Abstract
Testicular torsion is an emergency, mainly in newborn and adolescent males, resulting in testicular ischemia. The current study aimed to evaluate the effect of Idebenone (IDE) on testicular torsion/detorsion (T/D) in juvenile rats. Thirty-two rats were randomized into: (1) the sham group: rats received sham operations with no other interventions; (2) the IDE group: rats received idebenone (100 mg/kg, i. p) without T/D; (3) the T/D group: rats underwent torsion for 2 h and detorsion for 4 h; and (4) the IDE+ T/D group: rats received IDE 1 h before T/D. Testicular malondialdehyde (MDA), total nitrite/nitrate (NOx), total antioxidant capacity (TAC), tumor necrosis factor-α (TNF-α), caspase-3, sirtuin type 1 (Sirt1), serum interleukin-1β (IL-1β), total cholesterol, and testosterone were measured. Histological changes, nuclear factor (erythroid-derived 2)-like-2 factors (Nrf2), and proliferating cell nuclear antigen (PCNA) immuno-expressions were assessed. T/D displayed an increase in MDA, NOx, TNF-α, caspase-3

Highlights
(1) Testicular torsion is an emergency mainly in newborn and adolescent males, (2) Idenenone ameliorates testicular ischemia reperfusion injury in juvenile rats, (3) Idenenone exhibits anti-inflammatory, anti-apoptotic and antioxidant properties, (4) Idenenone regulates the Sirt1/Nrf2/TNF-α signaling pathway.
Introduction
A severe acute emergency, mainly in newborn and adolescent males, is testicular torsion. It occurs when the spermatic cord is distorted, resulting in obstruction of the blood supply and testicular ischemia. 1 Early management is essential to prevent subfertility, developing into testicular atrophy and infertility. Ischemia and reperfusion (I/R) are the main pathophysiological consequences of testicular torsion, which are caused by the twisting of the spermatic cord followed by detorsion. 2 Reperfusion of the testes following torsion causes testicular injury that is detrimental and more severe than ischemia. Testicular damage varies based on the torsion’s severity and duration.3,4
Germ cell death that occurs with T/D is due to a decrease in the level of oxygen supply relative to metabolic demands, thus accumulating toxic compounds with the reduction of stored ATP. 5 On the other hand, testicular detorsion after torsion increases proinflammatory cytokines, e.g. interleukin-1β and TNF-α, which recruit neutrophils to infiltrate the testicular parenchyma. 6 Neutrophils are the main source of reactive oxygen species (ROS) in ischemic injury, damaging proteins, lipids, and DNA. 7
Nuclear factor (erythroid-derived 2)-like-2 factors (Nrf2) shows cytoprotective effects against oxidative stress and inflammation as it has a role in the elimination of free radicals. 8 Also, Sirtuin type 1 (Sirt1) is a member of the vast Sirtuin family that regulates cell death/survival and stress responses through histone deacetylase activity. 9 Modulation of these parameters could explain how testicular T/D develops and ameliorates it.
Idebenone (IDE), a potent antioxidant, is a synthetic quinone with fewer and shorter lipophilic ends than coenzyme Q10 (CoQ10). IDE is a vital cell membrane antioxidant. It is effective in hypoxia as its antioxidant effect prevents lipid peroxidation and removes oxygen-free radicals. It protects cells from mitochondrial damage and apoptosis in human umbilical vein endothelial cells. 10 Also, IDE inhibits oxidative stress following endothelial cell injury and limits atherogenesis. 11 Interestingly, IDE can activate Nrf2 and its dependent genes. 12 IDE has an anti-inflammatory effect on lupus, neuroinflammation, ulcerative colitis, sepsis, nano-toxicity, atherosclerosis, and drug-induced inflammation. 13
Unfortunately, no proven appropriate therapy for testicular T/D does not involve surgical reduction. Even though research is focused on developing new prospective pharmacological treatments in clinical practice, there is still work to be done. Given previous reports on the efficacy of IDE, the current study was designed to investigate the ameliorating action of IDE in a model of testicular I/R injury caused by testicular T/D in juvenile rats, as well as the possible mechanisms mediating its action, with a focus on the Sirt1/Nrf2/TNF-α signaling pathway.
