Abstract
Bleaching agents containing a high concentration of H2O2 in the dental market lead to formation of reactive oxygen species, which have genotoxic effects. However, ozone bleaching, one of the most effective oxidants known, stimulates blood circulation and immune response and thus it has strong antimicrobial activity against viruses, bacteria, fungi, and protozoa. For these reasons, one of our hypothesis was ozone bleaching would reduce local and systemic DNA damage in the body. Hence, we aimed to determine the oxidative DNA damage biomarker levels in serum, saliva, and gingival crevicular fluid (GCF) by measuring 8-hydroxy-2′-deoxyguanosine (8-OHdG) after different bleaching methods.
Forty-eight volunteers who requested dental bleaching were divided into three treatment groups (n = 16). Group 1: ozone bleaching with the ozone-releasing machine; Group 2: chemical bleaching with 40% hydrogen peroxide (H2O2) gel; Group 3: 40% H2O2 gel activated with the diode laser. Initial and post-operative (immediately after bleaching and two weeks later) color measurements were performed with a spectrophotometer. The color changes were calculated with the CIEDE2000 (ΔE 00) formula. 8-OHdG levels in serum, saliva, and GCF samples were determined with ELISA. All three treatments resulted in efficient and statistically similar bleaching. The 8-OHdG levels in the serum and saliva were not affected by all bleaching methods (p > 0.05), but a temporary increase was observed in the GCF for chemical and laser-assisted groups except the ozone group (p > 0.05). According to the findings, chemical and laser-assisted bleaching can affect DNA damage locally but not systemically. Bleaching with ozone may eliminate this local DNA damage.
Keywords
Introduction
If today’s people are asked to list their priorities for life, the aesthetics of teeth described as the desire to look beautiful would be placed in the front rows. This situation makes dental aesthetics an important parameter in dentistry. 1 Bleaching is a conservative treatment of these aesthetic applications. 2 When the issue is evaluated on the demand and supply axis, increased demand for whiter teeth has naturally made the subject popular in terms of both dentists and manufacturers. Unfortunately, under the general principle of all good things bring with them a set of handicaps or problems. Although many methods have been developed and successfully applied in the treatment of tooth discoloration, in the literature, the existence of many studies on some negative effects of tooth bleaching indicates that the techniques still do not reach the perfect form. 3,4
Hydrogen peroxide (H2O2) is the main active chemical component, which is known to have high oxidation capacity and is widely used in tooth bleaching treatment. 5 The mechanism of tooth bleaching is not yet fully understood. It is assumed that free radicals generated following the break-down of H2O2 disintegrate chromophores into smaller molecules reflecting less light and subsequently results in whitening, on the other hand, it can cause harmful effects to the pulp tissue. 4,6
For many years, instead of suggesting alternative treatment protocols that may be both less harmful and more effective, the question of how we can reduce the side effects of H2O2 has been investigated such as pH of the agent, application technique, concentration, heat or light activation, absorption of the wavelength by the target tissue. 4 Laser activation leads to the release of more free radicals from the bleaching agent and providing penetration to tooth tissue in this way results in a faster bleaching process. 7 However, lasers should be used in the correct parameters. Because it causes irreversibly pulp damage if the temperature rise is increased by 5.6°C. 8
In recent years, using Ozone gas (O3) can be used as an alternative to H2O2 in the bleaching process due to having strong oxygenation (ability to form oxygen) and oxidation (ability to form oxidation-reduction reaction) ability. 9,10 However, it does not seem reliable enough because of the following two shortcomings; a) ozone gas can lead to oxidative damage and cell death, this handicap has not yet been tested on teeth and associated structures, b) the efficacy of O3 on tooth bleaching has not been compared to other agents in vivo. 10
The free oxygen radicals formed during the dental bleaching process-induced oxidative stress which leads to intense cell death, cell differentiation, or stimulus in pulp tissue 11 and, these radicals can cause oxidative damage to lipids, proteins, and DNA. 12 The most commonly used biomarker of DNA damage through modified bases is the concentration of 8-OHdG. 12,13 Increasing levels of 8-OHdG have been stated in body fluids and tissues in diseases such as diabetes, 14 cancer 14 and periodontitis. 15
The present study aims to evaluate the 8-OHdG levels, which are an indicator of cell damage, in serum, saliva, and GCF following different bleaching methods and to determine the most clinically effective method.
