Abstract
High-mobility group box 1 (HMGB1) is a nuclear non-histone protein, playing a critical role as a mediator between innate and acquired immunity; when released extracellularly, it coordinates the cellular stress response (under necrosis, bacterial lipopolysaccharide stimulation) and acts as an inflammatory marker and cytokine. The aim of the study was to demonstrate whether HMGB1 is over-expressed in chronic middle-ear pathologies and whether the entity of expression and the localization are correlated with the degree of the inflammatory reaction, thus suggesting that HMGB1 may play a crucial role in chronic inflammatory disorders of the middle ear, as already demonstrated in other airway diseases.
We analyzed 30 samples of middle-ear mucosa in patients affected by chronic suppurative otitis media with ear drum perforation with/without cholesteatoma and otosclerosis as control. The distribution of HMGB1 was evaluated as nuclear, cytoplasmic, and/or extracellular staining.
The inflammatory cells observed in the biopsies were mostly lymphocytes and plasmacells. A statistically significant difference in inflammation score between otosclerosis and chronic otitis samples (P < 0.01; Anova test) and between otosclerosis and cholesteatoma samples (P < 0.05; Anova test) was observed; the HMGB1 positivity was in accordance with the density of the inflammatory infiltrate.
HMGB1 is over-expressed in chronic middle-ear pathologies and may play a role in the progression of the inflammatory process from recurrent acute otitis media to chronic suppurative otitis media.
Introduction
Otitis media is defined as the inflammation/infection of the middle-ear mucosa; under this definition, several clinical entities are grouped: acute otitis media (AOM); recurrent acute otitis media (rAOM); otitis media with effusion (OME); chronic suppurative otitis media (CSOM) with/without cholesteatoma; and atelectasis of the tympanic membrane. According to Paparella, 1 these different clinical forms should be considered as stages along a continuum tending towards irreversible modifications. AOM and rAOM are an example: AOM is the acute phase of middle-ear infection, often associated with the tympanic membrane perforation. When AOM does not heal, it can reversibly become OME (fluids in the middle ear and absence of symptoms/signs of acute inflammation) or result in chronic perforation recurring four to six times in six months (rAOM), leading irreparably to CSOM.
Recently, the importance of defective innate immunity in otitis-prone children or in participants affected by chronic diseases of the nose and paranasal sinuses has been stressed: the innate mucosal immune system plays a defensive role by recognizing damage-associated molecular patterns (DAMPs) via pattern-recognition receptors (PPRs) such as toll-like receptors (TLRs).
Activation of TLRs by DAMPs induces innate immune response and the most frequent reaction is inflammatory infiltration and release of inflammatory cytokines. A recent study has shown that TLRs play a critical role in the inflammatory response to bacteria in otitis media. Proinflammatory cytokines (TNF-α, IL-1b, IL-6, IFN-γ) were up-regulated in CSOM patients whereas TLR2, TLR4, and TLR5 were significantly lower when compared with the chronic otitis media (COM) group or healthy participants. 2 High-mobility group box 1 (HMGB1) protein is a non-histone nuclear protein playing a critical role as prototypic DAMP, released by epithelial and immune cells with the task of coordinating the cellular stress response to necrosis and/or bacterial lipopolysaccharide stimulation. Extracellular HMGB1 functions as an immune adjuvant to trigger a robust response of T cells, dendritic cells, and endothelial cells. In addition, activated immune cells and endothelial cells also secrete HMGB1, creating a positive feedback loop that causes the release of additional cytokines and chemokines. All of these characteristics make HMGB1 a critical molecular target in multiple human diseases including sepsis, ischemia, immune disorders, neurodegenerative diseases, metabolic disorders, and cancer.
As regards the ear/nose/throat (ENT) districts, recent studies have focused on the up-regulation of HMGB1 in nasal mucosa of patients with chronic rhinosinusitis with/without nasal polyposis.3–7
The first aim of this study is to demonstrate that HMGB1 is over-expressed in chronic middle-ear pathologies, such as in other chronic inflammatory processes of the ENT district; the second aim is to investigate if the entity of expression and the localization of HMGB1 protein is correlated with the degree of the inflammatory reaction thus suggesting that HMGB1 may play a crucial role in the progression of chronic inflammatory disorders of the middle ear, towards chronic, more severe, and irreversible clinical pictures as already demonstrated in other upper airway diseases.
Material and methods
We examined middle-ear mucosa biopsies of 30 patients (19 women, 11 men) with cholesteatoma (ten cases), COM (ten cases), and otosclerosis (ten cases). Patients with otosclerosis were used as controls. The surgical operation was performed by the same surgeon at “Le Scotte” Hospital, Siena, Italy.
