Abstract
The aim of the present study was to study the effects of arachidonic acid (ARA) in a rat brain ischemia/reperfusion model induced by middle cerebral artery occlusion (MCAO). A total of 50 rats were randomly divided into five groups: control group, MCAO group, MCAO + ARA 0.3 g/kg group, MCAO + ARA 1 g/kg group, and MCAO + ARA 3 g/kg group. The MCAO + ARA groups received ARA by intraperitoneal injection daily for 14 consecutive days, while the rats in the control and MCAO groups were given equivalent volume of saline. We detected the Morris water maze test and pathological changes to investigate the ischemia/reperfusion injury. The protein levels of tumor necrosis factor-alpha and interleukin-6 in the hippocampus were detected by enzyme-linked immunosorbent assay kits. In addition, the activities of superoxide dismutase, glutathione peroxidase, and malondialdehyde were assayed in hippocampus homogenates to evaluate the oxidative stress after ischemia/reperfusion. The results indicated that ARA administration decreased biochemical parameters of inflammation and oxidative stress. Morris water maze test and histopathological examination further verified the protective effects of ARA on ischemia/reperfusion injury rats. These findings demonstrated that ARA could protect MCAO-induced brain injury rats by inhibition of inflammation and oxidative stress, suggesting that it may have potential as a therapy for cerebral ischemia/reperfusion injury.
Introduction
Ischemic stroke is a major cause of brain dysfunction and adult disability worldwide. 1 –3 Ischemic stroke occurs when a major cerebral artery is blocked by a thrombus or embolism. 4 This blockage impairs neuronal function and causes cell death and subsequent neurological dysfunction. Middle cerebral artery occlusion (MCAO) caused extensive death of hippocampal Cornu Ammon 1 (CA1) pyramidal neurons and a permanent long-term potentiation impairment in Schaffer collateral-CA1 synapses. 5 Previous studies showed that MCAO followed by reperfusion resembles the scenario of human ischemic stroke and it has been widely used to study ischemia mechanisms and potential interventions. 6,7 Although extensive studies on ischemia/reperfusion have been performed, an effective therapy remains to be found.
Reperfusion injury encourages several pathological roles. 8,9 Among them, inflammatory and oxidative stress have been identified in ischemia/reperfusion injury. Inflammation is one of the main causes of ischemic brain damage, 10 and it is well established that oxidative stress is significantly activated after ischemic injury. 11 Previous research has shown that anti-inflammatory and antioxidant agents had potent role in treating the cerebral ischemia/reperfusion injury. 12,13 Therefore, potent anti-inflammatory and antioxidant agents’ interference may be beneficial in the treatment of cerebral ischemia/reperfusion injury.
Arachidonic acid (ARA), which is a main constituent of biomembranes, is one of the major components of hippocampus and important in brain function. 14,15 Previous studies have demonstrated that ARA has anti-inflammatory and antioxidative effects. 16,17 We postulated that ARA would protect cerebral ischemia/reperfusion injury in rats. The Morris water maze test, histopathologic changes, inflammatory responses, and oxidative stress status were analyzed.
Methods
Animals
Adult male Wistar rats (290–330 g) were obtained from the Experimental Animal Center of Suzhou Aiermaite Technology Co. Ltd. (SPF grade, Certificate No. SCXK20140007). The rats were housed in a room with a temperature of 22°C ± 2°C, a relative humidity of 50% ± 10%, 12-h light/dark cycle, and free access to water and food.
Ethics statements
All animal experiments were performed in accordance with the National Institute of Health Guide for the Care and Use of Laboratory Animals. All procedures in this study, including the use of animals, were approved by the Institutional Animal Care Committee of Jinan Central Hospital.
Experimental procedure
Rats were randomly assigned to five groups: (a) control group, (b) MCAO group, (c) MCAO + ARA 0.3 g/kg group, (d) MCAO + ARA 1 g/kg group, and (e) MCAO + ARA 3 g/kg group. Each group consisted of 10 rats.