Materials and methods
Ethics
Animal procedures and treatments were performed in accordance with the Institutional Ethical Committee’s (Faculty of Medicine, Minia University, Egypt) requirements for the care of experimental animals and the NIH Guide for the care and use of laboratory animals (Approval number 7892021).
Chemicals
IDE was obtained from Santhera pharmaceuticals, Switzerland. Total antioxidant capacity (TAC) was measured colorimetrically using commercial kits (Biodiagnostic, Egypt). Interleukin-1β (IL-1β) was measured using ELISA kits from (Nanjing jiancheng Bioengineering, China), tumor necrosis factor- α (TNF-α) was assessed using ELISA Kits from (Elabscience Biotechnology Co Ltd, USA), and Caspase-3 was assessed using ELISA kits from (Cusabio, USA). Testosterone was measured using ELISA kits from (DRG, Germany), and total serum cholesterol was quantified colorimetrically using commercial kits (Biomed, Egypt). Carboxymethylcellulose (CMC) was obtained from (El-Nasr Pharmaceutical Co, Egypt). Rabbit anti-beta Actin antibody (1:1000, ab8227, abcam) and Rabbit anti-Sirt1 antibody (1:1000, ab189494, abcam) were used.
Animals and experimental design
Animals
Pregnant Wister female rats were kept individually under ordinary laboratory circumstances, including 12 h light: darkness cycles (light on at 06:00 h), controlled temperature (23 ±2°C), and free access to food and water. After parturition, the newborn rats were weaned at 21 days of age, randomized into four groups of eight males each, and used for experimentation.
Experimental design
Thirty-two rats were divided into four groups (8 rats/group): (1) the sham group: rats received sham operations with no other interventions; (2) the IDE group: rats received idebenone (100 mg/kg, i. p) (11) without T/D; (3) the T/D group: rats underwent torsion for 2 h and detorsion for 4 h (1); and (4) the IDE+ T/D group: rats received IDE 1 h before T/D (1).
The T/D procedures were 6 h long. To anesthetize rats, pentobarbital sodium, 50 mg/kg, was given intraperitoneally. All surgical procedures were performed by the same surgeon in sterile settings. A left-sided ilioinguinal incision was made to expose the left testis. The left testis was exposed through the incision, and an 11–0 atraumatic silk suture was inserted through the tunica albuginea in the sham group. A 4–0 silk suture was used to seal the incision and return the left testis to the scrotum. The left testis was rotated 720° counterclockwise in the T/D group to induce testicular ischemia, and the left testis was sutured to the scrotal wall with an 11–0 atraumatic silk suture. The fixing stitch was removed after 2 h of ischemia. The left testis was freed clockwise to induce reperfusion (maintained for 4 h), and bilateral orchiectomies were conducted at the end of the experiment.14,15
Sample collection
Rats were euthanized at the end of the experiment (after 6 h from the initiation of the T/D treatment) under anesthesia by using urethane hydrochloride (1 g/kg i. p.). Blood samples were obtained at room temperature from the rats' abdominal aortas in heparinized syringes. It was then centrifuged for 15 min at 4000 g. (Janetzki T30 centrifuge, Leipzig, Germany). After that, the serum layer was removed and stored at −80°C for biochemical analysis. Testis was divided into three parts. The first part was immersed in Bouin’s fluid for histological examination. The second part was used for immunoblotting. The third part was homogenized in 20% w/v ice-cold phosphate buffer (pH 7.4) centrifuged at 4000 g for 15 min. The supernatant was kept at 80°C for further analysis.
Biochemical analysis
Assessment of testicular markers
Total serum cholesterol and testosterone were measured following the manufacturer’s instructions.
Assessment of oxidative stress parameters
To assess testicular T/D oxidative stress biomarkers, lipid peroxidation was quantified as a thiobarbituric acid-reacting substance and displayed as equivalents of malondialdehyde (MDA) using 1,1,3,3-tetra methoxy propane as a standard. 16
Total nitrite, the stable oxidation end product of nitric oxide (NOx), was estimated after reducing nitrate to nitrite by copperized cadmium. The concentration of nitric dioxide (NO2–) was based on the Griess reaction, the reaction of nitrite with a mixture of naphthyl ethylenediamine and sulfanilamide. 17
TAC was assessed colorimetrically using commercial kits (Biodiagnostic, Egypt).
