Abstract
Tobacco use may initiate the process of oral carcinogenesis with clinically undetectable changes. Smoking cessation may prevent its progression. The objective of this study was to evaluate the association between DNA ploidy and micronucleus (MN) frequency in chronic smokers. Three groups were evaluated: Smoker Group, Former Smoker Group and Control Group. Exfoliative cytology was performed on the lateral border of the tongue and mouth floor. MN and DNA ploidy analyses were performed, as well as the correlation between the variables. The data showed a difference between the groups for the total MN (p = 0.0227), and the Smoker group had the highest mean (4.22 ± 4.12). The three groups did not differ statistically from each other on ploidy evaluation (p-value > 0.05). There was also an association between aneuploidy and increased MN frequency in the Former Smoker group (p = 0.0036). In conclusion, these results point out that there is a relationship between the frequency of MN and aneuploidy in former smokers. Moreover, smoking cessation, even for a short period of time, may promote the decrease of MN frequency caused by tobacco use.
Introduction
The tobacco epidemic is one of the greatest public health concerns the world has ever faced. 1 An estimated 1.3 billion people worldwide use tobacco products, 80% of whom are in low- and middle-income countries. 2
Biomonitoring using exfoliative cytology can identify the initial changes in tissues, caused by the initiation of oral carcinogenesis leaded by the tobacco consumption, when these are clinically undetectable. 3 Thus, the presence of aneuploidy and the investigation of micronuclei (MN) are excellent biomarkers of genotoxicity promoted by tobacco and alcohol use, and additionally, they are also simple, non-invasive, and reliable studies. 4
The micronuclei (MN) are extranuclear cytoplasmic bodies, formed from fragments of chromosomes or whole chromosomes, which were excluded in the anaphase during nuclear division, and which are induced by various genotoxic agents. 5 Also, it has been demonstrated in carcinogenesis that accumulated genetic and epigenetic alterations can lead to fragility of the chromosomes, either in the form of numerical or structural rearrangement, and can be demonstrated as a DNA of abnormal or aneuploid content. 6,7
The DNA of normal cells is diploid, since aneuploidy means a larger or smaller number of chromosomes generated by their instability. Aneuploidy can occur for a variety of reasons, including defects in the cell cycle and in response to DNA damage. Thus, the abnormality of DNA ploidy is considered a marker of alterations in the cellular genome, and results in a worse prognosis for carcinomas. 8,9,10
In view of the above, the objective of the present study was to evaluate the association between DNA ploidy and the frequency of MN in chronic smokers; moreover, whether this association can be modified by cessation of smoking. The authors’ hypothesis is that chronic smokers have greater MN formation frequency, and that smoking cessation impacts genotoxicity caused by tobacco use.
Methodology
Characteristics of the sample and exfoliative cytology
Seventy patients were assigned into groups as follows: a) Smoker Group: 18 chronic smokers, with consumption equivalent to or greater than 10 cigarettes/day for more than 10 years, attending the Outpatient Smoking Cessation Program of the Heart Institute (Incor), Faculty of Medicine, University of São Paulo (FMUSP), coordinated by Dr. Jaqueline Scholz; b) Former Smoker Group: 25 individuals undergoing smoking cessation treatment with abstinence for at least 1 year and a maximum of 2 years, with sex and age equivalent to smoker group, attending the Outpatient Smoking Cessation Program of the Heart Institute (Incor), Faculty of Medicine, University of São Paulo (FMUSP), coordinated by Dr. Jaqueline Scholz; c) Control Group: 27 nonsmokers, attending the outpatient clinic of the Department of Biosciences and Diagnostics of the Institute of Science and Technology (ICT), UNESP, São José dos Campos, São Paulo, coordinated by Dr. Janete Dias Almeida.
Inclusion criteria for all groups were the absence of a history of oral malignancy, absence of any visible clinical signs of change in the buccal mucosa, and non-use of other forms of tobacco consumption, illicit drugs, or alcohol abuse. 3 The sample size was defined based on previous research and MN test literature. 3,4
Exfoliative cytology was performed from the lateral border of the tongue and mouth floor of patients, without previous use of mouthwashes, according to Campos Fontes, 11 and then they were washed and fixed by alcoholic spray and then stained by the Feulgen method.
