Abstract
Background
Lymphocytes are a group of white blood cells with a variety of roles their integrity is crucial for the body’s immune responses. Cadmium, a heavy metal and environmental pollutant, is known as a toxicant to exert its adverse effects on some sort of cells including blood cells.
Research Design
In this study, human lymphocytes were divided into 3 groups: (1) lymphocytes at 0-h, (2) lymphocytes at 24 h (control), (3) lymphocytes treated with cadmium chloride (15 μM). Lymphocyte viability and plasma membrane integrity were assessed in these groups. In addition, the occurrence of apoptosis was investigated by assessment of nucleus diameter and flow cytometry. Activation of caspase-3 was also detected by immunocytochemistry.
Results
Result showed that lymphocyte’s viability and plasma membrane integrity decreased in lymphocytes treated with cadmium as compared with the control group. Decreased nucleus diameter and result of flow cytometry demonstrated cadmium-induced apoptosis in human lymphocytes. Furthermore, lymphocytes treated with cadmium displayed intensely activated caspase-3 immunoreactivity in their cytoplasm.
Conclusion
In conclusion, cadmium not only negatively effect on viability and plasma membrane, but also induces caspase-dependent apoptosis in human lymphocytes.
Introduction
White blood cells are a heterogeneous group of nucleated blood cells that play an essential role in phagocytosis and the body’s immune system, defending against infection. Lymphocytes, a group of white blood cells, are comprised of three subtypes, including T cells, B cells, and natural killer cells (NK cells). T and B cells are responsible for cell-mediated, cytotoxic adaptive immunity and adaptive, humoral immunity, respectively, while NK cells are involved in innate, antigen-independent immunity. 1 Because lymphocytes play a role in defending the body against both infection and foreign particles, their integrity seems crucial for the body’s immune responses. 2
Over the past century, global industrialization has seriously polluted the environment with toxic heavy metals such as mercury, lead, and cadmium. 3 Numerous studies have demonstrated that heavy metals exert adverse effects on the immune system, leading to dysfunction and decreased cells involved in the immune system (e.g. NK cells, B and T lymphocytes). They also alter immunoglobulins levels as well as trigger inflammatory reactions.4,5
Cadmium is a major industrial and environmental pollutant widely used in the smelting, batteries, pigment, plastic industries, alloys, soldering, and irrigation. 6 Cadmium has a biological half-life (15–20 years), and humans are exposed to this toxicant through contaminated drinking water and food, air, and smoking. 7 Then, accumulated cadmium in the body damages various organs such as the liver, kidneys, lungs, pituitary-hypothalamic axis, and the male and female reproductive system. 8 Cadmium is also detected in the blood, where it mainly affects nucleated cells, in particular lymphocytes.9,10 Accordingly, cadmium was shown to decrease mature lymphocytes, increase immature lymphocytes, and reduce the activity of the immune system in rats. Additionally, cadmium by inducing oxidative stress exerts lipid peroxidation and disrupts the membrane function of rat lymphocytes. 11 Moreover, a significant decrease in viability, changes in the morphology of the nucleus, and reduction of mitochondrial membrane potential were reported in cadmium-treated human lymphocytes. 12
Cadmium causes DNA damage and apoptosis in chicken spleen lymphocytes in a dose- and time-dependent manner.13,14 Several studies also showed that cadmium induces morphological features of apoptosis, including cell shrinkage, nuclear condensation, and DNA damage in the human T lymphocyte cell lines.15,16 Depault and co-workers demonstrated that cadmium at a dose of 0–150 μM significantly increased DNA fragmentation on both metaphase and interphase chromosomes of human lymphocytes. 17 In addition, cadmium induces apoptosis in human peripheral blood lymphocytes via the p53. 18
Cadmium has toxic and carcinogenic effects in rodents and humans. However, apoptogenic or anti-apoptogenic effects of this environmental pollutant are controversial. This study was conducted to investigate the effects of cadmium on the vital parameters of human lymphocytes. We also studied the mechanism of cadmium-induced apoptosis in these cells.