To the best of our knowledge, this study is the first in vivo study indicating the changes of 8-OHdG levels with different bleaching methods and including the recovery time of such oxidative stress. The null hypotheses were as follows: Chemical and laser-assisted bleaching would enhance the 8-OHdG levels in serum, saliva, and GCF immediately after bleaching and two weeks later. Ozone bleaching would not enhance the 8-OHdG levels in serum, saliva, and GCF immediately after bleaching and two weeks later. Ozone bleaching method would be as effective as other bleaching methods.
Materials and methods
This study was conducted at University, Faculty of Dentistry, in the Department of Restorative Dentistry and was approved by the Ethical Committee of University (Approval date and number: 2020/251).
Patients
For a significance level of 5% (α = 0.05) was considered at (1-β) 0.80, the minimum sample size determined was 14 for an effect size of 0.38 by using G*Power 3.1.9 software. Assuming any discontinuing patient during follow-up, two drop-out subjects were added to each group.
Forty-eight participants whose ages ranged between 18 and 28 were included. The relevant teeth were free of any restorations and/or caries. The participants have also had good oral hygiene and no periodontal disease. All were non-smokers. Pregnant or breast-feeding women, severely stained teeth such as tetracycline staining and fluorosis, previously bleached teeth were excluded. On the other hand, equal grouping was tried by taking into consideration the individual characteristics of the participants such as gender, age ratios, and similar tooth color distribution.
Study design
The processes and procedures applied before the in-office bleaching and the characteristics of these systems are given in Tables 1 and 2, respectively.
Processes and procedures applied before the start of office bleaching for all groups.

Parameters of bleaching methods application.

Forty-eight patients were divided into three treatment groups (n = 16). Group 1 (G1): bleaching with ozone; Group 2 (G2): chemical bleaching with 40% H2O2 gel; Group 3 (G3): bleaching with 40% H2O2 gel with diode laser activation. Each bleaching treatment was performed in two clinical sessions with a 3-day interval. The tested treatments were performed as follows.
Group 1: High-concentration ozone (600,000 ppm-µg) was delivered to the tooth surface in a controlled manner with an ozone-releasing machine (Ozonytyron XP-OZ MIO International, Munich, Germany). Ozone was administered for 15 min to each jaw. This was repeated two times. Therefore, the first visit lasted approximately 1 h. For this application, custom trays that can be attached to the ozone machine were produced. A second, similar session was applied 3 days later.
Group 2: For this group, 40% H2O2 bleaching gel (Opalescence Boost whitening system, Ultradent, South Jordan, Utah, USA) was applied according to the manufacturer’s instructions. A lip retractor (OptiView lip and cheek retractor, Kerr Dental, Orange, California, USA) and suction were used for isolation and to avoid any soft tissue damage. Gingival dam (OpalDam Green, Ultradent, South Jordan, Utah, USA) was applied throughout the gingival margin and cured with a light source (VALO Cordless, Ultradent, South Jordan, Utah, USA). Then, a 1 mm-thick layer of bleaching gel was applied to the buccal surface for 15 min. The gel was completely cleansed using an air-water spray and suction. The same procedure was repeated for all teeth three times per session (3 times × 15 min). A second, similar session was applied 3 days later.
Group 3: All steps for this group were the same as those for Group 2 except that 40% H2O2 containing bleaching gel was activated with a 980 nm diode laser (Gigaa Dental CHEESE, Wuhan, China). The bleaching gel remained on the tooth for 10 min while laser activation was performed for each quadrant by using a special bleaching headpiece (5.85 cm2 spot size) at an output power of 7 W for 12 s. All gel remnants were cleansed with an air-water spray. Then, this cycle was repeated two more times, thus completing the first session. A second, similar session was performed 3 days later.
