Abstract
Cryptococcosis is a neglected fungal disease. The scarcity of studies on oral cryptococcosis is certainly due to rarity and/or underreporting of the disease, especially in Brazil. We describe an example of orofacial cryptococcosis affecting a 57-year-old man after heart transplantation, who presented with multiple erythematous ulcers and erosions distributed in the chin, nasal cavity, labial mucosa, hard palate, and buccal vestibule. Computed tomography revealed opacities and micronodules in the lungs. Histopathological features of the oral and pulmonary lesions were compatible with Cryptococcus spp. Amphotericin B and fluconazole were used for treatment during hospitalization and itraconazole for prolonged therapy after hospital discharge. The patient has been under follow up for 6 months without signs of disease. According to a review conducted in PubMed, Web of Science, Scopus, Embase, and LILACS for data analysis of oral cryptococcosis, 26 reports were described in the literature. Predilection for men was observed (85%), with a male:female ratio of 5.5:1. The mean age of the individuals was 49 ± 15.3 years. Oral cryptococcosis mostly presented as an ulcer (n = 17). The palate and tongue were the most affected sites (n = 9 for each). Amphotericin B was the primary therapy utilized in most patients. Seventeen (65%) individuals survived. Knowledge of the clinicodemographic aspects of oral cryptococcosis is important for clinicians in decision making and surveillance.
Introduction
Cryptococcosis is an infectious disease acquired through the inhalation of spores of Cryptococcus sp. typically found in decaying wood, tree hollows, bird droppings, soil, water, and certain foods.1–3 The 2 most common species of Cryptococcus that cause disease in humans are C. neoformans and C. gattii. 4 This disease is a threat to immunocompromized individuals, especially those who have human immunodeficiency virus/acquired immune deficiency syndrome (HIV/AIDS). Sporadic examples of cryptococcosis have also been documented in healthy individuals.1,3 About 152,000 individuals are diagnosed with HIV-associated cryptococcal meningitis annually worldwide. 5 In the United States, the annual incidence of cryptococcosis among persons with HIV/AIDS is between 2 and 7 individuals per 1000 inhabitants, and the overall incidence is 0.4 to 1.3 individuals per 100,000 inhabitants. 6 Nevertheless, in the United States and Brazil, representative estimates of the incidence of cryptococcosis have been limited because few states have included this condition in their local records of public health-reportable diseases.7–9 Additionally, about 80,000 humans die each year from cryptococcosis, a fact that ranks it as the fifth deadliest infectious disease in absolute numbers of deaths. 5 Despite this, cryptococcosis is among the most poorly funded diseases in the world and is not addressed in the World Health Organization's portfolio of neglected tropical diseases.10,11
Emerging data have highlighted the heterogeneity of cryptococcosis according to serum cryptococcal antigen prevalence in HIV-positive cohorts. 12 For instance, C. neoformans var. grubii (serotype A) is the predominant global cause of HIV-associated cryptococcal meningoencephalitis, but in China this organism often infects apparently immunocompetent hosts. 13 It has been suggested that C. neoformans is responsible for more than 90% of cryptococcosis reports in Latin America—predominantly in people with HIV/AIDS, while C. gattii usually occurs in healthy individuals. 14 Hence, the relative contribution of host and pathogen genotypes underlying these global patterns of disease remains to be defined. 12
Cryptococcosis is a significant opportunistic infection among solid-organ transplant recipients and those on widespread use of immunosuppressive drug therapies.2,15 In about 60% of solid-organ transplant recipients with cryptococcosis, the infection is limited to the lungs, and the transplants are mainly renal, cardiac, and pulmonary. 16 Solid-organ transplant recipients are less likely to have central nervous system involvement or disseminated cryptococcosis. 15 In turn, the oral manifestations of cryptococcosis in solid-organ transplant and hematopoietic stem cell recipients need to be better understood, as only 2 examples of patients with oral involvement have been published in the literature to date.17,18
The diagnosis of cryptococcosis is supported by routine culture techniques, histopathology with the aid of special stains, and serological tests.1,2 In addition, molecular methods for the diagnosis and characterization of Cryptococcus have been employed, such as polymerase chain reactions, Sanger sequencing, multilocus sequence typing, whole genome sequencing, and quantitative trait loci mapping. 19 However, disseminated cryptococcosis involving the oral cavity can be challenging to diagnose, mainly due to its nonspecific clinical features. In this article, we report an example of a heart transplant recipient who presented with disseminated cryptococcosis involving the orofacial region. The clinicodemographic aspects, differential diagnosis, and management were discussed in light of a comprehensive literature review.
Clinical Report
A 57-year-old Brazilian man was admitted to the Emergency Department of Hospital das Clínicas, Universidade Federal de Minas Gerais, Belo Horizonte, Brazil, with a 15-day history of diarrhea, dyspnea, odynophagia, headache, and nasal obstruction. Painful palatal ulcers of unknown duration were also reported. His medical history was significant for heart transplantation due to ischemic heart disease 38 months earlier, in addition to chronic kidney disease, arterial hypertension, and diabetes mellitus, for which he was on daily treatment with tacrolimus (1 mg), mycophenolate mofetil (500 mg), metformin (1 g), clonidine (0.2 mg), atorvastatin (20 mg), omeprazole (20 mg), citalopram (20 mg), acetylsalicylic acid (100 mg), and clopidogrel (75 mg). In his dental history, residual fragments of roots of the upper right canine and first and second premolars had been extracted, in addition to a biopsy of an ulcer on the left palatal region performed 9 days before admission to the hospital (Figure 1A). The microscopic findings were compatible with a fragment of gingival tissue with an unspecified inflammatory process.
