Abstract
Purpose
To investigate the effects of subconjunctival bevacizumab, ranibizumab, and aflibercept in an experimental corneal neovascularization model.
Materials and methods
The eyes of 24 rats were chemically cauterized and randomly divided into four groups: bevacizumab group (0.05 mL/1.25 mg bevacizumab), ranibizumab group (0.05 mL/0.5 mg ranibizumab), aflibercept group (0.05 mL/1.25 mg aflibercept), and control group (0.05 mL saline solution). Plasma vascular endothelial growth factor (VEGF) levels were among the major measurement outcomes to assess corneal neovascularization. The collected plasmas were analyzed using the SIGMA RAB0511 Rat VEGF Elisa kit. The PCR technique and VEGF amplification procedures were used for molecular analysis. Each cornea was removed and histologically examined on day 21. Corneal images were evaluated by image analyzer software.
Results
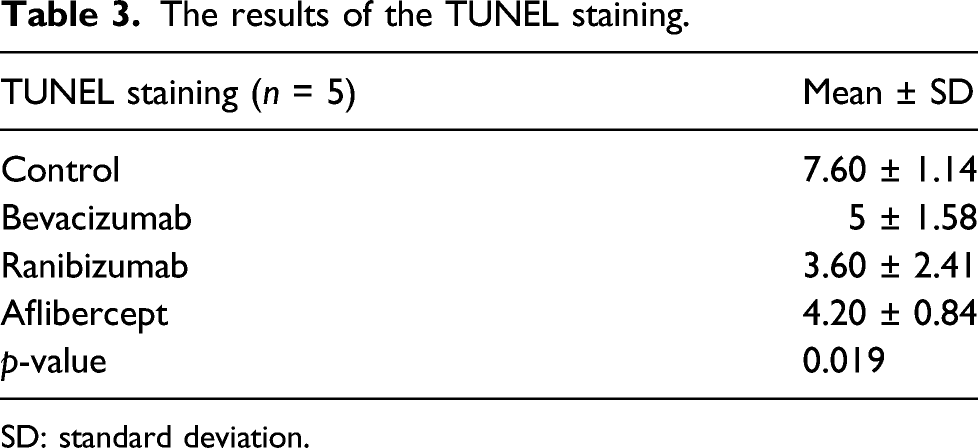
In the post-injection period, the number of major corneal arteries decreased significantly in the injection groups when compared to the control group (p = 0.037), but no statistically significant differences were noted among the injection groups (p > 0.05). The aflibercept group had the lowest area of neovascularization. Immunohistochemical staining revealed substantially lower VEGF percentages in neovascularized arteries of the injection groups than the control group (p = 0.015). In TUNEL staining, the mean TUNEL value (number/1hpf) was substantially greater in the control group than in the injection groups, but the mean TUNEL values were similar between the injection groups (p = 0.019, p > 0.05, respectively). No statistically significant differences were observed between the groups in terms of corneal surface area with increased cellularity, edema, and inflammation (p = 0.263). The mean plasma VEGF concentration in the control group was statistically greater than those in the injection groups (p = 0.001).
Conclusion
Subconjunctival bevacizumab, ranibizumab, and aflibercept crossed the blood and seemed to be effective in inhibiting corneal neovascularization without causing epitheliopathy in an experimental rat model compared to the controls. However, no significant results were noted between these three anti-VEGF molecules.
Keywords
Introduction
The cornea is a transparent connective tissue that is avascular and non-lymphatic in nature. 1 Corneal avascularity is required to maintain good visual acuity. 2 Corneal vascularization is possible when an imbalance between angiogenic and antiangiogenic stimuli results in an increase in proangiogenic factors (such as vascular endothelial growth factor (VEGF), basic fibroblast growth factor, and matrix metalloproteinases 1-2) and an antigenic deficiency (such as soluble VEGF receptor 2, angiostatin, endostatin, and pigment epithelium-derived factor).3,4 This condition may be caused by contagious, traumatic, toxic, ischemic, degenerative, or inflammatory problems.5,6 Although the etiology is unknown, VEGF certainly plays a significant role in corneal neovascularization (NV), both in the human and experimental cornea. 7
Corneal NV is a leading cause of blindness, which affects 4.14% of individuals with eye problems, or more than one million people in the United States each year. 8 Corneal NV has been recently treated with laser photocoagulation, topical steroid and non-steroid anti-inflammatory drops, fine needle diathermy, photodynamic therapy, and limbal, conjunctival, or amniotic membrane transplantation.5,8 However, none of these treatments have shown a permanent recovery.
