Abstract
The exposure to ionizing radiation has become inescapably because of increased dependence on radiation to execute works in different fields and also its influences on biological systems. Thus, the current study aimed at examination of the radio-protective effect of the natural ashwagandha (Ag) against acute and chronic doses of γ-radiation on liver and spleen of rats. The impact of Ag was inspected in rats exposed to acute exposure of 8 Gy (single dose) or to chronic exposure of 8 Gy (2 Gy every other day for 4 times). The data obtained reveals significant amelioration of the redox status (MDA, GSH and ROS) in spleen and liver tissues of rats treated with Ag and exposed to the 2-different modes of γ-radiation. Besides, the changes in inflammatory responses assessed by measurements of IL-17, IL-10 and α7-nAchR are less pronounced in rats received Ag and γ-radiation compared to irradiated rats. Further, the measurements of tissues structural damage markers (MMP-2, MMP-9 and TIMP-1) pointed to benefit of Ag against γ-radiation. The histopathological investigation of spleen and liver tissues confirmed this ameliorating action of Ag counter to γ-radiation hazards. It could be suggested that Ashwagandha could exerts radio-protective influences because of its antioxidants and anti-inflammatory capabilities.

Introduction
Exposure to ionizing radiation (IR) has become unavoidable due to its widely use in multiple fields such as medicine field (medical imaging diagnosis, and radiotherapy), sterilization of food raw materials and equipment in the development of the industry (in which radiation create negative effects that cannot be disregarded, such as radiation from electronic equipment). 1 Direct or indirect irradiation (through radiolysis of water producing reactive oxygen species, ROS), interact with macromolecules of cells (as DNA, lipid and proteins) causing oxidative damage to tissues 2 and finally organs dysfunction, through a series of biochemical and physical disturbances. This disturbance is observed by increased production of ROS leading to lipid peroxidation, oxidation of DNA and proteins as well as activation of pro-inflammatory factors 3 that subsequently led to decreased immunity and occurrence of diseases. 1
Liver, a second largest body organ, is responsible for metabolic activities, nutrient storage, and detoxification as well as maintaining homeostasis. 4 Also, liver is a pivotal player for immunologic and inflammatory responses. 5 The exposure of liver to number of risk factors such as alcohol, drugs, environmental pollutants, and radiation have a capability to inducing oxidative stress by generating free radicals that eventually resulted in various forms of severe liver damages.4,6-8 Additionally, the spleen as a largest secondary lymphoid organ in the body have a wide range of immunologic functions that makes the spleen play a central role in regulating the inflammation-related immune response besides its role in haematopoiesis.9-11
Withania somnifera, commonly known as Ashwagandha (Ag), is very revered herb and is one of the most important herbs of Ayurveda (the traditional method of medicine in India) used for its health benefits. As, the Ag is known to have different biological effects such as; anti-inflammatory, 12 antitumor, 13 anti-diabetic, 14 antioxidant, 15 cardio protective 14 and anti-stress effect. 16 Saleem et al. 17 revealed that withanolide is the pharmacologically active steroidal lactones present in W. somnifera.
Thus, the present study has been designed to investigate the effect of Ashwagandha (Ag) on damage induced by gamma irradiation in liver and spleen tissues. In order to verify this goal, this study monitored the oxidative stress status (measuring malondialdehyde; MDA, reduced glutathione; GSH and reactive oxygen species; ROS) and inflammatory status (measuring interleukin-17; IL-17, IL-10 and protein expression of α7 subtype of cholinergic nicotinic acetylcholine receptors; α7-nAchR) with biochemical structural damage marker (protein expression of Matrix Metallopeptidase-2; MMP-2, MMP-9 and tissue inhibitor of metalloproteinases; TIMP-1) in irradiated rats model and confirmed with histological examination of liver and spleen tissues.
Materials and Methods
Materials
The ashwagandha root extract used in this study is a vegetarian standardized extract produced by GNC Herbal plus ® and is distributed by General Nutrition Corporation Pittsburgh, PA 15222. The herbal supplements are provided in the form of capsules each one contains 470 mg ashwagandha root extract (Withania somnifera) and the active ingredient of this supplement is withanolides (1.5%). The other chemicals and reagents used in this study are the products of Sigma-Aldrich Chemical Co USA.