Assessment of inflammatory and apoptotic parameters
Serum IL-1β, testicular TNF-α, and caspase-3 were quantified following the manufacturer’s instructions of their kits.
Immunoblotting of sirt1
Testis homogenates (50 μg of total proteins) were boiled for 5 minutes with 2-mercaptoethanol containing loading buffer and then applied to 12% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) to do running for 2 hours at 100 V. After electrophoresis, proteins were applied to polyvinylidene fluoride (PVDF) membranes. Blocking for 1 hour was done in a Tris-buffered saline (TBS-T) solution, which contained 5% (w/v) non-fat milk and 0.05% Tween-20. Incubation with primary antibodies (1:1000) for rabbit antiSirt1 and β-actin overnight at 4°C was done. A secondary antibody of horseradish peroxidase-conjugated polyclonal goat anti-rabbit immunoglobulin was added at a dilution of 1:5000 in blocking buffer. Chemiluminescence was used for the visualization of bands with the help of chemiluminescence kits using a luminescent image analyzer (LAS-4100, Fujifilm Co, Japan). Applying Image J Software, protein bands of different groups were quantified as fold changes relative to the sham group after normalization to β-actin.
Histological procedures
Histological study
Testicular tissue sections from all groups were fixed in 10% formalin, embedded in paraffin, and dehydrated in ascending concentrations of ethyl alcohol (70%–100%). 5 µm tissue sections were stained with hematoxylin and eosin (H&E). 18
Immunohistochemical study
Immunocytochemical staining was performed according to the manufacturer’s instructions using rabbit polyclonal anti-nuclear factor erythroid two- related factor 2 (anti-Nrf2) antibody (ab92946, abcam) as a marker for oxidative stress and rabbit polyclonal anti-proliferating cell nuclear antigen (Anti-PCNA) antibody (ab18197, abcam) as an indicator for cellular proliferation.
4μm sections were cut for immune labeling. The procedure was performed following the manufacturer’s instructions. Briefly, sections were deparaffinized in xylene, rehydrated in descending concentrations of alcohol, and immersed in 0.1% hydrogen peroxide for 15 min to prevent endogenous peroxidase activity. For antigen retrieval, the slides were boiled in a 10 mm citrate buffer solution (pH 6) for 10–20 min, then cooled at room temperature for 20 min. Sections were washed with phosphate buffer and incubated in the ultra-vision block at room temperature for 5 min to prevent non-specific background staining. Sections were incubated with the anti-Nrf2 antibody and the anti-PCNA antibody for 30 min at room temperature. The reaction was seen using an ultra-vision one detection system, HRP Polymer, and (diaminobenzidine) DAB Chromogen (Thermo Fisher Company, USA). After fulfillment of the reaction, counterstaining was performed using Meyer’s hematoxylin and dehydrated by exposure to ascending grades of alcohol, then cleared by xylene. A coverslip using permanent mounting media was put on. The changes in the immuno-histochemical reaction of the testicular tissue sections of the treated animals were observed compared with the corresponding tissues of the control rats. The same technique was performed on negative control slides except for the primary antibodies.
Photography
The Olympus light microscope B X 51 (Olympus, Japan) connected to a computer program, LC micro application software, at the Histology and Cell Biology Department, Faculty of Medicine, Minia University, was used for examining and capturing images for the histological and immunohistochemical sections. Slides were photographed using an Olympus high-resolution color digital camera. Images were saved as jpg and processed using Adobe Photoshop seven to standardize brightness, contrast, and background color, then printed.
Morphometric study
A morphological study was done using image J software on equally magnified images captured randomly from five non-overlapping fields of each rat of the studied groups. Image J 22 software was used to measure the mean area fraction of Nrf2 and PCNA immune reactivity. The mean area fraction was measured in a standard measuring frame from five photomicrographs of each rat using a magnification of X 400. These areas were masked by a red binary color that could be measured by the computer system as follows: (1) Software converted the image type to an 8-bit greyscale. (2) The image was then color thresholded to select only the color of interest, the brown color of the immunopositivity. (3) A red binary color then masked the color to measure area fraction, which is the percentage of the pixels in the brown color highlighted in red.
