Abstract
Wood smoke (WS) contains many harmful compounds, including polycyclic aromatic hydrocarbons (PAHs). WS induces inflammation in the airways and lungs and can lead to the development of various acute and chronic respiratory diseases. Pulmonary fibroblasts are the main cells involved in the remodeling of the extracellular matrix (ECM) during the WS-induced inflammatory response. Although fibroblasts remain in a low proliferation state under physiological conditions, they actively participate in ECM remodeling during the inflammatory response in pathophysiological states. Consequently, we used normal human lung fibroblasts (NHLFs) to assess the potential effects of the PAHs-containing wood smoke extract (WSE) on the growth rate, total collagen synthesis, and the expression levels of collagen I and III, matrix metalloproteinase (MMP)-1, MMP-2, MMP-9, tissue inhibitor of metalloproteinase (TIMP)-1, TIMP-2, and the transforming growth factor (TGF)-β1. We also assessed MMPs activity. The results showed that WSE induced a trimodal behavior in the growth rate curves in NHLFs; the growth rate increased with 0.5-1 % WSE and decreased with 2.5% WSE, without causing cell damage; 5-20% WSE inhibited the growth and induced cell damage. After 3 hours of exposure, 2.5% WSE induced an increase in total collagen synthesis and upregulation of TGF-β1, collagen I and III, MMP-1, TIMP-1, and TIMP-2 expression. However, MMP-2 expression was downregulated and MMP-9 was not expressed. The gelatinase activity of MMP-2 was also increased. These results suggest that WSE affects the ECM remodeling in NHLFs and indicate the potential involvement of PAHs in this process.
Keywords
Introduction
One of the polluting sources of the ambient air is the smoke derived from the combustion of biomass, mainly wood smoke (WS), from domestic incineration and forest fires. WS is a global risk factor for poor health that can lead to oxidative stress, inflammation, and extracellular matrix (ECM) remodeling, which can trigger various lung diseases, including bronchitis, pneumonia, acute respiratory failure, and chronic obstructive pulmonary disease (COPD). 1 According to current World Health Organization estimates, about 52% of the world’s population uses solid biomass fuels for their heating and cooking needs. This impacts more than 2.4 billion people,2,3 generally including the poorest populations in the world, in approximately 90% of households in rural areas, which is responsible for approximately 4 million deaths each year.1,4,5 In forest fires, the smoke is derived from the incineration of various plant species; it contains a complex mixture of toxic molecules, particles, and harmful gases and represents a significant risk of exposure in ordinary people and firefighters. 6
WS contains approximately 400 gases, chemicals, and particles released during combustion, the majority of which are very similar to those present in cigarette smoke (CS). Selected analysis has shown WS to contain high levels of carbon monoxide, nitrogen oxides, dioxins, and polycyclic aromatic hydrocarbons (PAHs). PAHs in WS depend on the source of wood, the conditions of the incineration, and the combustion phase. WS composition also differs from the smoke released when biomass is incinerated, including coal, leaves, crops, or animal waste.7,8,10-14
PAHs are ubiquitous environmental pollutants generated primarily by the incomplete combustion of biomass and other energy sources.12-15 PAHs present in the atmosphere are distributed between gas and particle phases (G/P partitioning) and adsorbed mainly on particulate matter (PM) ≤ 2.5 μm (PM2.5). PAHs are of primary interest due to their toxic properties.7,8,12-18 Most PAHs are hydrophobic and stored in the fatty tissues of various organs; however, some are hydrophilic.12-18 Some PAHs are carcinogens, with many being genotoxic and mutagenic. The carcinogenicity of PAHs is associated with the complex nature of these molecules, especially with their increasing number of aromatic rings. Benzo [a] pyrene (BaP) is one of the most abundant and dangerous PAHs due to its high carcinogenic, genotoxic, and mutagenic effects.7,8,12-15 The toxicity of PAHs in mammals is mediated almost exclusively through the aryl hydrocarbon receptor (AhR) signaling. AhR is a ligand-activated transcription factor that is expressed in many cells.16-18 PAHs synergistically and negatively affect respiratory physiology by inducing an inflammatory response, oxidative stress, and ECM remodeling.7,8,12-19 PAHs also affect different cell types in vitro, including endothelial, epithelial alveolar bronchial and bronchiolar cells, macrophages, and fibroblasts.19,20
Studies carried out with healthy volunteers exposed to components of the WS are complex and produce partial information, but very accurate; 21 in vitro studies using cells exposed to WSE reduce the experimental complexity and provide more detailed molecular and cellular information of the toxic.8,12,16-18,22 Some studies have evaluated the effect of WSE on several cell types;20,21 but only in two studies have cultured fibroblasts exposed to WSE.9,25
Fibroblasts are actively involved in interstitial ECM remodeling. They synthesize most of the ECM molecules, including collagen, matrix metalloproteinases (MMPs), and tissue inhibitors of metalloproteinases (TIMPs); fibroblasts also maintain lung with their specific physiological and mechanical properties.19-21,26-28 MMPs are a family of Ca2+ and Zn2+-dependent endopeptidases involved in the ECM remodeling. Most MMPs are secreted as inactive zymogens (pro-MMPs) that require extracellular activation, and their activity is tightly regulated by TIMPs. Transforming growth factor-β1 (TGF-β1) regulates the expression of MMPs and TIMPs at the transcriptional level. 27
Emerging evidence indicates that WS induces an inflammatory response, but also generates various molecular events involved in the development of lung diseases, where the remodeling of the lungs actively occurs. 29 In this sense, the use of the extract derived from the WS (WSE), as a source of damage to normal human lung fibroblasts (NHLFs), could be used as a model to understand the modulating effect of WS on ECM remodeling in these cells. Accordingly, we hypothesized that PAH-containing WSE may induce a decrease in the growth rate, accompanied by the upregulation of the mRNA expression levels of collagen I and III, MMP-1, MMP-2, MMP-9, TIMP-1, TIMP-2, and TGF-β1.
The present study was performed to evaluate the effects of PAHs present in the WSE on the growth rate, total collagen synthesis, expression levels of TGF-β1, collagen I and III, MMP-1, MMP-2, MMP-9, TIMP-1, and TIMP-2, and gelatinase activity in NHLFs.