Antiangiogenic therapy promises the treatment of corneal NV in the future as VEGF levels have been observed to increase during angiogenesis in the cornea.1,5,8 Bevacizumab, ranibizumab, and aflibercept are the anti-VEGF drugs that have been currently used for the clinical treatment of corneal NV. Bevacizumab (Avastin, Genentech, San Francisco, California, USA) is a full-length humanized anti-VEGF monoclonal immunoglobulin G1 antibody against all isoforms of VEGF. 9 This antibody combines with VEGF, which may help to form abnormal blood vasculature and reduce vascular permeability, thus deactivating VEGF. Bevacizumab was first authorized by the US Food and Drug Administration (FDA) for the treatment of metastatic colon cancer. Meanwhile, it was also successful in the treatment of a variety of neovascular ocular disorders and has been used off-label to treat neovascular age-related macular degeneration. 5
Ranibizumab (Lucentis, Genentech, South San Francisco, California, USA) is a recombinant DNA-based antibody particle capable of inhibiting all physiologically active isoforms of VEGF as well as the active proteolytic component of VEGF. 10 Aflibercept (Eylea, Berlin, Germany) is a receptor fusion protein fused to the Fc domain of human IgG1. It is composed of the third domain of human VEGF receptor-2 and the second domain of human VEGF receptor 1. It functions as a receptor decoy for all VEGF-A isoforms. Additionally, the interaction of VEGF-B and placental growth factors 1 and 2 can enhance the angiogenic response.11,12 It has the greatest affinity of all anti-VEGFs tested so far. The FDA has authorized ranibizumab and aflibercept for the treatment of macular degeneration.
Several different studies are reporting how efficient these three molecules are in the treatment of corneal NV.13–18 However, the effects of these three molecules on the experimental corneal NV have not been compared simultaneously by measuring the plasma concentration of these three anti-VEGFs and using immunohistochemical staining methods. This study aims to not only compare the inhibitory impact of subconjunctival bevacizumab, ranibizumab, and aflibercept injections on the experimental corneal NV model by determining plasma VEGF levels, but also to assess therapy response by studying CD31 and VEGF levels and using TUNEL immunohistochemical staining.
Methods
This study included 24 right-hand side corneas of 24 male Wistar albino rats weighing 250–300 g. The experimental protocol was approved by Abant İzzet Baysal University, Medical School Experimental Animals Local Ethical Committee (Approval number: 2016-49). Subjects were administered and maintained according to the principles of the Association for Research in Vision and Ophthalmology for use of animals in ophthalmic and vision surveys. Each subject was held in different cages under approved circumstances.
The subjects were randomly assigned to one of four groups (control group-saline, bevacizumab group, ranibizumab group, or aflibercept group), each of which included six rats. Only the right eyes were cauterized for the induction of corneal NV. The procedures were performed under topical anesthesia (proparacaine hydrochloride, 0.5%) and general anesthesia (intraperitoneally administered mixture of 50 mg/kg ketamine hydrochloride and 5 mg/kg xylazine hydrochloride). Silver nitrate was utilized in conjunction with a 2 mm diameter chemical applicator stick to cauterize the corneal centers immediately after the anesthesia. These silver nitrate sticks (75% silver nitrate and 25% potassium nitrate) were employed on the corneal centers for 10 s. Following cauterization, the corneas and fornices were cleaned with a balanced salt solution to eliminate any residual chemicals. The first day was considered the day of corneal cauterization. Ophthalmological examinations and weight controls of the rats were maintained at regular intervals. All rats were re-examined under topical and general anesthesia on day 10 to evaluate corneal NV.17,19 The number of major vessels formed on the neovascularized corneas was counted after photographing with a Nikon digital camera (×25 magnification).