Animals
To conduct the experiments for this study, the adult male Swiss White Albino rats were adopted as experimental animals. The necessary number of rats weighing 120–150 g was obtained from the animal breeding unit of NCRRT-Egyptian Atomic Energy Authority. Upon receiving the experimental animals, they are left for acclimatize for a period of 1 week concurrent with the presence of standard commercial pellet diet and water ad libitum. The care and use of experimental animals of this study were done complied with the recommendations of National Institute of Health (NIH no 85:23, revised 1996) and in the obedience of guidelines adopted by the NCRRT ethic committee (Ref No: 38 A/21).
Radiation Facility
Experimental animals were exposed to γ-radiation doses, the subject of the current study, using the radioactive source, cesium-137, in the Canadian Gamma Cell-40, located in NCRRT- Egyptian Atomic Energy Authority. The dose rate during executing the experiment was 0.621 Gy/sec.
Experimental design
Rats were divided into six equal groups (6 rats/group) as follow: Group (1) Control (C): Rats of this group are normal rats just received physiological saline and not exposed to γ-radiation. Group (2) Ashwagandha (Ag): Rats of this group are received oral treatment of Ag at dose 300 mg/kg. wt/day 18 for 15 successive days. Group (3) Irradiated-Single dose (Rs): Rats of this group are exposed to a single dose of γ-radiation (8 Gy) at day 15 from the zero time of the experiment. Group (4) Ag+Rs: Rats of this group received Ag as in group 2 and exposed to a single dose 8 Gy as in group 3. Group (5) Irradiated-fractionated dose (Rf): Rats of this group are exposed to fractionated dose of γ-radiation (2 Gy every other day for 4 times) start from day 9. Group (6) Ag+Rf: Rats of this group are received Ag as in group 2 and exposed to fractionated dose of γ-radiation as in group 5.
On the 16th day from the zero time of the experiment and after rats being fasted overnight, they were anesthetized and sacrificed. Blood samples were collected through cardiac perforation by syringes. Tubes contain blood samples were left to coagulate and, centrifuged to separate the serum for biochemistry assessments. After scarification of animals, liver and spleen were excised, washed in ice-cold saline and divided to two parts: one part subjected for biochemistry measurements and the other part for histopathological examinations.
Biochemical Assay
The oxidative status was evaluated in serum and tissues (liver and spleen) via measuring the malondialdehyde (MDA) (as indicator of lipid peroxidation), 19 and reduced-glutathione (GSH) (as indicator of antioxidant level). 20 Activities of Aspartate amino-transferase (AST) and alanine amino-transferase (ALT) was determined in serum according to the method of Reitman and Frankel 21 to check liver functional status. 22 All photometric methods were done using (Thermo Electron UV-Visible Spectrophotometers U.S.A). The level of ROS (CAT#MBS169261), IL-17 (CAT#MBS164772) and IL-10 (CAT#MBS702776) was estimated by ELISA kits My BioSource according to the manufacturer’s instructions, for the enzyme-linked immunosorbent assays via ELISA microplate reader (DV990 BV 416; Gio. DE VITA and CO., Rome, Italy).
Western blot analysis