19
Measuring the mean number of PCNA immunopositive cells was assessed by counting at x400 magnification in five non-overlapping fields for the same slide of each animal. 20
Statistical analysis
Results were expressed as the mean ± SE.M. The one-way analysis of variance (ANOVA) was performed and followed by Tukey’s test to analyze the data for statistically significant variance. GraphPad Prism software was used for statistical calculations (version 5.01 for Windows, GraphPad Software, San Diego, California, USA (www.graphpad.com).
Results
Effect of Idebenone on testicular markers in juvenile rat testicular T/D
Effect of IDE on testicular markers in juvenile rat testicular T/D.

Data are shown as the mean ± SEM (n = 8 rats). Significance was set at p < 0.05.
asignificant difference compared to the control,
bsignificant difference compared to the IDE group,
csignificant difference compared to the T/D group. T/D= torsion/detorsion; IDE= idebnenone
Effect of Idebenone on oxidative stress parameters in juvenile rat testicular T/D
Effect of IDE on oxidative stress parameters in juvenile rat testicular T/D.
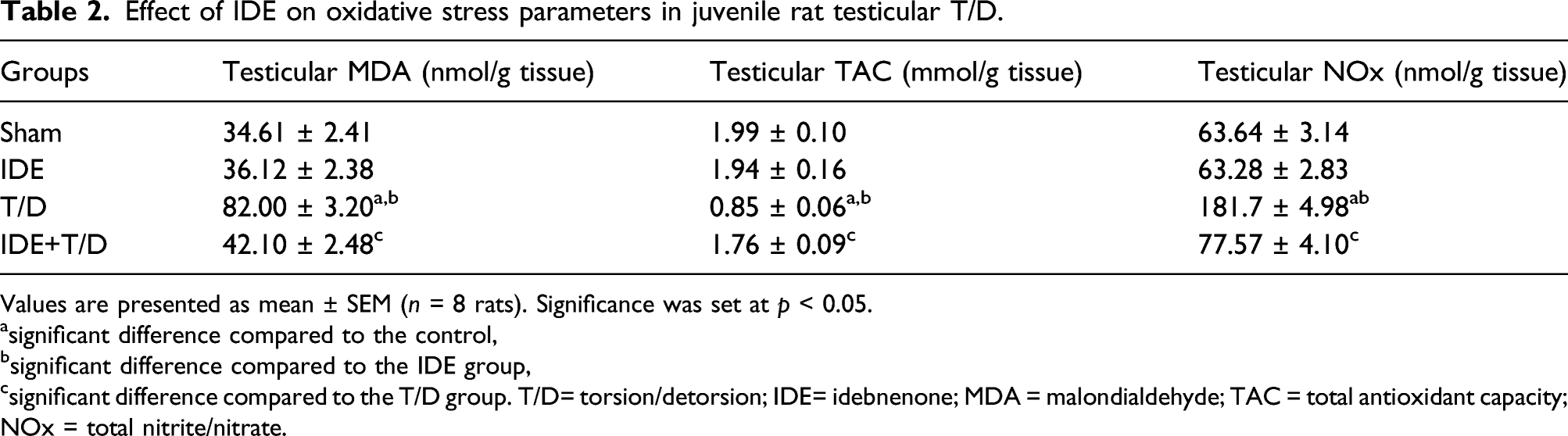
Values are presented as mean ± SEM (n = 8 rats). Significance was set at p < 0.05.
asignificant difference compared to the control,
bsignificant difference compared to the IDE group,
csignificant difference compared to the T/D group. T/D= torsion/detorsion; IDE= idebnenone; MDA = malondialdehyde; TAC = total antioxidant capacity; NOx = total nitrite/nitrate.
Effect of Idebenone on inflammatory and apoptotic parameters in juvenile rat testicular T/D
Effect of IDE on inflammatory and apoptotic parameters in juvenile rat testicular T/D.