The effective ophthalmic doses of anti-VEGF molecules have been reported in different concentrations for bevacizumab, ranibizumab, and aflibercept.20,21 So, the administered doses of these three molecules were different in nature. Therefore, the study aimed to compare the effects of the molecules for the recommended doses, not to compare the dose-dependent effectivity of the molecules. The process of medication dosage adjustment was performed similarly to previous studies.20,21 Using a 27-gauge needle, 0.05 mL isotonic saline solution, 0.05 mL bevacizumab (Avastin; 5 mg/0.05 mL), 0.05 mL ranibizumab (Lucentis; 0.5 mg/0.05 mL), and 0.05 mL aflibercept (Eylea; 40 mg/mL) were administered subconjunctivally to the control, bevacizumab, ranibizumab, and aflibercept groups, respectively. Chemical cauterization and subconjunctival injections were applied under sterile conditions by the same physician. Burns were graded uniformly according to Mahoney and Waterbury’s definition.
22
The vascularized area differences between the four groups are represented in Figure 1. View of rat cornea on the 10th day of study: (a) control group, (b) bevacizumab group, (c) ranibizumab group, and (d) aflibercept group.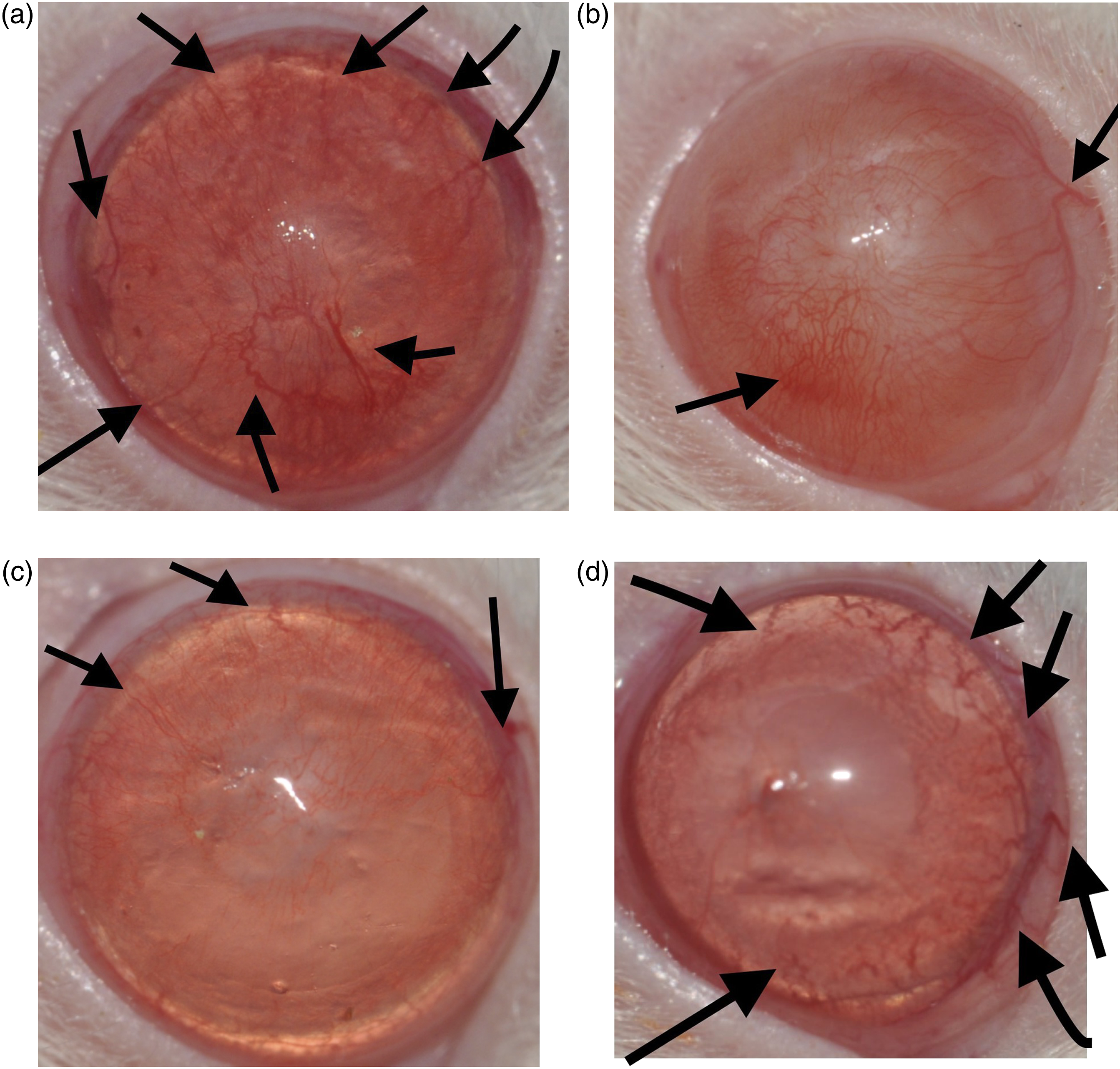
The neovascularization area was measured in pixels and the ratio of the neovascularization area to the total corneal area was defined in % using image analyzer software (Pixcavator Image Analyzer; Intelligent Perception, Huntington, West Virginia, USA). The samples were taken immediately before scarification in order to determine the plasma levels of VEGF. The samples were placed in 0.5 mL ethylenediaminetetraacetic acid (EDTA) capillary tubes. Their blood samples were centrifuged at +4°C for 20 min at 2000 r/min. Obtained plasma samples were kept at −70 and assessed using Rat VEGF Elisa kit (@SİGMA RAB0511). Polymerase chain reaction (PCR) technique was used for molecular analysis. The results of the VEGF amplification process (pg/mg) were compared within the groups. A high dosage of intraperitoneal pentobarbital was injected into all rats on day 21. The number of major vessels established on the corneas was counted after a second photographing.
