Abstract
Aim
Both MFGE8 and HMGB1 were vital players for aneurysmal subarachnoid hemorrhage. However, whether HMGB1 was served as the downstream target of MFGE8 was unknown. To test this new mechanism, we performed the SAH model in rats.
Method
All treatments were injected intraventricularly into the right lateral ventricles. SAH grade, brain water content, and neurological function scores were evaluated. HMGB1 expression was studied by double immunofluorescence staining. HE and Nissl’s staining were performed to observe the pathological change. Inflammatory factors were measured by ELISA method.
Results
High expression of MFGE8 could improve neurological function and reduce the brain edema and pro-inflammatory factors. Injection of rhMFGE8 inhibited HMGB1. To further verify the regulation of MFGE8 in HMGB1, we used rhHMGB1 and glycyrrhizin, and the results indicated MFGE8 produced excellent effect on SAH rats via inhibiting HMGB1.
Conclusion
In a word, MFGE8 improved EBI caused by SAH, depending on HMGB1 that was the potential mechanism.
Keywords
Introduction
Aneurysmal subarachnoid hemorrhage (SAH) is more common in young patients, accounting for 5–7% of strokes. 1 Although the neurocritical care management achieved excellent improvement with decreased case fatality over the past few decades, SAH still remains a devastating disease with high incidence. 2 The functional independence was recovered for only two-thirds of survivors at 1 year after the bleeding. 3 Sudden headache occurs after SAH, but there was no onset pattern reported in patients. 4 In recent years, the hotspot of extensive research is focused on early brain injury (EBI) within 3 days after the bleeding. In 2004, Kusaka et al. presented EBI for the first time, 5 which is the main predictor of the poor prognosis of SAH patients. 6 The potential mechanisms of EBI induced by SAH are multifactorial, such as oxidative stress, neuronal apoptosis, blood–brain barrier disruption, brain edema and neuroinflammation, and so on.6–8
As a multifunctional glycoprotein, milk fat globule-epidermal growth factor-factor 8 (MFGE8) is secreted by mononuclear cells. 9 MFGE8 is abundantly expressed in various tissues of the whole body, which has been corroborated to take part in multiple pathophysiological functions, such as inflammation response, 10 angiogenesis, 9 frobsis, 11 adaptive immune responses, 12 and phagocytosis of apoptotic cells.13,14 At present, only a few studies have focused on how MGFE8 affects SAH, which reported the effect of MFGE8 on improving EBI after SAH through anti-inflammation and anti-apoptosis. 15 In addition, MFGE8 exerted anti-inflammatory role, 16 attenuated microthrombosis, 17 maintained vascular endothelial integrity, 18 and inhibited neuron apoptosis, 19 in SAH model. From a vascular point of view, MFGE8 maintained vascular endothelial integrity, further to reduce degree of EBI after SAH via activating integrin β5 pathway. 18 Reduced oxidative stress induced by MFGE8 was found in a rat model of SAH through integrin β3-independent pathway. 20 MFGE8 predicted the occurrence of delayed cerebral ischemia (DCI) after aneurysmal SAH. Based on the published studies described above, MFGE8 is likely to be an underlying therapeutic target for SAH patients.
High mobility group box-1 (HMGB1) is widely disturbed in nuclear and cytoplasm of mammalian cells. 22 Activated by LPS, TNF or IL-1β, macrophages, monocytes, pituicytes could secrete HMGB1, a potent pro-inflammatory cytokine,23,24 and necrotic or damaged cells also passively produce HMGB1.25–27 Many evidences have stated HMGB1-mediated responses in SAH. HMGB1-mediated neuroinflammation28–30 has been studied, indicating that inhibition of HMGB1 mitigated inflammatory response after SAH rat model. Serum HMGB1 predicts DCI following aneurysmal SAH. 31 Many evidences showed that HMGB1 inhibition is likely to reduce brain injury following aneurysmal SAH. 32 Nevertheless, there was no literature reported on how MFGE8 mediated HMGB1 to affect EBI after SAH. In the present study, we established subarachnoid hemorrhage in rat model to study how the MFGE8 improved EBI induced by SAH through HMGB1. Rats received an intraventricular injection of recombinant human MFGE8 (rhMFG-E8) and glycyrrhizin (HMGB1 antagonist), and the pathological injury repair and HMGB1 expression were assessed. We further used recombinant human HMGB1 (rhHMGB1) to detect if the effect of MFGE8 on EBI after SAH was modulated by HMGB1.