The Feulgen method consists in washing the smears with alcohol 95% after the initial fixation, and then transferring the samples directly to hydrochloric acid (HCl) 5 N at room temperature for 5 mins. The material was transferred and incubated in Schiff reagent for 90 mins. at a temperature of 4°C. Then, three consecutive washes were performed in distilled water. After immersion, they were washed three times with absolute alcohol. The slides were clarified with xylol and assembled with Entellan® (Merck, Germany) and coverslips.
Tobacco consumption
Tobacco consumption variables analyzed were: number of cigarettes smoked per day, duration of consumption, smoke load, carbon monoxide (CO) expired concentration, and the Fagerström Test for Cigarette Dependence (FTCD). It is important to emphasize that the former smokers’ data was obtained from the files, and it refers to tobacco consumed before smoking cessation, except for CO expired concentration, which was analyzed at sample collection (duration of abstinence).
For the smoker and former smoker groups, the cigarette consumption assessment was supplemented with CO concentration data of exhaled air, using piCO + Smokerlyzer® equipment (Bedfont Scientific Ltd, UK). 12 According to the parameters provided by the manufacturer, abstinent individuals present a CO concentration < 6 parts per million (Smokerlyzer, 2017). Therefore, CO determination is also performed to verify the accuracy of information regarding the smoking cessation rate of participants in the former smoker group.
The evaluation of nicotine dependence was performed using the FTCD. 13
DNA ploidy analysis
The ploidy analysis followed the parameters described by Lima et al., 3 in the Automated Cellular Imaging System (ACIS III, DAKO, Glostrup, Denmark). This equipment converts the image of the selected cell nuclei into pixels and quantifies it in the form of an integrated optical density (IOD) value. With IOD values, the evaluated case can be classified as diploid (DI = 0.9–1.1), moderate aneuploidy (DI = 1.1–1.3), severe aneuploidy (DI > 1,8) and tetraploid (DI between 1.8 and 2.2). To perform the cell counting, an area of interest was chosen, composed of the highest cellularity, without overlapping or clustered cells. A total of 100 peripheral blood lymphocytes were used as control and 300 epithelial cells were selected individually for each sample.
MN evaluation