Clinical aspects of orofacial cryptococcosis. (A) In the first evaluation, there was generalized gingival swelling, erythema in the apical region of the right upper premolars, in addition to root remnants of the first and second right premolars. A crateriform ulcer covered by a purulent membrane was also observed in the region of the hard palate. (B) Extraoral aspects showing an ulcer on the chin covered by crust and erythema. (C) Deep erythematous and irregular ulcer covered by a purulent membrane in the region of the upper labial mucosa. (D) Appearance after extraction of the first and second right premolars revealing a swollen, nonhealed alveolar region covered by a purulent membrane and sutures in place. Hematoma in the adjacent attached gingiva was also noted. In the extraoral view, an ulcerated lesion covered by a crust was observed on the nasal mucosa. (E) Intraoral appearance after an incisional biopsy in which an erythematous and swollen crateriform ulcer was observed, covered by a purulent membrane in some regions. (F) Erythematous and swollen marginal gingiva extending from the left canine to the first and second premolars. Aspect of the condition in the (G) hard palate and in the (H) marginal gingiva region with spontaneous bleeding.
The first clinical suspicion was COVID-19 due to the aforementioned general status of the individual and the country's scenario, which had high rates of notifications due to new variants; however, the results were negative for SARS-CoV-2. In parallel, the lesions in the palatal region became more exuberant, with a necrotic and infected appearance; thus, piperacillin/tazobactam (4 g/0.5 g for 10 days) and a mouthwash with nystatin (100,000 IU for 2 weeks) were initiated. Despite the negative cytomegalovirus antigenemia test, ganciclovir (200 mg/day) was prescribed for 12 days due to the individual's immunosuppression status. Since the individual developed thrombocytopenia and the cytomegalovirus polymerase chain reaction was negative, therapy with ganciclovir was discontinued. An in-depth workup was then performed to determine whether the orofacial manifestations reflected a spread of an infectious disease, as detailed in Table 1.
Workup Performed at Hospital Admission and During Hospitalization of the Patient With Cryptococcosis.
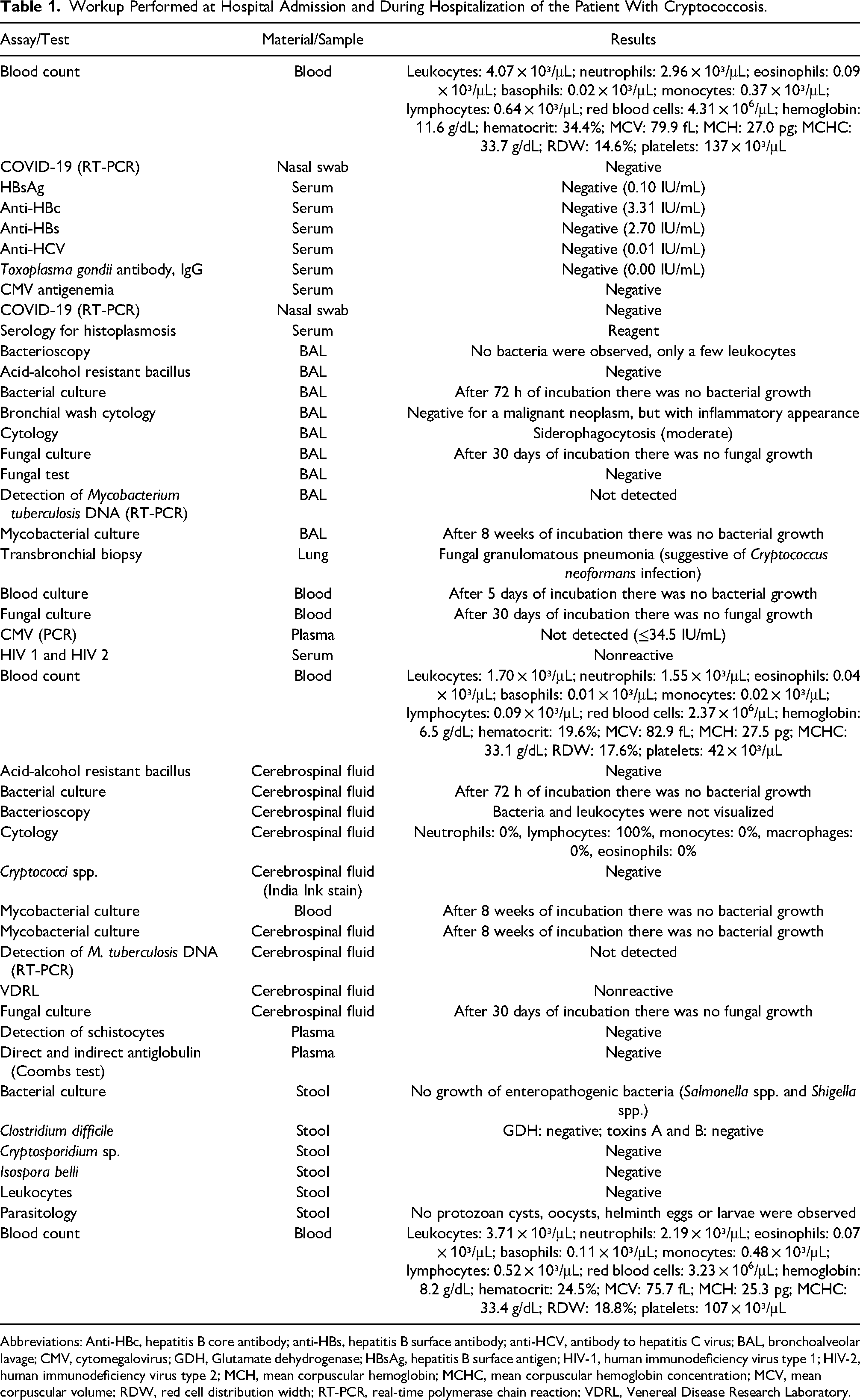
Abbreviations: Anti-HBc, hepatitis B core antibody; anti-HBs, hepatitis B surface antibody; anti-HCV, antibody to hepatitis C virus; BAL, bronchoalveolar lavage; CMV, cytomegalovirus; GDH, Glutamate dehydrogenase; HBsAg, hepatitis B surface antigen; HIV-1, human immunodeficiency virus type 1; HIV-2, human immunodeficiency virus type 2; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; MCV, mean corpuscular volume; RDW, red cell distribution width; RT-PCR, real-time polymerase chain reaction; VDRL, Venereal Disease Research Laboratory.