Material and methods
SAH rat model
Male adult Sprague–Dawley rats with weight of 200–220 g were provided from Jinan Pengyue Experimental Animal Breeding Co, Ltd (Permission Number: SYXK(Lu)20180030). First, rats need acclimatization for 7 days in an SPF housing room with temperature of 25°C and humidity of 55%, and the light/dark cycle of 12/12 h. During the adaptive housing, rats were free access to food and water. All experiments were approved by the Institutional Animal Care and Use Committee of Yantaishan hospital (Approval number 2022012), which in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
Aneurysmal subarachnoid hemorrhage (SAH) model was established via endovascular perforation reported previously 33 with slight modification. Briefly, rats were given an intraperitoneal injection of 3% pentobarbital sodium (45 mg/kg), and left carotid artery was exposed. A sharped 4-0 nylon sature was inserted into external carotid artery, and then go further into internal carotid artery. When the resistance occurred, the sature moved 3 mm to ultimately puncture the bifurcation of the anterior and middle cerebral artery. Rats in the sham group carried out the same surgery except for puncture. After SAH, the rats were continued to remain in the rearing environment described above. During the trial period, survival status of rats in each group was recorded.
Experimental designs and administration
In Experimental 1, we randomly divided the rats into four group, 15 rats for each group. (1) Sham group: the rats were normally housed after the surgery. (2) SAH + vehicle: the rats were given 3 μL PBS at 1 h after SAH. (3) SAH + 1 μg 16 rhMFGE8 (2767-MF-050, R&D) group: the rats were administrated with 1 μg rhMFGE8 (1 μg in 3 μL sterile PBS) at 1 h after SAH. (4) SAH + 5 μg rhMFG-E8 group: the rats were administrated with 5 μg rhMFGE8 (5 μg in 3 μL sterile PBS) at 1 h after SAH. All rats received intraventricular injection using a stereotaxic apparatus. A hole was drilled, and 10-μL hamilton syringe was introduced into the right lateral ventricles. The coordinates were as follows: 1.0 mm posterior and 1.5 mm lateral to the bregma, 4.0 mm below the dural layer. Then, rhMFGE8 was injected into lateral ventricles at a slow rate of 0.5 μL/min. Ten minutes later, the pillow was pulled out, and the burr hole was sealed using bone wax.
In Experimental 2, we randomly set four groups, 15 rats for each group. (1) SAH + vehicle and (2) SAH + 5 μg rhMFG-E8 group: the rats in two groups were given same treatment as described above. (3) SAH + 5 μg rhMFG-E8 + 3.3 μg rhHMGB1 (ab285780, Abcam) group: the rats were treated simultaneously with 5 μg rhMFG-E8 and 3.3 μg 34 rhHMGB1 (3.3 μg in 3 μL sterile PBS) at 1 h after SAH. (4) SAH + 5 mg/kg glycyrrhizin (CDS020796, Sigma-Aldrich) group: 5 mg/kg 35 glycyrrhizin were given 24 h before (precondition) and 1 h after SAH. All rats received intraventricular injection using a stereotaxic apparatus.
Neurobehavioral testing
The short-term behavioral evaluation was performed by a blinder observer. Rats in each group were assessed at 24 h after SAH by the modified Garcia score and beam balance test according to the reported methods.36,37 For the modified Garcia score with maximum score of 18, six tests are scored, including spontaneous activity (0–3 score); spontaneous movement of all limbs (0–3); forepaw outstretching (0–3); climbing (1–3); body proprioception (1–3); and response to whisker stimulation (1–3). Beam balance test is used to evaluate the walk ability of rats on a 15 mm-wide beam within 1 min. The range of score is 0–4 based on the walking distance. Three consecutive tests are carried out. The score is expressed as the mean of the triplicate experiments.
SAH grade
At 24 h after SAH, the rats were sacrificed under anesthesia via intraperitoneal injection of 3% pentobarbital sodium (45 mg/kg). The high resolution photos of brain base of rats in each group were taken immediately, and then evaluated by a blind investigator as previously reported. 38
Briefly, the brain base is divided into 6 segments, and the sum of six segments is total score with the range from 0 to 18. Score 0–7: mild SAH, 8–12: moderate SAH, and 13–18: severe SAH. When the total score is less than or equal to 7, the rats are excluded because of mild bleeding.
