Abstract
Background:
Renal ischemia-reperfusion (IR) injury is one of the main leading causes of acute kidney injury associated with inflammation, oxidative stress and cell apoptosis. We studied the effects of prazosin, as a specific blocker of α1-AR, on renal IR injury.
Methods:
Rats were divided into normal control; untreated IR and prazosin-treated IR (1 mg/kg body weight). Prazosin was administered by intraperitoneal injection 30 min prior to IR induction. The level of urea/creatinine and oxidative factors were detected by colorimetric methods. Apoptosis-associated factors, inflammatory, and signaling proteins were analyzed in renal tissue. The abnormalities of renal histopathology were detected by immunohistochemistry.
Results:
Administration of prazosin to IR rats ameliorated serum urea and creatinine and IR-induced histopathological damages. Lipid peroxidation was significantly improved after treatment by prazosin in IR injury rats, however, antioxidant status was not affected. Rats subjected to IR injury activated Bax protein and NF-κB mediated inflammatory response. Moreover, treatment with prazosin inhibited renal NF-κB activation, resulting in a significant decline in pro-inflammatory cytokine of IL-6.
Conclusion:
These findings suggest that prazosin could be a good candidate to attenuate renal IR injury due to its ability to modulate renal function, apoptosis and inflammation.
Introduction
Ischemia and reperfusion (IR) renal injury is the leading cause of acute renal failure, which may result in secondary tissue injury and acute inflammation. In detail, IR injury is caused by blood blockage to a specific organ and the restoration of blood flow and re-oxygenation state. Several cellular and molecular events are involved in IR, including inflammatory responses, release of reactive oxygen species (ROS), an increment of cytokine release, which eventually lead to mal-function of tissues. 1,2 Indeed, ischemia is a hypoperfusion state in which an unavoidable event occurs in various conditions such as kidney transplantation. 3 The final phase of ischemia arises during reperfusion. Reperfusion, also known as the effector step of ischemic injury, is more destructive and causes renal tissue damages by several pathologic mechanisms including releasing free radicals, infiltration of leukocytes into the renal graft, inflammation and apoptosis stimulation. 4 The outcomes after the reperfusion phase are more destructive. At reperfusion phase, restoring blood flow to ischemic organ supplies excess of oxygen which generate an increased level of ROS. ROS can cause oxidative stress and subsequent events, including inflammatory responses, DNA damage and endothelial dysfunction. 5,6 Release of pro-inflammatory cytokines (IL-1, TNF-α, and IL-6) can stimulate innate immunity leading to kidney rejection. 7 Together, IR can cause renal tissue damages by several pathologic mechanisms, including releasing of free radicals, infiltration of leukocytes into the renal graft, inflammation, and apoptosis stimulation. 8 Inflammatory cytokines and oxidative stress may cause cellular damage. 9 –11 Various renoprotective drugs have been supposed to overcome the damaging effects of IR. 12 It has been found that alpha1-adrenoceptor (α1-AR) is one of the functionally relevant adrenoceptor subtypes in the renal vasculature of the rat. 13
In fact, adrenoceptors are G protein coupled receptors, expressing in arterioles of kidney and participate in a variety of physiological renal functions and inhibition of α1-AR exhibit preventing impact on renal injury. 14
Several lines of studies have demonstrated that blocking of α1-AR decreases the level of stress-induced pro-inflammatory cytokines such as IL-1, IL-6 and increases the production of anti-inflammatory factors such as IL-10. 15 Besides that, blockade of α1-AR has been reported to reduce the effects of renal function and hemodynamic changes induced by stimulation of renal nerve and injection of norepinephrine (NE) in the kidney. 13 Therefore, the evidence has confirmed the effectiveness of α1-AR antagonists in the treatment of kidney against renal failures such as IR injury. 16
Prazosin is a selective antagonist of α1-AR, having an effective antihypertensive impact in patients with chronic renal failure. Furthermore, it has exhibited the protective effects of prazosin on cardiomyocytes against oxidative stress, inflammation, and apoptosis by enhancing expression and activity of Extracellular Signal-Regulated Kinase ½(ERK1/2). Activation of ERK is able to maintain the process of mitosis, cell growth, and differentiation. 17,18 Several lines of studies have confirmed that increased activation of ERK1/2 could have renoprotective effects against IR. 19 Previous studies have confirmed that MEK-ERK-mediated signals play a key role in ischemia-induced apoptosis by regulation of Bax and Bcl2 expression. 20
Oxidative stress is maliciously generated by renal IR and it is able to induce apoptosis. 3 Apoptosis is plainly associated with the downregulation of anti-apoptotic protein such as Bcl-2, over expression of pro-apoptotic protein such as Bax and amplification of active Caspase-3. 21
Although different studies have shown the efficacy and renoprotective effect of prazosin against IR-induce adrenal injury, 22 no studies have evaluated direct renoprotective effects of prazosin in factors involved in apoptosis, inflammation response, and oxidative stress on IR status. The main objective of the present research was to study the protective effect of prazosin on IR injury in kidneys of rats by evaluation of involving factors in oxidative stress, apoptosis, and inflammation process.