Methods
Isolation of lymphocytes
Peripheral blood samples were prepared from healthy donors (16–34 years old) and used for isolating lymphocytes according to the protocol described by Dagur et al. 19 All patients signed written informed consents before the beginning of the study. The local ethics committee approved the experiments at Arak University of Medical Sciences, Arak, Iran (IR.ARAKMU.REC.1398.076). Briefly, 5 mL of the human peripheral blood was added to 5 mL phosphate-buffered saline (PBS). The samples were genteelly mixed and carefully transferred to a tube containing 4 mL Ficoll (Lymphodex) (Inno-Train, Tehran, Iran) and centrifuged at 600 g for 15 min. After centrifugation, peripheral blood lymphocytes enriched in the interface between the plasma and Ficoll were carefully gathered. The samples were then washed with PBS and centrifuged (5 min, 400 g). Then, the supernatant was discarded, and the pellet was resuspended in culture medium RPMI-1640 (Bio-Idea, Tehran, Iran) containing fetal bovine serum (FBS, 0.5%, Gibco, Life Technologies, USA). Finally, lymphocytes were counted by a Neubauer hemocytometer and calculated as the number of cells/ml.
Groping and designing experiments
The suspension containing lymphocytes and the medium (1 × 106 cells/ml) was transferred to a 24-well plate and divided into three groups (n = 6 per group): (1) lymphocytes at 0-h, (2) lymphocytes at 24 h (control), (3) lymphocytes treated with cadmium chloride (Sigma, USA, 15 μM). The lymphocytes of groups 2 and 3 were then placed in a CO2 incubator for 24 h at 37°C. Cadmium was dissolved in PBS, and the control group received the corresponding amount of PBS. To determine an effective concentration of cadmium and a sufficient incubation time, the lymphocytes were incubated with different concentrations of cadmium chloride (5, 10, 15, 25, and 30 μM) for 12, 24, 48, and 72 h at 37°C. The viability of lymphocytes was then determined using MTT, 3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyl tetrazolium bromide, assay. According to our results (data not shown), 15 μM of cadmium chloride and 24 h were considered as an effective concentration and sufficient incubation time respectively. This concentration and incubation time were then used for all experiments.
Assessment of viability
The MTT assay was used to assess the viability of lymphocytes according to a method described by Mosmann. 20 In brief, 20 μl of MTT (Sigma, USA) stock solution (5 mg/mL medium) was added to each sample containing 1 × 105 cells and incubated at 37oC in a CO2 incubator for 4 h. Then, 100 μL dimethyl sulfoxide (DMSO) was added to the samples to extract purple MTT formazan. Finally, absorbances were measured using an ELISA reader (SCO diagnostic, Germany) at 545 nm. The percentage of viable cells was then calculated.
Assessment of plasma membrane integrity
To evaluate plasma membrane integrity, a combination of acridine orange (Merck, Germany) and propidium iodide (Sigma, USA) was used. 21 Briefly, acridine orange (5 μg/mL) was added to the samples and incubated for 15 min at 37°C. Then, propidium iodide (5 μg/mL) was added to the mixture and incubated for 5 min at 37°C. The suspension was placed on a glass slide and cover-slipped. Digital photographs were taken using an Olympus camera (DP72, Japan) attached to an Olympus fluorescence microscope (Japan) at ×1000 magnification. A total of 200 lymphocytes were then counted per slide, and the percentage of lymphocytes with intact plasma membrane was recorded. In this method, the nucleus of lymphocytes with intact plasma membranes was stained green, while those with damaged plasma membranes were stained red.
Measurement of nucleus diameter
Lymphocytes stained with acridine orange (see above) were photographed using an Olympus camera (DP72, Japan) attached to an Olympus fluorescence microscope (Japan). Lymphocyte nuclei diameter was measured in all groups in 20 cells using Motic software.