Materials and Methods
Preparation of the WSE
The WSE was obtained from the complete incineration of 5 g of wood from the branches of the Casuarina equisetifolia tree (hardwood), which was concentrated in 100 mL of Ham’s F-12 Nutrient Mixture (F-12)
Extraction and Culture of NHLFs
The NHLFs used in this study were derived from the lung of a subject after brain death. The subject was a healthy man of 20 years old at the time of death, who provided written and informed consent. Fibroblasts were obtained as previously described.26,28 Briefly, the tissue was cut into small pieces; subsequently, enzymatic digestion was carried out to obtain the pulmonary fibroblasts, with 1x ethylenediaminetetraacetic acid (EDTA): trypsin (0.05 mg/mL trypsin: 0.02 mg/mL EDTA) (Sigma-Aldrich Corporation, St. Louis, MO, USA), incubating them for 20 minutes at 37°C in Ham’s F-12 Nutrient Mixture (F-12) serum-free. The cells were recovered by centrifugation at 1200 rpm and 17°C for 10 minutes. The cells were cultured in 25 cm2 tissue culture flasks using Ham’s F-12 Nutrient Mixture (F-12), supplemented with 10% FBS (Thermo Fisher Scientific Inc, Carlsbad, CA USA), 100 U/mL of penicillin, 100 mg/mL streptomycin, and 2.5 μg/mL of amphotericin B (Sigma-Aldrich Corporation, St. Louis, MO, USA). Cells were maintained in a humidified incubator with a 95% air/5% CO2 atmosphere at 37°C. The medium was changed every other day.
Additionally, we proceeded to verify that the cells utilized in this study are fibroblasts. Cells were analyzed by immunocytochemistry and cytofluorometry, which demonstrated that the cells showed the typical characteristics of fibroblasts. Cells were stained with specific antibodies for fibroblasts markers, as well as with the pan-cytokeratin antibody showing the characteristic of epithelial cell phenotype. Specifically, NHLFs showed positivity for Thy-1/CD-90, vimentin, type I collagen, PDGFRβ, and negativity to pan-cytokeratin, which corroborates that they were indeed fibroblasts (data not shown). 28
Model of Exposure to WSE in NHLFs
WSE exposures were performed when the cells reached 75% confluence; concentrations of WSE ranged from 0.5-20%, and were performed on cells cultured in 1% FBS. The cells were lysed to obtain total cellular extracts for western blotting or to obtain total RNA for quantitative reverse transcription-polymerase chain reaction (qRT-PCR) analysis. Conditioned media were collected to measure the total collagen content and for gelatin zymography.
Growth Rate Assay
The growth rate assay helps to measure the number of metabolically active cells over time, resulting from the balance between the rates of cell death and cell proliferation. Fibroblasts were plated in 96 well culture plates at a cell density of 1 × 104 cells/well and synchronized with serum-free medium for 24 hours. The medium was then replaced with Ham’s F-12 Nutrient Mixture (F-12), supplemented with 1% FBS and antibiotics. NHLFs cultures were exposed to six different concentrations of WSE, 0.5, 1.0, 2.5, 5.0, 10, and 20%, for 4, 8, 16, 24, 48, and 72 h. Control cells were incubated in Ham’s F-12 Nutrient Mixture (F-12), supplemented with 1% FBS and antibiotics. The growth rate was monitored up to 72 hours, using the cell proliferation reagent WST-1 (Roche, Mannheim, Germany) as previously described.26,30 Absorbance at 450-620 nm was taken as the increase in the percentage of growth rate related to basal values (time zero).
Based on the results from the growth rate and the microscopic examination of the cells, was determined that the use of a dose of 2.5% WSE with 1% FBS; incubating cells for 24 and 48 hours was appropriate for the most experiments. qRT-PCR was performed at 3 hours according to the standardization tests. Gelatin zymography was performed in the same conditions but with serum-free culture media.
Total Collagen Content
Collagen content is a measurement of the new total collagen synthesized and secreted into cell culture medium. This evaluation measures all types of collagen and was performed with the quantitative Sircol Soluble Collagen Assay (Biocolor Ltd, Carrickfergus, Northern Ireland), according to the manufacturer’s specifications. 31 The concentration of proteins in cell culture medium was evaluated using the method of Bradford, with the Bradford Assay Reagent (Bio-Rad Laboratories Inc, Hercules, CA, USA). 32 The results were expressed as the mean ± standard deviation of μg collagen/mg protein.
Gene Expression by qRT-PCR
Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used as endogenous control, utilizing previous standardization to qRT-PCR assays.33,34 Indeed, GAPDH was chosen among three possible human RNAs, rRNA 18S (Hs03928985_g1), RNA Polymerase II Subunit A (POLR2A; Hs00172187_m1), and GAPDH (Hs02786624_g1), for which was verified that the treatments did not modify significantly the ΔCt of these RNAs. It was done using same the same experimental conditions described here; control cells and cells exposed to 1 and 2.5% WSE cultured in media with 1% FBS by 3 hours and the ΔCt was determined. A low variability was observed in the ΔCt for GAPDH, which showed 23.23 ± 2.10 in the control, 21.83 ± 1.13 with 1% WSE, and 21.06 ± 0.74 with 2.5% WSE. All qPCRs were performed in a mixture of 10 μL: 2 μL of cDNA and 8 μL of 2X PCR Master Mix (Applied Biosystems). The PCR conditions were 2 minutes at 94°C, followed by 40 cycles of 15 seconds at 94°C and 60°C for a minute. Three independent experiments were performed in triplicate, and the results were expressed as the mean ± standard deviation (SD), of 2^-ΔCt of the target gene, normalized to the mRNA expression of GAPDH (ΔCT = CT (target gene) - CT (a reference gene)).