The cerebral ischemia was induced by MCAO as described previously. 7 In brief, the rats were anesthetized with chloral hydrate (400 mg/kg, intraperitoneal [i.p.]), a midline incision was made to expose the left common carotid artery in the neck region. A 3-0 monofilament nylon suture (4.0 cm in length; Ethicon; Cincinnati, Ohio, USA) was inserted into the external carotid artery lumen through a small nick to block the middle cerebral artery. Two hours after the induction of ischemia, the filament was slowly withdrawn and the animals were then returned to their cages for a period of 22 h of reperfusion. All the procedures were performed with the exception of filament insertion in sham control rats.
The rats in the three ARA groups (MCAO + ARA 0.3 g/kg, MCAO + ARA 1 g/kg, and MCAO + ARA 3 g/kg) received ARA by i.p. injection daily for 14 consecutive days, while the rats in control and MCAO groups were given equivalent volume of saline.
Morris water maze test
In order to assess the spatial learning and memory of the experimental animals, we performed the Morris water maze test. 18 The apparatus consisted of a circular water tank (180 cm diameter and 60 cm diameter). The water temperature was kept at 24°C ± 0.5°C. A hidden submerged platform was placed in one quadrant 2 cm below water surface. The time of locating the submerged platform was measured. Every rat was subjected to four swimming trials in morning and afternoon time period for five consecutive days. Each trial started from a different quadrant. On testing day 6, the platform was removed and the animals were placed into the water maze for 120 s.
Histological examination
After Morris water maze test evaluation, the rats were killed by an overdose of pentobarbital. The brain tissue was fixed in 10% formalin solution, embedded in paraffin wax, and coronal sections of 5 μm thickness were made. After staining with hematoxylin and eosin (H&E), the pathologic changes in the brain tissues were observed under a light microscope at 200× magnifications (DP73; Olympus, Tokyo, Japan).
Assay of cerebral infarction
The brain tissue was processed by following the above-mentioned method and coronal sections of 2 mm thickness were made, placed in 1% 2,3,5-triphenyltetrazolium chloride solution, incubated for 15 min, and then placed in 4% formaldehyde solution overnight. Cerebral infarction area (infarct area/area at risk × 100%) was calculated.
Measurement of inflammatory mediator levels
The brain tissues were homogenized in physiological saline solution and centrifuged at 3000 r/min for 10 min at 4°C. The supernatant was collected and the levels of interleukin-6 (IL-6) and tumor necrosis factor-alpha (TNF-α) were measured using enzyme-linked immunosorbent assay (ELISA) kits according to the manufacturer’s instructions (Nanjing Jiancheng Co., Nanjing, China).
Measurement of biomarkers of oxidative stress
The activities of superoxide dismutase (SOD), glutathione peroxidase (GSH-Px), and malondialdehyde (MDA) in brain tissue were measured by ELISA kits.
Statistical analysis
Statistical analysis was performed using SPSS 17.0 and data were reported as the mean ± standard deviation. Differences between groups were analyzed by one-way analysis of variance followed by Scheffe test. P < 0.05 was considered statistically significant.
Results
Morris water maze test analysis
The escape latency to find the hidden platform in all rats were shown in Table 1. Among the five groups, rats in MCAO group spent the statistically longest time to search for the platform hidden in water (p < 0.05). The results indicate that MCAO led to impairment in spatial learning and memory, and the treatment with ARA significantly attenuated the performance impairment in the test trials.
Effects of ARA on the escape latency to find the hidden platform in Morris water maze test.
