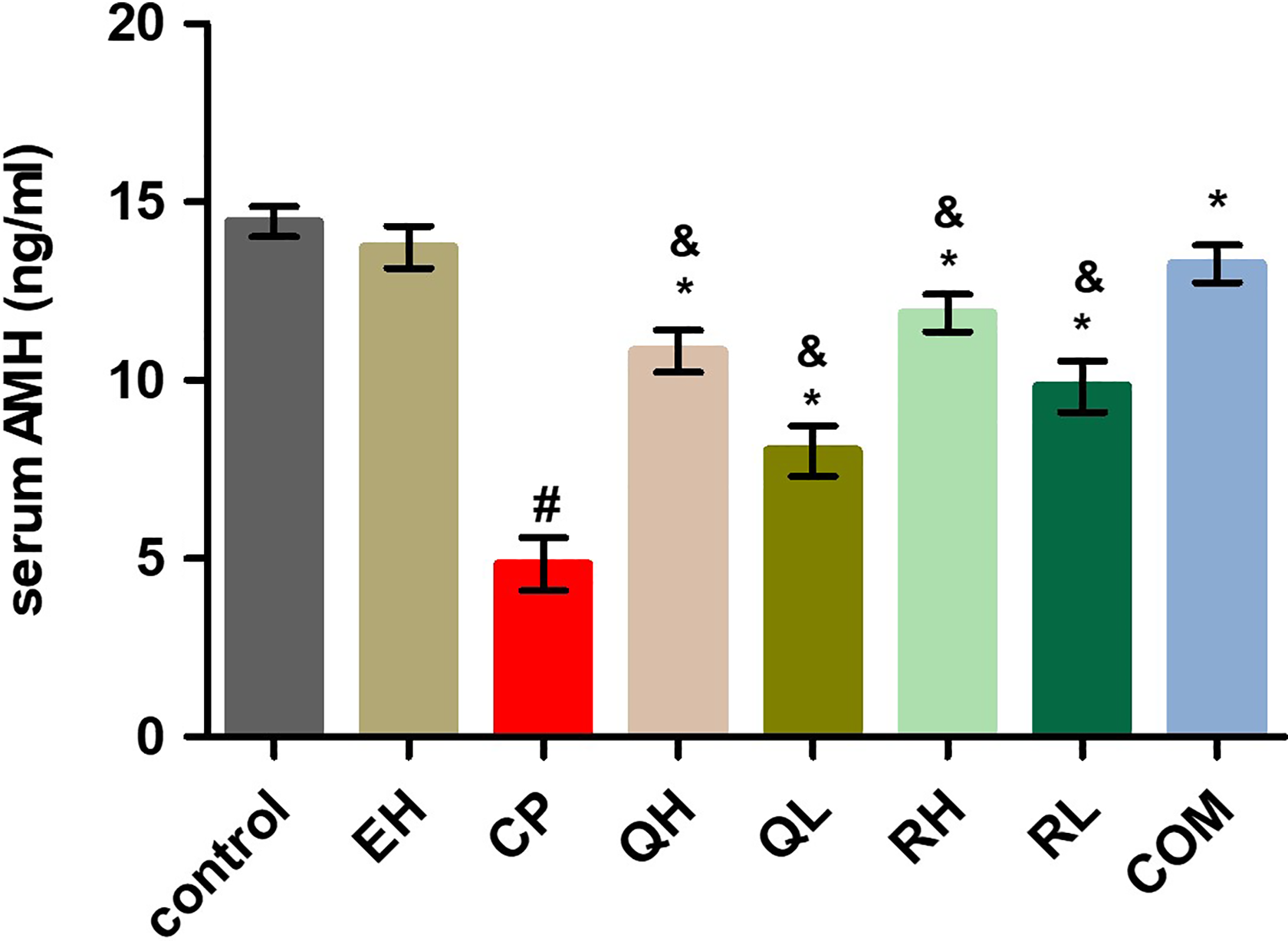
AMH level was assessed in different groups. Data were represented as mean ± standard deviation (n = 10).