Color examination
The change in color of teeth was measured with a spectrophotometer device (VITA Easy Shade Advance, Zahnfabrik H.Rauter, Bad Säckingen, Germany) by touching the tip of the device to the middle third of the buccal surface at 3-time intervals: before treatment, immediately, and 2 weeks after treatment. The spectrophotometer was calibrated before each measurement. At each measurement, L*, a*, and b* values were recorded and calculated with the formula of CIEDE2000: 16
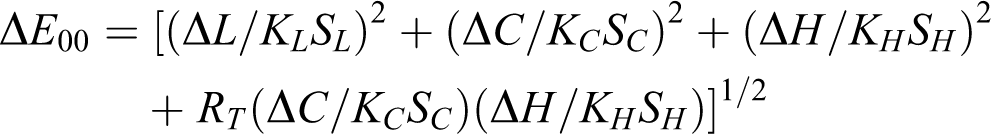
The parametric factors used in the color difference formula are KL = 1, KC = 1 and KH = 1 (corresponding to lightness, chroma and hue, respectively) for CIEDE2000 (1:1:1). 17 The 50:50% perceptibility threshold (PT) is the color difference between two objects that can be perceived by 50% of observers, and the 50:50% acceptability threshold (AT) is the color difference accepted by 50% of observers. For CIEDE2000, 50:50% PT is ΔE 00 = 0.8, whereas 50:50% AT is ΔE 00 = 1.8 under simulated clinical settings. 18
Biochemical examination (GCF, saliva, serum)
All parameters were evaluated at three time points: before treatment (T1), immediately after treatment (T2), and 2 weeks after treatment (T3). After the bleaching treatment, saliva samples were collected.
Unstimulated saliva samples were collected from all patients, placed in centrifuge tubes, and then centrifuged at 10,000 rpm for 10 min. Afterward, GCF samples were obtained from six teeth from the bleaching area (maxillary anterior teeth, from canine to canine) using sterilized paper strips (PerioPaper, Oraflow, Plainview, New York, USA). These paper strips were placed inside the gingival pocket until slight resistance was felt, and the strips remained there for 30 s. Each GCF sample was then placed in an Eppendorf tube.
After the saliva and GCF sample collection, peripheral venous blood samples were obtained from all patients. The samples were then centrifuged at 3000 rpm for 5 min to isolate the serum from the blood. The 8-hydroxydeoxyguanosine (8-OHdG) levels of each group at each time are detailed in Table 3.
Comparison of 8-OHdG levels on serum, saliva, and GCF (mean ± SD).
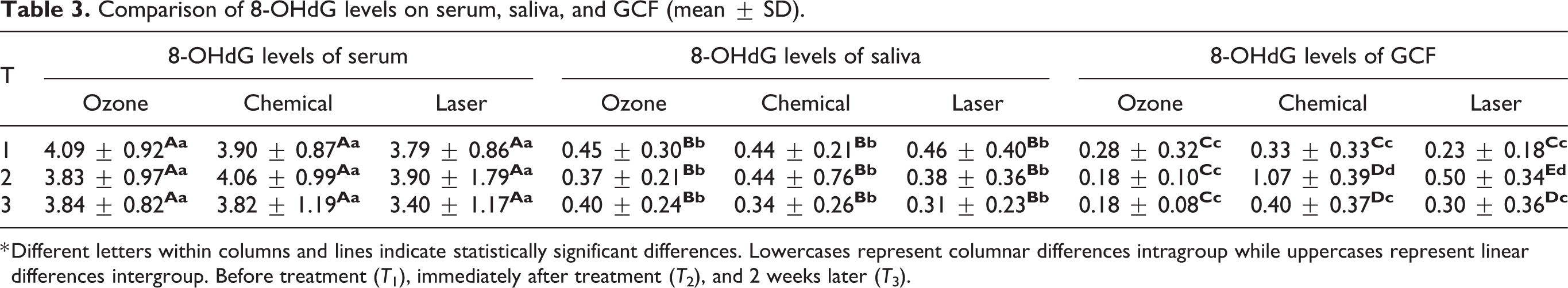
* Different letters within columns and lines indicate statistically significant differences. Lowercases represent columnar differences intragroup while uppercases represent linear differences intergroup. Before treatment (T 1), immediately after treatment (T 2), and 2 weeks later (T 3).