Patient assessment was based on age range, clinical history, symptoms and severity evaluation, hearing loss, previous surgery and recurrences. The mean age of patients enrolled in this study was 47.3 years (age range, 18–75 years).
Prior to surgery, the patients were subjected to an ENT visit: for each patient, an audiometric evaluation was performed. In addition to this, patients were evaluated using a Visual Analogue Scale (VAS) graded from 0 to 10 (0 was absence of symptom and 10 the highest intensity) for pain, hearing loss, vertigo, tinnitus and otorrhea.
During surgery, small mucosa samples were taken from the promontory between the oval and round window.
The tissue samples, always taken by the same surgeon, were fixed in 10% neutral formalin and paraffin-embedded. After routine processing, hematoxylin and eosin (H&E) stain was performed on 5-μm-thick sections. Furthermore, immunohistochemical assay was carried out for each case, using the rabbit polyclonal antibody HMGB1 antibody (Ab 18256) at 1:500 diluition (Abcam, USA). An automated stainer (Leica Bond III) was used. Heat-induced antigen retrieval was performed in citrate buffer pH 6. Specimens were counterstained with Mayer’s Hematoxylin and 3,3’ diaminobenzidine (DAB) was used as chromogen.
Both the histopathological and immunohistochemical evaluations were performed by two blinded pathologists. The study was approved by the Local Ethics Committee at the University of Siena (Comitato Etico di Area Vasta Sud Est – Azienda Ospedaliera Universitaria Senese – 22.04.14) and was performed in accordance with the Helsinki Declaration recommendations. All patients gave informed, signed consent to participate in the study.
The inflammatory cells were characterized histologically, then a score was given to each case according to the intensity of the inflammation (0 = no inflammation; 1 = low inflammation, < 20% of the cells in the examined section; 2 = moderate inflammation, 20–50% of the cells; 3 = intense inflammation, > 50% of the cells).
The distribution of HMGB1 was evaluated as nuclear, cytoplasmic, and/or extracellular staining and the positivity was estimated as a percentage of the total staining in the examined area, with the summarized total staining in the area equal to 100%.
All of the values were presented as the mean ± standard deviation (SD). The data were analyzed by the analysis of variance (ANOVA). A value of P < 0.05 was considered statistically significant.
Results were expressed as the mean values of the evaluations from the two observers.
Results
The clinical characteristics of the three groups are presented in Table 1.
Demographic and clinical characteristics of the enrolled participants: 30 enrolled patients (19 women, 11 men; age range, 18–75 years), ten affected by otosclerosis, ten by chronic suppurative otitis media with ear drum perforation (CSOM), and ten by cholesteatoma.

Hearing loss was expressed by air–bone gap (ABG): the difference between the threshold by bone conduction and air conduction.
All patients underwent surgery. Otosclerosis predominantly affects fertile women: this is the reason why age and sex were significantly different in this group from others.
The inflammatory cells observed in the biopsies were mostly lymphocytes and plasmacells; neutrophils were present in CSOM cases and in two cholesteatoma cases. In one cholesteatoma case eosinophils were also observed.
The mean inflammation cells score was 0.5 for otosclerosis cases, 2.3 for CSOM cases, and 1.9 for cholesteatoma cases (Figure 1). In other words, inflammation is low in otosclerosis and high in cholesteatoma and CSOM.

Semi-quantitative analysis of intensity of inflammation. Intensity of inflammation is measured as a score in middle ear mucosa of three pathology groups (Anova test). Inflammatory infiltrate is very low in otosclerosis (0.5%) and high in COM (2.3%) and cholesteatoma (1.9%).
As the graphs demonstrate (Figure 1), there is a statistically significant difference in inflammation score between the otosclerosis and CSOM samples (P < 0.01; Anova test) and between otosclerosis and cholesteatoma samples (P < 0.05; Anova test). No statistically significant difference in inflammation score was observed between cholesteatoma and CSOM samples (P > 0.05).
The HMGB1 distribution in inflammatory cells was nuclear or cytoplasmic. An extracellular distribution was observed in only two CSOM cases.
HMGB1 positive inflammatory cells comprised a range of 0–50% in otosclerosis samples, 40–90% in CSOM samples, and 0–80% in cholesteatoma samples. The HMGB1 positivity was in accordance with the density of the inflammatory infiltrate.
The HMGB1 expression in epithelial cells was always nuclear and there was no statistically significant difference in the three groups (Figure 2).