On the third day after initial presentation, our dental team requested evaluation of the individual's oral cavity. Extraoral examination revealed the absence of submandibular or cervical lymphadenopathy. However, a crusted ulcer with a well-defined erythematous base located in the chin measuring approximately 1.0 × 1.0 cm (Figure 1B) and ecchymoses and crusts in the right nasal cavity were observed. Intraoral examination revealed generalized poor oral hygiene. An ulcerated lesion with irregular borders and covered by exudate in the upper labial mucosa was also observed. It measured 1.5 × 1.0 cm and was painful on palpation (Figure 1C). In addition, red-purple and enlarged buccal gingiva was detected, with multiple shallow ulcerations and erosions extending from the hard palate to the buccal vestibule. Sutures and fibrinopurulent tissues were observed at the site of previous extractions. Mild discomfort on palpation and recurrent spontaneous bleeding were reported (Figure 1D-H).
Given the clinical appearance of the manifestations and the medical history of this patient, the differential diagnoses included orofacial ulcerative processes that are more likely to occur in immunocompromised individuals. These conditions can be broadly categorized as having infectious, inflammatory/autoimmune, and neoplastic etiologies. Regarding infectious diseases, fungal, bacterial, protozoal, and viral processes were the main ones, including histoplasmosis, aspergillosis, acquired oral syphilis, atypical/typical mycobacteria infection, mucocutaneous leishmaniasis, herpes simplex virus, and herpes zoster, respectively. Sarcoidosis and granulomatosis with polyangiitis (Wegener granulomatosis) were also listed in the context of inflammatory/autoimmune conditions. Malignant neoplasms such as leukemic/lymphoid infiltration, Kaposi sarcoma, and post-transplant lymphoproliferative disorder—a malignant process best classified as a heterogeneous group of diseases ranging from an indolent polyclonal lymphocytic proliferation to aggressive non-Hodgkin lymphomas—were also considered.
An incisional biopsy of the ulcerations on the palatal gingiva of the premolars was performed under local anesthesia. Microscopically, the tissue fragment revealed a granulomatous inflammation composed of a lymphocytic infiltrate and scarce areas of necrosis. Mitotic figures and angiotropism were not observed (Figure 2A-B). Grocott-Gomori methenamine silver staining revealed yeasts with round-to-ovoid structures, measuring approximately 5 to 10 µm in diameter, surrounded by a clear halo representing the capsule. These characteristics were compatible with Cryptococcus spp. (Figure 2C).
Histopathological features of oral cryptococcosis. (A) Oral mucosa lined with stratified squamous epithelium, hyperparakeratinized and with areas of acanthosis. An intense predominantly mononuclear inflammatory infiltrate was observed in the lamina propria (hematoxylin and eosin, original magnification 10×). (B) In the detail, round structures are observed (hematoxylin and eosin, original magnification 40×). (C) Grocott-Gomori silver methenamine staining confirms the presence of a fungal structure, measuring approximately 5 to 10 µm in diameter, compatible with Cryptococcus spp. (original magnification 100×).
Computed tomography of the chest disclosed multiple irregular reticulonodular opacities of predominantly central and symmetrical distribution, notably in the upper lung lobes (Figure 3). A transbronchial biopsy revealed a lymphocytic infiltrate with areas of necrosis and deposition of hemosiderin material, and hyperplastic epithelioid cells containing round or oval diaphanous structures (Figure 4A). These Grocott-Gomori methenamine silver-stained structures showed a brown-black color and were morphologically characterized by encapsulated structures of globular or ovoid shape, 5 to 10 µm in diameter, surrounded by a clear halo consistent with Cryptococcus spp. (Figure 4B).