Histopathological and immunohistochemical methods
The enucleated eye materials obtained from the rats were labeled in accordance with the group numbers, and they were delivered to the pathology laboratory with capped containers containing 10% formaldehyde (Figure 2). Eyes with routine tissue monitoring procedures on Leica brand tissue monitoring device were embedded in paraffin blocks via a Thermo scientific brand device. Sections with a thickness of 5 microns were obtained with the Leica brand microtome device and put into lysine-coated slides. While routine H&E (hematoxylin–eosin) staining was applied to the first sections; CD31 (PECAM-1, Clone JC70, sc-376764, Santa Cruz Biotechnology, CA, USA) and VEGF (C-1 clone, sc-7269; Santa Cruz Biotechnology, CA, USA) were stained in the other sections using Ventana brand automated immunohistochemical device. Terminal deoxyribonucleotide transferase-mediated dUTP-digoxigenin nick end labeling (TUNEL) was stained manually. TUNEL staining was performed using the ApopTag kit (Oncor, Purchase, New York, USA). Briefly, sections (4 μm) were deparaffinized in xylene, rehydrated in decreasing concentrations of ethanol, boiled in Citra (Biogenex, San Raman, California, USA) for 10 min, and digested in 0.5% pepsin for 60 min at 37°C, before endogenous peroxidase was blocked in 3% hydrogen peroxide. Three different dilutions (1:7, 1:11, and 1:16) of terminal deoxynucleotidyl transferase (TdT) in reaction buffer (containing a fixed concentration of digoxigenin-labeled nucleotides) were applied to serial sections for 1 h at 37°C, before the slides were placed in stop/wash buffer for 10 min. Following washes, a prediluted anti-digoxigenin peroxidase-conjugated antibody was applied for 30 min. Apoptotic cells were detected after incubation in the 3,3-diaminobenzidine (DAB) chromogen (DAKO, Carpinteria, California, USA) for approximately 6 min and slides were counterstained with methyl green (Sigma, St Louis, Missouri, USA).
23
X40, H&E; normal anterolateral site of the enucleated eye materials in H&E sections; lens (yellow circle); lens epithelium (yellow arrow); iris (blue arrows); a small part of retinal layers (red arrows); ciliary body (white star); normal corneal area adjacent to the cauterized study corneal centers (orange square); corneal epithelium (green arrow); and corneal endothelium (pink arrow).
Stained glasses were closed with a cover glass with the help of balsam and analyzed histopathologically and immunohistochemically with Olympus Bx 51 brand light microscope. In H&E sections, corneal edema and density and types of inflammatory cells and stromal cellularity rates were assessed according to international histopathologic study scoring standards.24,25 Corneal stromal edema was scored from 0 to 3 according to the density (0: no edema, 1: slight edema, 2: moderate edema, and 3: severe edema). Inflammatory cell density was determined in the affected area and inflammatory cell types were described in percentages. Inflammation cells were counted in 1 HPF (high power field, X400) area and then they were numbered. In the affected area, stromal fusiform cells in the corneal stroma were scored according to the cell cellularity from 0 to 3 (0: normal, 1: slightly increased cellularity, 2: moderately increased cellularity, and 3: severely increased cellularity). The percentage of the affected corneal surface area (area of increased vascularity, edema, and cellularity) to the entire corneal area was expressed as the percentage.