The slides were examined under light microscopy using an optical microscope, initially with 40x magnification, and for confirmation of the micronuclei, magnification was increased to 100x. The analysis included only the cells with intact nuclei, with a smooth and distinct nuclear perimeter, and that had defined cytoplasm. For counting of the MN, the criterion used was the presence of a surrounding homogeneous halo, representative of a membrane, less than 1/3 of the diameter of the associated nucleus, intensity of Feulgen staining similar to the nucleus, and focal plane under microscopy, 14 with no connection to the core (Figure 1).
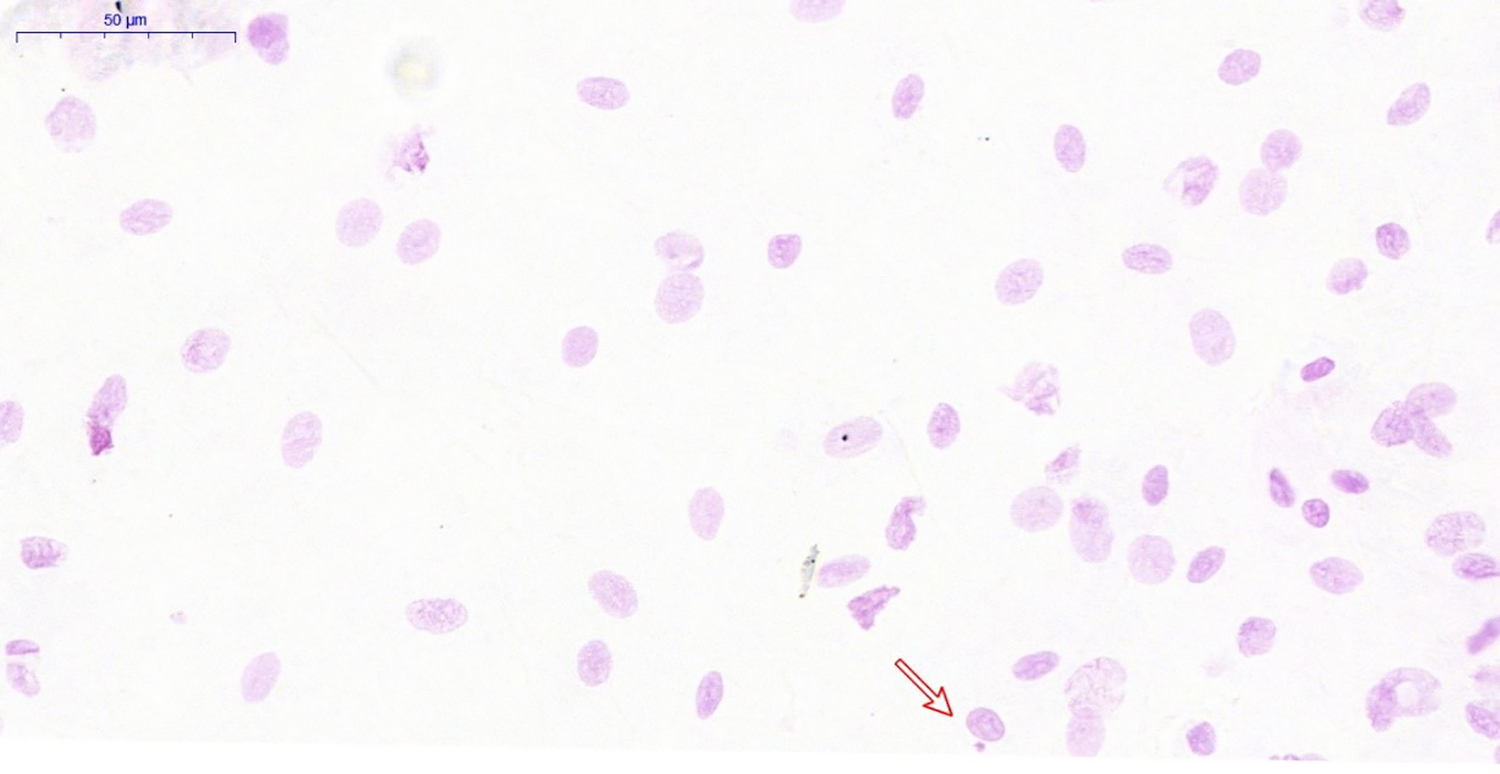
Slide examination. Red arrow points to a micronucleated cell. 40× magnification.
Data analysis
Minitab and Graph Pad Prism 6 statistical software programs were used for the data analysis. Descriptive analysis of the tobacco consumption, MNs and ploidy were by mean and standard deviation calculation. The Mann–Whitney test was used to compare smokers and ex-smokers in data on tobacco consumption, CO and FTCD. The Kruskal–Wallis test was used to evaluate the comparison between the groups on MN and all ploidy variables, complementing with Dunn’s test. Fisher’s test was used for the association between MN and diploid or aneuploid cells, and Spearman’s correlation for the correlation between MN and ploidy. For the non-parametric data, the Mann–Whitney, Kruskal–Wallis, and Dunn tests were performed. A level of significance of 5% was adopted in all the tests.
Results
Profile of the samples
The analysis about sample profiling shows that in the smoker group 10 men and 8 women, 16 men and 9 women in the former smoker group and 12 men and 15 women in the control group were evaluated. Regarding age, the mean in the smoker group was 54.39 ± 8.08 (standard deviation), in the former smoker group it was 59.24 ± 9.45 (standard deviation) and in the control group 54.96 ± 12.50 (standard deviation). The analysis of the educational profile, based on data collected from each participant, showed that 35.7% of the smokers had high education level (post-secondary education such as university or other academic studies—complete or incomplete) and 35.7% had high school level (complete or incomplete). In the former smoker group, 60% of the participants had elementary and middle school level (complete or incomplete), and only 20% had high education level (complete or incomplete). In the Control group, 68.2% had high school level (complete or incomplete).
Descriptive statistics of the tobacco consumption of individuals in the smoker and former smoker groups, using the Mann–Whitney test, are presented in Table 1. A statistically significant difference was observed between the groups only for the concentration of exhaled CO (p < 0.0001).
Descriptive statistics of the tobacco consumption of individuals in the smoker and former smoker groups, according to the study variables.