Recovery of ovarian architecture
CP group revealed a significant decrease in the number of primordial follicles as compared to the normal control group. QH, RH, QL, RL, and COM groups revealed a significant increase in the number of primordial follicles as compared to the CP group. QH, QL, RH, and RL groups revealed a significant increase in the number of primordial follicles as compared to the COM group. CP group revealed a significant increase in the number of atretic follicles as compared to the normal control group. QH, RH, QL, RL, and COM groups revealed a significant decrease in the number of atretic follicles as compared to the CP group. QL and RL groups revealed a significant decrease in the number of atretic follicles as compared to the COM group. QH and RH groups revealed a nonsignificant decrease in the number of atretic follicles as compared to the COM group (Figure 3). Hematoxylin and eosin-stained ovarian sections, the control group showed normal morphology of the ovary with normal cyclic ovarian architecture. Many primordial follicles and growing primary follicles are observed. In the CP group, ovary sections showed an obvious reduction in the number of follicles and follicular atresia, which indicated CP-induced ovarian toxicity. This toxicity was reversed by quercetin and rosuvastatin treatment in QH, QL, RH, RL, and COM groups (Figure 4).

Follicles number of different groups: (a) PrF and (b) atretic follicles. Data were represented as mean ± standard deviation 35 (n = 10). ns: nonsignificant (p > 0.05); #: significant (p < 0.05) as compared with the control group; *: significant (p < 0.05) as compared with the CP group; &: significant (p < 0.05) as compared with the COM group; PrF: primordial follicles; CP: cyclophosphamide.

Hematoxylin and eosin sections (×100 magnification). (a) Control group: hematoxylin and eosin sections showing many PrF and growing PF. (b) EH group: hematoxylin and eosin sections showing an increase in the features of AF and the presence of obvious number of PrF and PF. (c) CP group: hematoxylin and eosin sections showing AF, corpus lutetium and a marked decrease in the number of the PrF and PF. (d) QH group: hematoxylin and eosin sections showing a decrease in the number of AF and an increase in the number of the PrF. (e) QL group: hematoxylin and eosin sections showing an increase in the number of the PrF. (f) RH group: hematoxylin and eosin sections showing a marked increase in the number of the PrF. (g) RL group: hematoxylin and eosin sections showing normal growing PF and SF and an increase in the number of the PrF. (h) COM group: hematoxylin and eosin sections showing a marked increase in the number of the PrF. PrF: primordial follicles; PF: primary follicles; AF: follicular atresia; CP: cyclophosphamide; SF: secondary follicles.
Amelioration of CP-induced inflammation and apoptosis
Immunohistochemical stained sections showed a significant increase in caspase, NF-κB immunoexpression, and score in the CP group compared with the control group. QH, RH, RL, and COM groups revealed a significant decrease in expression and score compared with the CP group, but QL group revealed a nonsignificant difference compared with the CP group. QH, QL, RH, and RL groups revealed a significant difference as compared to the COM group (Figure 5). Caspase and NF-κB immunohistochemistry slides of ovaries of different groups are shown in Figures 6 and 7, respectively.

Immunoexpression score of the ovarian tissue. (a) Caspase expression score was assessed in different groups. (b) NF-κB expression score was assessed in different groups. Data were represented as median (n = 10). ns: nonsignificant (p > 0.05); #: significant (p < 0.05) as compared with the control group; *: significant (p < 0.05) as compared with the CP group; &: significant (p < 0.05) as compared with the COM group; CP: cyclophosphamide; NF-κB: nuclear factor-kappa B.