Neovascularized vessel numbers in the cornea were counted in 1 HPF area with the help of immunohistochemically examined CD31. Immunohistochemical VEGF staining was assessed according to staining percentage on the neovascularized vessels (VEGF-positive vessel numbers/all neovascularized vessel numbers) and staining density on the positive vessel endothelium (0: no staining, 1: slight density, 2: moderate density, and 3: severe density). TUNEL staining, which stained the nuclei of epithelium, endothelium, and stromal cells, was counted in 1 HPF area. The results were summarized in tables and statistically analyzed.
Statistical analysis
Statistical analysis of the study was performed by using SPSS for Windows version 15. The comparison of the data was performed with the Kruskal–Wallis test. Different groups were determined by the Dunn test. The significance level of the p-value was set as <0.05.
Results
The percentage of neovascularization area and the number of blood vessels in different injection groups compared to the control group.

n: number; hpf: high power field. p <0.05.
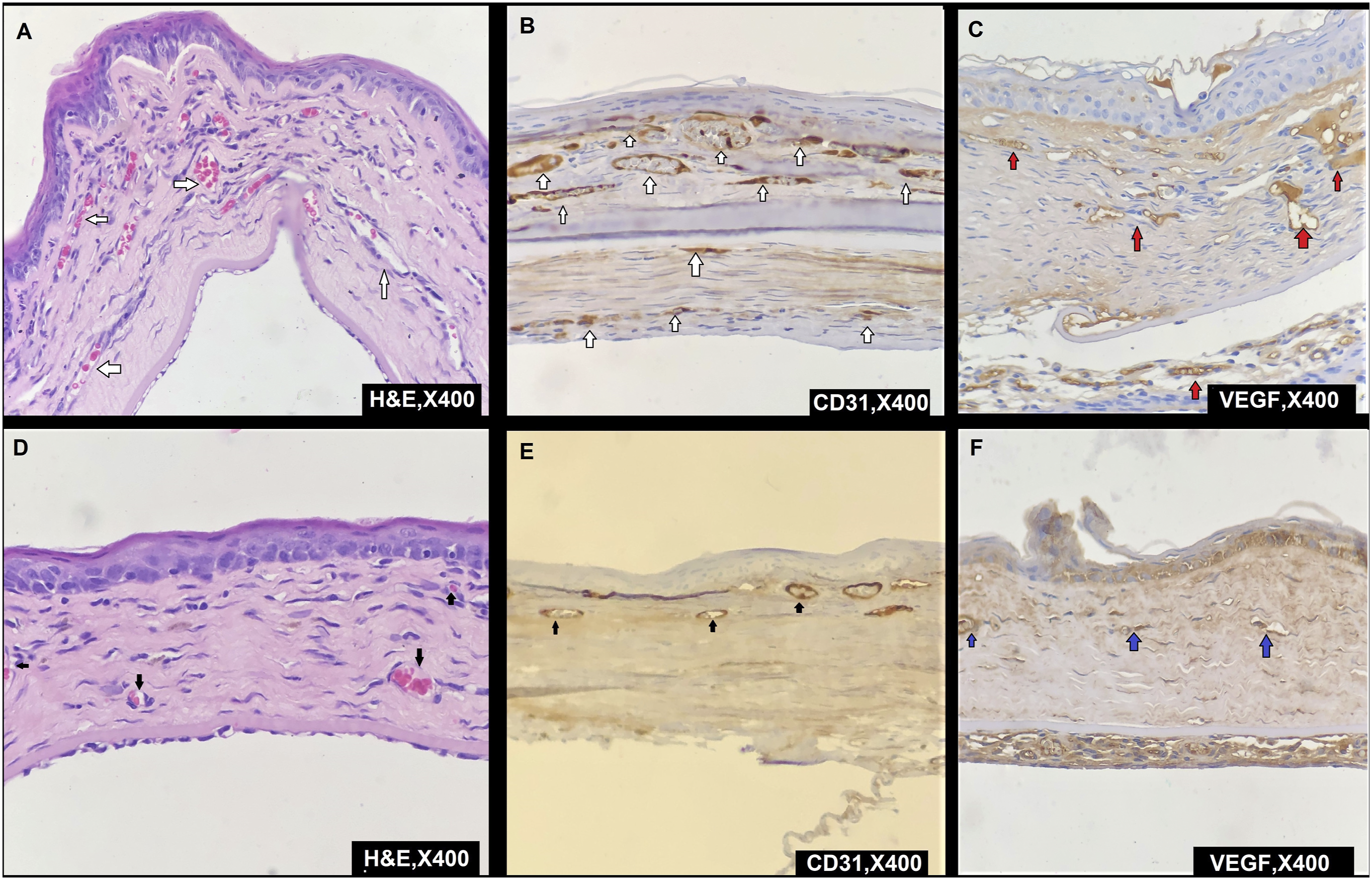
Neovascularization rates and VEGF staining levels in corneal specimens: (a) X400, H&E; increase in the number of neovascularized vessels in the control group (white arrows), (b) X400, CD31; increase in the number of neovascularized vessels in the control group with CD31 immunohistochemical staining (white arrows), (c) X400, VEGF; higher VEGF densities of neovascularized vessels in the control group with VEGF immunohistochemical staining (red arrows), (d) X400, H&E; reduction in the number of neovascularized vessels in the control group (black arrows), (e) X400, CD31; reduction in the number of CD31-labeled neovascularized vessels in the post-injection treatment groups with CD31 immunohistochemical staining (black arrows), (f) X400, VEGF; lower VEGF densities of neovascularized vessels in the control group with VEGF immunohistochemical staining (blue arrows). VEGF: vascular endothelial growth factor.
The results of the vascular endothelial growth factor staining.

VEGF: vascular endothelial growth factor.
The results of the TUNEL staining.
SD: standard deviation.

Stromal cellularity, edema, inflammatory cells, and TUNEL immunohistochemical staining in groups: (a) X400, H&E; stromal cellularity (yellow circle), edema (red star), and inflammatory cells (blue square) in the treatment groups, (b) X400, H&E; stromal cellularity (yellow circle), edema (red star), and inflammatory cells (blue square) in the control group, (c) X1000, H&E; increased eosinophils (orange arrows) in the control and ranibizumab groups, (d) X1000, H&E; other inflammatory cells; macrophage (black arrows), plasmacyte (blue arrows), and less eosinophils (orange arrows) in both groups, (e) X1000, TUNEL; increased