Values are presented as mean ± SEM (n = 8 rats). Results are considered significantly different when p< 0.05.
asignificant difference compared to control,
bsignificant difference compared to IDE group,
csignificant difference compared to T/D group. T/D= torsion/detorsion; IDE= idebnenone; TNF-α = tumor necrosis factor alpha; IL-1β = interlukin-1beta.
Effect of IDE on the expression of Sirt1 in juvenile rat testicular T/D
Figure 1 revealed a significant decrease in testicular Sirt1 expression in the T/D group relative to the control and IDE groups. The IDE+T/D group showed increased testicular Sirt1 expression compared to the T/D group. Effect of IDE on expression of Sirt1 in juvenile rat testicular T/D. Data are shown as the mean ± SEM (n = 8 rats). Significance was set at p < 0.05. (a) significant difference compared to the control, (b) significant difference compared to the IDE group, (c) significant difference compared to the T/D group. T/D= torsion/detorsion; IDE= idenenone; Sirt1= Sirtuin type 1.
Histological study for juvenile rat testicular T/D using hematoxylin and eosin
Regarding juvenile rat testicular tissue sections, those of the control and IDE groups were formed of closely packed rounded seminiferous tubules as shown in Figure 2. A and B. On the other hand, Figure 3. A and B showed examination of the T/D group tissue section. Meanwhile, Fig 3(C) revealed the IDE+T/D group tissue section. Representative photomicrographs of sections of juvenile rat testes of control (a) and IDE (b) groups revealing seminiferous tubules (T) with regular contour lined by stratified germinal epithelium and separated by interstitial tissue with immature Leydig cells (black star). Different stages of spermatogenic cells; spermatogonia type A (black arrow), type B (red arrow) and primary spermatocytes (leptotene, zygotene (l/z), pachytene (p) and diplotene (d)) are observed with presence Sertoli cells (elongated to polygonal nucleus with dispersed chromatin and a prominent nucleolus) between spermatogonia (white arrows). Spindle-shaped cells (dotted arrow) with dark flat nuclei are seen surrounding the basement membrane. (H&E ×400). Representative photomicrographs of sections of juvenile rat testes showing: (a) and (b) T/D group with distorted seminiferous tubules (T) with areas of ruptured and discontinuous basement membranes (dotted arrows). Many tubules show disorganized lining epithelium (green stars), other tubules with focal areas of loss of spermatogenic cells (arrow head) and degenerated cells with vacuolated cytoplasm and small dark nuclei (red arrows). Some tubules obliterated with numerous spermatogonia (yellow star). Some tubules showed nuclear remnants (yellow arrows) and sloughed germ cells and necrotic debris in the center of the tubules (black circles). Interstitial tissue shows shrinked darkly stained nuclei of immature Leydig cells (black star), acidophilic hyaline material (red star) and dilated congested blood vessel (red thick arrow). (c) IDE+T/D group with seminiferous tubules (T) restored to their normal histological architecture (regular contour and intact basement membrane with myoid cells surrounding the tubules (dotted arrow)) with interstitial tissue with immature Leydig cells (black star). Spermatogenic epithelium retains to its normal arrangement and thickness with type A (black arrow), type B (red arrow) spermatogonia and primary spermatocytes (p) with restoration of different stages of spermatogenesis. Notice presence of Sertoli cells in between spermatogenic cells (white arrows). Few small sized cells with darkly stained nucleus are observed within seminiferous tubules (blue arrows). (H&E ×400).

Immunohistochemical study
Immunohistochemical study using Nrf2 antibodies
Figure 4 revealed the Nrf2 immunohistochemical study in the control, IDE, T/D and IDE+T/D groups' testicular tissue sections. Representative photomicrographs of the juvenile rat testicular tissue sections immunostained with Nrf2 antibodies showing: (a) Control group with scattered Leydig cells (black arrows) and myoid cells (dotted arrow) with positive Nrf2 immune-reactivity. (b) IDE group with few Leydig cells (black arrows), myoid cells (dotted arrow) and germ cell (red arrows) with positive Nrf2 expression. (c) T/D group with strong positive Nrf2 expression in the Leydig cells (black arrows), myoid cells (dotted arrow) and germ cell (red arrows). (d) IDE+T/D group with downregulation of Nrf2 expression in the Leydig cells (black arrows), myoid cells (dotted arrow) and germ cell (red arrows). (Nrf2 antibodies x 400).