Evaluation of Protein Expression by Immunoblotting
The cells were lysed with the RIPA buffer (Sigma-Aldrich Corporation, St. Louis, MO, USA), plus a cocktail of serine and cysteine protease inhibitors (Calbiochem, San Diego, CA, USA) at 4°C. The protein concentration was measured using Bradford reagent (Bio-Rad Laboratories Inc, Hercules, CA). 32 Protein extracts, containing 30 μg/lane, were mixed (V/V) with Laemmli buffer and separated under reducing conditions on 10% SDS-PAGE. The separated proteins were electro-transferred to nitrocellulose membranes (Bio-Rad Laboratories Inc., Hercules, CA) at 35 mA overnight, and blocked with fat-free milk 5% in TBS/0.1% Tween (137 mM sodium chloride, 20 mM Tris, 0.1% Tween-20, pH 7.6). The membranes were incubated overnight at 4°C with the corresponding primary antibody: rabbit polyclonal anti-MMP-2 (Abcam ab97779; 1:1000 dilution), rabbit monoclonal anti-MMP-9 (Abcam ab76003 1:1000 dilution), rabbit monoclonal anti-TIMP-1 (Abcam ab211926; 1:1000 dilution), rabbit polyclonal anti-TIMP-2 (Abcam ab180630; 1:500 dilution), and rabbit polyclonal anti-β-actin (Abcam ab8227). The membranes were incubated 1 hour at room temperature, with anti-rabbit IgG (H+L) secondary antibody (Catalog # 31460; Thermo Fisher Scientific Inc, Waltham, MA. USA; 1:200 dilution), and subsequently washed 4 times for 10 minutes with PBS-T (10 mM sodium phosphate, 0.15 M NaCl, 0.05% Tween-20, pH 7.5). Immunodetection was performed by chemiluminescent reaction (West Pico Chemiluminescent Substrate (Thermo Fisher Scientific Inc., Carlsbad, CA USA). Images were processed using the Molecular Imager ChemiDoc XR. Image Lab v6.0 software was used for densitometry analysis (Bio-Rad Laboratories Inc, Hercules, CA, USA). The results were normalized against β-actin. 28
Gelatin Zymography
Gelatin zymography is an extremely sensitive and useful technique for measuring the relative amounts of active and inactive gelatinases (zymogen) in soluble-aqueous samples by measuring gelatin hydrolysis. The enzymes MMP-2 (gelatinase A) and MMP-9 (gelatinase B) are fractionated on SDS-PAGE and detected by including gelatin as a substrate in the gel, which is hydrolyzed producing two or three white bands, for each one, zymogen or active forms, after Coomassie blue staining.26,28 Briefly, cells were maintained in the Ham’s F-12 Nutrient Mixture (F-12) without FBS for 24 and 48 hours; the conditioned media were collected; and protein was measured with the Bradford method. 32 Samples containing 10 μg of protein were mixed with an equal volume of sample buffer and resolved, under non-reducing conditions on 7.5% SDS-PAGE, containing 1 mg/mL of gelatin as a substrate (Sigma, St. Louis, MO, USA; Cat. No. G-8150; Lot. 63H06591). Conditioned media from human lung fibroblasts were used as a positive control for MMP-2, while the obtained from U2-OS human cells were used as MMP-9 positive control.
Characterization of PAHs in the WSE