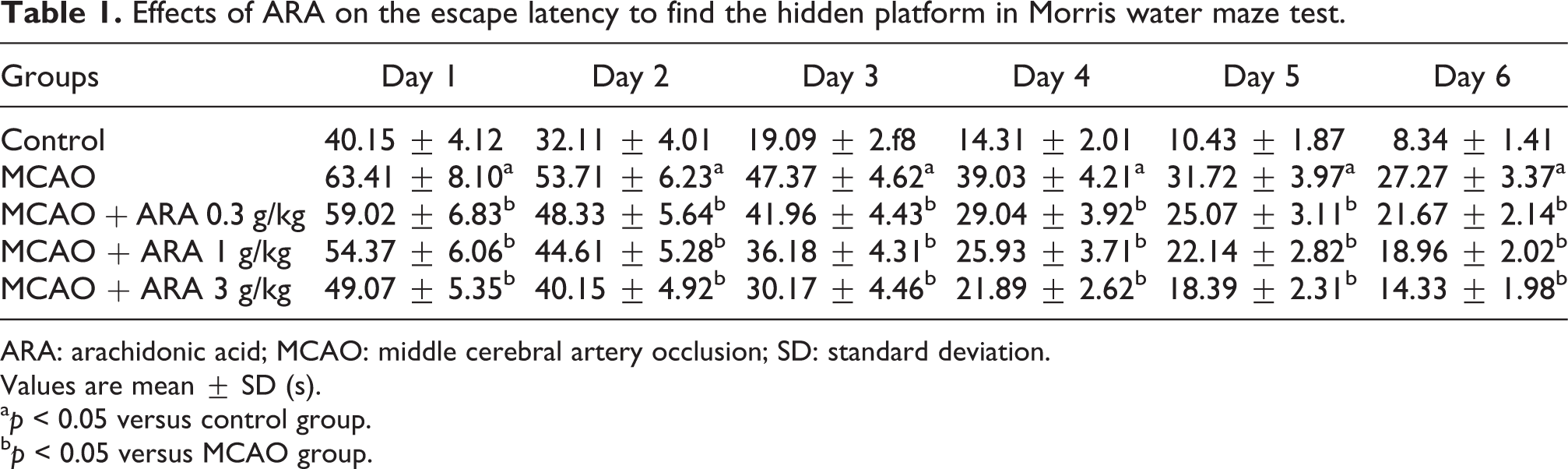
ARA: arachidonic acid; MCAO: middle cerebral artery occlusion; SD: standard deviation.
Values are mean ± SD (s).
a p < 0.05 versus control group.
b p < 0.05 versus MCAO group.
Effects of ARA on histopathology and cerebral infarction
To evaluate the effects of ARA on cerebral ischemia/reperfusion injury, histological examination in each group was performed in H&E-stained sections. The control group showed normal neurons and oligodendroglial cells with the characteristic perinuclear clearing. Abnormal changes like prominent intercellular and pericellular edema were observed in MCAO group. In contrast, these abnormalities were recovered in the ARA-treated groups, MCAO + ARA groups showed a mild intercellular and pericellular edema and a mild degree of glial cell infiltration at edge (Figure 1). In addition, cerebral infarction area was calculated in Figure 1.
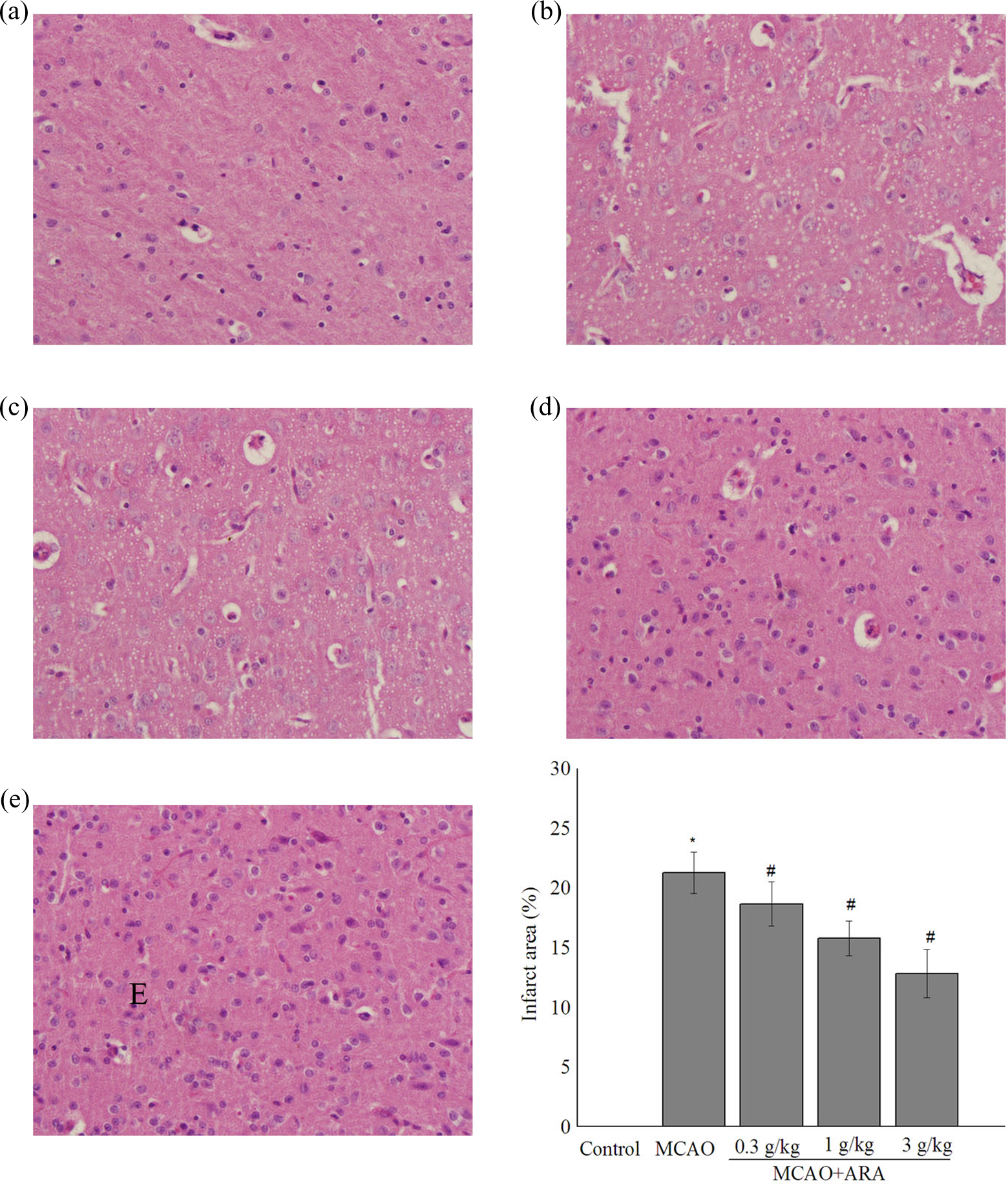