Brain water content
The wet-dry method is used to evaluate the degree of brain edema. After anesthesia, the brain tissues were collected, and the divided into left hemisphere, right hemisphere, cerebellum, and brain stem. After that, each part was weighed, and put in baking box at 105°C. After 72 h, we weighed each part and calculated the ratio of wet/dry according to the following formula:
Enzyme-linked immunosorbent assay
The pro-inflammatory factors in serum were measured by enzyme-linked immunosorbent assay (ELISA). Specific procedures were performed in strict accordance to the manufacturer instructions. All ELISA kits of IL-1β (H002), IL-6 (H007-1-1) and TNF-α (H052-1) were purchased from Nanjing Jiancheng Bioengineering Institute. And the result was presented as relative cytokine level (vs. Sham or SAH + vehicle).
HE staining
After dewaxing and hydration, 5-μm paraffin section was stained in hematoxylin for 10 min, and washed under flowing water for 3 min. After that, sections were differentiated by hydrochloric acid for several seconds followed by placed into tap water for 5 min. The slices were then passed sequentially through 75% alcohol (5 min), 85% alcohol (5 min), eosin (5 min), 95% alcohol (1 min × 2 times), 100% alcohol (10 min × 2 times), and xylene (10 min × 2 times). Finally, the sections were sealed by resinene. The images were observed under a microscope (LEICA DM 1000 LED).
Nissl’s staining
Five micrometers thick sections were placed into dimethylbenzene and alcohol for dewaxing and hydration, then stained with Nissl staining solution, and incubated for 10 min. Nissl differentiation solution was added on the sections, then the differentiation was observed under microscope until nuclei and particles were clear. The images were visualized under a microscope (LEICA DM 1000 LED).
Immunofluorescence assay
Double immunofluorescence staining was performed to detect HMGB1 in neuron of brain tissues. After dewaxing and rehydration treatment, the brain sections were subjected to antigen repair in a sodium citrate solution and blocked by 5% goat serum for 30 min. Then the sections were incubated with primary antibodies overnight at 4°C, secondary antibodies and DAPI at room temperature, and mounted by antifade solution. The information of primary and secondary antibodies was listed: rabbit anti-rats HMGB1 polyclonal antibody (1:150, 10829-1-AP, proteintech), mouse anti-rats NeuN monoclonal antibody (1:100, 66836-1-lg, proteintech), Cy3-labeled goat anti-rabbit IgG (H + L) (1:300, A0516, Beyotime), FITC-labeled goat anti-mice IgG (H + L) (1:500, A0568, Beyotime). The expression and location of HMGB1 were observed under laser scanning confocal system (LSM800, Zeiss, Germany). Mean fluorescence intensity of HMGB1 was analyzed by Image J 1.49p software.
TUNEL
The paraffin sections were dewaxed and hydrated, and then tunel staining was performed according to the instructions for tunel cell apoptosis detection kit (C1091, Beyotime). After sealing, the sections were observed via the microscope (LEICA DM 1000 LED). Tunel-positive ratio of apoptotic cells was analyzed by Image J 1.49p software.
Statistical analysis
Analysis of data in this article was performed by SPSS 19.0. The data appeared in the form of mean ± SD. The statistical difference in multiple groups was analyzed through one-way ANOVA followed by Tukey test. Significant difference was confirmed when p value was less than 0.05.
Results
Effects of exogenous MFGE8 on neurological functions and inflammatory response
The mortality rate of rats and number of excluded rats in each group.