To identify the PAHs contained in the WSE, 5 g of wood from the branches of C equisetifolia were incinerated and the resulting ashes were bubbled in 100 mL of deionized water, the same water that was used to prepare the culture medium of the cells, as was described previously. 10 The aqueous samples of the WSE were subjected to a liquid–liquid extraction process; 3 mL were mixed with 1 mL of dichloromethane (DCM) (HPLC grade, Merck & Co, Temecula, CA, USA), in duplicate. The excess of DCM was evaporated under a gentle stream of nitrogen gas (Infra 99.998%) to obtain 1 mL of the final organic extract. PAHs in this WSE were analyzed by gas chromatography with an automatic injector (Agilent 7890A GC System, Conquer Scientific, San Diego, CA, USA) coupled to a mass spectrometer (Agilent 5973 inert gas chromatograph/mass spectrometer (GC/MS), Conquer Scientific, San Diego, CA, USA), with a simple quadrupole and electronic ionization (70 eV), as described previously. 10 Briefly, the injector temperature was 300°C, the injection of 2 μL was carried out in “splitless” mode. Helium was used as the carrier gas (99.998%; Infra) at 1.2 mL/min. The temperature of the transfer line was 300°C; the ion source was 230°C; and the quadrupole temperature was 150°C. PAHs separation was carried out in a capillary column (DB-35 ms GC column, Conquer Scientific, San Diego, CA, USA) with a length of 60 m, 0.25 mm internal diameter, and 0.25 μm stationary phase thickness. The temperature ramp was 40°C increased by 50°C/min up to 110°C, the second increment at 5°C/min up to 30°C for 0 min, and the third increment at 20°C/min up to 335°C for 7.4 minutes. The total run time was 50 minutes. Chromatogram processing was carried out using the Data Analysis software (Agilent Chem Station, Agilent Technologies, Santa Clara, CA, USA).
Statistical Analysis
Results are expressed as the mean ± standard deviation, obtained from three independent experiments performed in triplicate. Differences between two groups were evaluated using an unpaired Student’s t-test, while those performed between more than two groups were analyzed using one-way analysis of variance (ANOVA), followed by Tukey-Kramer post hoc test at 95% significance, which was used for multiple comparisons. All analyses were performed using GraphPad Prism v. 6.1 software (GraphPad Software, Inc, San Diego, CA, USA). Differences were considered statistically significant at P < 0.05.
Results
WSE Has a Trimodal Dose-Dependent Effect on the Growth Rate in NHLFs
The growth rate is shown in Figure 1, which shows the behavior of the growth rate curves in the NHLFs exposed to different doses of WSE and the control curve. A trimodal effect was observed in the growth rate curves. With 0.5% and 1% WSE, the growth rate was increased at 48 and 72 hours, respectively (P < 0.05; Figure 1A), when compared with the control curve, which exhibited the same effect at 72 hours when it reached the normal stationary phase. In contrast, the 2.5% WSE growth rate decreased from 24 to 72 hours (P < 0.05; Figure 1A). However, with 5-20% WSE, an inhibitory effect on growth was observed, with values