numbers of nuclear TUNEL positivity in the cornea epithelium, stroma cells, and endothelium of vessels in the cornea of the control group (white arrows), (f) X1000, TUNEL; quite a few numbers of nuclear TUNEL positivity in the cornea epithelium and stroma cell in the cornea of post-injection treatment groups (blue arrows). TUNEL: terminal deoxyribonucleotide transferase-mediated dUTP-digoxigenin nick end labeling.
The rates of corneal edema and inflammatory cell density in hematoxylin–eosin sections and the assessment of cell types and stromal cellularity.

hpf: high power field.
The mean plasma values of the vascular endothelial growth factor presented as mean concentration ± standard deviation.

SD: Standard deviation
Discussion
The results of this study postulated that administration of subconjunctival anti-VEGF injections can prevent the development of corneal NV. Immunohistochemical evaluation of each injection group revealed a reduction in corneal NV, which was consistent with initial biomicroscopic examination. To the best of our knowledge, no reports have been documented comparing the effects of three distinct anti-VEGF injections on corneal NV in an animal corneal cauterization model. Photographs of rat corneas demonstrated no evidence of corneal epitheliopathy in any group of injection.
Considering the important role of VEGF in the development of corneal NV, the treatment of anti-VEGF antibodies seemed to be the best course of action. VEGF-A, -B, -C, and -D are all members of the human VEGF family. VEGF-A is considered the most important member of the family because of its role in pathological hemangiogenesis. 26 Bevacizumab is a recombinant humanized monoclonal IgG1 antibody. It specifically binds to VEGF-A and also inhibits the biological activity of VEGF-A. Thus, it can prevent neovascularization. 27 Ranibizumab is a recombinant and humanized monoclonal antibody fragment that inhibits angiogenesis by neutralizing all VEGF-A isoforms and degradation products. 28 Bevacizumab and ranibizumab both act by binding to antibodies. Aflibercept is a fusion protein that has two binding sites for VEGF receptors 1 and 2 and PIGF (another advanced proangiogenic agent). These components contribute to the higher binding capacity of aflibercept than the other two anti-VEGFs.26,27
Anti-angiogenesis against corneal NV was assumed as the comparable effect of anti-VEGFs in this study. It is known that various anti-VEGFs exhibit varying drug half-life, vascular permeability, and binding affinity for VEGF-A. We hypothesized that bevacizumab, ranibizumab, and aflibercept would have significantly different antiangiogenic effects on corneal NV. However, similar antiangiogenic effects were observed in three anti-VEGFs despite their significant results compared to the control groups.