The protein expression of MMP-2, MMP-9, TIMP-1 and α7-nAchR in both tissues liver and spleen was performed according to Omar et al. 23 via using a homogenization lysis buffer (Sigma-Aldrich, St Louis, MD, USA). After centrifugation of tissue lysate for 20 mints (at 8678g, at 4°C), it was collected and protein concentration was measured via a BCA-protein kit (Thermo Fisher Scientific). Then, from each sample, 7.5 μg of protein was denatured and loaded on to 8% of SDS–PAGE (sodium dodecyl sulphate-polyacrylamide gel electrophoresis). After that and throughout a semidry transfer apparatus (Bio-Rad, Hercules, Calif.), it was transferred to a nitrocellulose membrane (Amersham Bioscience-Piscataway-New Jersey-USA). The obtained membranes were incubated overnight at 4°C with milk blocking buffer (at 5%) containing the following: Tris-HCl (10 mmol/L at pH=7.4), NaCl (150 mmol/L) and TBST (Tris-buffered saline with Tween-20, 0.05%). Next, the membranes were washed with TBST and incubated with a dilution of anti-TIMP-1 (CAT# 580-RT-010, 21.5 kDa), anti-MMP-2 (CAT# 924-MP-010, 71 kDa) from R&D Systems, anti-MMP-9 (CAT# ab76003, 78 kDa, abcam) and anti-α7-nAchR (CAT# sc-58,607, 55 kDa, Santa Cruz Biotechnology) antibodies overnight at 4°C on a roller shaker. By the indicated primary antibody, the immunoblotting was done and then the appropriate horseradish peroxidase (HRP) conjugated goat immunoglobulin (Amersham Biosciences) followed it. The chemiluminescence detection was performed according to the manufacturer’s protocols of amersham detection kit, and then exposed to X-ray film. The protein level of each sample was quantified by densitometric analysis of the auto-radiograms through using a scanning laser densitometer (Biomed Instruments). The records obtained were assayed after normalization to β-actin protein expression (that used as the housekeeping protein). 24
Histopathological examination
Autopsy samples from the liver and spleen tissues of rats in different groups were taken and fixed in formalin saline (at 10%) for 24 h. Then, washing with tap water was done and for dehydration, a serial dilution of alcohol (methyl, ethyl and absolute ethyl) was used. After that, specimens were cleared in xylene and at 56°C in hot air oven for 24 h was embedded in paraffin. The paraffin bees wax of tissues blocks was prepared and sectioning at 4 mm thickness by slide microtome. Finally, the obtained tissue sections were collected on glass slides and de-paraffinised then stained by hematoxylin and eosin (H&E) stain for routine examination via the light electric microscope according to the method of Banchroft et al. 25
Statistical analysis
All statistical analysis was carried out using ANOVA (one-way analysis of variance) followed by Tukey-Kramer post hoc multiple comparisons test by Graph Pad Prism® version 8.00 (San Diego, CA, USA) software. The significant differences were considered statistically at p < 0.05. Data were presented as mean ± SEM (standard error of the mean).
Results
Protective effect of ashwagandha against oxidative stress induced by γ-radiation