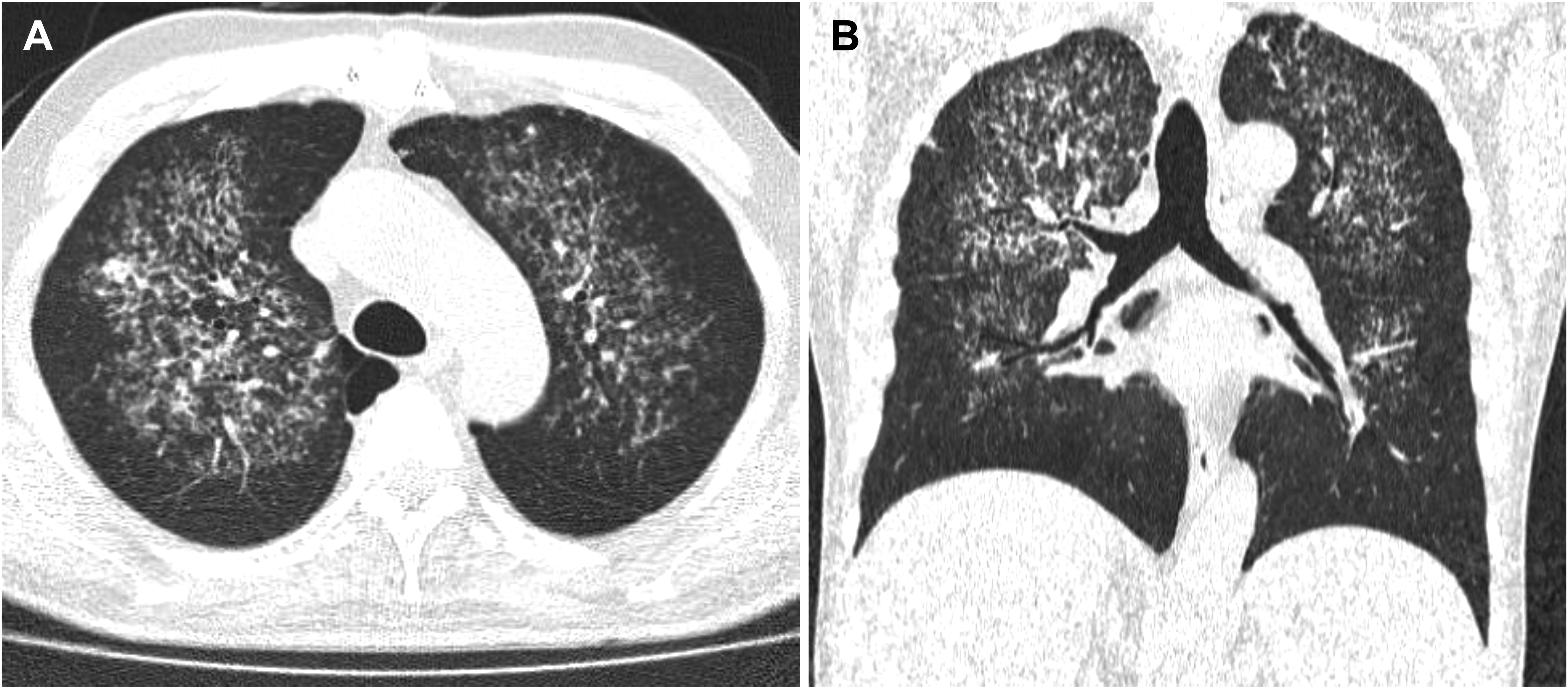
Computed tomography of the chest. (A) Axial and (B) coronal views reveal multiple reticulonodular opacities and irregular micronodules, centrally and symmetrically distributed in the upper lung lobes.
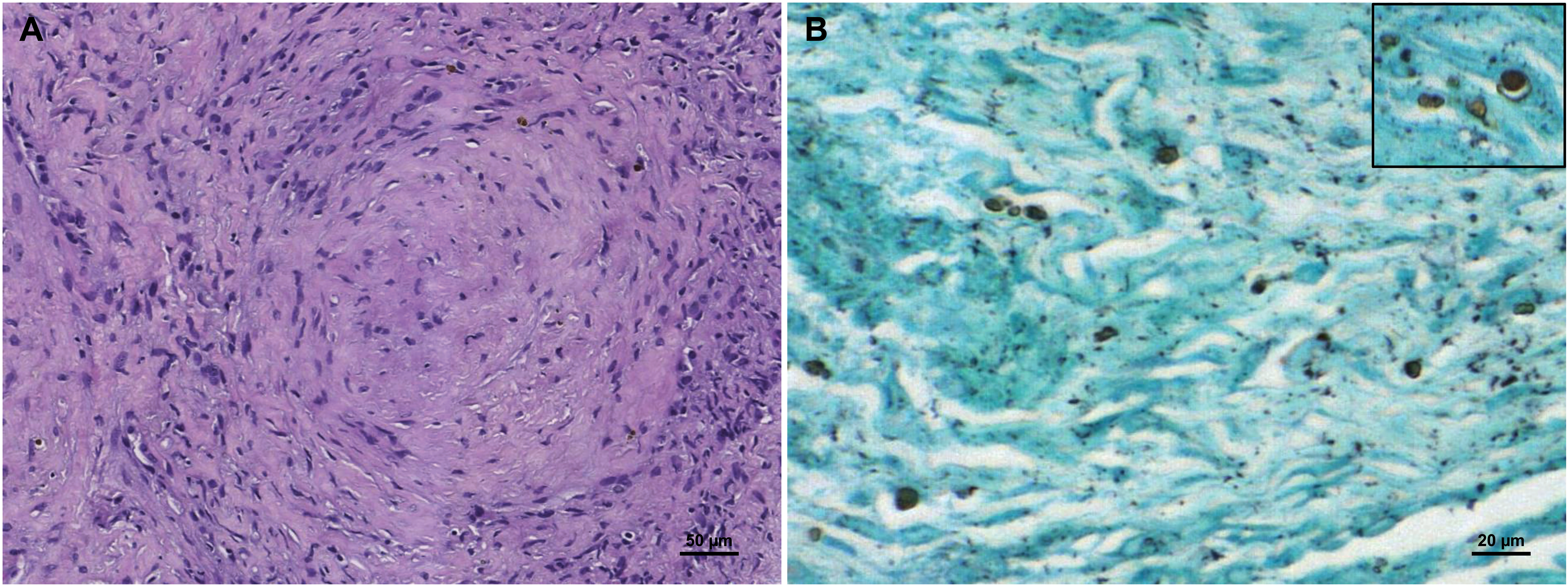
Histopathological findings of pulmonary cryptococcosis. (A) Lymphocytic infiltrate with areas of necrosis and deposition of hemosiderin material, hyperplastic epithelioid cells containing round or oval diaphanous structures (hematoxylin and eosin, original magnification 10×]. (B) Special staining revealed rounded structures brown-black in color. In the inner box, it is possible to observe morphological characteristics compatible with Cryptococcus spp. such as encapsulated structures of globular shape, surrounded by a clear halo, measuring from 5 to 10 µm in diameter (Grocott-Gomori silver methenamine, original magnification 20×).
The individual was instructed to perform oral hygiene with a 0.12% chlorhexidine digluconate mouthwash (3 times a day) for 14 days. As multiple sites of gingival bleeding were observed, topical tranexamic acid was introduced and acetylsalicylic acid was discontinued. Amphotericin