Hosseini et al. found that a single subconjunctival bevacizumab injection resulted in a 32% decrease in corneal NV and shorter arteries in rabbit corneas cauterized with alkali. 29 Manzano et al. observed a 40% reduction in NV areas compared to the control group after 7 days of topical bevacizumab 4 mg/mL application. 30 Barros and Belfort utilized modest doses of subconjunctival bevacizumab (0.05 mg) and found a 17.7% decrease in the NV area during the post-injection period. 31 In our study, the rate of decrease in the corneal NV area was found to be 39.5% in the bevacizumab group, 48.5% in the ranibizumab group, 48% in the aflibercept group, and 7.5% in the control group. The changes were statistically significant between the injection and control groups. Histological examinations revealed no statistically significant variations in the mean number of blood vessels between the groups. In histological sections, the corneal NV area was found to be the smallest in the ranibizumab group and the largest in the control group. Akar et al. evaluated the efficacy of subconjunctival bevacizumab, ranibizumab, and pegaptanib sodium in rats and found a substantial reduction in corneal NV of all three groups receiving anti-VEGF therapy compared to the control group. 32 Besides, it was found that bevacizumab was more effective than ranibizumab and pegaptanib sodium. These results were also histologically confirmed.
In their experimental study on rats, Türkçü et al. evaluated the efficacy of subconjunctival and topical ranibizumab. 19 They discovered that the blood vessels of the treatment groups shrank considerably more than those of the control group. However, they found no significant changes in the rate of NV between the treatment groups. Histopathological examinations revealed that the treatment groups had substantially fewer new vasculature and inflammatory cells than the control group. They examined CD34 and von Willebrand factor stainings, as well as group scores, and discovered that both stainings were substantially larger in the control group compared to the treatment groups. The average number of eosinophils was substantially higher in the control and ranibizumab groups than in the other two groups, but no significant difference was observed between the bevacizumab and aflibercept groups. There are limited studies on this subject in the literature. If the severity of inflammation remains constant but the number of inflammatory cells increases or decreases, this might be due to an increase or decrease in extravasation caused by the effect of the relevant drug on the inflammatory cells.
Ruti et al. compared the effects of topical ranibizumab and topical bevacizumab on corneal NV in rats. They demonstrated that the NV area in the aflibercept group was smaller than the control and bevacizumab groups on days 7 and 10. 17 They also found that staining was 29% in the aflibercept group, 92% in the bevacizumab group, and 86% in the control group, which were all statistically significant. CD31 immunofluorescence labeling showed that endothelial proliferation was not detected in the bevacizumab or control group; however, it was detected in the aflibercept group.
Avisar et al. found that CD31 staining of vascular endothelial cells increased corneal NV labeling of chemically damaged endothelial cells and then caused a substantial reduction in staining following bevacizumab injection. 3 However, they claimed that PCR studies on the efficacy of bevacizumab on corneal NV did not yield a conclusive result, but that there was a trend toward decreased VEGF mRNA levels in the injected subjects when compared to controls. Similarly, staining with CD31 reduced substantially in the injected groups in our study. However, no significant change in VEGF density was observed between the groups.
Dursun et al. have shown that subconjunctival bevacizumab and ranibizumab treatments may be beneficial in reducing corneal NV. 33 Additionally, they claimed that bevacizumab had a greater inhibitory impact on corneal NV than ranibizumab. They demonstrated that the longest neovascular sprout length was substantially shorter in the bevacizumab and ranibizumab groups than in the control group. Additionally, both treatment groups had substantially lower corneal edema scores than the control group. In contrast, our study found no significant difference in corneal stromal edema. We found that the mean VEGF level of the control group was statistically higher than those of the injection groups, whereas no statistically significant differences were noted between the injected groups.