Assessment of apoptosis by flow cytometry
Apoptosis was examined by a double-staining method using a FITC-Annexin V/propidium iodide (PI) apoptosis detection kit (Sigma, USA) according to the manufacturer’s protocol. Lymphocytes (1 × 105) in the control and the cadmium chloride groups were collected and centrifuged at 10,000 g for 6 min. Next, the supernatant was discarded, and the pellet was resuspended in 50 μl PBS. The samples were incubated with Annexin V and propidium iodide (PI) for 30 min at room temperature in the dark. After incubation, 500 μl PBS was added to the samples, genteelly pipetted, and analyzed by flow cytometry (CyFlow, PARTEC) using Flow jo 7.6.1 software. Cells with negative PI and Annexin V were scored as healthy cells, and PI negative and Annexin V positive cells were considered apoptotic cells, whereas those cells that were positive for both PI and Annexin V were considered necrotic cells.
Immunocytochemistry for caspase-3
Lymphocytes in the control and the cadmium chloride groups were smeared on poly-L-lysine-coated glass slides, air-dried, and fixed in 4% paraformaldehyde in PBS for 10 min. The slides were washed with PBS (3 × 5 min) and incubated with anti-rabbit cleaved caspase-3 antibody (Cell Signaling and Technology, USA) at 1:100 dilution, in a moist chamber at 4°C, overnight. After washing with PBS (3 × 5 min), the slides were incubated with Alexa Fluor®488 anti-rabbit IgG (Invitrogen, USA), labeled secondary antibody at 1:500 dilution, at room temperature for 1 h. Next, the slides were washed with PBS (3 × 5 min) and counterstained with nuclear stain, Hoechst 33,342 (10 μg/mL PBS, Sigma, USA), for 1 min at room temperature. The slides were then washed with PBS (3 × 5 min), mounted in glycerol/PBS solution (1:1), and cover-slipped. Finally, the slides were photographed using an Olympus camera (DP72, Japan) attached to a fluorescence microscope (Olympus, Japan) at ×1000 magnification.
Statistical analysis
Data were expressed as mean ± standard deviation (SD). One-way analysis of variance (ANOVA) followed by Tukey’s post hoc test was used to assess the statistical significance among different groups. A p < 0.05 was considered statistically significant.
Results
Evaluation of viability and plasma membrane integrity
viability and plasma membrane integrity in human lymphocytes treated with cadmium chloride (15 μM) for 24 h.
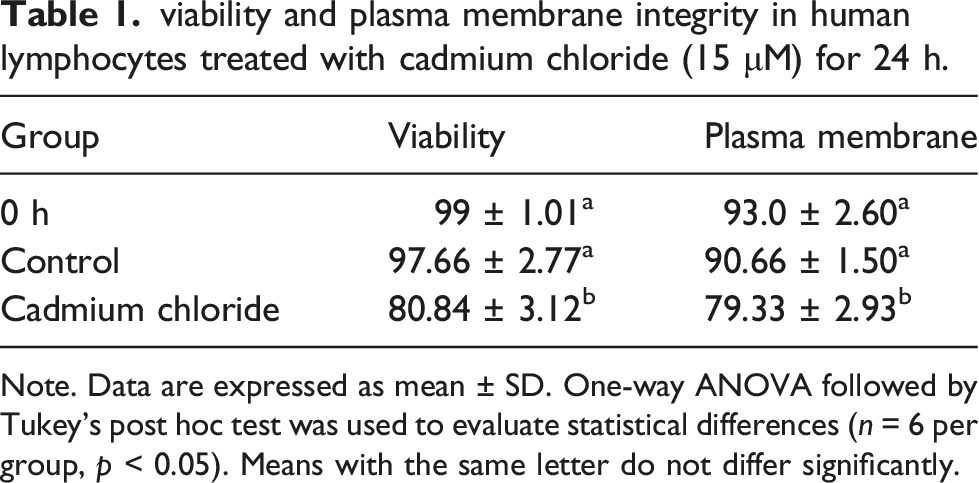
Note. Data are expressed as mean ± SD. One-way ANOVA followed by Tukey’s post hoc test was used to evaluate statistical differences (n = 6 per group, p < 0.05). Means with the same letter do not differ significantly.