CO: carbon monoxide; ppm: parts per million; FTCD: Fagerström test for cigarette dependence. Mann–Whitney test; mean ± standard deviation [median]. * Represents significant difference between groups (p < 0.05).
Ploidy evaluation
The DNA ploidy evaluation was done according to the following: Smoker group (n = 18): 15 cases of the mouth floor and 3 of the lateral border of the tongue were analyzed; Former Smoker Group (n = 25): 15 cases of the mouth floor and 10 of the lateral border of the tongue; Control group (n = 27): 12 of the mouth floor and 15 of the lateral border of the tongue.
The mean values of the DNA index (DI), coefficient of variation (CV) and integrated optical density (IOD), as well as the comparison between DI, CV and IOD, performed using the Kruskal–Wallis test (α = 5%), can be seen in Table 2. The three groups do not differ statistically from each other (p > 0.05). The graph on Figure 2 illustrates the comparison of the results between groups.
Comparison between MN frequency and DI, CV and IOD in the smoker, former smoker, and control groups.

MN: micronucleus; DI: DNA index; CV: coefficient of variation; IOD: integrated optical density. Kruskal–Wallis test (α = 5%); *Represents significant difference between groups (p < 0.05).

Graph showing the comparison of MN frequency and DNA ploidy between groups using the analyses of Kruskal–Wallis with Dunn Test as post hoc method for multiple comparisons. H = coefficient of the Kruskal–Wallis test (p < 0.05); A and B represents the homogeneous groups from the Dunn’s test. MN: micronucleus; DI: DNA index; CV: coefficient of variation; IOD: integrated optical density.
MN evaluation
The averages for total MN found and for the total of micronucleated cells, as well as the comparison between the groups performed using the Kruskal–Wallis test (α = 5%), can be seen in Table 2. The test indicated a statistically significant difference (Figure 2).
Correlation between DNA ploidy and MN frequency
The relationship between the variables studied was established by means of the Spearman correlation coefficient (Table 3). Statistically significant correlations were verified only between the variables of the former smoker group. In this group, the moderate correlations between Total MN and DI (p = 0.0065) and IOD (p = 0.0253) were directly proportional. Among the MN and CV total (p = 0.0284), the correlation was inversely proportional.
Correlation matrix between total MN and DNA ploidy for the smoker, former smoker, and control groups.

R: Spearman correlation; MN: micronucleus; DI: DNA index; CV: coefficient of variation; IOD: integrated optical density. Spearman correlation. *Represents significant difference between groups (p < 0.05).
Association between DNA ploidy and MN frequency
Fisher’s exact test (α = 5%) was performed to verify if there is a significant association between the classification of ploidy of the cells and the frequency of MN. The results of this analysis can be seen in Table 4.
Association of ploidy data and MN frequency in smoker, former smoker, and control groups.

MN: micronucleus. The following were adopted: ≤ for less than or equal to the average of the frequency of MN, > for greater than the average frequency of MN. *Represents significant difference between groups (p < 0.05).
A significant association between ploidy and MN frequency was observed only in the former smoker group (p = 0.0036), with diploid cells being associated with the lowest MN frequency, and aneuploidy associated with higher MN frequencies. The same result was found for both the total of micronucleated cells and the total MN (p = 0.0036).
Correlation between the quantitative data of tobacco consumption and age versus MN frequency and DNA ploidy
The degree of correlation between MN, DNA ploidy, data of age and tobacco consumption (cigarettes smoked per day, duration of consumption, smoke load, CO expired concentration, and FTCD) was observed using the Spearman correlation test. The results of these analyses configure the correlation matrix. The results showed: a) Smoker group: no significant correlation between tobacco consumption and age with MN frequency and neither with DNA ploidy; b) Former smoker group: no significant correlation between smoking and age data with MN frequency. Correlation inversely proportional between FTCD and MN frequency (p = 0.035). Significant correlation between the values of FTCD and DI (p = 0.053), CV (p = 0.032) and IOD (p = 0.030). For the CV, this correlation was directly proportional; c) Control group: significant correlation between age and IOD (p = 0.038), which was positive, and directly proportional. The control group was correlated only with age, since these individuals never consumed cigarettes.