Effects of ARA against MCAO-induced pathological changes and cerebral infarction area. (a) Control group, (b) MCAO group, (c) MCAO + ARA 0.3 g/kg group, (d) MCAO + ARA 1 g/kg group, and (e) MCAO + ARA 3 g/kg group. *p < 0.05 versus control group and # p < 0.05 versus MCAO group. ARA: arachidonic acid; MCAO: middle cerebral artery occlusion.
Effect of ARA on cerebral inflammatory parameters
The levels of TNF-α and IL-6 in brain tissues of each experimental group were summarized in Figure 2. TNF-α and IL-6 levels were significantly increased in MCAO group as compared to control (from 0.18 ± 0.02 ng/mg to 0.32 ± 0.01 ng/mg for TNF-α and from 0.28 ± 0.02 ng/mg to 0.71 ± 0.05 ng/mg for SOD, both p < 0.05). In ARA-treated groups, TNF-α and IL-6 levels were significantly reduced.

Effects of ARA on the levels of (a) TNF-α and (b) IL-6 in brain tissues. Data are shown as mean ± SD. *p < 0.05 versus control group, # p < 0.05 versus MCAO group. ARA: arachidonic acid; MCAO: middle cerebral artery occlusion; TNF-α: tumor necrosis factor-alpha; IL-6: interleukin-6; SD: standard deviation.