B lipid complex (5 mg) plus fluconazole (800 mg) was started, but a rapid development of an adverse reaction to this therapeutic regimen was noted. Drug administration was replaced with a reduction in infusion rate and, 1 h before the procedure, analgesics (dipyrone or paracetamol), diphenhydramine and pethidine were recommended for 2 weeks. At this time, erythema associated with ocular hemorrhage was observed and involvement of the central nervous system was hypothesized, but a cerebrospinal fluid smear was negative by India ink staining. Peaks of fever and neutropenia were observed, and granulokine® (300 mu/mL) was prescribed. Due to persistent diarrhea, meropenem (2 g) and metronidazole (2 g) were also established for 7 and 10 days, respectively.
On the 36th day of hospitalization, the individual was discharged with prescribed use of itraconazole (200 mg) twice daily for 3 months, followed by once daily for 12 months. After 6 months of follow up, complete healing of the oral manifestations was observed (Figure 5).
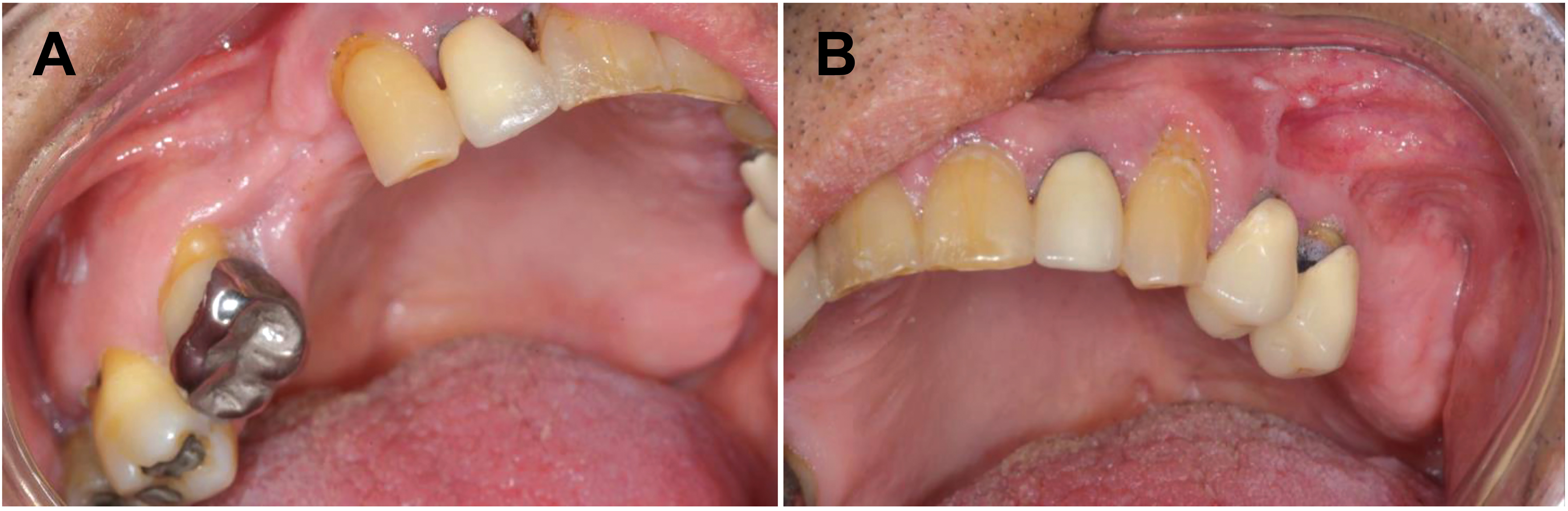
Six months of follow up. Clinical healing and absence of signs of recurrence in the hard palate and alveolar region.