Discussion
Studies regarding the injury caused by smoking and the persistence of such damage after cessation of smoking are important considering that chronic smokers are a risk group for oral squamous cell carcinoma (SCC). MNs are of great importance in the identification and quantification of the genotoxic action of carcinogens and mutagens. 7 DNA aneuploidy is frequently found in carcinomas, and it also appears to be associated with lesions with malignant transformation potential. 6 This study was conducted with the opportunity to evaluate the association between the frequency of MN and the presence or absence of aneuploid DNA in smokers and former smokers compared to nonsmokers.
Although controversial, the analysis of the educational profile corroborates studies showing that smokers, in general, have a higher educational level. 14
In the data on tobacco consumption, the Smoker and Former Smoker groups presented similar characteristics, except for the concentration of exhaled CO, which was higher in smokers than in former smokers. This is because CO is one of the by-products of cigarettes that has a short plasma half-life (approximately 1 to 4 hours). 15 Some studies have shown that smoking is one of the main factors responsible for the increase in exhaled CO concentration. 16 Therefore, the more the individual smokes cigarettes, the greater the concentration in the body. Since individuals in the Former Smoker group were abstinent, significantly lower levels of CO were observed compared to the Smoker group, with concentrations below 6 ppm. The calibration of the CO level using the Smokerlyzer® PICO device was adopted as a method for easy application and low cost, regardless of sample variability. 17
The abnormality of DNA ploidy is considered a marker of changes in the cellular genome and results in a worse prognosis for carcinomas. 7,8 In the present study, no difference was observed in the degree of DNA ploidy between the groups. Other studies also did not find a direct association between smoking and the presence of aneuploidy. 5,9,18 This suggests that aneuploidy may be related to age and genomic instability, since a high incidence of DNA aneuploidy has already been observed in young patients with SCC in the tongue, with a higher prevalence in nonsmokers. 9 On the other hand, some studies have shown that smoking causes an early appearance of DNA ploidy and that it may worsen the prognosis for smoking patients with SCC. 19 Another study confirmed that the survival of patients with gastric cancer was higher in low ploidy groups, while in the high ploidy groups this survival decreased. Another point stated in the same study was that cancers with a higher degree of aneuploidy manifest a higher incidence of vessel invasion and lymph node metastasis. 20
Regarding MN evaluation, a statistically significant difference was observed between the groups, both for the total number of MN and for the total number of micronucleated cells; smoking being associated with higher MN frequencies. These findings are biologically coherent and are compatible with the literature, in which some authors also verified in their respective studies a direct relationship between the frequency of MN and tobacco consumption. 21,22
Like Namala et al., in the present study we verified that cells of the control group occasionally presented MN, but MN frequency was statistically higher in the smoker group, confirming that tobacco consumption increases MN frequency. 5 This represents the cytogenetic damage caused by smoking as being quite significant. On the other hand, results of the former smoker group show that smoking cessation may promote the reversibility of genotoxicity.
MNs are derived from chromosomal instability during cell division, and are related to the initiation process of carcinogenesis, suggesting recent DNA damage, 13 while changes in DNA ploidy are related to later stages in the progression of carcinogenesis. 3 In the smoker and control groups no correlation was observed between DNA ploidy and MN frequency in this study. Such correlation may not have occurred in most groups, since the formation of MNs predates the stages in which changes in DNA content occur. However, damage to DNA content may not yet have occurred in the micronucleated cells evaluated in this study.
Although DNA ploidy and MN frequency did not correlate in the smoker and control groups of this study, there was a directly proportional correlation between the DI and IOD variables and MN frequency in the former smoker group. In addition, diploid cells were associated with a lower frequency of MN, whereas aneuploid cells were associated with higher MN frequencies only in the former smoker group. This finding may be related to the degree of genotoxicity damage of the studied cells, since both MN and DNA ploidy are markers of cytogenetic alterations. Therefore, a cell of normal content (diploid) is less prone to form MN during its nuclear division.