Evaluation of plasma membrane integrity in human lymphocytes using acridine orange (green) and propidium iodide (red) staining. (a) lymphocytes at 0-h (b) control group (24 h) (c) cadmium chloride-treated lymphocytes (15 μM). Arrow: lymphocyte with the intact plasma membrane, arrow head: lymphocyte with the damaged plasma membrane. Magnification: 200×.
Evaluation of nucleus diameter
Lymphocytes incubated for 24 h (control group) showed no significant change in the nucleus diameter compared with 0-h lymphocytes. However, the nucleus diameter of lymphocytes treated with cadmium chloride (15 μM) significantly decreased (p < 0.001) compared to the control group (Figure 2). Evaluation of nucleus diameter in human lymphocytes at 0-h, control, and cadmium chloride-treated group (15 μM) after 24 h. Data are expressed as mean ± SD. One-way ANOVA followed by Tukey’s post hoc test was used to evaluate statistical differences (n = 6 per group, p < 0.05). Means with the same letter do not differ significantly.
Evaluation of apoptosis
According to the flow cytometry dot plot (Figure 3), 2.199% of lymphocytes underwent apoptosis in the control group, whereas in the cadmium-treated lymphocytes (15 μM), cadmium chloride induced apoptosis in 66.1% of the cells. Therefore, a significant increase (p < 0.001) was found in apoptotic lymphocytes in the cadmium chloride group compared to those of the control group. Flow cytometry dot plot with 4 parts showing the percentage of dead cells in Q1, the percentage of apoptotic cells in Q2 and Q3, and living cells in Q4 in the control group (a) and cadmium-treated group (b). In human lymphocytes treated with cadmium chloride (15 μM, 24 h), apoptosis increases to 66.1% (51 + 15.1), which is significantly different compared to the control group (2.199%) (n = 6 per group, p < 0.05).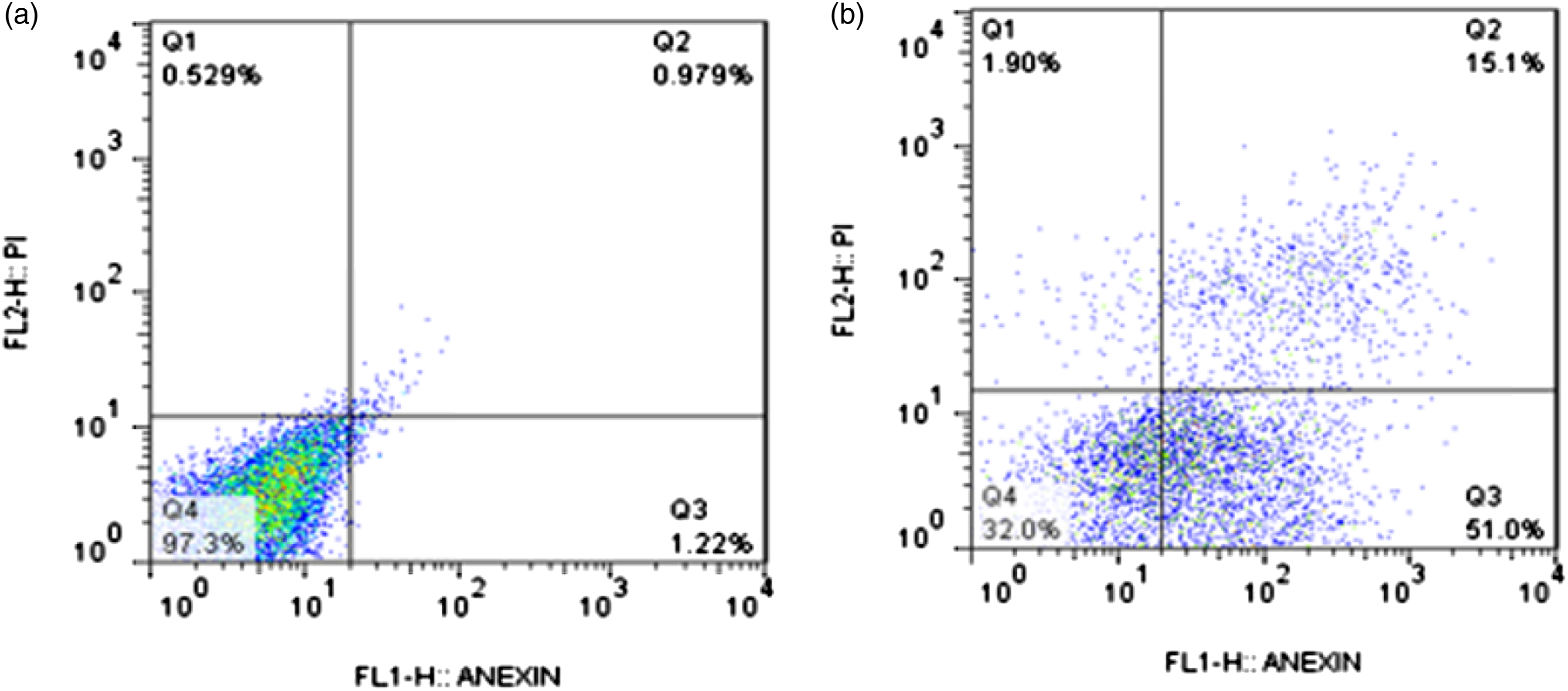
Evaluation of cleaved caspase-3 immunoreactivity
In slides stained with cleaved caspase-3 antibody, no immunoreactivity was found at both 0-h and the control group. However, in lymphocytes treated with cadmium chloride (15 μM) for 24 h, intense immunoreactivity was detected for the antibody in the cytoplasm of lymphocytes (Figure 4). Immunolocalization of cleaved caspase-3 antibody at 0-h, 24-h (control), and cadmium chloride (15 μM) groups using counterstaining for cleaved caspase-3 antibody (green) and nuclear stain Hoechst 33,342 (blue). In the cleaved caspase-3 column, no immunoreactivity was observed at 0-h and the control group. Lymphocytes treated with cadmium chloride (15 μM) for 24 h displayed intense immunoreactivity for the antibody in their cytoplasm. Magnification: 400×.
Discussion