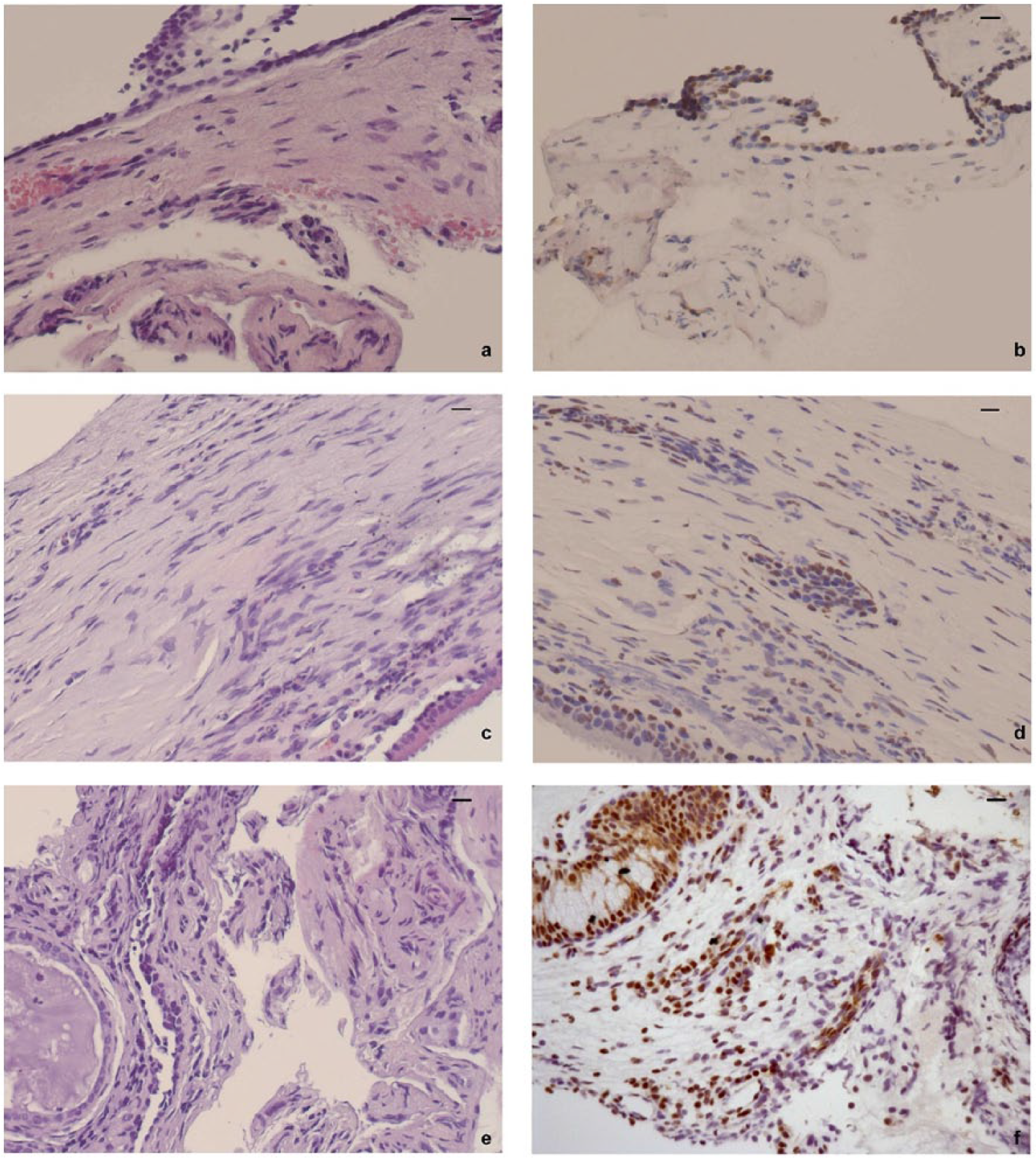
Inflammatory infiltrate and HMGB1 positivity in epithelial and endothelial cells of middle-ear mucosa in patient with (a, b) otosclerosis, (c, d) cholesteatoma, and (e, f) COM. Scale bars: 10 µm. Each section was stained with H&E and subjected to immunohistochemistry (IHC) for HMGB1 detection. (a) Otosclerosis : cubical epithelium and underlying chorion with a minimal inflammatory infiltrate. H&E, 200×; (b) HMGB1 positivity in epithelial and endothelial cells. IHC, 200×; (c) ciliated pseudostratified columnar epithelium and underlying chorion with inflammatory infiltrate composed by lymphocytes, plasmacells, histiocytes, and polymorphonucleated. H&E, 200×; (d) nuclear positivity in epithelial and inflammatory cells. IHC, 200×; (e) copious inflammatory infiltrate, composed of lymphocytes, plasmacells, and neutrophils. Also present is a gland with cubical epithelial cells, correlated to inflammation. H&E, 200×; (f) strong HMGB1 positivity in epithelial and inflammatory cells with prevalently nuclear distribution. IHC, 200×.