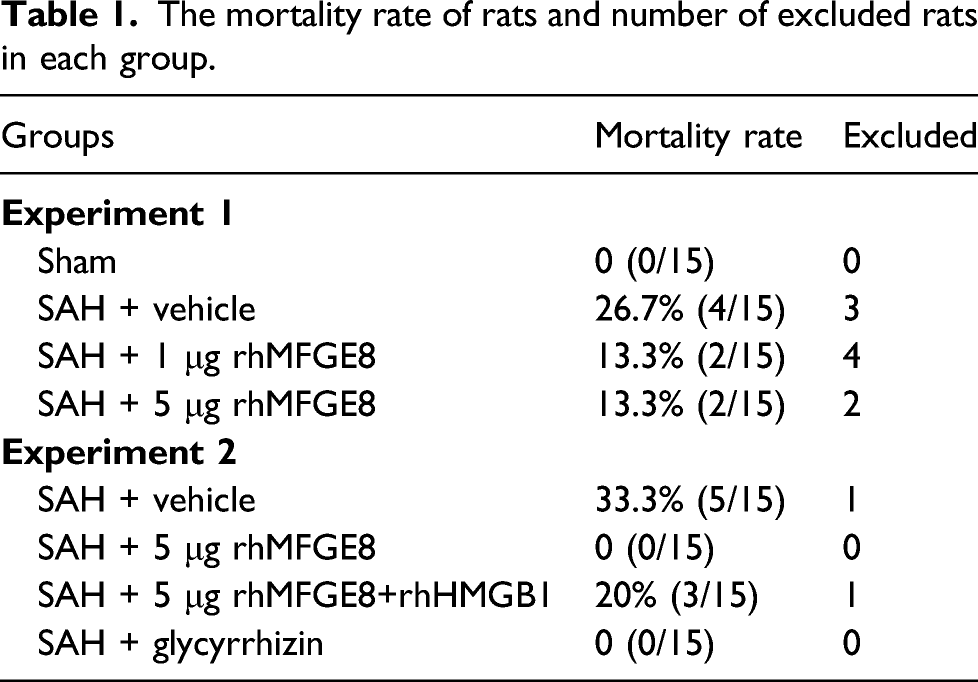
The photos in Sham and SAH + vehicle group were taken to intuitively show severity of brain and successful construction of SAH rats (Figure 1a). Except for excluded rats, SAH grade of rest rats in SAH + vehicle, SAH + 1 μg rhMFGE8, SAH + 5 μg rhMFGE8 groups have no remarkable difference. Rats in Sham group did not have any blood clots (Figure 1b). The brains were dried in an atmosphere of 105°C for 3 day, and weighed prior to and after drying. The calculated percentage of brain water content showed the degree of brain edema (Figure 1c). The brain water content in SAH + vehicle group was higher than Sham group (p < 0.01). After addition of rhMFGE8, a lower percent was observed with a dose-dependent manner, particularly in high-dose rhMFGE8. The short-term neurological functions were evaluated by modified Garcia scoring system and beam balance test (Figure 1d). After SAH, modified Garcia scores and beam balance scores were lower than Sham group (p < 0.01). A gradually increasing scores occurred after administration of rhMFGE8, especially 5 μg rhMFGE8. There was a significant difference between SAH + vehicle group and SAH + rhMFGE8 group (p < 0.01). Meanwhile, we also tested inflammatory factors (IL-1β, IL-6, and TNF-α) in serum (Figure 1e). There was an elevated inflammation with increasing level of IL-1β, IL-6, and TNF-α in SAH + vehicle group. There was a decreased level of the aforementioned pro-inflammatory factors after intraventricular injection of rhMFGE8. Effects of exogenous MFGE8 on neurological functions and inflammatory response. (a) Representative images of brains in Sham group and SAH + vehicle group. (b) SAH grades were evaluated in each group without significant difference. (c) The percentage of brain water content in each group was calculated 24 h after SAH. (d) The effects of recombinant human MFGE8 antibody on short-term neurobehavioral outcomes: Modified Garcia scores and beam balance scores. (e) The effects of rhMFGE8 on inflammatory factors IL-1β, IL-6, and TGF-α in serum. The result was presented as relative cytokine level (vs. Sham). Sham-*p < 0.05 and **p < 0.01; SAH + vehicle: ^p < 0.05 and ^ ^p < 0.01; SAH + 1 μg rhMFGE8: #p < 0.05 and ##p < 0.01.