Materials and methods
Reagents
Prazosin was provided by Tocris Bioscience (Ellisville, MO). Stock solutions of drug was prepared in distilled water and injected intraperitoneally.
Animals and experimental groups
For this study, 21 Male Wistar rats (230 ± 15 g) were provided from Pasture Institute (Tehran, Iran). All experimental rats were maintained according to the standards of the National Institutes of Health for Laboratory Animals Care and Use (22°C, with 60 ± 5% humidity, and 12/12 h light/dark cycle) for 7 days.
23
During this period of time, the animals were fed with ad libitum and had free access to water. The rats were randomly distributed into three equal groups: Group I: normal control group (sham); Group II: non-treated IR group; Group III: IR pre-treated with prazosin at 1 mg/kg body weight.
24
In brief, groups I and II just were fed by a standard diet (no treatment) and group III was treated a single intraperitoneal injection with prazosin 30 min before induction of IR injury.
Induction of IR injury
In order to induce IR, according to the previous published research, the animals were anesthetized with xylazine and ketamine (10 and 90 mg/kg, IP) through i.p injection. 25 We performed unilateral IR injury protocol in left kidney. In the control group (sham), just renal vascular (arteries and veins) manipulation (without clamp) was performed, whereas in the other two groups, the abdominal area was incised and renal vascular were blocked for 45 min by non-traumatic vascular forceps. Ischemic was approved by observing the red-colored kidneys turned to pale. Then, hemostatic forceps were removed and the abdominal area was stitched. Then all animals were returned into their own houses. After 6 h reperfusion, blood samples were collected directly from the left ventricle of experimental animals for subsequent biochemical analysis. Finally, rats were euthanized and the kidney in all animal groups was separated and immersed in liquid nitrogen and stored at −80°C for further analyses.
Measurement biochemistry
Blood samples were kept at 37°C to be clotted and then centrifuged to separate the serum. Serum samples were applied to measure creatinine and urea by commercially available colorimetric assay kits (BioMerieus). The biochemical analysis was performed using an autoanalyzer (Olympus AU 5400, Tokyo, Japan).
Histopathological examination
To study histopathological changes of kidneys in response to the administrated prazosin, the kidney tissues were separated and fixed by formalin (10% neutral) then embedded in paraffin. The prepared tissues were sectioned and stained by hematoxylin and eosin (H&E). A light microscope (Olympus, Tokyo, Japan) was applied for histopathological examination. The results were graded based on the scale of severity of observed changes.
