Abstract
T-2 toxin pre-disposes individuals to osteoarthritis, Kashin–Beck disease (KBD). The major pathological change associated with KBD is the degradation of the articular cartilage matrix. Herein, we investigated the key molecules that regulate T-2 toxin-mediated cartilage degradation. Potential KBD treatments were also investigated. Sprague Dawley rats were divided into the T-2 toxin group and the control group. The T-2 toxin group received 100 ng/g BW/day, whereas the control group received a similar dose of PBS. The expression of matrix metalloproteinase-13 (MMP-13) and TGF-β receptor I/II (TGF-βRI/II) was analyzed using immunohistochemical staining. C28/I2 chondrocytes were exposed to TGF-βRI/II binding inhibitor (GW788388) for 24 h before incubation in different T-2 toxin concentrations (0, 6, 12, and 24 ng/mL for 72 h). The expression of mRNA for TGF-βRI/II, MMP-13 and proteins for MMP-13, and Smad-2 in chondrocytes were analyzed using RT-PCR and western blot, respectively. Safranin O staining revealed that T-2 toxin treatment modulated the expression of articular cartilage matrix. On the other hand, T-2 toxin treatment sharply increased the expression of MMP-13, TGF-βRI, and TGF-βRII in the rat cartilages. Interestingly, blocking the TGF-βRs-smad 2 signaling pathway using GW788388 abrogated the effect of T-2 toxin on upregulating MMP-13 expression. The expression of MMP-13 in chondrocytes induced with T-2 toxin is regulated via the TGF-βRs signaling pathway. As such, inhibiting the expression of TGF-βRs is a potential KBD treatment.
Introduction
T-2 toxin is the most toxic trichothecene, which is a group of mycotoxins mainly produced by members of the Fusarium genus. These mycotoxins present a potential threat to animal and human health throughout the world. 1 T-2 toxin causes diverse toxic effects in animals such as weight loss, decreases in erythrocytes and leukocytes, hypoglycemia, as well as liver and stomach damages. 2 Trichothecenes have been detected in several field crops (wheat, maize, barley, oats, etc.) and processed grains (malt, beer, bread, etc.). These mycotoxins are generally toxic to humans.3,4 T-2 toxin is thought to predispose individuals to Kashin–Beck disease (KBD), a chronic and endemic type of osteochondropathy in China.5, 6 After the dietary change from excessive consumption of grains combined with nutritional supplementation, the incidence of KBD in endemic areas decreased dramatically from 12.31% in 2000 to 4.21% in 2007. 7 Even so, the incidence of KBD is still considerably high in western China.8,9 Given that it is practically impossible to transfer all residents in endemic areas to better regions, there is need to understand how T-2 toxin mediates KBD development, which can reveal corresponding treatment strategies.
The pathological features of KBD include the degeneration and death of chondrocytes in the deep layer of cartilage and an altered cartilage matrix metabolism. Research shows that type II collagen is under-expressed in individuals with KBD, whereas an opposite trend is true for matrix metalloproteinases (MMPs). Overall, these indicators represent the degradation of the extracellular matrix (ECM) in the articular cartilage of these patients.10,11 MMP-13 is the major enzyme that catalyzes cartilage degradation. Compared with other MMPs, the expression of MMP-13 is more restricted to connective tissues 12 and targets collagen II in cartilage for degradation. 13 T-2 toxin can alter the expression of collagen II and its regulatory enzymes MMP-13 in KBD rat models. 14 Further in vivo and vitro studies revealed that T-2 toxin induces degradation of cartilage matrix by upregulating MMP-13 expression. 15 It also induces apoptosis in human chondrocytes by upregulating the expressions of apoptosis genes.16,17 These findings underline the role of T-2 toxin in the degradation of matrix cartilage in KBD patients. However, T-2 toxin-mediated KBD pathogenesis is not well understood.
Transforming growth factor-
Methods
Experimental Rats
Sprague Dawley (SD) rats were purchased from the Experimental Animal Center of Xi’an Jiaotong University. In order to mimic the ages of the human adolescents who are the most susceptible population to KBD, all of the animals used in this study were initially 1 month old and weighed from 60 g to 80 g. T-2 toxin was purchased from the Toronto Research Chemicals (Toronto, Canada). T-2 toxin was dissolved in absolute ethanol to a concentration of 5 mg/mL and then diluted with 0.9% normal saline. As our previous study, a daily dose of 100 ng/g of BW was selected for the experimental rats, 20 which altered cartilage matrix metabolism like KBD. The rats were randomly divided into two groups. The experimental group (T-2 toxin group, n = 6) received a daily dose of 100 ng/g of BW, whereas the control group (n = 6) received 0.9% normal saline. In both cases, the treatments were administered intragastrically for 4 weeks as previously described. 21 The rats used in the study were 4 weeks old when the treatments started and they grow quickly until they are 8 weeks old, which is similar to the growth spurt seen in adolescent-aged children between 5 years and 15 years old found in KBD areas.