Effects of rhMFGE8 on damage of neurons and HMGB1 expression in the cortical region
Next, we analyzed the neuron injury induced by SAH via HE assay (Figure 2a). SAH establishment gave rise to early brain injury. In SAH + vehicle, a serious pathological change, including wrinkled nuclei, vacuolization, and deformed neuronal cells, were found in the cortical area of SAH rats. The administration of rhMFGE8 significantly improved the damage existed in brain tissues. Low-concentration rhMFGE8 could slightly decrease the damaged neurons, while the significant improvement was found after injection of 5 μg rhMFGE8. As shown in Figure 2b, HMGB1 was overexpressed in brain tissues after SAH. However, we observed a significant reduction of influorescence intensity of HMGB1 after rhMFGE8 treatment, expecially high-dose rhMFGE8. These results indicated that MFGE8 has a protective effect on damaged neuron caused by SAH through increasing HMGB1 expression. Effects of rhMFGE8 on damage of neurons and HMGB1 expression in the cortical region. (a) Pathological condition was assessed by HE staining. The scale bars were 200 and 100 μm. (b) Expression of HMGB1 was measured by immunofluorescence staining. Quantified data showed as mean fluorescence intensity, which was analyzed by Image J 1.49p. The scale bar was 20 μm. Sham-*p < 0.05 and **p < 0.01; SAH + vehicle: ^ ^p < 0.01.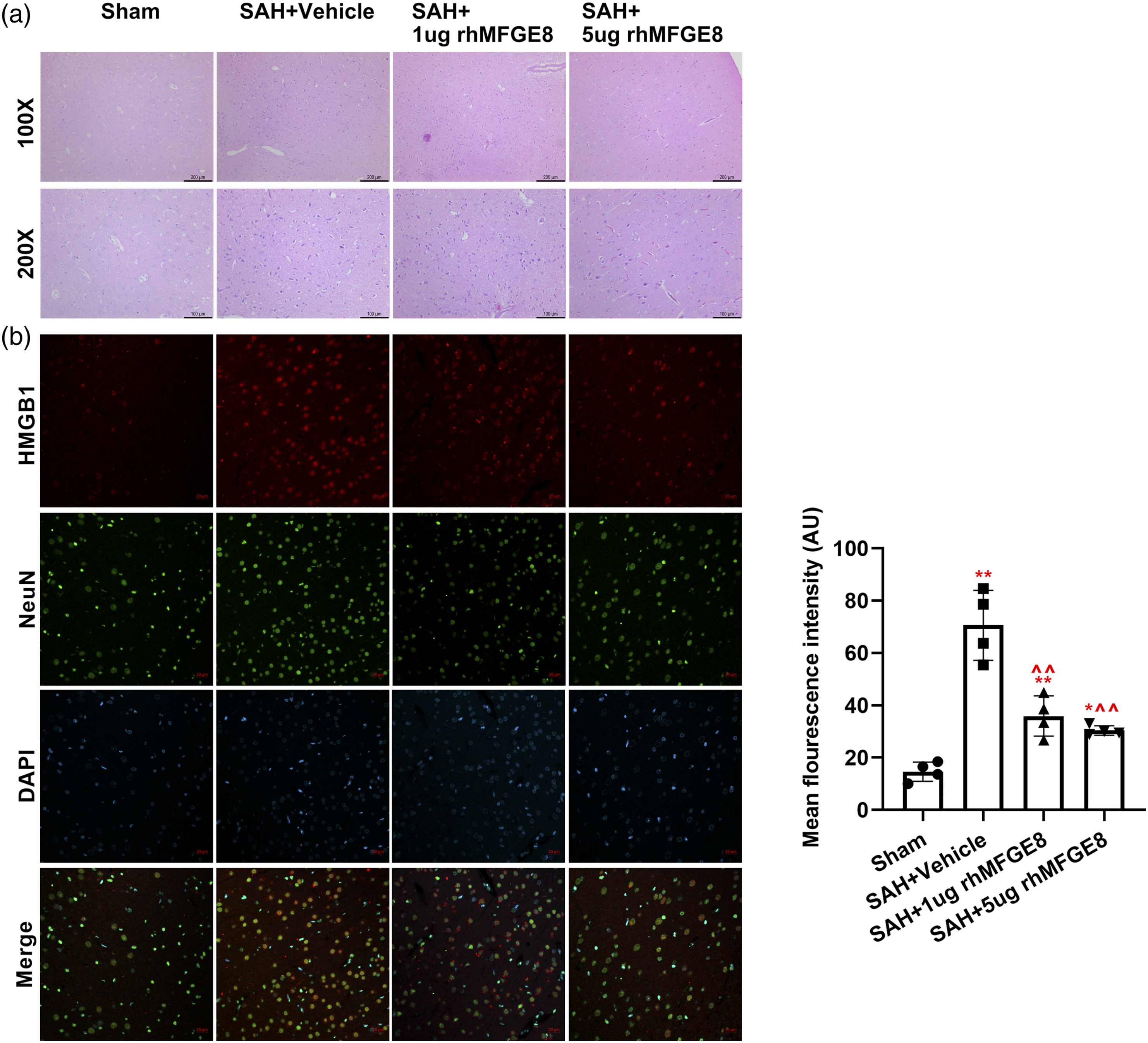
HMGB1 expression affected by MFGE8 was checked. And the results of double immunofluorescence staining showed that HMGB1 was expressed in the neuronal cell nuclei, and mean fluorescence intensity of HMGB1 in SAH + vehicle was significantly higher than sham group (p < 0.01). However, the expression of HMGB1 was downregulated by rhMFGE8, indicating the regulation of MFGE8 in HMGB1 (Figure 3). Administration of exogeneous HMGB1 abolished the neuroprotective role of MFGE8. (a) SAH grades in each grade with no significant differences between all the groups. (b) Brain water content in four parts of the brain tissue. (c) Modified Garcia scores and beam balance scores. (d) Inflammatory factors detected by ELISA method in each group. The result was presented as relative cytokine level (vs. SAH + vehicle). SAH + vehicle: ^p < 0.05 and ^ ^p < 0.01; SAH + 5 μg rhMFGE8: #p < 0.05 and ##p < 0.01.