Caspase immunohistochemical expression of the ovarian tissue in different groups (streptavidin biotin 100×). (a) Mild expression of caspase in control group (score 1). (b) Moderate expression of caspase in EH group (score 2). (c) Very dense expression of caspase in CP group (score 4). (d) Dense expression of caspase in QH group (score 3). (e) Very dense expression of caspase in QL group (score 4). (f) Moderate expression of caspase in RH group (score 2). (g) Dense expression of caspase in RL group (score 3). (h) Mild expression of caspase in COM group (score 1). CP: cyclophosphamide.

NF-κB immunohistochemical expression of the ovarian tissue in different groups (streptavidin biotin 100×). (a) Mild expression of NF-κB in control group (score 1). (b) Moderate expression of NF-κB in EH group (score 2). (c) Very dense expression of NF-κB in CP group (score 4). (d) Dense expression of NF-κB in QH group (score 3). (e) Very dense expression of NF-κB in QL group (score 4). (f) Moderate expression of NF-κB in RH group (score 2). (g) Dense expression of NF-κB in RL group (score 3). (h) Mild expression of NF-κB in COM group (score 1). NF-κB: nuclear factor-kappa B; CP: cyclophosphamide.
Ehrlich tumor volume measurement
CP group revealed a nonsignificant decrease in tumor volume as compared to the EH group. QH, RH, RL, and COM groups revealed a nonsignificant decrease in tumor volume as compared to the CP group, and QH, QL, RH, and RL groups revealed a nonsignificant decrease in tumor volume as compared to the COM group (Figure 8).
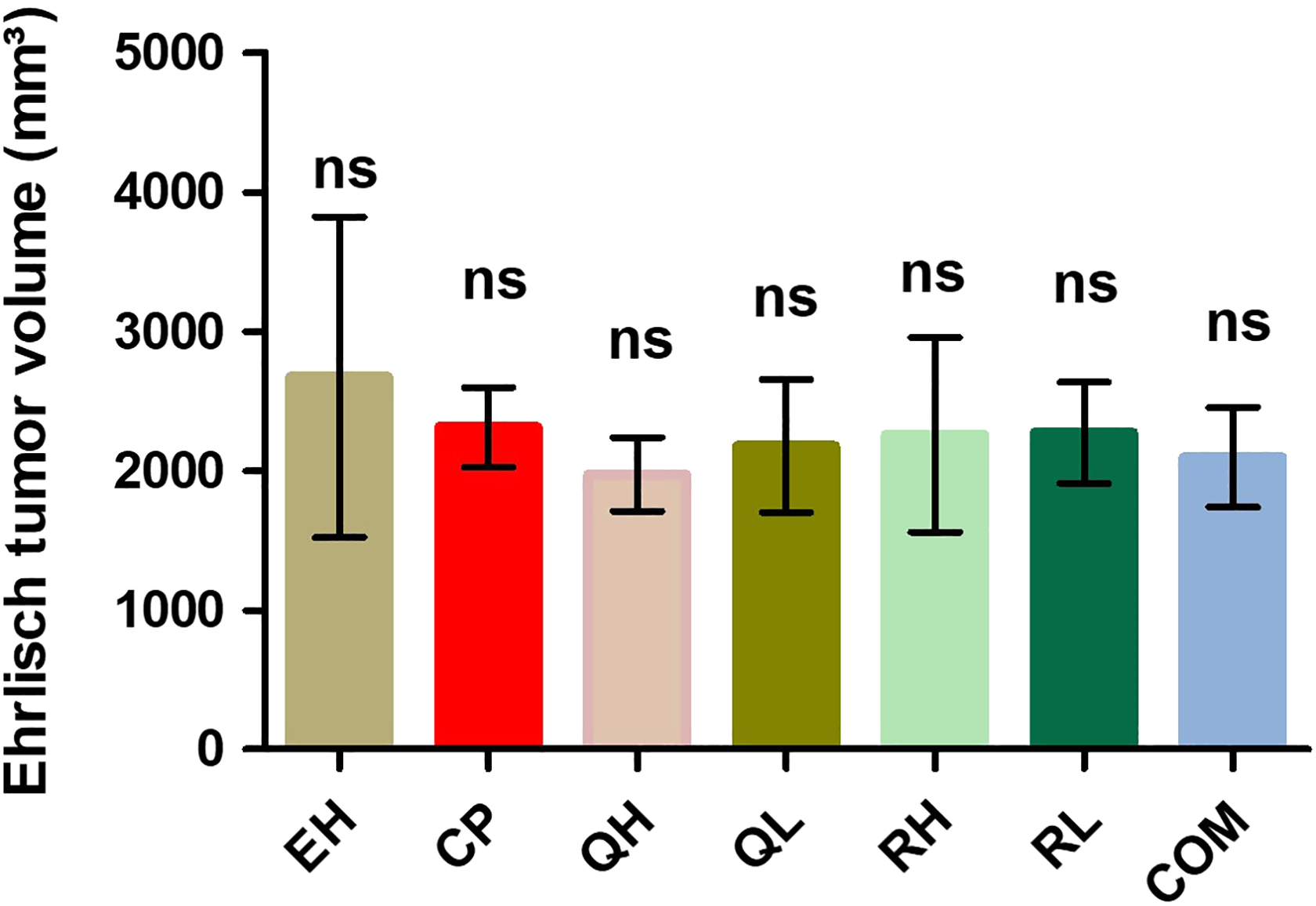
Tumor volume was assessed in different groups. Data were represented as mean ± standard deviation 35 (n = 10). ns: nonsignificant (p > 0.05) as compared with the EH group, with the CP group, and with the COM group. CP: cyclophosphamide.
Histopathological observation of Ehrlich tumor