Effect of ARA on cerebral oxidative stress parameters
As shown in Figure 3, the MDA levels increased significantly and SOD and GSH-Px levels decreased significantly in MCAO group as compared to the control group (from 188.21 ± 16.85 nmol/g to 640.42 ± 43.11 nmol/g for MDA, from 7.16 ± 0.32 U/mg to 4.13 ± 0.18 U/mg for SOD, and from 22.37 ± 2.02 U/mg to 7.26 ± 0.84 U/mg for GSH-Px, p < 0.05). Treatment with ARA increased the levels of SOD and GSH-Px, simultaneously decreasing the level of MDA (p < 0.05).
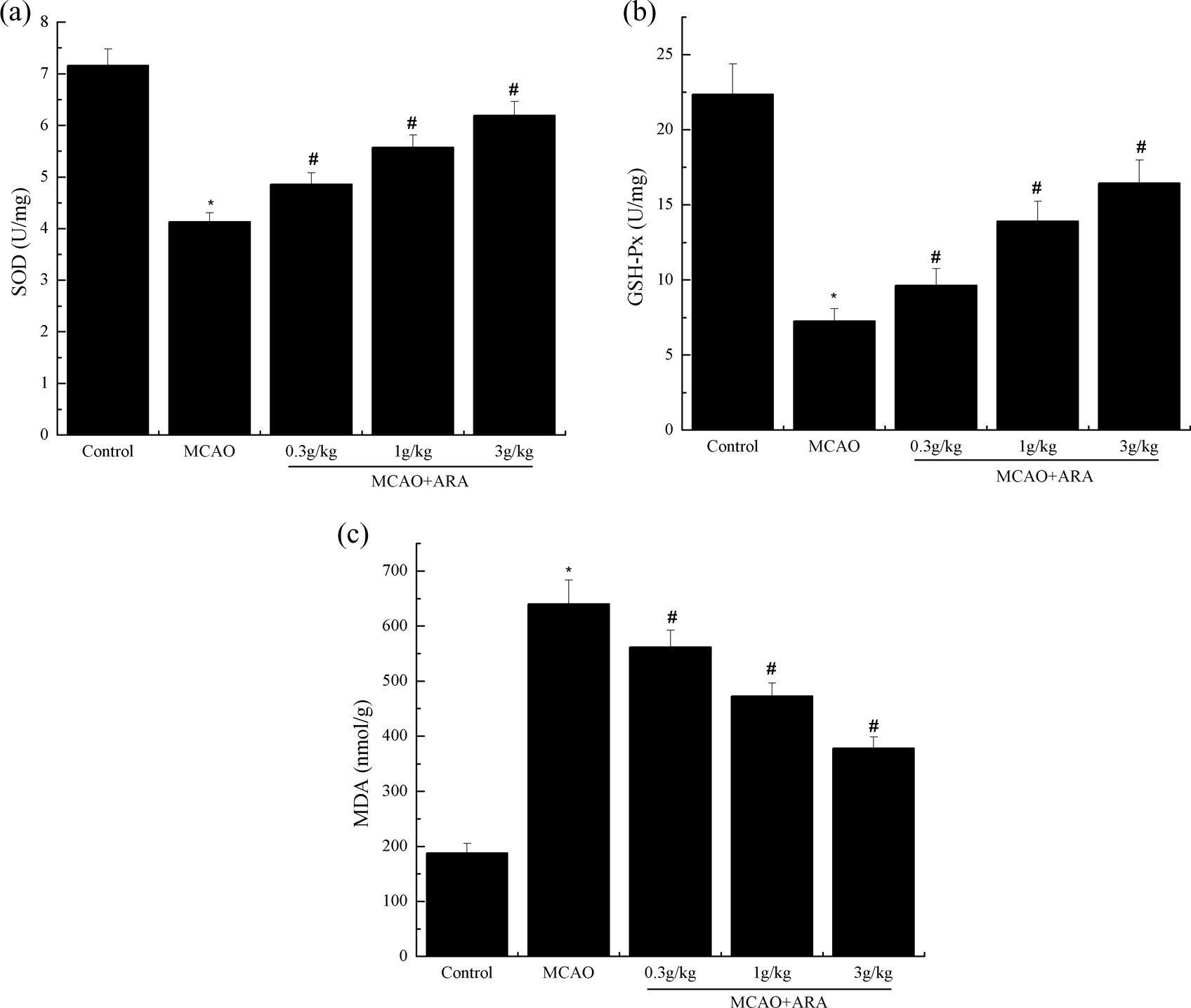
Effects of ARA on the levels of (a) SOD, (b) GSH-Px, and (c) MDA in brain tissues. Data are shown as mean ± SD. *p < 0.05 versus control group, # p < 0.05 versus MCAO group. ARA: arachidonic acid; SOD: superoxide dismutase, GSH-Px: glutathione peroxidase; MDA: malondialdehyde; MCAO: middle cerebral artery occlusion; SD: standard deviation.