The results of the present study illustrated that cadmium chloride not only reduced the viability and plasma membrane integrity but also induced caspase-dependent apoptosis in human lymphocytes. Oxidative stress by exerting an imbalance between the production of reactive oxygen species (ROS) and the capacity of the antioxidant defense system damages living cells and reduces their viability. 22 Heavy metals such as cadmium may reduce cell viability by increasing ROS and decreasing antioxidant defense enzymes, including catalase, glutathione peroxidase, and superoxide dismutase. 23 Oxidative stress by inducing lipid peroxidation disrupts the integrity of the plasma membrane and the membrane of organelles such as mitochondria, affecting cell function and viability. 24 Numerous studies showed that mitochondria can be the first target for cadmium toxicity.25–27 Cadmium can accumulate in the mitochondria and binds to sulfhydryl groups of mitochondrial enzymes. 28 It leads to alter mitochondrial oxidative phosphorylation and reduces ATP production. 29 Reduced ATP production will decrease the energy required for the biological activity and lymphocyte viability. Plasma membrane integrity is one of the key players that can indirectly affect cell viability. 30 There is evidence that cadmium directly inhibits the Na+/K+ pump in various cells and tissues.31–33 Considering that these pumps consume about one-third of the cell’s ATP to balance intracellular ions and maintain cell volume, depletion of cytosolic ATP following mitochondrial damage interrupts plasma membrane potential, and therefore disrupt the integrity of the plasma membrane.34,35 In addition, the high level of unsaturated fatty acids increases the susceptibility of the plasma membrane to lipid peroxidation. 36 The cascade of lipid peroxidation alters membrane fatty acids, leading to increased non-specific permeability of the membrane to ions, the inactivation of membrane receptors, carriers, and enzymes.37,38 Disruption of plasma membrane integrity can affect several cellular processes, including cell death. 39 The plasma membrane is a dynamic structure that plays important roles in chemical reactions. Many chemical compounds, including cadmium, impair plasma membrane to initiate cell inflammatory responses, leading to cell death. 40 The plasma membrane dysfunction can occur during apoptosis and necrosis. 41 Therefore, the investigation of phosphatidylserine of the plasma membrane is one of the methods to detect the occurrence of apoptosis. 42 In normal cells, the presence of phosphatidylserine is limited to the inner part of the plasma membrane, while during apoptosis the plasma membrane symmetry is lost and this phospholipid is transferred to the outer part of the plasma membrane. 43 The flow-cytometer can then detect serine on the outside of the plasma membrane of apoptotic cells by binding annexin V labeled to a fluorescent dye. 44 Both flow cytometry results (changes in plasma membrane phospholipids) and the morphological features of apoptosis (nuclear and chromatin condensation) could indicate the induction of apoptosis in the lymphocytes. Cadmium mediates apoptosis through both caspase-dependent and independent manners. 