Determination of IR-induced oxidative stress status in kidney
The kidney sample of animals was divided into small pieces, put in a cold solution of 0.25 M sucrose and blotted on a filter paper. The prepared tissues were homogenized in 10% cold Tris–hydrochloride buffer using a tissue homogenizer (Remi Motors, Mumbai, India). Then, centrifuged for 15 min at 7000 rpm at 0°C. The final supernatant was used for detection of superoxide dismutase (SOD), lipid peroxidation for MDA (malondialdehyde) content, glutathione peroxidase (GPx), reduced glutathione (GSH), total antioxidant capacity (TAC) and catalase (CAT) activity by the colorimetric methods. The SOD activity was determined based on pyrogallol autoxidation, as previously developed method by Marklund and Marklund. 26 The CAT activity was detected by H2O2 consumption and measured using the Claiborne method at 240 nm. 27 Subsequently, GSH level was investigated based on the previously defined method and described as Nm/MG-PRnm. 28 GPx activity assay was conducted using an assay kit provided from Northwest (Life Science Specialises, LLC). 29,30
Assessment of apoptosis and inflammation factors in renal and serum
Western blot analysis was conducted to determine the expression of apoptosis-related factors (Pro- and cleaved caspase-3, Bcl-2, and Bax), inflammatory cytokines (IL-1b, IL-6 and TNF-a) and other signaling proteins including MMP2, MMP9, IkB-α/phosphorylated IkB-α (pIkB-α) and NF-kB/ phosphorylated NF-kB(pNF-kB) as previously described method. 31 In brief, renal tissues were homogenized in cold RIPA lysis buffer containing 1% cocktail as a protease inhibitor. After centrifugation at 12,000 rpm for 10 min, the quantity of total protein in the supernatants was determined using a BCA protein assay kit (Pierce Biochemical, Rockford). 32
Sixty microgram protein of each sample was loaded on 10% SDS-PAGE. 33,34 The separated protein on the gels was transferred to polyvinylidene fluoride membrane. The skim milk (5%) was used to block the membranes. Then, the membranes probed with the following primary antibodies:
Mouse Monoclonal BCL2 (sc-492 Santa Cruz Biotechnology), BAX (sc-7480, Santa Cruz Biotechnology, Inc.), caspase 3 (SC-7272, Santa Cruz Biotechnology, Santa Cruz, CA), Cleaved caspase (SC-56052-Santa Cruz, CA), IL-6 (sc-28343; Santa Cruz Biotechnology, Santa Cruz, CA, USA), TNF-α (sc-130349 Santa Cruz biotechnology, Inc.), IL1-B (sc-32294, Santa Cruz Biotechnology), p-NF-kB (ab16502, Abcam), MMP2 (sc-10736, Santa Cruz Biotechnology, USA), MMP9 (sc-393859, Santa Cruz Biotechnology, USA), p-IKB1 (Elabscience Biotechnology), and β-actin (sc-47778, Santa Cruz Biotechnology Inc).
After this stage, the membranes were washed with Tris–buffered salin–Tween 20 (TBST) at three times for 5 min, and then, the membranes were probed with secondary goat anti-mouse HRP-conjugated antibody (SC-2031, Santa Cruz Biotechnology Inc) at room temperature for 2 h. At the final stage, the membranes were washed and protein bands were visualized using enhanced chemiluminescence detection kit (Roche, UK). Finally, imageJ 1.6 software was used to measure the intensity of protein bands. β-actin control was used to normalize the signal intensity of each band.
Statistical analysis
All experiments were done in triplicates and data were depicted as mean ± SD. The statistical analysis was carried out using Graphpad prism software 6.01. One way analysis of variance (ANOVA) was used to compare laboratory data according to Tukey’s test. A value of p < 0.05 was considered statistically significant. * indicates comparison with sham control group, # indicates comparision with IR-induced group.
Results
Effect of prazosin on histological abnormalities in renal IR-induced rats
The histological changes in all experimental groups have been shown in Figure 1. In the rats subjected to renal IR injury, morphological damage was revealed when compared with the sham group (p < 0.001). Treatment with prazosin attenuated the morphological abnormalities in IR-induced rats as compared with untreated IR group, however, the injury score not reduced in a significant manner.

Evaluation of histopathological abnormalities in IR-induced renal injury. (a) H & E staining (400×) of kidney sections in the sham, IR, and IR + prazosin group are depicted. (b) Injury scores were graded based on the scale of severity of observed changes. ***p < 0.001 vs. sham control group, H&E: hematoxylin and eosin; IR: ischemia–reperfusion group, (scale bar = 25 μm); ns: non-significant. All values are expressed as mean ± SD.
Prazosin administration enhanced renal function after IR Injury
To investigate renal function, the level of urea, creatinine and total protein were detected in serum samples of three experimental groups. Rats exposed to IR injury revealed a high level of serum urea (p < 0.001) and creatinine (p < 0.01) as compared to the sham group, indicating a kind of dysfunction in the IR-induced rats. Following prazosin administration, the level of urea and creatinine were reduced significantly when compared with the untreated IR-induced rats (p < 0.01 and p < 0.001, respectively) (Figure 2(a) and (b)). In fact, prazosin at 1 mg/kg body weight was effectively decreased urea and creatinine level to the average level of sham groups. Moreover, the level of total protein was reduced in the IR-induced group (p < 0.01), however, no significant change was observed between prazosin-treated and untreated IR groups (Figure 2(c)).