In liver, the data of Ag group revealed a non-significant change in ROS, MDA and GSH levels when compared to control as observed in data presented in Figure (1). Irradiation of rats caused oxidative stress confirmed by a significant elevation (p<0.05) of MDA (by 1.55 fold change and 1.77 fold change), ROS levels (by 2.11 fold change and 2.53 fold change) with a significant decline (p<0.05) in GSH contents (by 40.25% and 42.05%) as a response to irradiation induced oxidative stress in Rs and Rf groups, respectively compared to control group Figure (1). This effect was significantly improved with concurrent Ag treatment, showed by significant decrease (p<0.05) in MDA (by 42.47% and 38.02%) and ROS levels (31.37% and 35.18%) with significant increase (p<0.05) in GSH content by 1.43 fold change and 1.71 fold change compared to Ag+Rs and Ag+Rf groups, respectively as compared to Rs and Rf group, respectively Figure (1). Protective effect of ashwagandha against oxidative stress induced by γ-radiation in liver and spleen tissue. Each value represents Mean± SEM (n=6). Columns denoted with ״a״ significant from C, ״b״: significant from Ag, ״c״ significant from Rs, ״d״ significant from Ag+Rs, ״e״ significant from Rf and ״f״ significant from Ag+Rf, at (p <0.05). C: normal rats, Ag: rats treated with Ag, Rs: rats exposed to 8 Gy, Ag+Rs: rats treated with Ag and exposed to 8 Gy, Rf: rats exposed to (2 Gy every other day for 4 times) and Ag+Rf: rats treated with Ag and exposed to (2 Gy every other day for 4 times).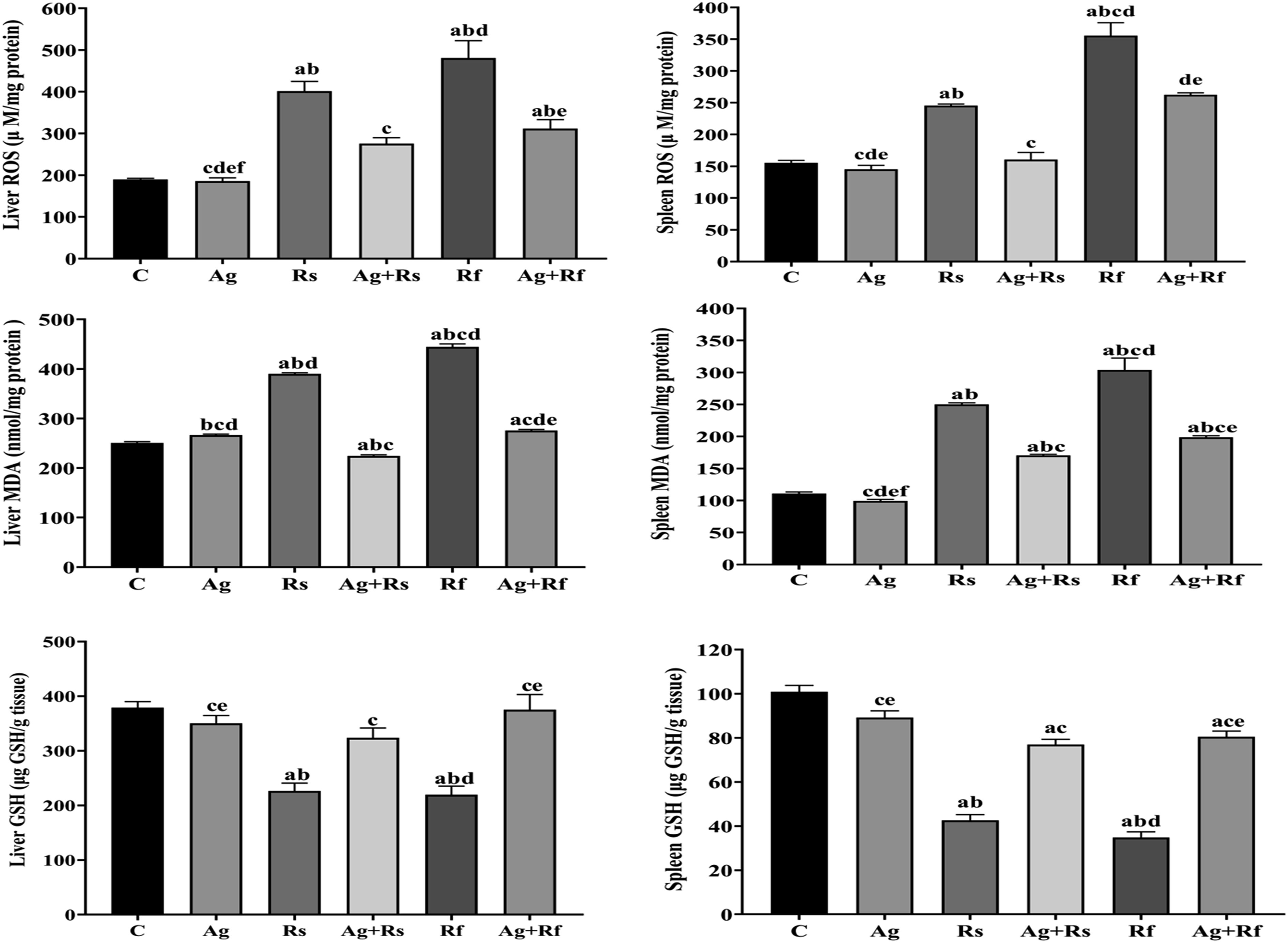
In spleen, the data of Ag group showed a non-significant change in ROS, MDA and GSH levels when compared to control Figure (1). In irradiation groups, the data of Rs and Rf groups observed a significant elevation (p<0.05) in MDA (by 2.26 fold change and 2.75 fold change) and ROS (by 1.58 fold change and 2.28 fold change) with significant reduction (p<0.05) in GSH values (by 57.78% and 65.41%) as compared to control group. The oxidative stress status was significantly enhanced with Ag treatment as showed by significant decrease (p<0.05) in MDA (by 31.89% and 34.59%) and ROS levels (by 34.62% change and 26.16% change) associated with a significant increase (p<0.05) in GSH activity (by 1.81-fold change and 2.30-fold change) in Ag+Rs and Ag+Rf groups respectively, when compared to Rs and Rf group, respectively Figure (1).
Data presented in Figure (2) illustrated the oxidative stress of Ag group in serum that revealed a non-significant change in ROS, MDA and GSH levels as compared to control group Figure (2). A significant increase (p<0.05) in MDA (by 3.00 fold change and 3.21 fold change) and ROS levels (by 2.09 fold change and 2.69 fold change) with a significant decrease (p<0.05) in GSH content (by 63.27% and 55.56%) were observed in Rs and Rf groups compared to control group Figure (2). In Ag+Rs and Ag+Rf groups, the results showed a significant decrease (p<0.05) in MDA (by 42.90% and 38.07%) and ROS levels (by 38.50% and 44.54%) respectively, linked with a significant increase (p<0.05) in GSH activity (by 2.39-fold change and 1.89-fold change) respectively, when compared to Rs and Rf groups respectively, Figure (2). Protective effect of ashwagandha against oxidative stress and inflammation induced by γ-radiation in serum. Each value represents Mean± SEM (n=6). Columns denoted with ״a״ significant from C, ״b״: significant from Ag, ״c״ significant from Rs, ״d״ significant from Ag+Rs, ״e״ significant from Rf and ״f״ significant from Ag+Rf, at (p <0.05). C: normal rats, Ag: rats treated with Ag, Rs: rats exposed to 8 Gy, Ag+Rs: rats treated with Ag and exposed to 8 Gy, Rf: rats exposed to (2 Gy every other day for 4 times) and Ag+Rf: rats treated with Ag and exposed to (2 Gy every other day for 4 times). Defensive effect of ashwagandha against inflammation induced by γ-radiation.