Rat cadavers were obtained after euthanasia by intraperitoneal injection with an overdose of barbiturate (200 mg/kg). The knee joints were processed for histopathological evaluation. The protocol for this study was approved by the ethics committee of Xi’an Jiaotong University, and the experiments were conducted in line with the Helsinki Declaration.
Chondrocyte culture
The C28/I2 human chondrocyte was sourced from the Weill Medical College of Cornell University (New York, USA). The cells were cultured in Dulbecco’s Modified Eagle Medium F-12 (DMEM/F-12) (HyClone, Logan, Utah, USA) supplemented with 10% fetal bovine serum (FBS, Gibco, USA) and a mixture of 1% penicillin and streptomycin (penicillin: 100 U/mL, streptomycin: 100 μg/mL).
Cell viability assay
C28/I2 chondrocytes were cultured in 96-well plates as described above. MTT assay was then performed using the MTT assay kit (Abcam, Cambridge, MA, USA), following the manufacturer’s instructions. The 96-well plate was read at 590 nm using a microplate reader (Tecan, Männedorf, Switzerland).
T-2 toxin was dissolved in absolute ethanol to a concentration of 5 mg/mL and then diluted with PBS. According our previous studies,22,23 we selected 0, 6, 12, and 24 ng/mL of T-2 toxin, which reduced C28/I2 chondrocytes viability by 10%, 25%, and 50% for subsequent experiments.
To determine the concentration of TGF-βRs inhibitor (GW788388) in C28/I2 chondrocytes, the cell viability of chondrocytes was detected after the addition of 0–100 μM GW788388 for 24 h. GW788388 was purchased from MCE (Monmouth Junction, NJ, USA).
Histology
Rat knee joints and engineered cartilaginous tissues were fixed in 4% paraformaldehyde, embedded in paraffin, and sliced into 5 μm sections. Safranin O-Fast green staining was used to evaluate the general tissue structure. Masson staining and picrosirius red staining were used to evaluate extracellular matrix.
Assessment of gene expression
Total RNA from chondrocytes (C28/I2) was extracted using TRIzol (Invitrogen, Life Technologies, Karlsruhe, Germany). First-strand cDNA was synthesized using the RevertAid First Strand cDNA Synthesis kit (Thermo Fisher Scientific, Waltham, MA, USA). The relative level of gene expression was quantified using reverse transcription polymerase chain reaction (RT-PCR). The Fast Start Essential DNA Green Master reagent (Roche, Mannheim, Germany) was used. Details of the primers used in this study are shown in Supplementary Table 1.
Protein expression
To assess the expression of/the effect of TGF-βRs and MMP-13, immunohistochemical staining (IHC staining) and western blot assay were used. TGF-βRI (Santa Cruz, Dallas, TX, USA), TGF-βRII (Santa Cruz, Dallas, TX, USA), and MMP-13 (Proteintech, Rosemont, IL, USA) were incubated overnight at 4°C according to the manufacturer’s protocol. The tissue sections of rats were then stained using diaminobenzidine, observed under a light microscope (Olympus Corporation, Japan), and photographed using a digital camera (Canon Corporation, Japan). Cells in six random fields of the three articular cartilages were labeled and counted using Image J software (NIH, USA), under 400×/200× magnification. The images were analyzed independently by two scientists blinded to the experimental conditions.
The expression of MMP-13 and Smad-2 in the C28/I2 chondrocytes was analyzed using the western blot assay. The cells were incubated for 30 min at 37°C and thereafter overnight at 4°C in media supplemented with Smad-2 (Abcam, Cambridge, MA, USA) and MMP-13 (Proteintech, Rosemont, IL, USA). GAPDH (Bioss, Beijing, China) was used as the internal control.