Saliva, GCF, and serum samples were stored at −80°C until biochemical analysis. 8-OHdG levels in saliva, serum, and GCF were measured biochemically by using Northwest kit (NWLSS 8-OHdG ELISA High Sensitivity kit, Northwest; Vancouver, Canada) according to the manufacturers’ protocol.
Statistical analysis
The Statistical Package for the Social Sciences (SPSS, version 22, IBM, Armonk, New York 10504, NY, USA) was used for the statistical analysis. As descriptive statistics, numerical variables were summarized as mean (±standard deviation). In the intragroup comparison, Friedman and Wilcoxon’s tests were used for non-normally distributions, while repeated variance analysis and paired t-test were used for normal distributions independent variables. In the intergroup comparison, ANOVA and the LSD tests were used for normal distributions, while the Kruskal–Wallis and Dunn’s tests were used for non-normally distributions in independent variables. The statistical significance level (p) was set to <0.05.
Results
GCF, saliva, and serum analysis
Intragroup comparison
Salivary and serum 8-OHdG levels: None of the bleaching techniques showed a significant difference immediately after treatment and 2 weeks later (p > 0.05).
GCF 8-OHdG levels: Ozone bleaching did not show a significant difference (p > 0.05) immediately after treatment and two weeks later. Chemical bleaching and laser-assisted bleaching led to significant increases in the GCF 8-OHdG levels (p < 0.05) immediately after treatment. However, these increased levels eventually returned to degrees similar to the pre-operative ones (p > 0.05) 2 weeks later.
Intergroup comparison
The salivary and serum 8-OHdG levels did not significantly differ across the three bleaching techniques at each time (p > 0.05).
There was no significant difference in the baseline GCF 8-OHdG levels among all groups (p > 0.05). By contrast, immediately after treatment, the GCF 8-OHdG levels significantly differed among all groups (p < 0.05). The GCF 8-OHdG levels after ozone bleaching showed significant differences compared with the other groups 2 weeks after the treatment (p < 0.05).
Color analysis
L*, a*, b* values at each time are presented in Table 4.
The mean values ±SD and comparison of L*, a*, b*.

* Different letters within columns and lines indicate statistically significant differences. Lowercases represent columnar differences intragroup while uppercases represent linear differences intergroup. Before treatment (T 1), immediately after treatment (T 2), and 2 weeks later (T3).
Intragroup comparison
For all groups, L*, a*, and b* measurements immediately after treatment (L 2, a 2, b 2) and 2 weeks later (L 3, a 3, b 3) were significantly different from baseline (L 1*, a 1*, b 1*) (p < 0.05).
Similarly, for all groups, there were no significant differences between immediately after treatment (L 2, a 2) and 2 weeks later (L 3, a 3), while for b* values were observed significant differences between b 2* and b 3* in group 2 and 3.
Intergroup comparison
Among all groups, there was no significant difference in L*, a* and b* values in all (p > 0.05) for all measured time.
The ΔE 00 values obtained with the CIEDE2000 formula at each time interval are presented in Table 5.
Comparison of bleaching efficiency for all groups (mean ± SD).

* Different letters within columns and lines indicate statistically significant differences. Lowercases represent columnar differences intragroup) while uppercases represent linear differences (intergroup).
For all inter and intra groups comparisons, ΔE001 values (before treatment - immediately after) were similar to ΔE 002 (before treatment –2 weeks later) (p > 0.05).