Defensive effect of ashwagandha against inflammation induced by γ-radiation
In liver, the protein levels of IL-17, IL-10 and protein expression of α7-nAchR obvious a non-significant change in Ag group when compared with control group Figure (3). While, the results of radiation exposure in Rs and Rf groups manifested a significant raise (p<0.05) in IL-17 (2.26-fold change and 2.53-fold change) respectively, as well as a significant diminish (p<0.05) in IL-10 (56.11% and 59.78%) respectively, and in protein expression of hepatic α7-nAchR (82.76% and 79.30%) respectively, when compared to control group Figure (3). In Ag+Rs and Ag+Rf groups, the Ag treatment shown a significant decline (p<0.05) in IL-17 (38.20% and 31.77%) respectively, with a significant height (p<0.05) in IL-10 (1.94 fold change and 2.07 fold change) respectively, and protein expression of hepatic α7-nAchR (3.64 fold change and 2.88 fold change) respectively, as compared to Rs and Rf groups, respectively Figure (3). Defensive effect of ashwagandha against inflammation induced by γ-radiation in liver and spleen tissue. Each value represents Mean± SEM (n=6). Columns denoted with ״a״ significant from C, ״b״: significant from Ag, ״c״ significant from Rs, ״d״ significant from Ag+Rs, ״e״ significant from Rf and ״f״ significant from Ag+Rf, at (p <0.05). C: normal rats, Ag: rats treated with Ag, Rs: rats exposed to 8 Gy, Ag+Rs: rats treated with Ag and exposed to 8 Gy, Rf: rats exposed to (2 Gy every other day for 4 times) and Ag+Rf: rats treated with Ag and exposed to (2 Gy every other day for 4 times). Modulatory effect of ashwagandha against structural damage induced by γ-radiation.
The inflammatory status of spleen in Ag group showed a non-significant change in protein levels of IL-17, IL-10 and protein expression of splenic α7-nAchR when compared with control group Figure (3). Whereas, the data of Rs and Rf groups displayed a significant increase (p<0.05) in IL-17 (1.83-fold change and 2.17-fold change) respectively, associated with a significant decrease (p<0.05) in IL-10 (44.17% and 52.40%) respectively, and in protein expression of splenic α7-nAchR (76.49% and 72.19%) respectively, as compared to control group Figure (3). Administration of Ag revealed a significant reduction (p<0.05) in IL-17 (33.72% and 31.64%) respectively, connected with a significant elevation (p<0.05) in IL-10 (1.43 fold change and 1.93 fold change) respectively, and protein expression of splenic α7-nAchR (2.82 fold change and 2.75 fold change) respectively, Ag+Rs and Ag+Rf groups as compared to Rs and Rf groups, respectively Figure (3).
Additionally, as shown in Figure (2), the systemic inflammation of Ag group displayed a non-significant change in IL-17 and IL-10 levels of serum when compared to control group. But, the serum of rats exposed to γ-radiation in Rs and Rf groups showed a significant increase (p<0.05) in IL-17 by 2.68 fold change and 3.09 fold change and decrease in IL-10 by 58.72% and 65.62% respectively, compared to control group Figure (2). While, Ag administration in Ag+Rs and Ag+Rf groups manifested a significant decreased (p<0.05) in IL-17 concentrations, by 44.45% change and 36.77% change, respectively accompanied with a significant increased (p<0.05) in IL-10 (1.89-fold change and 2.06-fold change, respectively) when compared to Rs and Rf groups, respectively Figure (2).