Literature Review
Electronic searches were performed in August 2022 to identify articles on oral cryptococcosis, without time or language restrictions, in PubMed, Web of Science, Scopus, Embase, and Latin American and Caribbean Health Sciences Literature (LILACS). The following combination of terms was employed: (cryptococcosis OR cryptococcus OR cryptococcal) AND (oral cavity OR mouth OR oral mucosa OR oral mucosal OR buccal mucosa OR buccal mucosal OR floor of the mouth OR lip OR lips OR tongue OR alveolar process OR alveolar ridge OR gingiva OR palate OR hard palate OR soft palate OR jaw OR jaws OR mandible OR maxilla). A search update was performed in December 2022. Abstracts of all articles identified through the searches were independently examined by 2 authors (ACVPS and JAAA).
Eligibility criteria were studies in which complete clinicopathological data on oral cryptococcosis were available. For studies that appeared to meet the inclusion criteria or for those with insufficient data in the abstract, the full report was obtained to permit a clear decision. Exclusion criteria were laboratory studies, letters to the editor, and expert opinions, unless any of these types of publications provided sufficient data of interest. The following data were retrieved from each of the studies identified: author(s), year of publication, country, patient sex and age, anatomical location, clinical aspects, symptoms and time of evolution of the condition, extraoral manifestations, systemic characteristics, associated conditions, diagnostic rendering, treatment, and outcome/follow-up period. Disagreements were resolved by discussion between the authors. A third author (TAS) verified the data extraction. Data were descriptively analyzed using Microsoft Office Excel 2019 (Microsoft® software, Redmond, WA, USA).
The electronic searches yielded 808 articles (PubMed: 168, Web of Science: 154, Scopus: 330, Embase: 125, and LILACS: 31). Twenty-six studies comprising 26 affected individuals met the eligibility criteria and were analyzed.17,18,20–43 The articles were from the United States,17,20–26,28,32–36 Peru, 29 Brazil, 30 Colombia, 38 Trinidad and Tobago, 41 Argentina, 43 Germany, 27 The Netherlands, 18 Malaysia, 31 Japan,37,40 Indonesia, 39 and Australia 42 (Table 2).
Clinicopathological Data, Management and Outcomes of Reports of Oral Cryptococcosis Published in PubMed, Web of Science, Scopus, Embase, and LILACS.
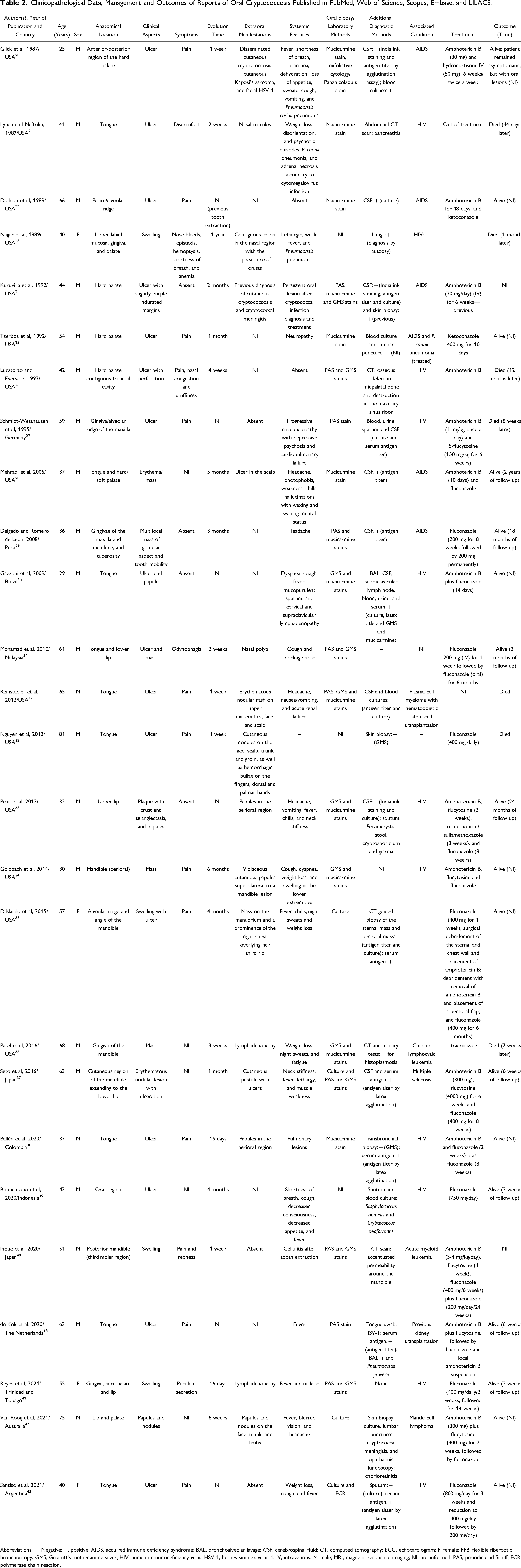
Abbreviations: −, Negative; +, positive; AIDS, acquired immune deficiency syndrome; BAL, bronchoalveolar lavage; CSF, cerebrospinal fluid; CT, computed tomography; ECG, echocardiogram; F, female; FFB, flexible fiberoptic bronchoscopy; GMS, Grocott's methenamine silver; HIV, human immunodeficiency virus; HSV-1, herpes simplex virus-1; IV, intravenous; M, male; MRI, magnetic resonance imaging; NI, not informed; PAS, periodic acid-Schiff; PCR, polymerase chain reaction.