Lima et al. confirmed a higher rate of aneuploidy in former smokers compared to smoking patients, which appears to generate controversy. 3 As DNA content analysis is done by converting the cell nucleus image into pixels, degeneration in the cells found in the most superficial layer of the epithelium 23 may interfere with this quantification. Ma et al. suggest that the presence of inflammatory infiltrate, the overlap of cells in the smears, and the presence of necrotic cells may also produce false-positive or false-negative results. 24 Smoking cessation promoted a decrease in MN, as observed in the present study. This fact may have promoted the correlation and association only in the former smoker group, since it suggests that the micronucleated cells that persisted after smoking cessation are those that suffered greater damage. This means that they already presented, besides the presence of MN (considered premature lesion of carcinogenesis), alterations in the content of DNA ploidy (aneuploidy). That is to say, the hypothesis is that in the former smoker group, the remaining damaged cells are the ones with the greatest damage, whereas in the smoker group, there are probably micronucleated cells that have not yet undergone changes in their DNA ploidy content, and cells that have already undergone this change, therefore establishing no correlation or association.
Regarding the inversely proportional correlations between ploidy, MN and FTCD (which quantifies the user’s dependence on nicotine), we suggest the hypothesis advocated by some authors 25,26 concerning a certain protective effect of nicotine on the oral mucosa, which explains why the greater the dependence of these individuals, the greater their nicotine intake, the greater the protection and lower genotoxicity.
Chen et al. carried out a study comparing the effects of nicotine-free cigarettes, with low nicotine load and conventional cigarettes and found that nicotine-free cigarettes had a greater inhibitory or retarding effect on cell growth. The study also demonstrated that such an effect is observed due to the action of nicotine on the apoptotic pathways, attenuating this effect and consequently the cytotoxicity. Finally, the authors further noted that cigarettes without nicotine or low alkaloid loading have the same mutagenic potential as conventional cigarettes. 25
Nersesyan et al. demonstrated in their study that the presence of cytogenetic alterations increases considerably with daily exposure to tar and decreases simultaneously with the absorption of nicotine. 26 These findings suggest that nicotine protects cells against carcinogens contained in tobacco smoke, which is in agreement with the fact that we did not find significant values of cytogenetic changes in the smoker group in our study, but we did in the former smoker group. Authors hypothesize that since the former smoker is no longer under the protective effect of nicotine, their cells may be more susceptible to cytogenetic changes due to tobacco substantivity.
However, the directly proportional correlation between age and ploidy in the control group makes biological sense, since the increase in the integrated optical density (IOD) indicates aneuploidy; a fact related to age, as found in the literature. 21,27
The present study used exfoliative cytology to analyze mucosa cells, thus only the intermediate and superficial cells were examined, this being one of the limitations of this technique. We cannot guarantee that the results of the analysis of the basal cells would be the same, considering the degree of keratinization and maturation of superficial cells. Despite the limitations, the work ends with pertinent contributions about former smokers.
In conclusion, these results point out that there is a relationship between the frequency of MN and aneuploidy in former smokers. Moreover, smoking cessation, even for a short period of time (abstinence for at least 1 year and at most 2 years), may promote the decrease of genotoxicity caused by tobacco use.
Footnotes
Authors’ note
The authors declare that part of data from DNA ploidy was published in: J Oral Pathol Med 2017; 46: 961–966.
Acknowledgement
A.C. Camargo Cancer Center for the assignment of the ACIS III equipment (Dako Corp.) for the evaluation of DNA ploidy.
Author contributions
BSC: substantial contributions to acquisition of data and analysis and interpretation of data; drafting the article; LATS: substantial contributions to acquisition of data, and analysis and interpretation of data; revising it critically for important intellectual content; BFDCC: substantial contributions to acquisition of data and analysis of data; revising it critically for important intellectual content; MGOA: substantial contributions to acquisition of data; CFLC: substantial contributions to conception and design; revising it critically for important intellectual content; IB: substantial contributions to analysis and interpretation of data; JRS: substantial contributions to acquisition of data; JDA: substantial contributions to conception and design; revising it critically for important intellectual content; final approval of the version to be published.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The present study was approved by the Human Research Ethics Committee of the São Paulo State University (UNESP), Institute of Science and Technology, São José dos Campos (ICT-UNESP) under protocol nº 1.033.312/2015 PH/CEP. All patients were invited to participate and, those who agreed signed a free informed consent form and underwent intra- and extra-oral clinical examination.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the São Paulo Research Foundation (FAPESP) (grants #2012/05371-4, #2013/09910-0, #2016/24047-4) and the National Council for Scientific and Technological Development (PIBIC – CNPq/UNESP), Brazil ID: 27956.