Discussion
The middle ear is endowed with several mechanisms of defense against invading pathogens, pollutants, and allergens: the anatomic characteristics of the Eustachian tube (ET) in the first years of life, the mucociliary apparatus of its mucosa, and the secreted mucus and its content of soluble chemical factors such as surfactant proteins, lactoferrin, interferon, and defensins. 8
In addition, different defects of both innate and acquired immune system have been advocated as predisposing factors for developing rAOM/COM. 9
Our study was aimed at evaluating the possible role of HMGB1 protein in middle-ear pathologies and the correlation between HMGB1 and the degree of inflammation. It must be underlined that the measurement of HMGB1 was performed directly at the level of the target organ, the middle-ear mucosa; thus, our findings reflect the local inflammatory reaction. First, we found that cholesteatoma and CSOM samples have higher HMGB1 concentrations than otosclerosis samples. And this finding is in agreement with the specificity of the otosclerotic disorder limited to the bony tissue. For this reason, otosclerosis samples were used in our research as controls.
In addition, in both inflammatory diseases of the middle-ear mucosa, the pathogenic mechanism is different between CSOM and cholesteatoma: cholesteatoma is a highly keratinizing process where the inflammatory infiltrate and the release of cytokines is the molecular responses to cell damage/necrosis. CSOM is an inflammatory/infective process primed by otopathogens.
The paper by Szczepanski et al. 8 strengthens our hypothesis: by histological analysis they demonstrated that HMGB1 protein and its major receptor RAGE are higher expressed in cholesteatoma samples than in normal skin. In addition, in an ex vivo study they showed that HMGB1 prevents human immortalized keratinocyte (HaCaTs) cell apoptosis: HMGB1 release from damaged and/or necrotic cells and its binding to RAGE receptor is the molecular signaling for cellular hyperplasia, proliferation, and cholesteatoma growth.
The different localization of HMGB1 reflects this situation: in fact, it is always nuclear in the epithelial cells of all our samples (cholesteatoma, COM, and otosclerosis); in inflammatory cells it is both nuclear and cytoplasmic, but only in COM samples is the positivity extracellular. In other words, even if no statistically significant difference in inflammatory score and HMGB1 positive inflammatory cells is observed between cholesteatoma and COM samples, only COM samples show higher active progression of the inflammation through the extracellular release of this mediator.
Coming back to the Paparella hypothesis on a continuum which links the different clinical pictures of otitis media, in the light of our results and current literature,9,10 we could postulate that different immune defects of the innate immunity, with specific TLRs or cytokines pattern, drive the response of “otitis prone” children towards cure or more severe forms of otitis media.
Conclusion
There are numerous studies showing up-regulation of HMGB1 in nasal mucosa in patients with chronic rhinosinusitis with/without nasal polyposis. The middle ear is part of the rhinopharyngotubaric unit and so it can be postulated that, in the same way, middle-ear diseases are characterized by up-regulation of HMGB1 and inflammatory infiltrate. This study demonstrates that HMGB1 is over-expressed in chronic middle-ear pathology and the entity of expression correlates with the degree of the inflammatory reaction thus suggesting that HMGB1 may play a crucial role in the progression of inflammatory disorders of the middle ear towards chronicity and a more severe clinical picture.
From a pharmacological point of view, HMGB1 is the best studied HMG protein and, thanks to its ubiquity, could be a suitable target for the treatment of several inflammatory and infectious disorders in the different organs of the body.
Antibodies against HMGB1 can reverse sepsis caused by peritonitis in mice; 11 therefore, the identification of inhibitors of HMGB1 might have significant therapeutic importance in all the chronic inflammatory processes of upper airways including the middle ear. Among inhibitors of HMGB1, glycyrrhetic acid (GA) has been proven to be effective in adults and children with chronic inflammatory disorders of the upper airways having nasal obstruction as the main symptom: in contrast to corticosteroids or antihistamines, it appears to be well tolerated and there are no side effects in humans.12,13
The identification and validation of biomarkers to support the assessment of novel therapeutics is the new frontier in pharmacological researches. Further studies are needed to deepen our knowledge on HMGB1 protein function and its role in the innate immunity mechanisms; however, this protein, thanks to its ubiquity, its characteristics as extracellular cytokine, and in the light of recent researches, appears as a reliable inflammatory biomarker of upper airway diseases severity and a good target for therapy.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