The mean age of affected participants was 49 (±15.3) years, ranging from 25 to 81 years, with a peak in the fourth and fifth (n = 6/23%, each) and seventh (n = 6/23%) decades of life. Men were the most affected (n = 22/85%). The palate (n = 9) and tongue (n = 9) were the most common sites. Ulcers (n = 17), followed by swelling/mass (n = 9), papule (n = 3), erythema (n = 2), plaque (n = 1), and nodule (n = 1) were the clinical findings. In 8 individuals, more than 1 concomitant oral manifestation was reported. Pain/discomfort was reported in 14 patients. The evolution of the lesions ranged from 1 to 48 weeks (median: 4 weeks). Special stains were used to identify Cryptococcus, such as Grocott-Gomori methenamine silver (n = 13), mucicarmine (n = 13), and periodic acid-Schiff (n = 10). Confirmation of Cryptococcus by molecular analysis was reported in 1 study. 43
Fever (n = 11) and cough (n = 6) were the main general features described. Sixteen patients were positive for HIV/AIDS. Other associated conditions were chronic lymphocytic leukemia (n = 1), acute myeloid leukemia (n = 1), mantle cell lymphoma (n = 1), plasma cell myeloma (n = 1), and multiple sclerosis (n = 1). Two patients had a history of renal (n = 1) and hematopoietic stem cell (n = 1) transplantation. Cryptococcus was positive in cerebrospinal fluid samples (n = 9), serum antigens (n = 5), blood cultures (n = 3), and skin biopsies (n = 3). Serological tests with latex agglutination (n = 4) were used in serum and/or cerebrospinal fluid samples.30,37,38,43
Regarding treatment, amphotericin B was used in 15 patients, associated with fluconazole (n = 10), flucytosine (n = 7), and ketoconazole (n = 1). Also, fluconazole (n = 6), itraconazole (n = 1), and ketoconazole (n = 1) were used as monotherapy. Seventeen individuals remained alive, 7 died, and 2 articles did not provide information on outcome.
Discussion
Data on the oral manifestations of cryptococcosis are sparse in the literature, with 26 reports documented hitherto.17,18,20–43 Men were affected in almost 85%, with a peak incidence in the fourth, fifth, and seventh decades of life. It is known that individuals vulnerable to cryptococcosis are those with advanced HIV/AIDS, either because they are unaware of their serological status or because they are not on adequate antiretroviral therapy. 44 A previous systematic review reported that cryptococcal disease is responsible for 19% of AIDS-related deaths worldwide. 44 Furthermore, cryptococcosis is the third most common invasive fungal infection in solid-organ transplant recipients. 2 In line with the present report, cardiac transplant recipients with cryptococcosis are mostly middle-aged and older adult males using tacrolimus as immunosuppressive therapy. 45 Evidence indicates that the period of infection usually occurs between 16 and 21 months after solid-organ transplant. 2 Although in our patient the infection occurred 36 months after heart transplantation, the occurrence of oral cryptococcosis has been documented at 11 and 264 months after hematopoietic stem cells and kidney transplants, respectively.17,18
Cryptococcosis primarily presents with pulmonary and neurological systems.1,3 Although the infection begins in the lungs, cryptococcal meningitis is the most common form of disease presentation, 46 representing about 98% of those infected in Brazil. 8 Despite this, it is worth mentioning that it was only since 2020 that the notification of patients with cryptococcosis became mandatory in the Brazilian System of Reportable Diseases (SINAN). 47 Individuals with HIV show greater involvement of the central nervous system (68%) when compared to non-HIV nontransplanted (39%), and transplant recipients (32%); in turn, the last 2 groups have more frequent localized pulmonary infection. 48 In instances of neurological involvement, symptoms such as headache, altered mental status, and fever are more common, whereas in pulmonary involvement, fever, chills, cough, night sweats, and dyspnea are reported.2,8 Our review identified central nervous system involvement in 9 individuals with oral manifestations.17,20,22,24,28–30,33,37
Although the cerebrospinal fluid sample from the present patient was negative for cryptococcal infection, ocular erythema, and hemorrhage were detected, suggesting involvement of the central nervous system. Cryptococcal meningitis associated with ocular abnormalities may clinically manifest as papilledema, loss of visual acuity in 1 or both eyes, and abducens nerve palsy. 49 However, none of these last features were present in our patient. A meta-analysis found a sensitivity and specificity of 98.8% and 99.3%, respectively, for cryptococcal antigen in the cerebrospinal fluid of adults living with HIV. 50 This approach may be a safe and effective alternative to the use of culture and India ink staining for establishing the diagnosis of cryptococcal meningitis, particularly in settings where lumbar puncture is feasible but where laboratory equipment is limited. 50