Modulatory effect of ashwagandha against structural damage induced by γ-radiation
To study the effect of γ-radiation on liver injury, the protein expression of MMP-2, MMP-9 and TIMP-1 in liver tissue with liver function enzymes in serum (ALT and AST) was detected. In the Ag group, the results revealed a non-significant change in those indicators when compared to control group (Figure 2 and 4). Whereas, the finding of these indicators in Rs and Rf groups displayed a significant enlarge (p<0.05) in expression of MMP-2 (4.13 by fold change and 4.52 fold change, respectively), MMP-9 (6.41 by fold change and 5.85 fold change, respectively), ALT (4.69 by fold change and 4.17 fold change, respectively), AST (2.33 by fold change and 2.13 fold change, respectively) with a significant decrease (p<0.05) in TIMP-1 (77.29% and 71.37%) as compared to control group (Figure 2 and 4). The data observation of Ag+Rs and Ag+Rf groups showed a significant modulation (p<0.05) in these indicators after Ag administration as follow; decrease in MMP-2 (53.95% and 46.81%, respectively), MMP-9 (54.41% and 50.48%, respectively), ALT (51.39% and 55.73%, respectively), AST (44.22% and 35.82%, respectively) and increase in TIMP-1 (2.68 fold change and 2.52 fold change, respectively) as compared to when compared to Rs and Rf groups, respectively (Figure 2 and 4). Modulatory effect of ashwagandha against structural damage induced by γ-radiation in liver and spleen tissue. Each value represents Mean± SEM (n=6). Columns denoted with ״a״ significant from C, ״b״: significant from Ag, ״c״ significant from Rs, ״d״ significant from Ag+Rs, ״e״ significant from Rf and ״f״ significant from Ag+Rf, at (p <0.05). C: normal rats, Ag: rats treated with Ag, Rs: rats exposed to 8 Gy, Ag+Rs: rats treated with Ag and exposed to 8 Gy, Rf: rats exposed to (2 Gy every other day for 4 times) and Ag+Rf: rats treated with Ag and exposed to (2 Gy every other day for 4 times). The data of β-actin (housekeeping) were found previously in Figure 3.
In the spleen, the structural damage of tissue in Ag group revealed a non-significant change in the protein expression of MMP-2, MMP-9 and TIMP-1 when compared to control group Figure (4). Whilst, the detection of these proteins in Rs and Rf groups manifested a significant augment (p<0.05) in expression of MMP-2 (by 6.47 fold change and 6.98 fold change, respectively), MMP-9 (by 7.61 fold change and 7.70 fold change, respectively) with a significant decrease (p<0.05) in TIMP-1 (43.79% and 70.00%) as compared to control group Figure (4). On the other hand, the Ag+Rs and Ag+Rf groups showed a significant improvement (p<0.05) in these proteins after Ag administration as follow; decrease in MMP-2 (54.89% and 53.63%, respectively), MMP-9 (50.00% and 49.59%, respectively) and increase in TIMP-1 (1.58 fold change and 2.65 fold change, respectively) when compared to Rs and Rf groups, respectively Figure (4).
Histopathological examination of liver and spleen
The histopathological examination of the liver and spleen tissues of different animal groups was showed in Figure 5. Histopathological examination of Liver and Spleen in different animal groups. The tissues sections stained with hematoxylin and eosin, magnification ×400 (liver; H&E x 400) and ×200 (spleen; H&E x 200). C: normal rats, Ag: rats treated with Ag, Rs: rats exposed to 8 Gy, Ag+Rs: rats treated with Ag and exposed to 8 Gy, Rf: rats exposed to (2 Gy every other day for 4 times) and Ag+Rf: rats treated with Ag and exposed to (2 Gy every other day for 4 times).
Liver tissue: The C-group showing normal hepatic parenchyma; note the organized hepatic cords with normal polygonal hepatocytes and central nucleus (thick arrows), the normal central vein (c) and blood sinusoids (thin arrows) (Lesion Score: 0). The Ag-group showing normal hepatic parenchyma; note the organized hepatic cords with normal polygonal hepatocytes and central nucleus (thick arrows), the normal central vein (c) and blood sinusoids (thin arrows) (Lesion Score: 0). The Rs-group showing Severe inflammatory reaction; note the congested hepatoportal blood vessel (arrow head) and blood sinusoids (*), together with mononuclear cells infiltration (thin arrows). The hepatocytes are atrophied (thick arrows) (Lesion Score: ++). The Ag+Rs group showing amelioration in the inflammatory reaction into normal hepatic parenchyma; note the organized hepatic cords with normal polygonal hepatocytes and central nucleus (thick arrows), the normal central vein (C) and blood sinusoids (thin arrows) (Lesion Score: 0). The Rf-group showing diffuse inflammation and degeneration; note the congestion of the blood sinusoids (*) and the leucocytic cells infiltration in the portal tract (thick arrows), together with vacuolated hepatocytes (thin arrows) (Lesion Score: +++). The Ag+Rf group showing amelioration in the degeneration with slight inflammatory reaction; note the organized hepatic cords with normal polygonal hepatocytes and central nucleus (*), the hepatoportal blood vessel is congested (arrow head) (Lesion Score: +).