Administration of exogeneous HMGB1 abolished the neuroprotective role of MFGE8
To verify the protective role of MFGE8 in EBI induced by SAH, exogeneous HMGB1 antibody and antagonist glycyrrhizin were used. SAH grade was also estimated according to the described the code of points. A non-significant variability was found in all groups (p > 0.05). Brain water contents in left-, right hemisphere, and cerebellum were analyzed, demonstrating a significant decrease in SAH + 5 μg rhMFGE8 and SAH + glycyrrhizin groups compared with SAH + vehicle group (p < 0.01), and an evident increase in SAH + 5 μg rhMFGE8 + rhHMGB1 compared with SAH + vehicle group and SAH + 5 μg rhMFGE8 (p < 0.01 or p < 0.05). We found a non-significant change of brain stem in all groups. Additionally, modified Garcia scoring system and beam balance test were also evaluated by a blinder researcher. The results showed that recombinant human HMGB1 antibody could impaired the neurological protective effect of rhMFGE8, which could reduce the scores with a remarkable divergence compared with SAH + vehicle group and SAH + 5 μg rhMFGE8 group (p < 0.01). While downregulation of HMGB1 by glycyrrhizin could evidently ameliorate neurological functions with the increasing scores compared with SAH + vehicle (p < .01). For inflammation, inhibiting HMGB1 could depress the level of pro-inflammation factors, while injection of rhHMGB1 could neutralize anti-inflammatory effect of MFGE8. The above results suggested that MFGE8 could control neurological functions through inhibiting HMGB1.
HMGB1 blemished the beneficial effect of MFGE8 on neuronal damage in the cerebral cortical region
To further explore whether the pathological condition regulated by MFGE8 was related to HMGB1, HE staining (Figure 4a) and Nissl’s staining (Figure 4b) were performed. There was more serious injury after co-administration of rhMFGE8 and rhHMGB1 compared with SAH + vehicle and SAH + 5 μg rhMFGE8 groups. A significant improvement was observed in SAH + glycyrrhizin group. HMGB1 blemished the beneficial effect of MFGE8 on neuronal damage in the cerebral cortical region, demonstrating by HE staining (a) and Nissl’s staining (b). The scale bars were 100 and 200 μm in a and 100 μm and 50 μm in b.