Discussion
In the present study, we demonstrate that ARA could protect cerebral ischemia/reperfusion injury in rats. Administration of ARA markedly ameliorated MCAO, causing spatial learning and memory impairment, histopathological changes, deteriorated inflammation, and oxygenation. These findings indicated that ARA had a protective effect of MCAO-induced cerebral ischemia/reperfusion injury by inhibition of inflammation and oxidative stress.
ARA, which is a main constituent of biomembranes, is one of the major components of hippocampus and has strong positive effects on brain function such as synaptic plasticity, membrane fluidity, and neurogenesis. 14,15 Brain ARA level is a well-characterized form of synaptic plasticity and a strong candidate for a cellular mechanism of learning and memory and decreases with increasing age. 19 Both animal experiment and clinical experiment indicated that ARA plays a critical role in maintaining brain function. 20,21
Inflammatory reaction plays an important role in ischemia/reperfusion injury. 22 Proinflammatory cytokines, such as TNF-α, IL-1β, and IL-6, are the potential cytokines that initiate inflammatory reactions and induce the expression of other cytokines after ischemia/reperfusion injury. 23,24 In order to determine the relationship between ischemia/reperfusion injury and inflammatory reaction, our study determined the levels of TNF-α and IL-6 in ischemia/reperfusion rats. We noticed that the TNF-α and IL-6 levels were significantly increased in ischemia/reperfusion rats. In contrast, the levels of TNF-α and IL-6 were significantly decreased in ARA-treated ischemia/reperfusion rats. Those results indicate that ARA may be having cerebroprotective action through anti-inflammatory effect.
Lipid peroxidation release free radicals and the end product of which is MDA, which reflects the damage caused by reactive oxygen species. 25 SOD and GSH-Px are thought to be two important endogenous antioxidative enzymes that provide cellular protection against the apoptosis caused by oxygen-derived free radicals. 26,27 The present study determined marks of oxidative stress (MDA) and antioxidant defense (SOD and GSH-Px) in ischemia/reperfusion rats. Our result showed that with remarkable reduction in SOD and GSH-Px, the levels of MDA in ischemia/reperfusion rats were significantly increased. In contrast, SOD and GSH-Px levels increased significantly and MDA levels decreased significantly in ARA-treated ischemia/reperfusion rats. These results show that ARA strengthened the oxidative defense mechanisms and reduced lipid peroxidation. Therefore, we suggest that ARA has cerebroprotective action against cerebral ischemia/reperfusion injury through antioxidative mechanism.
In conclusion, our data indicate that ARA has protective effects against cerebral ischemia/reperfusion injury in rats, and the cerebroprotective effects may be mediated by antioxidant and anti-inflammatory mechanisms. The findings further support the possible use of ARA as a therapeutic agent to ameliorate ischemic stroke.
Footnotes
Author contribution
Yun Qu and Hui-Long Zhang contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