Preparation of engineered cartilage
Demineralized bone matrix gelatine (BMG) scaffolds were harvested from four freshly euthanized adult New Zealand white rabbits (from the Experimental Animal Centre of Xi’an Jiaotong University) as previously described. 24 We have detected the type II collagen and proteoglycan of engineered cartilages in vitro and in vivo.25-27 They all showed that engineered cartilages are functional. The BMG scaffolds were sterilized for 48 h using ethylene oxide and thereafter pre-incubated for 24 h at 37°C in PBS. Then, 1.0×106 chondrocytes re-suspended in 100 μL DMEM/F-12 medium were seeded in centrifuge glass tubes containing BMG graft. After 4 h, 4 mL of culture medium containing 10% (v/v) FBS was carefully added to the chondrocytes–cancellous BMG constructs and cultured for another 14 days. The medium was changed every 2 days. On day 14, the engineered cartilage tissues were treated with T-2 toxin at 12 ng/mL and TGF-βRs inhibitor at 20 μM for an additional week. T-2 toxin and TGF-βRs inhibitor were diluted with culture medium.
Statistical analysis
Quantitative data are expressed as mean ± standard deviation (SD) from three independent experiments. Statistical analyses were performed using one-way analysis of variance (ANOVA) and Student’s t-test. Statistical significance was set at P < 0.05.
Results
Morphology of articular cartilage from rat treated with T-2 toxin
Safranin O staining revealed that compared with normal cartilage (Figure 1(a)), T-2 toxin leaded the degradation of ECM, which is mainly composed of several collagens (Figure 1(b)). Safranin O staining in the articular cartilage. a: articular cartilage from control rat; b: articular cartilage from rat treated with T-2 toxin (a daily dose of 100 ng/g of BW for 4 weeks).
T-2 toxin increases the expression of MMP13, TGF-βRI, and TGF-βRII in rat articular cartilage
IHC staining revealed that T-2 toxin treatment significantly increased the expression of MMP-13 and TGF-βRI/II in the articular cartilage (Figure 2). MMP-13 is the major enzyme that hydrolyzes type II collagen, the major collagen in articular cartilage. IHC staining further revealed that both TGF-βRI and TGF-βRII are localized in the membrane, adjacent to the cytoplasm (Figure 2). IHC staining in the rat articular cartilage. a, d, g: articular cartilage from control rat; b, e, h: articular cartilage from rat treated with T-2 toxin (a daily dose of 100 ng/g of BW for 4 weeks). A, B: representative images showing MMP-13 staining. d, e, g, h: representative images showing TGF-βRI and TGF-βRII staining. c, f, i: quantitative data of MMP-13, TGF-βRI, and TGF-βRII (* P< 0.05 versus control group, n = 6).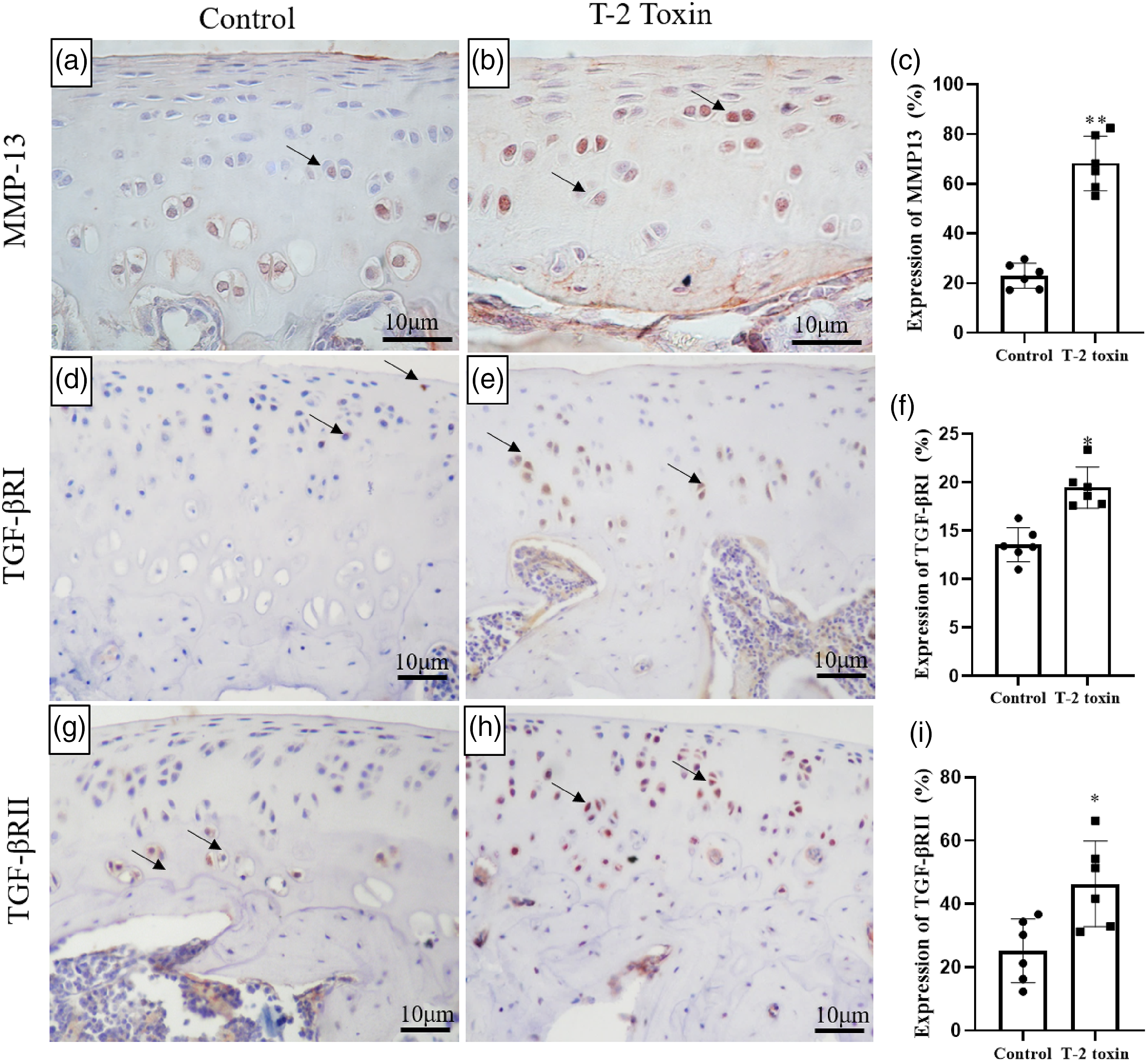