Discussion
An unavoidable consequence of aerobic metabolism is the production of reactive oxygen species (ROSs). ROSs are classified as either endogenous or exogenous. ROSs of endogenous origin are produced during routine metabolic processes in the body’s normal use of oxygen. However, exogenous free radicals are produced after exposure to negative external factors, such as heat, trauma, smoking, radiation, dental materials, infection, or excessive exercise. 19 Normally, there is a balance between the formation of these free radicals and the antioxidant system’s removal of them from the body. Oxidative stress develops when this balance shifts to the overproduction of ROSs and their non-removal by the antioxidant system. Oxidative stress has a destructive effect on nucleic acids, proteins, and lipids. 20
Vital bleaching using H2O2 gel is a frequently preferred conservative aesthetic treatment. It easily decomposes to ROSs, 21,22 which consist of superoxide (O2-), hydroxyl (HO), peroxyl (ROO), and alkoxyl (RO). 23 ROSs penetrate hard dental tissue, breaking down the chemical bonds in the chain of chromogens. 22,24 When high concentrations of H2O2 are applied, a huge number of ROSs immediately diffuse throughout coronal hard tissues and may lead to severe pulpal inflammation, mutations, protein denaturation, cell fragmentation of pulp tissue, and enzyme inactivation sensitivity. 25,26 This also results in an imbalance between ROSs and endogenous/exogenous antioxidants. 27
Light sources are used to accelerate the chemical reaction that occurs during the bleaching process. 19 For a diode laser, many different wavelengths are available. However, the 980 nm diode laser is the only one approved by the Food and Drug Administration for dental bleaching due to its superior absorption by water. 28 Previous studies have determined that activation methods increase the efficiency of H2O2 and shorten the time required for bleaching. 7,29,30 On the contrary, in some studies, it has been shown that activation methods accelerate the reaction but do not affect the bleaching activity. 31 –33 However, laser-assisted causes an increase in ROS formation and acceleration of oxidation-reduction reactions. 33,34 This results in creating a successful bleaching effect. 35 On the other hand, increased ROS production can lead to cellular damage on the dental tissue or the surrounding tissues. 36 The ROSs produced by bleaching agents facilitate dental tissue permeability and the diffusion of oxidizing substances from dentinal tubules, which can reach the periodontium. 25 Kawamoto and Tsujimoto 37 have shown that ROSs that come into contact with cells can interact with deoxyribonucleic acid (DNA) and cause oxidative damage, such as strand and chromosomal breakage. However, Rezende et al. 38 suggested that in-office bleaching did not cause DNA damage to the gingival tissue during the bleaching period may be oxidative DNA lesions induced by H2O2 are repaired by DNA repair systems. Furthermore, Lu et al. 39 reported that DNA repair systems can eliminate possible damage.
DNA damage caused by H2O2 is largely repaired by the body’s defense systems. However, the long-term effects of DNA damage on pulp tissue due to frequent or prolonged use of bleaching agents is still unknown. 40 Human saliva can be used as a biomarker which is an indicator of oxidative stress in the oral cavity. 41 GCF is a body fluid containing serum, leukocytes, structural cells, and microorganisms. It can be considered an indicator of inflammation occurring in routine dental procedures. 42 –44 The most common stable product of oxidative DNA damage caused by ROSs is 8-OHdG, which has been shown to increase in GFC, bodily fluids, and tissues in many diseases. 45 –47 According to our results, 8-OHdG levels in serum and saliva were not affected by the bleaching method, but a temporary increase in GCF was observed immediately after chemical bleaching and laser-assisted bleaching. This temporary effect was highest in the chemical group and, there was a significant difference between the other groups immediately after bleaching. The reason for this may be gel leakage into the gum tissue, since chemical whitening takes longer to apply. 48 The bleaching gel containing 35% H2O2 was activated by the diode laser at an output power of 7 W for 12 s. This protocol may promote the antioxidant systems as to bio-stimulated ROS-damaged. 49 On the other hand, these increased levels of 8-OHdG eventually returned to similar before bleaching levels of 8-OHdG 2 weeks later. Therefore, our first hypothesis that chemical and laser-assisted bleaching would enhance the 8-OHdG levels in serum, saliva, and GCF immediately after bleaching and 2 weeks later was partially rejected, because 8-OHdG levels in GFC decreased 2 weeks later. From a biological standpoint, the bleaching with H2O2 whether it is laser-activated bleaching or chemical bleaching regarding their clinical efficacy and potential toxic effects to the dental tissue shall be questioned. Although the increase of 8-OHdG is temporary, the probable side effects of short-term oxidative stress should also be taken into consideration.