There was a significant increase in the mean area fraction of Nrf2 expression in the T/D and IDE+T/D groups compared to the control and IDE groups. A significant decrease in the mean area fraction of the Nrf2 reaction of the IDE+T/D group was noticed relative to the T/D group (Figure 4).
Immunohistochemical study using PCNA
Figure 5 revealed PCNA immunohistochemical study in the control, IDE, T/D and IDE+T/D groups' testicular tissue sections. Representative photomicrographs of the juvenile rat testicular tissue sections immunostained with PCNA antibodies showing: (a), (b) control and IDE groups with strong nuclear PCNA immunoreactivity of different stages of spermatogenic cells (black arrows). (c) T/D group with negative PCNA expression in nuclei of numerous spermatogenic cells (black arrows) and faint expression in some germ cells (red arrows). D) IDE+T/D group with positive PCNA expression in nuclei of numerous spermatogenic cells (black arrows). (PCNA antibodies x 400).
There was a significant decrease in the mean number of PCNA positive cells in the T/D group compared to the control and IDE groups. A significant increase in the mean number of PCNA immunopositive cells was observed in the IDE+T/D group relative to the T/D group (Figure 5).
Discussion
A severe acute urological emergency caused by twisting the spermatic cord, called testicular torsion, results in an interruption in the blood supply and testicular ischemia, mainly in newborn and adolescent males. 1 Understanding the molecular mechanisms of testicular I/R injury is important to optimize therapeutic intervention. Overproduction of ROS and reactive nitrogen species (RNS) is the main cause of testicular injury. 21 The present study assessed the ameliorative action of IDE on testicular I/R injury in the rat T/D model.
According to the findings, the testicular injury was linked to a reduction in serum testosterone levels, indicating a functional impairment in response to testicular I/R. These results are in accordance with previous studies.1,3,22 It was also observed that hypercholesterolemia was present and this metabolic disturbance was in line with Elshaari et al., 2012 23 and Abdelzaher et al., 1 2020. The decreased testosterone level due to Leydig cell damage explains hypercholesterolemia. Histopathological alterations in tubular architecture, such as deformed seminiferous tubules with areas of torn and discontinuous basement membranes, showed testicular injury. Many tubules showed disorganized lining epithelium and many apoptotic cells. Some tubules are obliterated by numerous spermatogonia, with testicular injury, negative PCNA expression in the nuclei of numerous spermatogenic cells, and faint expression in many germ cells.
Propitiously, the present study revealed that IDE could ameliorate the functional and metabolic disorders related to testicular I/R. This was confirmed by histopathological examination, which showed normal histological architecture of the seminiferous tubules, regular contouring, intact basement membranes with myoid cells surrounding it, and many spermatogenic cells exhibiting positive PCNA nuclei.