After addition of HMGB1 agonist and antagonist, we detected the HMGB1 expression (Figure 5). The quantified data showed that glycyrrhizin and rhMFGE8 after SAH could reduce the HMGB1 expression. But an elevated fluorescence intensity was induced in SAH + 5 μg rhMFGE8 + rhHMGB1 group. A significant difference existed between SAH + 5 μg rhMFGE8 group and SAH + 5 μg rhMFGE8 + rhHMGB1group (p < 0 .05). These results indicated that inhibition of HMGB1 could ameliorate brain injury, and MFGE8 played a protective role for EBI caused by SAH through inhibiting HMGB1. The regulation of MFGE8 in HMGB1 was verified by addition of recombinant human HMGB1 or HMGB1 antagonist glycyrrhizin. The quantified results of fluorescence image showed mean fluorescence intensity of HMGB1. The scale bar was 20 μm. SAH + vehicle: ^ ^p < 0.01; SAH + 5 μg rhMFGE8: #p < 0.05.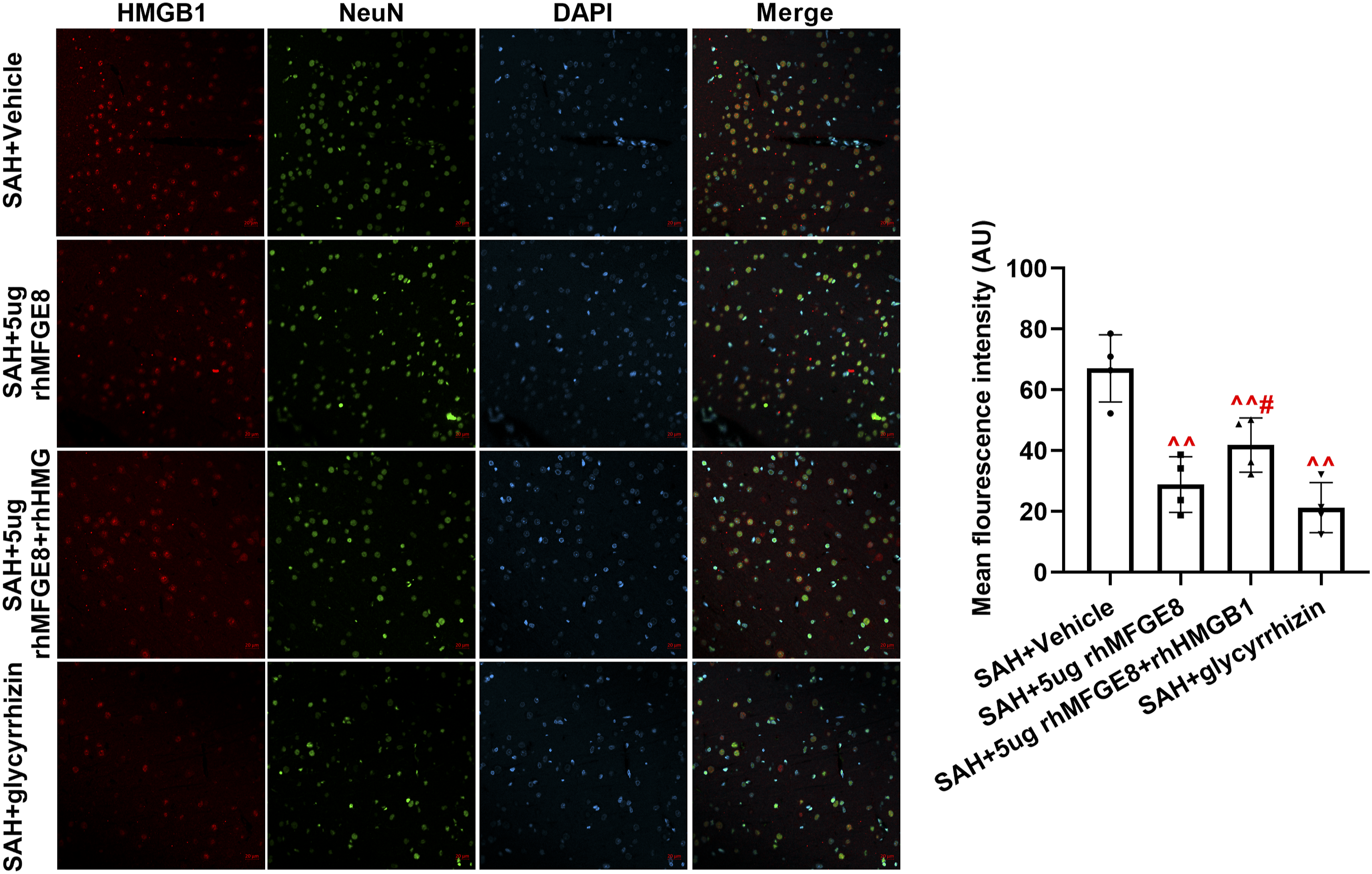
Discussion
In this study, we explored the neuroprotective role of MFGE8 through regulation of HMGB1 in experimental SAH rats. The novel findings of this study were as follows: (1) HMGB1, as the downstream target; (2) the new mechanism of MFGE8 on SAH rats is likely to be related to inhibition of HMGB1. SAH lead to upregulation of HMGB1, and inhibition of HMGB1 may promote neuronal regeneration after SAH. 34 Consistent with the above finding, we also found inhibition of HMGB1 by glycyrrhizin could increase neurological function scores, reduce cerebral edema and restrain inflammation, promoting recovery of EBI. However, whether the effective action of MFGE8 on SAH-induced EBI is closely associated with HMGB1 is unknown, and has not been reported. Through experimental exploration, we found that MFGE8 promote the recovery of neurological function after SAH, partly depending on low expression of HMGB1, which is the focus of this article. We observed how MFGE8 regulated HMGB1 through double immunofluorescence staining. A decreased expression of HMGB1 appeared after rhMFGE8 treatment, which indicated that MFGE8 downregulated HMGB1.