Roald et al. used intravitreal aflibercept to treat 16 individuals with naïve neovascular macular degeneration. 34 To summarize, aflibercept significantly reduced the plasma VEGF levels (which was 56% on the first day) immediately after the treatment. Despite the monthly intravitreal injection of aflibercept, plasma VEGF levels were 87% of baseline in the first month and 90% in the second month. One and a half months after the subsequent dosage, plasma VEGF levels improved significantly. Regardless of re-administration dosages of aflibercept, the main reason responsible for this increase in plasma VEGF values retained its ambiguity.
Park et al. found that VEGF mRNA levels were 0.227 in the 0.1% aflibercept group, 0.811 in the 0.01% aflibercept group, and 0.495 in the 0.1% bevacizumab group in a rabbit experiment examining the effects of different doses of topically administered aflibercept on corneal NV. 18 All treatment groups had substantially lower VEGF levels than the control group (1.491), (p = 0.031, p = 0.05). However, the difference between the 0.01% aflibercept, 0.1% aflibercept, and 0.1% bevacizumab groups was smaller than the difference between the 0.1% aflibercept and 0.1% bevacizumab groups. There was no significant difference between the 0.1% aflibercept and 0.1% bevacizumab groups.
Ahmed et al. conducted a study investigating how rabbits responded to bevacizumab after experimental corneal NV and the relation between the neovascularization and VEGF levels in the precorneal film and aqueous humor. 35 While NV in the control group increased linearly from 7 to 14, the VEGF concentrations in the precorneal layer and aqueous humor also increased. Up to the 14th day, VEGF levels in the precorneal film and aqueous humor were significantly lower (26.9%) in those who were administered 12.5 mg/mL topical bevacizumab with 8 h interval a day than in the control group.
Different from other studies, Turkcu et al. used ranibizumab in their study including five groups. The first group served as the control group and only teardrops were administered, the second group received topical ranibizumab two times a day, the third group received topical ranibizumab four times a day, and the fourth and fifth groups were injected subconjunctival 0.5 mg/0.05 mL, and 1 mg/0.1 mL ranibizumab at first, third and seventh days, respectively. In the histopathological investigation of hematoxylin–eosin, immunohistochemical CD34, and von Willebrand factor examinations; fewer inflammation cells, fewer vessels, and less fibroblast activity were examined in the treatment groups compared to the control group. 19 However, this current study uses three anti-VEGF molecules of ranibizumab, bevacizumab, and aflibercept and reports reduced vascularity after the usage of all three anti-VEGF molecules, yet it does not report any significant difference between the three molecules.
Both subconjunctival injections and topical applications are frequently used as easy-to-apply methods with minimal complications in ocular drug administration. Although subconjunctival injections may be a viable option for individuals who are non-compliant with topical therapy, they may cause potential adverse effects such as accidental globe perforation or an oculocardiac reaction. 36 In addition, the subconjunctival dosage must be administered by a physician, whereas the patient can apply topical administrations by herself/himself. Considerable limitations for topical application include formulation issues, poor water solubility, and poor solution stability, all of which contribute to vulnerability to bioactivity loss during prolonged storage. 37 In addition, long-term topical bevacizumab treatment may result in epitheliopathies and descemetocele.38,39
Conclusion
In a rat model of alkali burn-induced corneal NV, it is demonstrated that subconjunctival administration of bevacizumab, ranibizumab, and aflibercept effectively inhibited corneal NV, reduced inflammation, and fibroblast activity, and all three anti-VEGF molecules crossed to the blood. Although the therapeutic concentration of anti-VEGFs in the cornea was not established in our study, it is observed that single subconjunctival dosages of bevacizumab (0.05 mL/1.25 mg), ranibizumab (0.05 mL/0.5 mg), and aflibercept (0.05 mL/1.25 mg) were effective. However, we do not know the optimal subconjunctival dosage of bevacizumab, ranibizumab, or aflibercept to prevent corneal NV in this animal model. Currently, the off-label use of these drugs for corneal NV is prohibited. The main strength of this study is that it may be the first report comparing three anti-VEGF compounds simultaneously and utilizing TUNEL immunohistochemical labeling.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project is supported by Duzce University Research Fund Project [Grant Number: 2016.04.02.473].