Effect of prazosin administration on renal function of IR-induced rats. **p < 0.01 and ***p < 0.001 vs. sham control group. ## p < 0.01 and ###p < 0.001 vs. IR-induced group. IR: ischemia–reperfusion; PRA: prazosin. All values are expressed as mean ± SD.
Effect of prazosin on IR-induced oxidative stress
As shown in Table 1, renal IR injury resulted in a drastic increase in MDA level (3.80 ± 0.18 nmol/mg) when compared to the sham group (p < 0.001), whereas treatment with prazosin, decreased MDA level significantly in IR-induced rats as compared to the untreated IR group (p < 0.001). In addition, IR injury significantly reduced the antioxidant capacity of SOD, GSH, GPX, and TAC in the kidney of IR rats in comparison to the sham rats (p < 0.01, p < 0.001, p < 0.001 and p < 0.01, respectively), and treatment of IR-induced rats with prazosin increased the level of GSH as compared with untreated IR groups (p < 0.05), returning fairly to the level as prior to IR injury. However, the level of SOD, GPX and TAC did not demonstrate significant alteration in prazosin-treated IR-induced rats. The level of CAT did not differ in IR-induced and prazosin-treated group. Alterations in oxidative stress factors such as GSH, SOD and MDA confirmed that the induction of IR could alter the antioxidant capacity in IR injury. These findings indicated that prazosin can improve the antioxidant activity of enzymes in IR injury.
Evaluation of oxidative markers in prazosin-treated IR-induced rats.
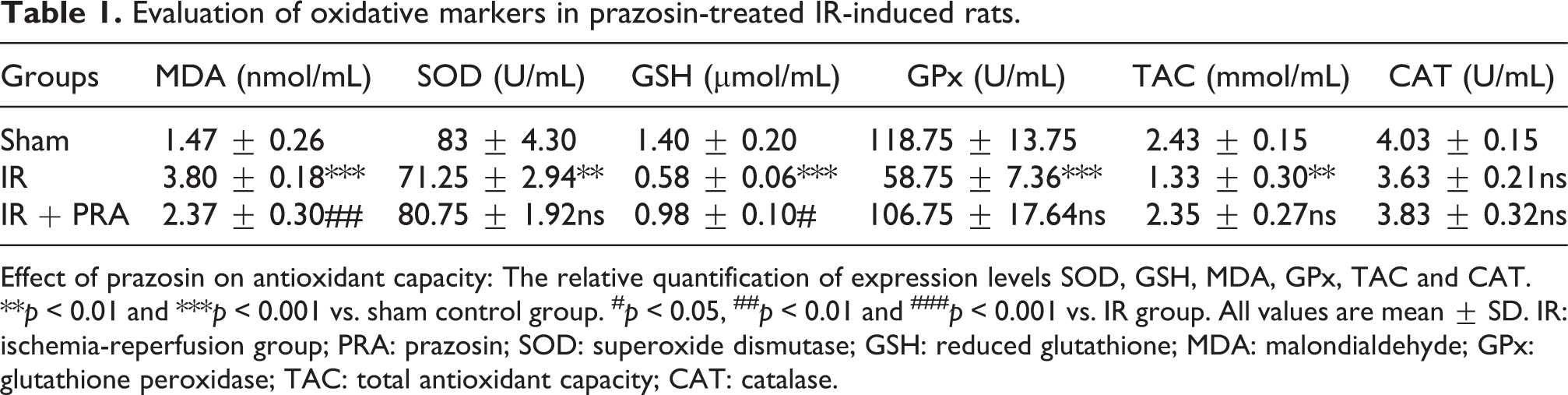
Effect of prazosin on antioxidant capacity: The relative quantification of expression levels SOD, GSH, MDA, GPx, TAC and CAT. **p < 0.01 and ***p < 0.001 vs. sham control group. # p < 0.05, ## p < 0.01 and ### p < 0.001 vs. IR group. All values are mean ± SD. IR: ischemia-reperfusion group; PRA: prazosin; SOD: superoxide dismutase; GSH: reduced glutathione; MDA: malondialdehyde; GPx: glutathione peroxidase; TAC: total antioxidant capacity; CAT: catalase.