A large number of studies authenticated the vital role of neuroinflammation and neuronal apoptosis in EBI after SAH.39–41 MFGE8, as a secretory glycoprotein, has attracted extensive attention, and provides neuroprotection through anti-inflammation, 39 anti-oxidative stress, 20 and especially apoptosis 42 in central nervous system (CNS) disease. Regulating inflammation could availably ameliorate the prognosis of SAH. 39 In this paper, we used ELISA to detect pro-inflammation factors (IL-1β, IL-6, and TNF-α) in serum. The results indicated that intraventricular injection of rhMFGE8 can significantly decrease the content of IL-1β, IL-6, and TNF-α. Meanwhile, we also added HMGB1 antagonist glycyrrhizin and recombinant human HMGB1 antibody to analyze the action of HMGB1 in the neuroprotection role of MFGE8 on EBI after SAH. We found that inhibition of HMGB1 could reduce inflammation, and administration of rhHMGB1 could weaken anti-inflammatory effect of MFGE8. Accumulating researches confirmed neuronal apoptosis could bring the harmful effect in the process of traumatic brain injury. 42 We performed TUNEL assay to explore the neuronal apoptosis. Injection of rhMFGE8 led to the decreased percentage of TUNEL-positive cell, although the opposite tendency occurred after co-administration of rhMFGE8 and rhHMGB1. These results suggested that MFGE8 improved EBI caused by SAH, depending on HMGB1 that was the potential mechanism. In addition, HE and Nissl’s staining were carried out to visualize the pathological injury of cerebral cortical area of SAH rats. Despite increasing studies have demonstrated the potential role of integrin β3/nuclear factor erythroid 2-related factor 2/heme oxygenase pathway, TIGβ5/PI3K/CXCL12 axis, and microglial M2 polarization, our study discussed a new mechanism of downstream of MFGE3 on anti-inflammatory and anti-apoptosis during SAH.
Although this study demonstrated the benefit of MFGE8 in a novel mechanism regulated via suppression of HMGB1, some limitations should be raised concern. First, MFGE8 display its characteristics of anti-inflammatory and anti-apoptosis following SAH in HMGB1-dependent behavior, however other characteristics have not been explored, such as anti-oxidative stress, neurovascular remodeling et al. Second, MFGE8/HMGB1 axis was discussed, without further investigation of the downstream target or pathway of MFGE8/HMGB1. Therefore, we need to deeply study other roles of MFGE8/HMGB1 system in SAH and the downstream target or signal pathway of HMGB1.
Footnotes
Author contributions
All authors were participated in this study. RM, LW and LG were involved in material preparation and purchase. Experiment operation was performed by JQ and WL. Data acquisition and analysis were performed by JQ, WL and LM. The first draft of the manuscript was written by JQ and LM. All authors examined and verified the previous versions of the manuscript, and approved the final manuscript. All authors have promised to take on public responsibilities for appropriate portions of the content.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
All experiments were approved by the Institutional Animal Care and Use Committee of Yantaishan hospital, which in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was granted by project ZR2020QH105 supported by Shandong Provincial Natural Science Foundation. The name of this project was HMGB1/RAGE signaling pathway in Early Brain Injury After Subarachnoid Hemorrhage in Rats. Number of funding sequence was 2779.