Hematoxylin and eosin-stained tumor mass sections, EH group showed wide proliferative areas of neoplastic cells with limited central necrosis. CP group showed extensive necrosis and decrease the proliferative areas with the appearance of the subcutaneous adipose tissue. Quercetin and rosuvastatin treatment in QH, QL, RH, and RL groups showed marked decrease of the neoplastic cells density within the proliferative nodule with the appearance of the multiple fat tissue and necrosis in between the nodules and the COM group showed very small proliferative foci of neoplastic cells with widely distributed necrosis within the mass (Figure 9).

Hematoxylin and eosin sections (×50 magnification) of Ehrlich tumor. (a) EH group showing wide proliferative areas of Ehrlich neoplastic cells mostly within the peripheral areas (white arrow) with limited central necrosis (black arrow). (b) CP group showing extensive necrosis (black arrow) and marked decrease in the proliferative areas (white arrow) with appearance of the subcutaneous adipose connective tissue. (c) QH group showing marked decrease of the neoplastic cells density within the proliferative nodule with appearance of the multiple fat tissue (white arrow) and necrosis in between the nodules (black arrow). (d) QL group showing marked decrease in neoplastic cells proliferation (white arrow) and wide areas of necrosis (black arrow). (e) RH group showing mild decrease of Ehrlich neoplastic cells proliferation (white arrow) and also mild to moderate increase of necrotic areas within the mass (black arrow). (f) RL group showing moderate decrease in the proliferative nodules with presence of fat tissues (white arrow) and noticeable central necrosis (black arrow). (g) COM group showing very small proliferative foci of neoplastic cells (white arrow) with widely distributed necrosis within the mass (black arrow). CP: cyclophosphamide.