GW788388 inhibits the TGF-βRs signaling pathway
To explore the role of TGF-βRI/II in KBD, we inhibited the TGF-βRs signaling using GW788388. To determine the concentration of GW788388 in C28/I2 chondrocytes, the cell viability was measured after the addition of 0–100 μM GW788388 for 24 h. The optimum GW788388 concentration was found to be 20 μM (Figure 3(a)), which was the maximum concentration caused no measurable toxicity. Establishment of inhibiting TGF-βRs signal in C28/I2 chondrocytes. A: selection of GW788388 concentration; B: TGF-βRI and TGF-βRII genes expression treated with GW788388 at 20 μM for 24 h; C: Smad-2 protein expression treated with GW788388 at 20 μM for 24 h (* P < 0.05 versus control group; ** P < 0.01 versus control group, n=3).
The effect on TGF-βRs signaling was assessed based on the expression of TGF-βRI/II and Smad-2. Chondrocytes were incubated with GW788388 at 20 μM for 24 h. We found that GW788388 significantly modulated the expression of TGF-βRI and TGF-βRII (Figure 3(b)). GW788388 treatment also modulated the expression of Smad-2 protein (Figure 3(C)).
Inhibiting TGF-βRs signaling abrogated the T-2 toxin-induced expression of MMP-13 in chondrocytes
After using GW788388 to inhibit TGF-βRs signal, C28/I2 chondrocytes were cultured in medium containing 0, 6, 12, and 24 ng/mL T-2 toxin for 72 hours. Compared with controls, T-2 toxin increased the expression of MMP-13 by almost 2.5 folds (Figures 4(b) and (c)). However, inhibition of TGF-βRs signaling pathway reversed this phenomenon (Figures 4(b) and (c)). MMP-13 expression after inhibiting TGF‐βRs signal in C28/I2 chondrocytes. After inhibiting TGF‐βRs signal with GW788388, C28/I2 chondrocytes were cultured in medium containing 0, 6, 12, and 24 ng/mL T-2 toxin for 72 hours. a: b: MMP-13 protein expression in various samples; c: MMP-13 gene expression in various samples (* P<0.05 vs 0 ng/mL T-2 toxin group; # P<0.05 vs 0 ng/mL T-2 toxin + GW788388. n = 4).
Inhibiting TGF-βRs signaling abrogated the T-2 toxin-induced degradation of collagen fibers in cartilaginous tissues
Cartilaginous tissues were treated with T-2 toxin to evaluate its role on the ECM degradation in chondrocytes. TGF-βRs signaling was inhibited using GW788388.