Effect of prazosin on IR-induced inflammation
To evaluate the effect of prazosin on IR-induced inflammation, the expression level of pro-inflammatory cytokines was investigated by western blotting. As shown in Figure 3, pro-inflammatory cytokines including TNF α, IL 6, and IL 1β were induced in IR rats as compared to sham rats and among these cytokines, TNF α and IL-6 were significantly produced in the IR group (p < 0.05). Treatment of IR rats with prazosin reduced the expression level of all observed inflammatory cytokines and among them, the level of IL-6 was dramatically decreased as compared to IR group (p < 0.01).

Effect of prazosin on IR-induced alterations in TNF α, IL 6, and IL 1β expression levels. (a) Representative western blotting analysis of inflammatory cytokines in renal tissues of rats. The relative quantification of expression levels (b)TNF α, (c) IL 6, and (d) IL 1β. *p < 0.05 vs. sham control group. ##p < 0.01 vs. IR-induced group. IR: ischemia-reperfusion; PRA: prazosin; ns: non-significant. All values are expressed as mean ± SD.
Effect of prazosin on IR-induced renal apoptosis
The impact of prazosin on apoptosis-associated proteins was determined by western blotting analysis. Induction of IR in rats up-regulated the expression level of pro-apoptotic protein of Bax (p < 0.001) and to some extent the active form of caspase3 (cleaved caspase-3) as compared to sham rats (Figure 4(b) and (f)). Administration of prazosin reduced the expression level of Bax and cleaved caspase-3 as compared to the untreated IR groups, however, the change were not significant. On the other hand, the expression of procaspase-3 and Bcl-2 (anti-apoptotic protein) significantly decreased in IR-induced rats as compared with untreated groups (p < 0.05 and p < 0.001). The treatment of prazosin remarkably increased Bcl-2 expression (p < 0.01) to the average level of the sham group (Figure 4(c) and (e)). The comparison of the relative density of Bax/Bcl-2 and cleaved/pro caspase3 in untreated and prazosin-treated IR rats indicated that prazosin can effectively reduce apoptotic promoting factors in IR-induced kidney damage.

Evaluation of pro- and anti-apoptotic factors in prazosin-treated IR rats. (a) Representative western blotting analysis. The relative quantification of expression levels (b) Bax, (c) Bcl-2, (d) Bax/Bcl-2, (e) pro-caspase3, and (f) cleaved-caspase3 and cleaved/Procaspase3. *p < 0.05, **p < 0.01, and ***p < 0.001 vs. sham control group. # p < 0.05 and ## p < 0.01 vs. IR-induced group. IR: ischemia-reperfusion group; PRA: prazosin; ns: non-significant. All data are expressed as mean ± SD.
Effect of prazosin on IKB/NF-κB signaling pathway in renal IR injury
To investigate the effect of prazosin on NF-κB signaling pathway in the rats subjected to IR injury, the protein levels of p-NF κB/NF-κB and p-IκBα/IκBα were detected by western blotting. Our observation depicted that the protein expression of NF-κB, p-NF κB, and p-IκB-α significantly went up in IR rats when compared to sham rats (p < 0.001, p < 0.001, p < 0.01), while treatment of IR rats with prazosin potently decreased the level of these proteins as compared to the untreated IR group (p < 0.05, p < 0.05 and p < 0.01, respectively) (Figure 5(b), (c), and e). Moreover, the expression level of IκB-α protein came down in IR rats as compared to that in the sham rats (p < 0.001) and treatment with prazosin increased its expression level in a significant manner (p < 0.05). These observations revealed that prazosin may have the renoprotective effect through the contribution in p-NF κB/NF-κB and p-IκBα/IκBα signaling pathways.

Effect of prazosin on prazosin on NF-κB signaling pathway in rats with IR. (a) Representative western blotting analysis. The relative quantification of expression levels (b) NF-κB, (c) p-NF-κB, (d) IκBα, and (e) p-IκBα. **p < 0.01 and ***p < 0.001 vs. sham control group. # p < 0.05 and ## p < 0.01 vs. IR-induced group. IR: ischemia-reperfusion group; PRA: prazosin. All data are expressed as mean ± SD.