Discussion
Several studies demonstrated that CP induces POF through accelerated follicular atresia. 36 The follicular atresia is induced by numerous mechanisms including oxidative stress with lipid peroxidation, 27 apoptosis, 37 and inflammation. 11 There is a variety of approaches aimed at fertility preservation or restoration during chemotherapy use and an increase in the survival rate of cancer cases. 38
Our study substantiates the idea that oxidative stress plays an important role in CP-induced POF. CP induced a state of oxidative stress in the ovary represented by the significant decrease in reduced GSH content and increase in lipid peroxidation represented by significant increase in MDA which agrees with Abdel-Raheem et al. 39 and Kabirian et al. 40 who demonstrated CP-induced oxidative stress. CP also increased caspase expression indicating follicular destruction attributed to enhanced apoptosis agreeing with Kalich-Philosoph et al. 37 who observed the appearance of pyknotic granulosa cell nuclei that proves apoptosis as one of POF mechanism. Consistent with Nayki et al. 41 who demonstrated CP-induced inflammation, CP in our study showed an increase in NF-κB expression indicating an inflammatory role in CP ovarian toxicity. These mechanisms participated in decreasing the ovarian reserve which in turn caused decrease in primordial follicles number and AMH level. This state of oxidative stress, apoptosis, and inflammation was reversed by quercetin and rosuvastatin.
To our knowledge, this is the first search to evaluate the protective potential of quercetin and rosuvastatin against CP-induced POF, and the results confirmed their protective effect against CP-induced POF without compromising its antineoplastic activity.
Quercetin choice, being a bioflavonoid, was based on its reliable antioxidant and anti-inflammatory effects, also having an antiapoptotic effect, 20 and was reported to repair DNA damage through epigenetic modification. 21 Moreover, quercetin ameliorated CP-induced toxicity in mice. 22 Rosuvastatin is one of the HMG-CoA reductase inhibitors. 42 It has pleiotropic effects rather than antihyperlipidemic such as its antioxidant, antiapoptotic effect, and anti-inflammatory. 25 Moreover, it induces epigenetic modifications of DNA to protect cells against DNA injury. 26 Because of the previous effects of rosuvastatin, it was chosen to ameliorate CP ovotoxicity, especially it previously ameliorated cisplatin-induced nephrotoxicity. 43
In our study, quercetin in a dose-dependent manner decreased significantly the oxidative stress in ovarian tissue, this was proved by a significant decrease in MDA level in the QH group, however, QL group showed a nonsignificant decrease in MDA level . QH group also showed a significant increase in GSH level, but QL group showed a nonsignificant effect. These results agree with Rinwa and Kumar, 44,45 who demonstrated the dose-dependent antioxidant effect of quercetin. The antiapoptotic effect of quercetin had been proved through the decrease in ovarian caspase-3 expression score, only significant with the high-dose quercetin. The result of QH correlates with Chow et al., 46 who showed that activation of the caspase-3 in H2O2-treated cells was significantly blocked via quercetin, while the result of QL was contradictory with Ahmad et al. 47 who demonstrated antiapoptotic effect of QL in brain cells and we can justify this by the much lower dose used in the current work compared to Ahmad et al. 47 The anti-inflammatory effect of quercetin is assessed through the decrease in NF-κB expression score, but there was significant decrease only with the high-dose quercetin and insignificant decrease with low-dose quercetin. The result of QH correlates with Ajibade et al. 48 who showed that quercetin can decrease NF-κB expression induced by toxic cobalt chloride. However, the result of QL disagrees with Ma et al.49 who demonstrated that QL inactivated NF-κB in livers of the CCl4-treated mice. We can attribute this controversy to the lower dose of the study of Ma et al. 49 is higher than the low dose used in our study. By all the previous mechanisms, quercetin showed improvement as regard to the amplitude of increase in functioning follicle count that is reflected on improved hormonal levels. These results agree with the previous finding of Erbas et al. 50 and Kamarzaman et al. 51 who found out that ovarian protection took place after restoring near-normal serum ovarian hormonal level and functioning ovarian follicles count.