lower than the control that were statistically significant from 24 at 72 hours, as compared with the control curve (P < 0.05; Figure 1A). Additionally, the decrease in growth rate with 2.5% WSE did not induce any morphological alterations at 24 hours (Figure 1C) or 48 hours (Figure 1D), when compared with the control (Figure 1B), where the typical elongated spindle shape of the fibroblasts was observed. However, an important cytotoxic effect was observed with 5-20% WSE, as shown in the representative image taken of fibroblasts exposed to 10% WSE for 24 hours. When the loss of elongated and spindle shape was evident, the cells no longer adhered to the culture plate and acquired a rounded abnormal shape. Cell debris was also noted, all of which are characteristic of cells in the death process (Figure 1E). Based on these observations, the majority of the remaining experiments were performed with 2.5% WSE. Wood smoke extract (WSE) induced a trimodal dose-dependent effect on growth rate curves in NHLFs, augmenting with 0.5-1%, decrementing with 2.5%, and inhibiting it with 5-20% WSE. Growth rate curves of NHLFs exposed to 0.5% WSE, analyzed at 4, 8, 16, 24, 48, and 72 hours (A). The values in Figure 1A represent the mean ± standard deviation, obtained from at least three independent experiments performed in triplicate (one-way analysis of variance (ANOVA), followed by Tukey-Kramer post hoc test). * P < 0.05, compared with the control. Representative image of the control cells (B), where it is observed the typical elongated fusiform appearance of fibroblasts. Cells exposed to 2.5% WSE for 24 hours, exhibiting similar morphology to control cells (C). Cells exposed to 2.5% WSE for 48 hours (D), also showing the typical characteristics of the fibroblast. Cells exposed to 10% WSE for 24 hours (E), where the loss of elongated and spindle-shaped was evident, and cells no longer adhered to the culture plate, acquiring a rounded abnormal shape and cell debris were also observed, characteristic of cells in the death process. Original augment 10x.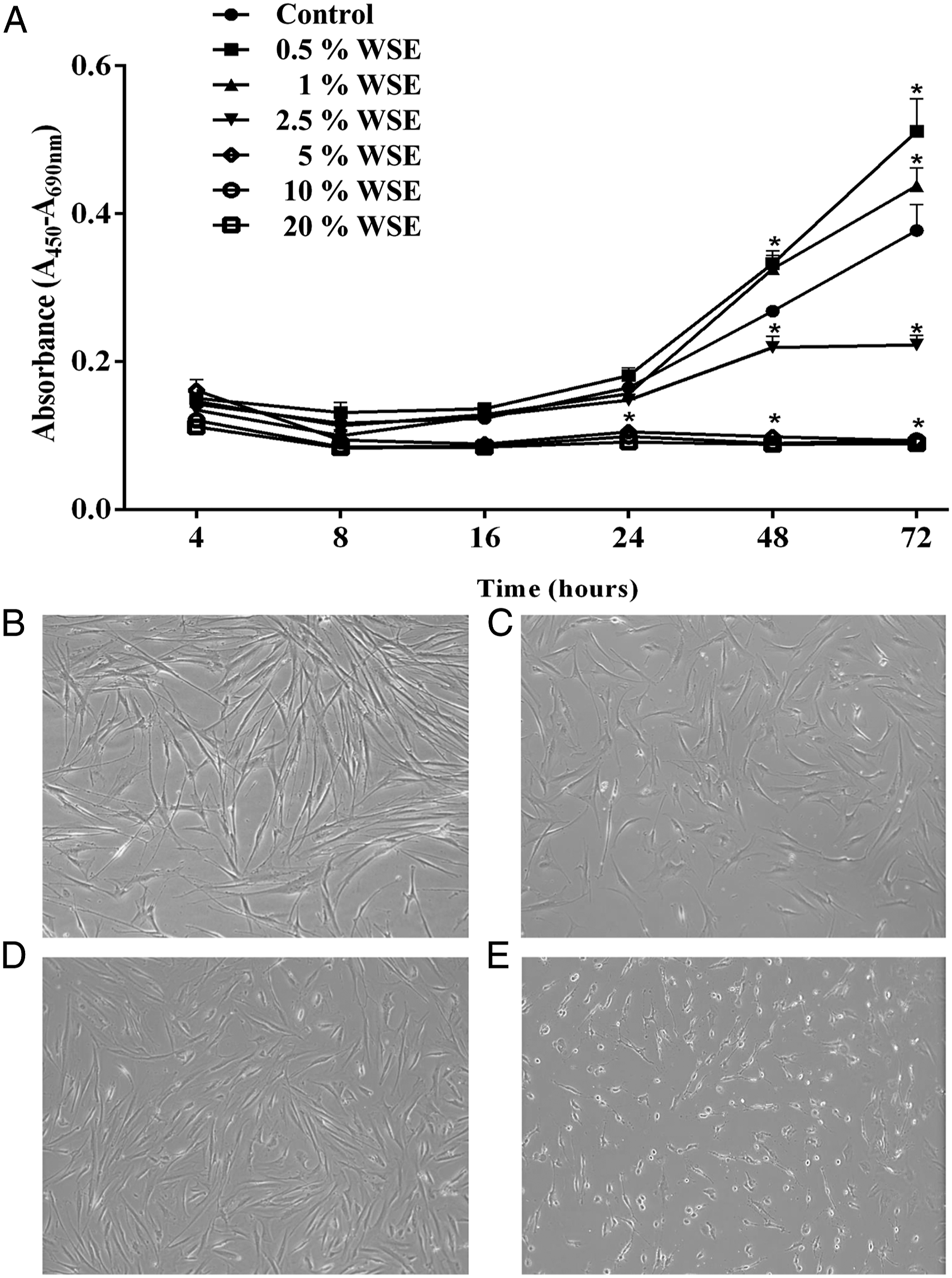
WSE Induces an Increase in the Synthesis of Total Collagen and Expression of Collagen I and III
Exposure to 2.5% WSE induced an increase in the total soluble collagen secreted into cell culture medium by NHLF, and this effect was also observed at 24 as at 48 hours (P < 0.05 and P < 0.01, respectively; Figure 2A). It also induced the upregulation in the mRNA of both fibrillary collagens, COL1A1 and COL3A1, when the cells were incubated with 2.5% WSE for 3 hours (P < 0.01) (Figures 2B and 2C). WSE increased the total collagen synthesis and upregulated the mRNA expression levels of collagen I and III in NHLFs. Quantification of total soluble collagen (A), COL1A1 mRNA expression (B), and COL3A1 mRNA expression (C). Total collagen synthesis was measured with Sircol reagent. The mRNA expression levels were measured by qRT-PCR in NHLFs exposed to 2.5% WSE incubated for 3 hours. The expression levels of the collagen mRNAs were normalized with that of the GADPH mRNA. The values represent the mean ± standard deviation, obtained from at least three independent experiments performed in triplicate. * P < 0.05; ** P < 0.01 compared with the control.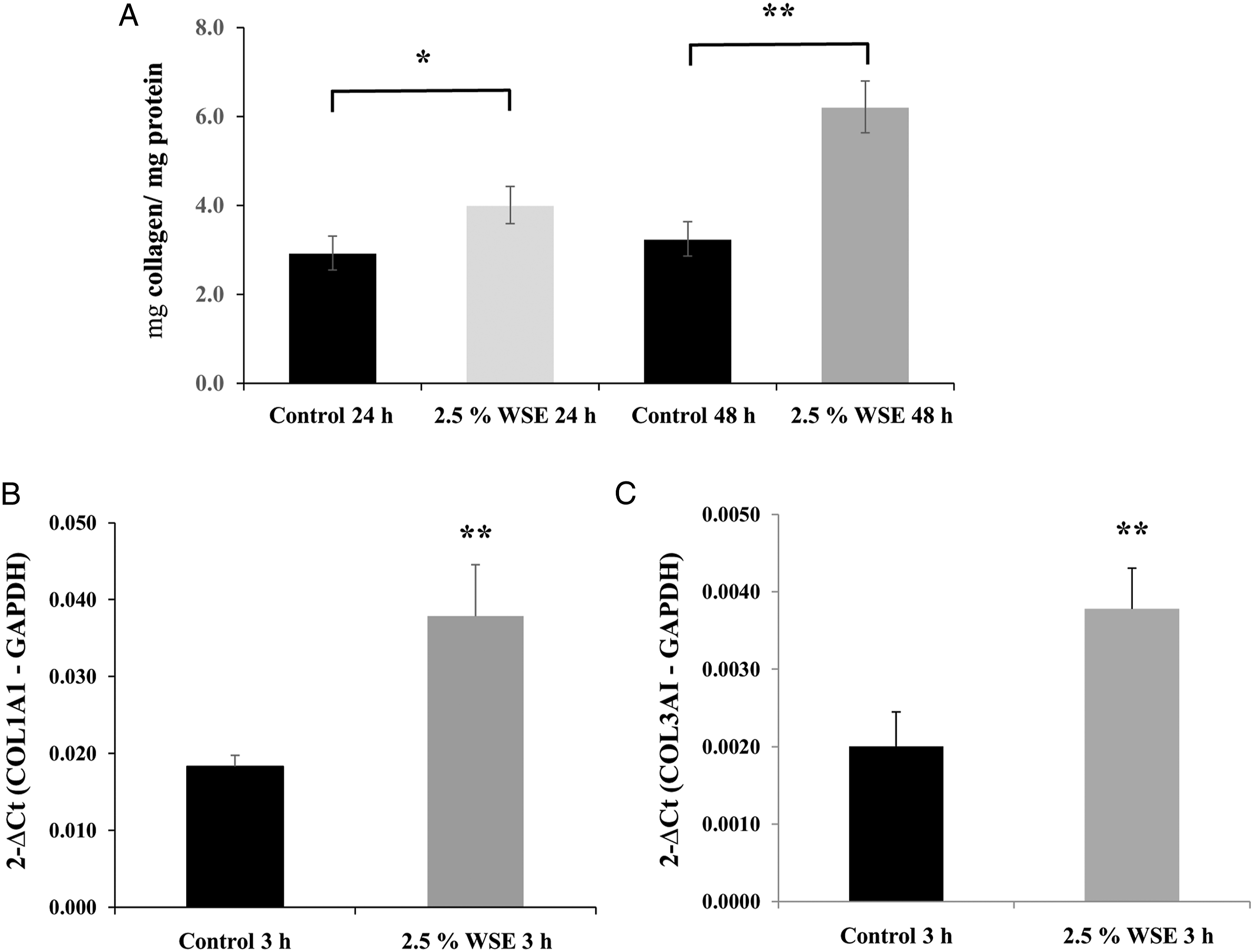