Effect of prazosin on the expression of matrix metalloproteinase proteins in renal IR injury
In order to study the MMPs protein level in IR rats, western blot analysis was conducted, too. As demonstrated in Figure 6, induction of IR injury in rats promoted the expression of MMP-2 and MMP-9 in a significant manner when compared to the sham group (p < 0.05). Treatment with prazosin showed a dramatic reduction only in MMP-9 expression level (p < 0.05). Prazosin reduced the expression of MMP-2 protein; however, we did not observe any significant change as compared to the untreated IR group.

Effect of prazosin on the expression level of MMP-9 and MMP-2 proteins in IR-induced rats. (a) Representative of western blotting analysis. The relative quantification of expression levels (b) MMP-2 and (c) MMP-9. *p < 0.05 vs. sham control group. # p < 0.05 vs. IR-induced group. IR: ischemia-reperfusion group; PRA: prazosin; ns: non-significant. All data are expressed as mean ± SD.
Discussion
IR is the leading cause of acute kidney injury, associated with high mortality and morbidity.
Several lines of studies have shown the effectiveness of α1-AR antagonists in the treatment of kidney against renal failures such as IR injury. 16 In the present study, we investigated whether prazosin can protect renal against IR injury in rats.
Urea and creatinine are unwanted metabolic products excreted by the kidney in normal condition. The high serum level of these factors reveals renal dysfunctionality. 35 It has been found that the production of high levels of urea could cause oxidative stress. 36 Furthermore, augmented levels of urea induce ROS-mediated mitochondrial apoptosis in the kidney. 37 In the current study, we determined effects of prazosin as a selective α1-adrenoceptor antagonist on IR-induced renal dysfunction. Interestingly, our findings showed that administration of prazosin showed nephroprotective effects. Prazosin efficiently reduced the serum level of urea and creatinine as compared to the untreated IR group, suggesting that prazosin markedly could overcome the deterioration of renal function. Previous studies have consistently confirmed the beneficial effects of different α-blockers such as doxazosin in preservation of renal function through increasing glomerular filtration and decreasing serum creatinine and urea level. 38
When the level of ROS production overwhelms the antioxidant capacity of the cell, this condition leads to a state of oxidative stress and contributes to I/R injury. Oxidative stress can directly affect biological molecules such as lipids, proteins, nucleic acids and carbohydrates and induce its injurious effects by changing the structure and the activity of molecules. Besides that, it is assumed that oxidative stress may induce its toxic effect indirectly through NF-κB activation. Different mechanisms have been studied to explain the effects of oxidative stress on the NF-κB signaling pathway. Most of the destructive effects of NF-κB activation on renal tissue are mediated by augmentation of inflammatory cytokines (e.g. IL-1β, IL-6 and TNF-α) and induction of apoptosis. 39 The role of ROS in NF-κB activation by inflammatory cytokines has been the focus of several studies. H2O2 is one of the molecules produced during the oxidative stress generated by inflammatory signals. It has been demonstrated that H2O2 could noticeably decrease the ability of TNF to induce IKK activity, which in turn resulted in the inhibition of I-κB degradation, NF-κB activation and translocation in the nucleus. 40 Therefore, agents with antioxidant activity can act as NF-κB inhibitor, preventing in this way renal I/R injury.
Alteration in MDA level and antioxidant activities are important parameters in oxidative stress. 4 In this study, prazosin treatment significantly reduced the MDA level and increased GSH level in IR injury, which demonstrates that prazosin is able to protect the kidney against oxidative stress-induced abnormalities in IR rats. Induction of inflammatory responses in IR injury occurs to restore the immune system to its initial status. IL-1, TNF-α, and IL-6 play a key role in the inflammatory state of an organ. 41 Moreover, TNF-α, and IL-1 are the typical activators of the NF-κB pathway, which plays an important role in the development of IR injury. 40 The results of present study indicated that the treatment of IR rats with prazosin declined expression level of pro-inflammatory cytokines in kidney as compared to the untreated IR.