Spleen tissue: The C-group showing normal splenic parenchyma; note the normal splenic follicles (arrow head), normal white bulb, and normal red bulb (arrows) (Lesion Score: 0). The Ag-group showing normal splenic parenchyma; note the normal splenic follicles (arrow head), normal white bulb, and normal red bulb (arrows) (Lesion Score: 0). The Rs-group showing Severe inflammatory reaction; note the congestion of the blood vessel (arrow head) and red bulbs (*), together with mononuclear cells infiltration (thin arrow), The splenic follicles are atrophied (thick arrow) (Lesion Score: ++). The Ag+Rs group showing amelioration in the inflammatory reaction into normal splenic parenchyma; note the normal splenic follicles (arrow head), normal white bulb, and normal red bulb (arrows) (Lesion Score: 0). The Rf-group showing focal inflammatory reaction and diffuse degeneration; note the focal leucocytic cells infiltration (arrow) and congested red bulbs (*). The splenic follicles are depleted and degenerated (arrow head) (Lesion Score: +++). The Ag+Rf group showing amelioration in the degeneration with slight inflammatory reaction; note the normal splenic follicles (arrow head). The red bulb is congested (*) (Lesion Score: +).
Discussion
Scientists working in the field of biological science pay attention to decrease the possible bad effects of using radiation on biological systems. For that, researches tried to find the best way to prevent the undesirable effects of radiation simultaneous with raising the benefits from using, depending on natural substances with plant origin. Thus, the present study aims to protect vital organs like liver and spleen against γ-radiation exposure (acute; Rs or chronic exposure; Rf) through exploring efficacy of ashwagandha (Ag) against the different mode of γ-radiation on biochemical and histological examination.
The obtained results indicate significant increase in the ROS and MDA levels associated with decrease in GSH levels of serum, liver and spleen in rats exposed to γ-radiation (acute or chronic) when compared to the ROS equivalent values in control rats (Figure 1 and 2). This hazard effect could be attributed to the mechanism of γ-radiation action that occurs through radiolysis of water (whereas cells and tissues constitute mainly of ∼80% water) and induces the production of ROS. 26 These radical species damage cellular components (lipid, protein and DNA), affect the antioxidant system (by plummeting the intracellular concentration of GSH, superoxide dismutase (SOD), and catalase activities) and can induce irreversible genetic defects causing a carcinogenesis, necrosis or apoptosis.26-27 Several studies have reported that the detrimental effects evolved after irradiation were connected to the status of oxidative stress and imbalance of oxidation/reduction (redox) system.28-30 It seemed that, the ROS generated after γ-radiation exposure attacks antioxidant molecules and resulted in consumption of liver and spleen contents of GSH in ROS neutralization process. In addition, the plenty of ROS interact with the lipid molecules that organized in the cellular structures of liver and spleen causes increasing in tissue contents of MDA which leads in turn to loss of cellular and tissues redox tone and finally the development of oxidative stress.7,9, 31-32 On the other hand, a significant improvement in the ROS generation, GSH and MDA levels was observed when Ag was administrated before exposure to γ-radiation (Rs) or before and during exposure to fractionated γ-radiation (Rf) in liver and spleen (Figure 1 and 2). The antioxidant capabilities of Ag that was observed in the current study could be contributed to its mechanisms of free radicals and ROS neutralization as well as obstruction of ROS production paths that enhanced after radiation exposure. Whereas, study of Shahriar et al. 33 stated that Ag might be considered as a potential source of antioxidants and thus could prevent many radical related disorders. Also, it was found that Ag induced a reduction in hepatic enzymes, MDA levels and DNA damage associated with elevation in SOD, glutathione peroxidase activities and GSH content in rats exposed to 6 Gy. 34