WSE Upregulates TGF-β1 and MMP-1 and Increases the Protein and Activity of MMP-2 in NHLFs
Quantitative reverse transcription-polymerase chain reaction analysis showed that after 3 hours of exposure to 2.5% WSE, there was a significant increase in the expression of TGF-β1 mRNA (P < 0.01; Figure 3A). Additionally, a statistically significant upregulation of MMP-1 (interstitial collagenase) mRNA expression was observed (P < 0.05; Figure 3B). In contrast to what was observed in MMP-2 mRNA, which was downregulated (P < 0.01; Figure 4A), 24 and 48 hours after exposure to 2.5% WSE, the expression of MMP-2 protein (gelatinase A) was increased (P < 0.01; Figures 4C and 4E). MMP-9 (gelatinase B) gene expression and protein were not detected (Figures 4B and 4D). WSE induced the upregulation of the mRNA expression levels of TGF-β1 and MMP-1 in NHLFs. Effect of 2.5% WSE on the mRNA expression of TGF-β1 (A) and MMP-1 (B). The mRNA expression levels were measured by qRT-PCR in NHLFs with 2.5% WSE after 3 hours of incubation. mRNA expression levels were normalized to that of GADPH mRNA. The values represent the mean ± standard deviation, obtained from at least three independent experiments performed in triplicate. * P < 0.05; ** P < 0.01 compared with the control. WSE induces the upregulation of MMP-2 mRNA and augments their protein level and activity in NHLFs, without effect on MMP-9. mRNA expression of MMP-2 (A) and MMP-9 (B). Western blot to MMP-2 (C) and MMP-9 (D). MMP-2 and MMP-9 gelatin zymography. The mRNA expression levels were measured by qRT-PCR in NHLFs treated with 2.5% WSE after 3 hours of incubation. The expression levels of the mRNAs were normalized with that of the GADPH mRNA, whereas western blots were normalized to β-actin. The values represent the mean ± standard deviation, obtained from at least three independent experiments performed in triplicate. The original images in Figures 4E and 4F were cropped to obtain only five tracks and standardize the figure. ** P < 0.01 compared with the control.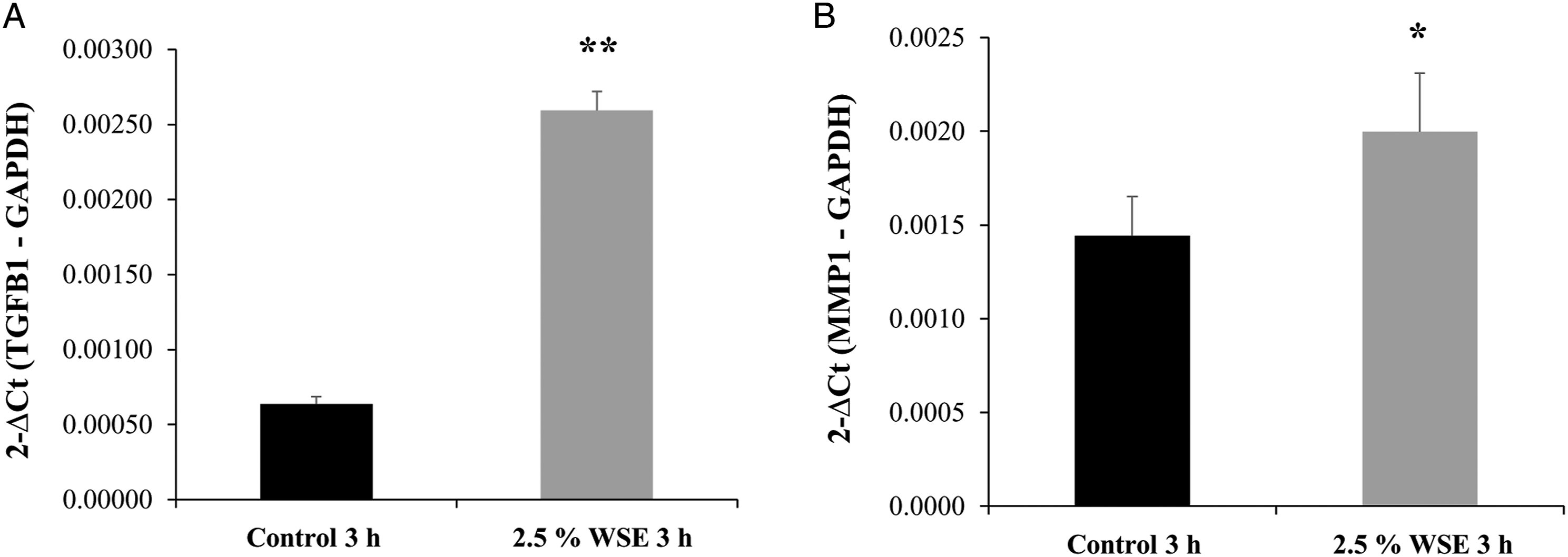
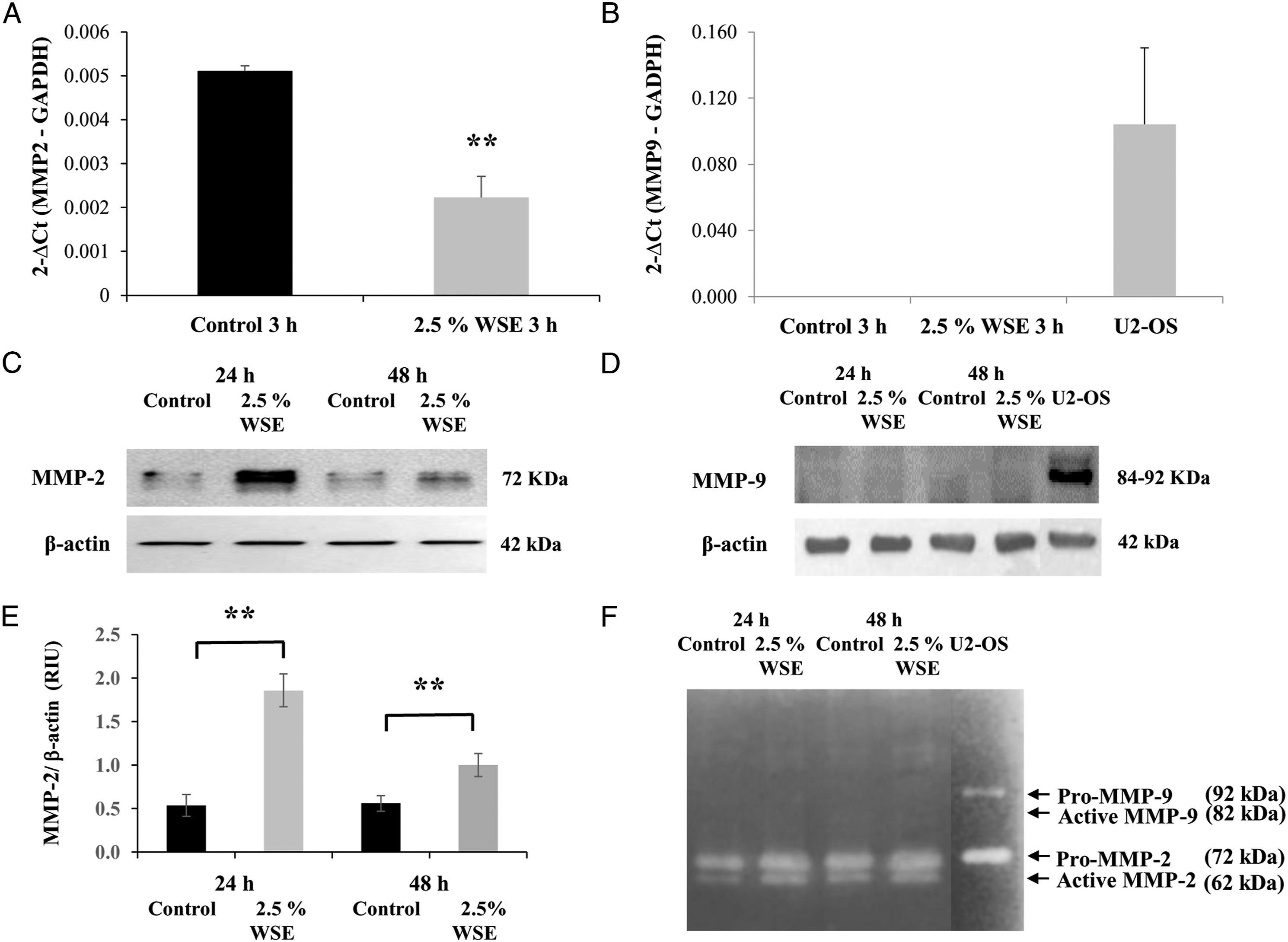
The gelatinolytic activities of MMP-2 and MMP-9 in the conditioned media were assayed by zymography, which confirmed that MMP-2, but not MMP-9, was increased by exposure to 2.5% WSE (Figure 4F). The clear bands of the zymogen Pro-MMP-2 (62 kDa) and its active enzyme MMP-2 (72 kDa) were augmented in comparison with the control after 24-48 hours (Figure 4F). However, in the case of MMP-9, clear bands corresponding to the zymogen Pro-MMP-9 (92 kDa) and its active enzyme MMP-9 (82 kDa) were absent; only the clear band of the culture medium from the positive control, U2-OS cell line, was observed (Figure 4F). These results support the fact that MMP-2 but not MMP-9 was affected by 2.5% WSE.