In contrast, ozone gas (O3), which has been used medically in the treatment of many illnesses, began to be used in dentistry for the sake of its strong oxygenation and oxidation features. 50 It is mentioned that O3 keeps away the compounds responsible for changing the color of teeth due to its oxidization capacity, which makes it an alternative bleaching method. 51 Ozone gas application is carried out with the help of a special device, and the concentration, time, and reach of the applied ozone are controlled. 52 Ozone gas produces powerful antioxidants, such as superoxide (O), fluorine, and persulfate. 50 However, there are few studies on bleaching with ozone. 53,54
On the other hand, there is no long-term clinical follow-up study of ozone bleaching treatment in the literature. The antibacterial effect of ozone 50 may support sustained whitening, because regression may be caused by bacterial adhesion to the tooth surface after bleaching with H2O2. 55
In the current literature, there is insufficient data to compare 8-OHdG levels following ozone bleaching, chemical, and laser office bleaching on serum, saliva, and GFC. Lima et al. 49 analyzed the levels of cytokines in GCF samples after applying in-office bleaching treatments containing 15% and 35% H2O2 and showed that H2O2-based gels do not cause inflammation at GFC levels. On the other hand, Colares et al. 56 investigated that H2O2 and nitric oxide levels in GCF at 15% and 35% H2O2 concentrations office bleaching application, and revelated the inflammation caused by 35% H2O2 is longer. Firat et al. 57 reported that chemical bleaching can be considered safer when compared on a light-activated bleaching system which could lead to increased proinflammatory cytokine in gingival tissue. Akbari et al. 58 examined possible systemic effects of the home bleaching and revealed that resulted in systemic oxidative stress.
Bleaching with ozone reduced 8-OHdG levels in GCF immediately after bleaching and, two weeks later. Furthermore, the GCF 8-OHdG levels were significantly lowest among all groups. We assumed that the decrease in 8-OHdG levels in GCF following ozone application may be based on the increase in antioxidant enzyme activity 50 and a reduction in inflammatory responses. 59 Thus our first hypothesis that Ozone bleaching would not enhance the 8-OHdG levels in serum, saliva, and GCF following bleaching was accepted.
The use of CIELab (ΔE ab) 60 and CIEDE2000 (ΔE 00) 61 to measure total color differences is very popular for evaluating color changes after dental bleaching. In this study, we used CIEDE2000, which has consistently performed better than other color difference formulae in predicting visual results. 62 The detectability threshold value was considered to be ΔE 00 > 1.8 for CIEDE. 18 In this study, effective whitening was observed in all groups when the data were calculated with the CIEDE2000 formula. In a previous study, Polydorou et al. 63 found that the 980 nm diode laser did not increase the efficacy of bleaching. This is in accordance with the findings of the present study, which found similar results following chemical and diode-laser-activated methods in terms of color change. Aykut-Yetkiner et al. 64 found that 35% peroxide has more bleaching capacities and provides better whitening than ozone because ozone cannot penetrate the tooth as extensively as other peroxide-based bleaching agents. However, some research shows that ozone has the potential to whiten teeth as effectively as H2O2. 51,65 In our study, the color change following the ozone bleaching was similar to bleaching methods including H2O2. Thus, our third hypothesis that the ozone bleaching method would be as effective as other bleaching methods was accepted.
In this study, we aimed to detect the 8-OHdG levels in serum, saliva, and GCF samples to determine the damage potential of using H2O2 and ozone for tooth bleaching. After a detailed literature review, no similar study investigating the effects of ozone and H2O2 on DNA damage by detecting 8-OHdG during the bleaching process was identified. However, this study has several limitations; i. The 8-OHdG which is an indicator of DNA damage due to the hydroxyl radical attack is also one of the biomarkers to measure oxidative stress. In our study, the other oxidative stress biomarkers such as malondialdehyde (MDA), total oxidant status (TOS), and total antioxidant status (TAS) could have been used so that the role of the 8-OHdG would have been confirmed ii. We could have assessed the oxidative damage biomarker by using different methods such as comet assay to check the ELISA method iii. cannot be standardized because the oral cavity is a multifactorial environment, and each patient has individual biological variations.
Conclusion
Chemical and laser-assisted bleaching cause local DNA damage. However, ozone bleaching eliminates this local DNA damage and reduces GCF 8-OHdG levels in two weeks, with bleaching effectiveness similar to that of H2O2. Therefore, ozone bleaching may enable patients to avoid undesired side effects of H2O2.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was self-funded by the authors.