Testicular T/D was related to an oxidative imbalance in the form of elevated MDA and NOx levels and a decrease in TAC levels in testicular tissue in the present research. Our findings are consistent with those previously published. Testicular I/R injury was linked to an increase in testicular MDA and NOx levels, according to Refaie and her coworkers 2017. 3 Also, Abdelzaher et al., 2020 1 stated that TAC levels were decreased with increased levels of MDA and NOx in the testicular T/D rat model. The imbalance between the synthesis of oxidants and the scavenger, the defense system component, causes oxidative stress. Attracted neutrophils to the T/D site produce ROS, leading to testicular injury. 24 ROS interacts with membrane lipids, causing lipid peroxidation, loss of tissue biological structures, and changes in membrane permeability and cell integrity. 25 MDA is a stable lipid peroxidation end product that can be used as an indirect biomarker of oxidative stress in testicular injury. 26
On the other hand, TAC levels decline as one or more sulfhydryl group residues in antioxidant enzymes, which are responsible for their catalytic activity, become inactive. 27 NO is also a very diffusible free radical. It can create peroxynitrite, a highly reactive compound that can harm DNA, RNA, proteins, and lipids. At high concentrations, it is harmful.28,29
IDE, a ROS scavenger, has antioxidant effects that prevent lipid peroxidation and inactivate ROS, protecting the cells from oxidative injury. 10 It improved oxidative stress parameters in the current results. IDE is a protective agent against increased lipid peroxidation. The reduction of oxidant stress and antioxidant enzyme elevation by IDE was concordant with many studies. Erb and his colleagues, 2012 30 supported our results as they suggested that short-chain quinones as IDE have a therapeutic role for disorders associated with complex mitochondrial dysfunction and/or oxidative stress. Avcı et al., 2021 31 showed that IDE ameliorates Parkinson’s disease in rats through decreasing lipid peroxidation. In line with IDE-dependent protection, IDE also affected RNS by reducing NOx levels in the present study. This whole effect is related to IDE augmentation of the antioxidant defense. 32
Excessive oxidative stress depletes Nrf2, the cytoprotective antioxidant protein, in testicular T/D injury. 33 The Nrf2 signaling pathway is activated by IDE, which benefits detoxification and antioxidant defense. IDE enhances Nrf2 and prevents T/D-induced Nrf2 depletion, enhancing the ability to withstand oxidative stress. 34
Proinflammatory cytokine synthesis is upregulated by oxidative stress. Increased proinflammatory cytokines such as TNF-α and IL-1β suggest a significant mechanistic link between oxidation and inflammation. 35 In this experiment, TNF-α and IL-1β levels in testicular T/D were significantly higher. Similarly, Raafat and her colleagues 2021 4 reported increased oxidation and inflammation in rats with testicular I/R injury. T/D allows neutrophils to transmigrate from the endothelium into the interstitium of the testes and proinflammatory cytokines to rise, resulting in an increase in ROS generation, which causes cellular dysfunction and death. 36
TNF-α is negatively influenced by Nrf2, indicating that IDE-induced downregulation of Nrf2 immuno-expression may aim at inactivating T/D-induced inflammatory signaling pathways. 37 The previous studies by Al-Rasheed and his colleagues, 38 Yan and his coworkers, 39 and Shastri et al. 32 reporting that IDE reduced the proinflammatory mediators are consistent with the current findings.
Emerging data indicates that cell death, necrosis, and apoptosis simultaneously occur in oxidative stress conditions like testicular I/R. Therefore, widespread spermatogenic cell apoptosis occurs, leading to impaired testicular function and male infertility. 4 Increased caspase-3 levels in testicular T/D revealed this theory in the current experiment. That is in line with Gerhardt et al., 40 who found that IDE inhibited the activation of pro-apoptotic factors in mice. Sadik and her coworkers 41 also supported our results as they reported that IDE ameliorated galactose-induced cataracts in rats through its antioxidant and antiapoptotic properties.
Stress response and longevity are regulated by Sirt1, a NAD-dependent protein deacetylase. 42 IDE activated Sirt1 in this study, which is suppressed by testicular T/D. Sirt1 inhibits caspase-3 production directly through deacetylation of it by its N-terminal domain. 42 On the other hand, it was reported that Sirt1 stimulated the Nrf2 pathway, reducing ROS generation in glomerular mesangial cells caused by advanced glycation end products. 43 These findings show that Sirt1 is an important regulator of inflammation, apoptosis, and antioxidant defense systems and helps prevent testicular T/D injury through IDE administration. Further studies are strongly recommended to examine IDE in different doses and its testicular-preserving properties in patients with testicular damage, particularly in newborn and adolescent males.
Conclusion
Through modulation of the Sirt1/Nrf2/TNF-pathway, IDE has profound ameliorative effects against testicular I/R injury by lowering oxidative stress, inflammatory processes, and apoptosis. We expect our results to pave the way for new research and therapeutic implications about IDE usage with different doses in preventing testicular damage, particularly in newborn and adolescent males.
Footnotes
Author contribution
WAY, NNW, and DAAL conceptualization performed the experiments, wrote, and revised the manuscript. ASAA and MABE conducted experiments. GMH did data analysis and manuscript revision. AFA performed and wrote the histopathological and immunohistochemical examination. MAF performed and wrote Western blot measurements. All authors read, revised, and approved the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