In our study, Rosuvastatin in a dose-dependent manner significantly decreased the MDA level . RH also significantly increased the GSH level, although RL showed a nonsignificant increase in the GSH level. These results agree with Ghaisas et al. 52 who demonstrated dose-dependent antioxidant effect of rosuvastatin. Correlation with Mostafa et al. 43 rosuvastatin decreased caspase-3 expression score. Also, inconsistent with Rashidian et al. 53 and Mostafa et al., 43 the anti-inflammatory effect of rosuvastatin is proved through decrease in NF-κB ovarian expression. Because of all previous mechanisms, rosuvastatin showed improvement as regard to the amplitude of increase in functioning follicle count that is directly reflected on improved hormonal levels. These results agree with the previous finding of Melekoglu et al. 38 who proved the ovarian protection through restoration of near-normal serum hormonal level and functioning ovarian follicles count.
However, RH showed more protective effect than QH against CP ovarian toxicity and this conclusion is dependent on restoration of ovarian follicles and AMH level, while the COM group showed more increase in functioning follicle count and improved hormonal levels. These findings can be attributed to additive antioxidant, anti-inflammatory, and antiapoptotic actions of QH and RH, which can be considered a marked pharmacodynamic interaction.
Regarding Ehrlich tumor histopathological finding of Miranda-Vilela et al., 54 CP showed more necrosis and less proliferation, which is a good cure sign. However, CP caused a nonsignificant tumor size reduction which is inconsistent with Kabel et al., 55 who showed a significant tumor reduction with the therapy, and this controversy can be attributed to longer duration of chemotherapy that was used in the previous study. But in our study, only single dose of chemotherapy (CP) was not enough to produce a significant difference in tumor volume.
In our study, quercetin was administrated IP as it has been proved that oral administration of quercetin failed to produce any improvement of tumor model. 56
Quercetin and rosuvastatin have antitumor effect against cancer cell line 57,58 and in correlation with Sanchez-Gonzalez et al. 59 and Kabel et al., 55 quercetin and rosuvastatin markedly decreased proliferating area and increased necrosis each alone or in combination. However, quercetin and rosuvastatin each alone or in combination caused a nonsignificant tumor size reduction. This disagrees with Wang et al. 60 and Hayashi et al. 61 This controversy can be attributed to the longer duration and higher dose of administrated therapy in the previous studies than our study.
Conclusions
The significant ovarian protection against CP by QH and RH is due to potentiation between their antioxidant, anti-inflammatory, and the antiapoptotic effects, besides their epigenetic modification properties. Regarding the cytoprotective effect of QH and RH and whether it could have the potential to compromise the antitumor effect of CP which need to be verified in further human clinical studies.
Supplemental material
Estrogen_level_in_supplementary_material_file - Effects of quercetin and rosuvastatin each alone or in combination on cyclophosphamide-induced premature ovarian failure in female albino mice
Estrogen_level_in_supplementary_material_file for Effects of quercetin and rosuvastatin each alone or in combination on cyclophosphamide-induced premature ovarian failure in female albino mice by MA Elkady, S Shalaby, F Fathi and S El-Mandouh in Human & Experimental Toxicology
Supplemental material
Supplementary_Material_Sage_SS_Edits - Effects of quercetin and rosuvastatin each alone or in combination on cyclophosphamide-induced premature ovarian failure in female albino mice
Supplementary_Material_Sage_SS_Edits for Effects of quercetin and rosuvastatin each alone or in combination on cyclophosphamide-induced premature ovarian failure in female albino mice by MA Elkady, S Shalaby, F Fathi and S El-Mandouh in Human & Experimental Toxicology
Footnotes
Acknowledgment
Special thanks to Dr Waleed Abdo and Dr Yomna Zamzam for helping me in carrying out the histopathological examination and helping me in photoimaging and in finishing this work. Special thanks to Dr Mohammed Balaha for helping me at the beginning of the work.
Author contributions
Conceptualization, MAE, FF, and SE; methodology, MAE and SS; writing—original draft preparation, MAE and SS; writing—review and editing, SS, FF, and SE; and supervision, SS, FF, and SE.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