Furthermore, the exposure of rats to γ-radiation leads to disorders in the pro-inflammatory and anti-inflammatory balances in serum, liver and spleen evidenced by increases in the pro-inflammatory; IL-17 and decreases of anti-inflammatory molecules; IL-10 and α-7nAchR (Figure 2 and 3). This might be the consequence of oxidative stress evolved after γ-radiation exposure. Whereas, Yahyapour et al. 35 reported that the change in redox status is responsible for the acute injuries induced by radiation as well as the early and late belongings encompasses inflammation, bystander effects, apoptosis and fibrosis. Thus, the exposure to γ-radiation induced a status of imbalance between pro-inflammatory (such as NF-κB and IL-6) and anti-inflammatory (IL-10 and α-7nAchR) secretion.29-30 Also, gamma irradiation endorses the secretion of inflammatory cytokine IL-17 that applies in a variety of biological effects by binding to its receptors which broadly expressed on the surface of numerous cells and has imperative roles in encouraging inflammation. 36 On contrary, the administration of Ag according to the present protocol reveals significant ameliorations in the inflammatory responses of rat’s groups exposed to γ-irradiation. This could be ascribed to the antioxidant role of Ag against oxidative stress as observed above, whereas oxidative stress is one of the main stimulants to inflammatory reactions. Furthermore, it was found that Ag exhibited anti-inflammatory effect via inhibit the release and suppress the expression of inflammatory cytokines.14,37 The α-7nAchR is a primary receptor of the CAP (cholinergic anti-inflammatory pathway) that reveals anti-inflammatory reactions as well as the immune-modulatory response to keep immune homeostasis. 30 The study of Gao et al. 38 found that the oxidative stress down-regulate α-7nAchR expression. Therefore, it could be explained the role of Ag in elevating the expression of α-7nAchR to its antioxidant abilities against ROS attacks and subsequent raised of oxidative stress. Whereas, the study of Han et al. 39 found that reduction of inflammatory response and oxidative stress play roles in α-7nAchR-protective action.
Matrix metalloproteinase (MMPs) plays a crucial role in diverse processes including physiological and pathological conditions. MMPs shares a structural homology through cleaving certain component of the extracellular matrix via a zinc-binding site since they are secreted in a latent state as zymogen then undergo activation to exhibit a particular function in tissue remodelling. MMPs are inhibited by some specific tissue inhibitors, namely TIMPs. 40 The disturbances occurred in oxidative stress and inflammatory responses in liver and spleen after γ-radiation exposure is accompanied with loosing of balances between those regulators MMP-2, MMP-9 and its inhibitors TIMP-1 expression. Galis and Khatri 41 related the imbalance between MMPs and TIMPs, and the promotion of vascular remodelling to the induction of oxidative stress and the development of inflammatory responses. 30 Also, Knight et al. 42 establish that unresolved inflammation is a significant predictor of this status of imbalance. Additionally, it was found that IL-10 increases the production of TIMP-1 expression. 43 Therefore, it could be hypothesizing that the decreases of TIMP-1 expression associated with increase in MMP-2 and -9 expressions may cause tissue damage as observed in biochemical and histopathological investigation in the current study (Figure 4 and 5). Evidently, the histopathological examinations comprised in the present study might emphasis the biochemical changes discussed above and outlined the damages and changes induced by γ-radiation exposure in liver and spleen tissues architectures, which is confirmed with failure in liver functions enzymes (ALT and AST). 30 On the other side, administration of Ag before irradiation displays significant modulation of the biochemical and histopathological measurements encompassed in the current study (Figure 4 and 5). This could be explained in the light of Ag antioxidant and anti-inflammatory properties as reported previously in the finding of Kumar and Patnaik 44 and the study of Sajida and Prabhu. 45 This occurred through reduction of the ROS generation, prevention the accumulation of harmful products resulted from ROS oxidative attacks of cellular macromolecules, as well as other biochemical variations occurs as a cascade of inflammatory responses.
In conclusion, the administration of ashwagandha root extract might reduce inflammation and protect tissue structure of liver and spleen after exposure to γ-radiation. Ashwagandha reduce ROS released in abundant because of γ-radiation exposure, prevents exacerbation of oxidative situation which ended by fall down of antioxidant system and accumulation of toxic organic radicals (lipid peroxidation products). Subsequently, modulate the inflammatory cascades of oxidative stress that not preceded forward echoes on the balance between expression of MMP (−2 and −9) and TIMP-1 expression.
Footnotes
Acknowledgements
Authors acknowledge Dr Sherein Saied Abd-Elgayed (Professor of Histopathology, Pathology Department, Faculty of Veterinary Medicine, Cairo University, Egypt) for accomplishing the Histopathological examination in the current study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