WSE Upregulates the mRNA Expression Levels of TIMP-1 and TIMP-2 in NHLFs
Tissue inhibitors of matrix metalloproteinase-1 and tissue inhibitors of matrix metalloproteinase-2 are the specific tissue inhibitors of MMPs. WSE induced the upregulation of the mRNA expression levels of both TIMP-1 (P < 0.01; Figure 5A) and TIMP-2 (P < 0.01; Figure 5B) after 3 hours of incubation when compared with the control (Figures 5A and 5B). Additionally, the increase in TIMP-1 mRNA expression corresponded to increased TIMP-1 protein levels after 24 hours of exposure to WSE (P < 0.01; Figures 5C and 5E). No changes were observed in the level of TIMP-2 protein (Figure 5D). WSE induced the upregulation of the mRNA expression levels of TIMP-1 and TIMP-2 in NHLFs. Effect of 2.5% WSE on the expression of TIMP-1 (A) and TIMP-2 (B) mRNA Western blot of TIMP-1 (C) and TIMP-2 (D). The mRNA expression levels were evaluated by qRT-PCR in NHLFs with 2.5% WSE after 3 hours of incubation. The expression levels of the mRNAs were normalized to that of GADPH mRNA, whereas those of the western blot were normalized to those of β-actin. The values represent the mean ± standard deviation, obtained from at least three independent experiments performed in triplicate. ** P < 0.01 compared with the control.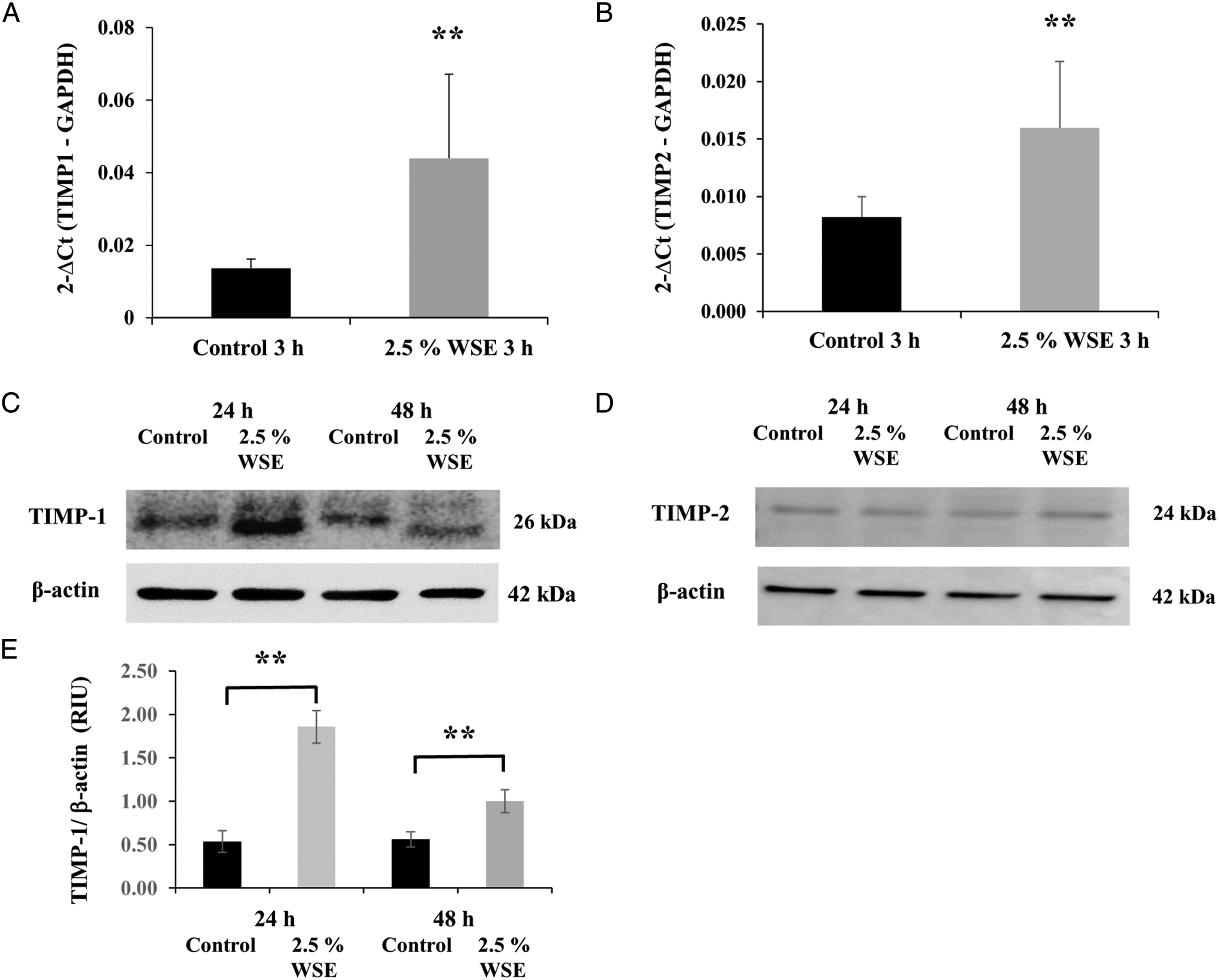
PAHs are Present in the Aqueous Phase of WSE
Concentrations of Polycyclic Aromatic Hydrocarbons Present in the Soluble Aqueous WSE From Casuarina equisetifolia.
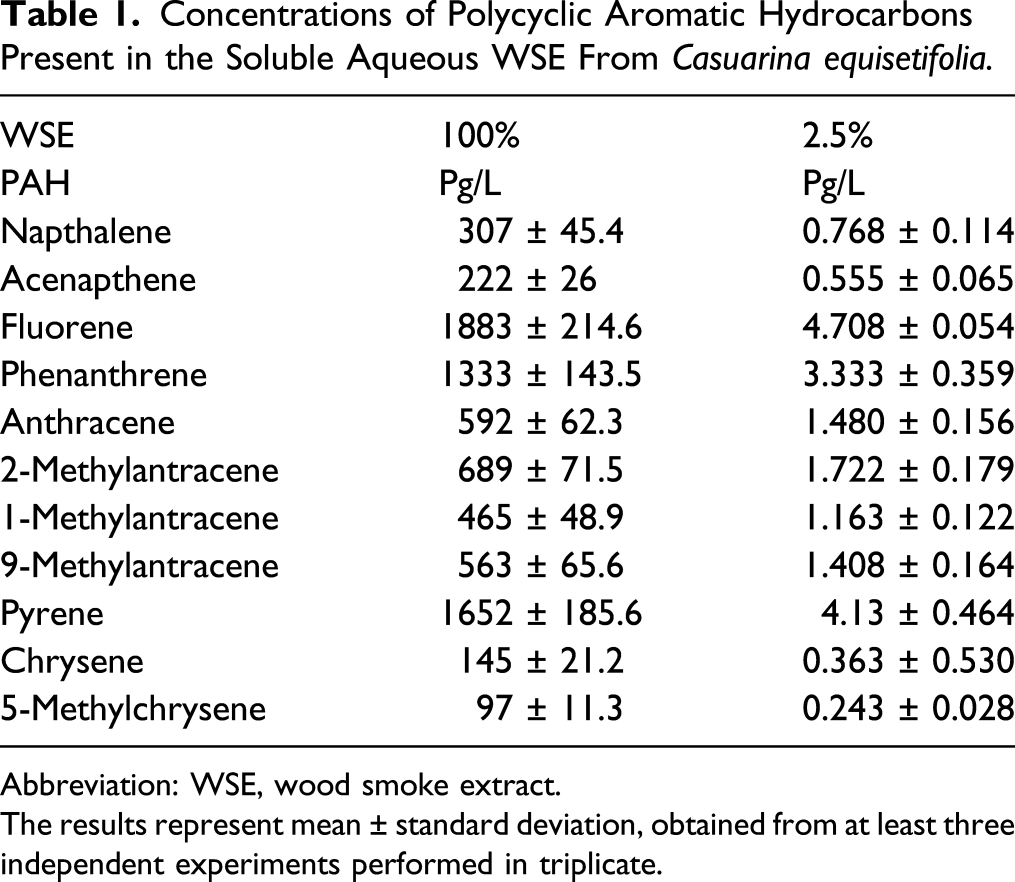
Abbreviation: WSE, wood smoke extract.
The results represent mean ± standard deviation, obtained from at least three independent experiments performed in triplicate.