Induction apoptosis is the other key contributor to the renal IR injury. 42 Apoptosis is related to downregulation of Bcl-2 (anti-apoptotic protein) and the high expression of Bax (pro-apoptotic protein), and amplified caspase-3 activity. 43 It is acknowledged that Bcl-2 is the potent inhibitor of apoptosis triggered by IR and involved in prolonged cell survival by impeding the apoptotic pathway. Moreover, it has been shown that dysfunctionality of the mitochondrial and Bcl-2 family proteins are the main mediator of the apoptotic process. 21 Consistent with these studies, our observations showed that IR strongly diminished Bcl-2 and increased TNF-α level which can be correlated with the caspase 3 pathway. Treatment with prazosin up-regulated Bcl-2 and hindered caspase 3 activity. Decreased level of Bax /Bcl-2 ratio and cleaved caspase 3 in prazosin treatment in IR rats indicate the potential of prazosin in reversing the mitochondrial apoptosis pathway during 6 h after reperfusion. Previous reports have also shown that mitochondria are the main organelles impaired in the epithelial cells by oxidative stress. Under oxidative stress condition, the expression of the p53 tumor suppressor gene increases, leading to the over expression of the downstream apoptotic proteins such as caspase3 and Bax. 44
Considering that NF-κB is a critical mediator for the regulation of inflammatory cytokines, it is suggested that inhibition of NF-κB activation can reduce inflammatory responses in IR injury. 45 The activation of NF-κB occurs through the dissociation from IκB (NF-κB inhibitory protein) via the phosphorylation of IKKα/β. 46 Based on these evidence, we investigated the effect of prazosin on IκB and NF-κB proteins. The findings of the current study demonstrated higher level NF-κB p-65, and p-IκB α/IκB α in renal tissue of the IR model rats than those in sham rats, confirming the presence of the inflammatory status of renal tissue of the IR-induced rats due to the active inflammation-related signaling pathway. Our observation revealed that prazosin administration could significantly suppress NF-κB protein in the treated IR rats. According to our findings and previous studies, it seems that one of the main mechanisms by which prazosin may inhibit NF-κB activation, is the inhibition of oxidative stress which induced by I/R injury. 39,40 Direct suppression of IκB protein phosphorylation may be the other mechanism of NF-κB inhibition by prazosin. However, there are not enough evidences to confirm this statement.
We finally elucidated whether the pro-inflammatory cytokines in IR rats have induced MMPs activation, we detected the expression of MMP-9 and MMP-2. A prior study has reported overexpression of MMP-2 and MMP-9 proteins in renal IR injury. Besides that, there was a direct relation between the severity of inflammation and level of these MMPs. 47 Based on these observations, our findings depicted the higher level of MMP-2 and MMP-9 in rats subjected to IR surgery. Interestingly, administration of prazosin down-regulated the expression level of MMP9 and MMP-2. Taken together with this view, these finding may be important in light of the impact of prazosin in blunting the detrimental effects of oxidative stress, inflammation and apoptosis that resulted from IR injury.
Since the higher number of rats leads to harder management the small number of examined animals may be considered as a limitation in this study which may caused to inadequate reports to draw a definitive conclusion. Another possible limitation that may exist in this study is insufficient information regarding the effects of prazosin. Further investigation need to fully assess the effectiveness of prazosin in terms of anti-inflammatory and anti-oxidative effects.
In the current study, to elucidate the renoprotective effects of prazosin, administration of it was done before IR injury. Further investigation is in demand to investigate the therapeutic effect of prazosin after the occurrence of IR injury. Moreover, it seems that is required to determine various dose and administration routes (e.g. gavage and injection) of prazosin, in regards.
Conclusion
Based on the present findings and as a concluding remark, it can be suggested that prazosin administration may have a beneficial and renoprotective effects on IR injury, which is a crucial concern for kidney functions. This effect of prazosin is probably mediated via modulating and interfering with a series of events, including inflammation and apoptosis pathways in renal tissues.
Footnotes
Acknowledgements
The authors would like to thanks the Molecular Medicine Research Center, Tabriz University of Medical Sciences, Tabriz, Iran. All researchers who have made considerable contributions, but who have not met the criteria for being authorship, are acknowledged here.
Author contributions
Conceptualization and experimental design: MMR; curation of experimental data and writing original draft: AB, YB, and AVN; investigating and visualizing the research: YB and EF; supervision: SM and SJ; writing, review and editing: SJ and SM, final editing: EF. All listed authors have read and approved the final manuscript.
SJ and SM equally contributed to this work.
Data availability
The data sets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
Ethical consent was given by Ethics Committee at Tabriz University of Medical Sciences, Tabriz, Iran with Ethic code No: IR.TBZMED.VCR.REC.1399.255, and Pazhoohan ID 65704.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The financial sponsorship of this work provided by Molecular Medicine Research Center, Tabriz University of Medical Sciences, Tabriz, Iran [Pazhoohan ID:65704].