We found T-2 toxin enhanced the degradation of collagen fibers (Figures 5(c) and (g)). The picrosirius red stains represent the collagen fibers. The staining intensity is directly proportional to the abundance and packing density of collagen fibers. On the other hand, GW788388 blocked the effect of T-2 toxin on the degradation of collagen fibers (Figures 5(d) and (h)). Masson’s trichrome staining for analysis of collagen fibers in the cartilage revealed comparable findings (Figure 5). Histological evaluation of engineered cartilaginous tissues treated with T-2 toxin and/or GW788388. The engineered cartilaginous tissues were treated with T-2 toxin at 12 ng/mL and TGF-βRs inhibitor at 20 μM for a week. a, b, c, d: Picrosirius red staining of engineered cartilaginous tissues; e, f, g, h: Masson staining of engineered cartilaginous tissues.
Discussion
KBD results from significant alterations in the chondrocyte.28,29 In this study, safranin O staining revealed that T-2 toxin increased the breakdown of collagen fibers in the articular cartilage (Figure 1(b)), an important pathological factor for KBD. T-2 toxin is the most toxic trichothecene, a group of mycotoxins that disrupts protein synthesis in eukaryotes.4,30
MMP-13 participates in cartilage degradation. MMP-13 is a uniquely important collagenase that promotes the irreversible degradation of collagen in the cartilage of patients with osteoarthritis (OA). In the present study, we found that T-2 toxin sharply increases the expression of MMP-13 in the rat cartilage (Figure 2). Cartilage consists of cells surrounded by a specialized extracellular matrix predominantly composed of type II collagen and proteoglycans. 31 MMP-13 is the most regulated collagenase. It is expressed by chondrocytes and it hydrolyzes type-II collagen more efficiently than any other collagenase. 32 T-2 toxin promotes cartilage degradation by increasing MMP-13 expression.
Proliferation, differentiation, migration, and apoptosis of cells as well as ECM synthesis and degradation are regulated via the TGF-β signaling pathway. Mice models revealed that inhibition of TGF-β signaling in chondrocytes upregulates the expression of Runx 2, MMP-13, and Adamts 5 in articular cartilage tissue, resulting in progressive OA development. 33 A rat model revealed that intra-articular injection of adeno-associated virus expressing αKLOTHO and sTGF-βR2 inhibits the release of matrix-degrading enzymes to the ECM and maintains the cartilage thickness in the OA rat model. 34 However, in this study, we found that T-2 toxin upregulates the expression of TGF-βRI and TGF-βRII in the cartilages tissues (Figure 2). In related research, Yin reported that T-2 toxin treatment dramatically increased the production of ROS in chicken hepatocytes. 35 Meanwhile, NO/ROS modulates the expression of TGF-βRs in human cartilage tissues. 36 In the present study, inhibition of the TGF-βRs-smad 2 signaling pathways using GW788388 (Figure 3) suppressed the effect of T-2 toxin in modulating the expression of MMP-13 (Figure 4). Research shows that the TGF-β/SMAD2/3 pathway exerts dual effects on the cartilage tissues. 18 Recent studies have provided substantial evidence that TGF-β/Smad signaling participates in OA development and progression. 37 Thus, inhibiting the TGF-βR1 suppressed MMP-13 expression in OA-mesenchymal stromal cells but stimulated its production in OA chondrocytes. 38 TGF-β/SMAD3 can decrease the miR-140 activity in human chondrocytes. 39 MiR-140 is a microRNA originally expressed in cartilage and modulates the expression of MMP13, a dis-integrin, and metalloproteinase with thrombospondin motifs 5. 40 Further experiments using genetic engineering confirmed that inhibiting TGF-βRs signal abrogated the effect of T-2 toxin-induced ECM degradation (Figure 5).
In summary, T-2 toxin induces CEM degradation by upregulating the expression of MMP-13 via the TGF-βRs signaling pathway. As such, the TGF-βRs signaling pathway is a potential target for KBD treatment.
Footnotes
Authors’ contributions
Jinghong Chen is responsible for the integrity of the work, from inception to finished article. Ying Zhang had full access to all of the data in the study and is responsible for the integrity of the data and accuracy of data analysis. Zhengzheng Li, Ying He, Yinan Liu, and Ge Mi were responsible for acquisition, analysis, and interpretation of data. All authors have read and approved the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the National Natural Science Foundation of China (No. 81872565, No. 81573102) and Shaanxi Science and Technology Department (No. S2021-JC-QN-1910). The authors gratefully acknowledge financial support from China Scholarship Council (No. 201706280086).