45 Therefore, in the present study, to find out the cadmium-induced apoptosis pathway in lymphocytes, the activated caspase-3 (as a key effector caspase in apoptosis) was investigated. Intensely activated caspase-3 reactivity in the cytoplasm of cadmium-treated lymphocytes could clarify the involvement of caspase-3 in the apoptosis of cadmium-exposed lymphocytes. During apoptosis, activated caspase-3 in the cytoplasm disrupts cell integrity by breaking down cytoplasmic proteins and cytoskeletal components such as fudrins and vimentins. 46 Activated caspase-3 can also proteolyze nuclear matrix proteins. 47 The nuclear lamina form structures near the nucleus membrane and around the chromatin. 48 Lamins are the targets of caspase digestion in apoptosis. 49 Breaking down of these and other nuclear skeletal proteins causes nuclear and chromatin condensation. 50 In addition, activated caspases-3 in the cytoplasm cleaves the inhibitor of caspase-activated DNase (ICAD) and activates caspase-activated DNAase (CAD). 51 After translocation to the nucleus, CAD cleaves the nuclear skeletal proteins, leading to the nucleus and chromatin condensation. 52
Apoptosis is a complex process that can independently and/or simultaneously occurs by multiple pathways.
53
More research is needed to confirm the mechanisms by which cadmium activates caspase-3 and induces apoptosis in human lymphocytes. However, Figure 5 suggests a possible mechanism involved in cadmium-induced apoptosis in the lymphocytes according to the previous studies. One possibility is that Cd disrupts cytosolic Ca+2 homeostasis through increasing Ca+2 released from intracellular Ca+2 sources such as the endoplasmic reticulum (ER).
54
Cadmium enhances intracellular Ca+2 levels via the G-protein-coupled receptor, resulting in activating phospholipase (PLC). This enzyme produces inositol 3 phosphates and IP3. IP3 triggers the release of more Ca+2 from IP3 gated Ca+2 channels (IP3 receptors, IP3R).55,56 Elevated calcium levels activate calcium-dependent proteases, in particular calpains.
57
This protease not only directly but also by activating caspase-3 cleave cytoplasmic and nuclear proteins, inducing apoptosis.
58
On the other hand, the increased intracellular calcium levels through caspase-12 activate caspase-3.
59
Cadmium also increases ROS levels and activates caspase-8 to damage mitochondrial.60,61 The dysfunction of mitochondria causes the release of apoptogenic factors including cytochrome c from mitochondria, resulting in the activation of caspase-3.
62
Assessment of apoptogenic factors such as cytochrome c, cytosolic calcium levels as well as the activity of other caspases can help to elucidate the exact mechanism(s) involved in the apoptosis of human lymphocytes treated with cadmium. Possible mechanisms involved in cadmium-induced apoptosis in human lymphocytes. Cd2+: cadmium; Ca2+: calcium; IP3R: inositol three phosphate receptor; PLC: phospholipase c; PIP2: phosphatidylinositol bisphosphate; IP3: inositol 3 phosphate.
Conclusion
Cadmium not only reduces viability and plasma membrane integrity but also induces caspase3-dependent apoptosis in human lymphocytes.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the grant from Arak University