Discussion
We tested the effect of water-soluble WSE from C. equisetifolia on NHLFs, to assess the potential effect on the growth rate, total collagen secreted, expression levels of collagen I and III, MMP-1, MMP-2, MMP-9, TIMP, TGF-β1, and gelatin zymography.26,28,30 C. equisetifolia is a tree distributed throughout Mexico, which is used as an energy source in various regions of the country.
Similar to this model, most CSE and WSE exposure studies use the proliferation curve, as an alternative to the growth rate, to define the concentration with which the cell growth will be stimulated.12,35-43 We observed that WSE induced a trimodal dose-dependent behavior in the growth rate curves of NHLFs. The lowest doses increased the growth rate, while higher doses inhibited it and caused cell damage. Moreover, 2.5% WSE decreased the growth rate without inducing cell damage; therefore, 2.5% WSE at 24-48 hours were used in the experiments, except in qRT-PCR, which was performed at 3 hours. The curves with 1-2.5% WSE were the typically reported for these cells, reaching the stationary phase at 72 hours.4,30 In contrast, the curves with 5-20% WSE were atypical and the cells suffered severe damage or death.26,30
The increase in growth rate with .5-1 % WSE agrees with the only study reported in cultured lung fibroblasts, where both WSE and CSE augmented their proliferation.14,37,38 But also with models using other cell types, such as macrophages, bronchial, bronchiolar and alveolar epithelial cells, and endothelial cells.23,36,39-43 A possible explanation for this fact is that exposure to low doses of toxic compounds can induce an acute-adaptive cellular response. In contrast, if the cellular adaptive response to toxins is unable to counteract the adverse exposure, as occurs with higher doses of WSE, the cells may become damaged and may die.19,23,42-44
Wood smoke extract-induced ECM remodeling in fibroblasts has been reported by others. One study on human lung fibroblasts from donors undergoing lung resection or lung transplantation exposed to WSE, and independently CSE, showed upregulation of perlecan and fibronectin. In addition, WSE increased the secretion of IL-8 and upregulation of ERK1/2, demonstrating that both extracts had similar effects in vitro. 25 Another study, using human lung fibroblasts obtained from several pulmonary disease states, showed that WSE induced the overexpression of IL-6 and IL-8, activating ERK, Smad, and p38 signaling pathways. 9 Our findings extend these observations to PAHs.
Matrix metalloproteinase-1 gene expression was upregulated in our model. MMP-1 is the main collagenase secreted by fibroblasts and type II pneumocytes. MMP-1 is significantly upregulated in pulmonary fibrosis and emphysema.43-45 MMP-1 was increased by the exposure to CSE in human fibroblasts and keratinocytes, where activates the AhR signaling pathway, suggesting a role for the PAHs contained in CSE. 16 In this way, a similar mechanism would occur with NHLFs exposed to the PAHs contained in the WSE, although this requires further investigation to confirm.
Matrix metalloproteinase-2 gene expression was downregulated, whereas its protein and activity were increased due to the WSE effect. This may be due to the transcriptional and translational mechanisms of regulation of gene expression and epigenetic mechanisms, especially by the stability of their mRNA. 27 In agreement with our model, MMP-2 was downregulated, decrementing their activity and also upregulating TIMP-2, in a dose-dependent manner by CSE in human lung fibroblasts. 38 Upregulation of MMP-1 and MMP-2 has been reported in several models of COPD induced by CS or WS as well as in humans.38-40 These features strongly support the occurrence of an important ECM remodeling phenomenon induced by WSE in NHLFs26-28 and are consistent with the observations made in animal models of COPD-induced with WS and CS.11,24,38,39
Transforming growth factor-β1, which is upregulated by WSE, is crucial since this cytokine is the main gene orchestrator in ECM remodeling. TGF-β1 regulates the differential expression of most MMPs and TIMPs in the physiological and pathophysiological processes in tissues and organs.27,46-48 In this sense, it seems that NHLFs could be stimulated in an autocrine manner by TGF-β1 upregulated the effect of WSE.27,38,39,44,45 Interestingly, alveolar epithelial cells exposed to WSE containing PAHs displayed disrupted epithelial barrier structure and function, through the p44/42 MAPK signaling pathway. 7 This could also be a mechanism by which the WSE acts in our model with NHLF; however, this evaluation requires a more extensive and focused experimental analysis.
Of the PAHs, found in the WSE of C. equisetifolia, only 5-methylchrysene is classified as possibly carcinogenic to humans; the remaining ten are currently characterized as non-carcinogenic. 13 However, these PAHs have been strongly associated with the development of metabolic alterations in cells,7,8 lung inflammation, ECM remodeling, and pulmonary diseases, including asthma and COPD.12-15,49,50
The effects documented in this study cannot be directly attributed to the reported PAHs. To determine the specific effects of PAHs, the experimental approach could involve the preparation of a mixture containing the same compounds and concentrations indicated in Table 1, followed by incubating the fibroblasts with this mixture and then using the same variables for evaluation to determine if the same effects are observed.
Finally, we acknowledge that this study used NHLFs derived from a single, apparently normal individual. Additional evaluation of the effects of WSE on NHLFs obtained from other individuals, especially those who have underlying lung disease, would be of interest.
In conclusion, this study shows some of the effects of WSE, which contains PAHs, on NHLF. WSE induced an increase, decrease, or inhibition of the growth rate in a dose-dependent manner. With 2.5% WSE, an increase in total collagen synthesis and upregulation of TGF-β1, collagen I and III, MMP-1, TIMP-1, and TIMP-2 was observed; however, MMP-2 gene expression was downregulated, and the protein level and activity were enhanced, with no effect MMP-9. Thus, these results suggest that WSE induces an ECM remodeling mechanism in NHLF, where the PAHs might have a potential role, which remains to be exhaustively analyzed.
Footnotes
Author Contributions
Recillas-Román, S. contributed to the acquisition, analysis, and interpretation of the data and drafted the article; Montaño, M and Ramos, C contributed to conception and design, contributed to acquisition, analysis, and interpretation, drafted the article, and critically revised the article; Ruiz, V. contributed to conception and design, contributed to the acquisition, analysis, and interpretation and critically revised manuscript; Pérez-Ramos, J. contributed to conception, design and contributed to acquisition, analysis, and interpretation; Becerril C., Herrera I., Amador-Muñoz, and Martínez-Domínguez YM. contributed to acquisition, analysis, and interpretation. All authors gave final approval and agreed to be accountable for all aspects of work, ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
The Ethics, Scientific, and Biosecurity Committees at Instituto Nacional de Enfermedades Respiratorias Ismael Cosío Villegas (INER) approved the present study. This study was performed under protocol number B 24-18.
Data Availability
The primary data supporting the results reported in this article can be provided by the corresponding author.