Oral cryptococcosis usually presents as multiple ulceration, nodules and a granulomatous appearance, with the palate and tongue being the main affected sites.17,18,20–26,28,30–32,38,41,42,43 As in the current report, cryptococcal infection can also present with nonhealing tooth extraction wounds.22,40 Najjar et al 23 documented an example of disseminated cryptococcosis involving the labial, nasal, and palatal regions in which the diagnostic difficulty was due to the concomitant presence of mature T/natural killer cell lymphoma. 23 On this basis, post-transplant lymphoproliferative disorder—a malignant process best classified as a heterogeneous group of diseases ranging from an indolent polyclonal lymphocytic proliferation to aggressive non-Hodgkin lymphomas—should also be considered in the list of differential diagnoses. 51 However, some conditions, including but not limited to herpesvirus infection, leishmaniasis, paracoccidioidomycosis, and acquired oral syphilis, also share clinical features with oral cryptococcosis and therefore should be considered. 52
For instance, herpesvirus infection often appears as ulcers that affect keratinized mucosa, such as the hard palate and gingiva. In immunosuppressed hosts, as in the present report, manifestations can also affect nonkeratinized mucosa. 53 Mucocutaneous leishmaniasis and oral paracoccidioidomycosis have a remarkable predilection for middle-aged adults and men.54,55 Despite this, oral leishmaniasis is not associated with pain and often affects sites close to the nasal region. About 35% of lesions involve the hard/soft palate, uvula and tonsils, while 4.1% occur in the gingivae. 54 In the chronic forms of paracoccidioidomycosis, 64% and 50% of individuals have the lungs and oral mucosa affected, respectively. 56 However, skin manifestations have been reported in 30% of affected patients. 56 Clinically, oral lesions generally appear in single or multiple forms as “mulberry-like” ulcerations that most commonly affect the gingiva/alveolar ridge, palate, lips/labial commissure, and buccal mucosa, accompanied by oral pain and hoarseness.54,55 In the present report, however, the classical moriform appearance and hemorrhagic dots of paracoccidioidomycosis were not observed.
Acquired oral syphilis can also be considered an illustration of the pitfalls set by the great imitator, besides the resurgence of this sexually transmitted infection. 57 The secondary stage is the one that would suit our patient. Recently, a South American study revealed that individuals aged 20 to 29 years are the most affected by that condition and about 12% of patients were in their sixth decade of life. 58 In contrast to the present report, the manifestations of secondary oral syphilis are mainly mucous patches, and the tongue, followed by the lip/labial commissure are the most affected sites. 58 Not least, some individuals in the secondary stage of acquired syphilis may exhibit palmoplantar involvement with erythematous-brownish papules and plaques surrounded by a desquamation collarette (Biett's collar). 58 Except for orofacial manifestations, no other cutaneous condition was observed in our patient. Ultimately, in instances of single ulcerated lesions with indurated borders on the tongue, clinicians may hypothesize conditions such as histoplasmosis, tuberculosis, and oral squamous cell carcinoma.52,59,60
The microscopic features of a cryptococcal lesion generally show a granulomatous inflammatory response to the organism. Cryptococcus spp. can change morphology in response to environmental conditions, a plasticity that may enhance its survival. Some morphotypes have been described, such as hyphae, pseudohyphae, seed cells, microcells (<2 µm), typical yeast cells (6-8 µm), and titan cells (>10 µm). 61 Staining with Grocott-Gomori methenamine silver, periodic acid-Schiff, Alcian blue, and mucicarmine is employed to identify the fungus.1,62 In pulmonary cryptococcosis, Grocott-Gomori methenamine silver staining is the most used and has shown 100% sensitivity, significantly higher than periodic acid-Schiff staining when compared to Alcian blue staining. 62 Our review identified that Grocott-Gomori methenamine silver and mucicarmine staining were the most employed in the diagnosis.17,21,22,24–26,28–31,33,34,36–38,40,41 Additionally, Fontana-Masson staining, which identifies melanin in the yeast cell wall, has also been used, but less frequently.1,3
As demonstrated in the present study and others,20,24,33 direct identification of globular and encapsulated cryptococcal yeast cells in body fluids is possible using India ink staining. 3 In Brazil and Nepal, for example, India ink staining has been the test of choice due to its simplicity for morphophysiological identification of Cryptococcus in cerebrospinal fluid smears, also in view of economic aspects such as limited resources in these countries.8,63 Indeed, the sensitivity of India ink staining is considered low, that is, 86%. 64 Nonetheless, agreement between cerebrospinal fluid cryptococcal antigen and reference-standard (cerebrospinal fluid culture) has been reported to be 98%. 50
Amphotericin B is currently the mainstream therapy for systemic mycosis infection, including cryptococcosis. Although little used in Brazil, another class of antifungals used is pyrimidine analogs (eg, 5-fluorocytosine) plus amphotericin B, instituted for 2 to 4 weeks. 65 According to the Infectious Diseases Society of America, 3 phases are recommended for the treatment of cryptococcosis. In the induction phase, amphotericin B plus flucytosine is recommended for at least 2 weeks; in consolidation, high-dose fluconazole for at least 8 weeks; and for maintenance, fluconazole at lower doses for 6 to 12 months.2,66,67 In Brazil, an important risk factor in the context of mortality from cryptococcosis is HIV/AIDS. 68 Transplant recipients and HIV-positive individuals had a similar 90-day mortality risk. 66 In line with our report, a previous study found that 3 of the 4 individuals who underwent heart transplantation and had cryptococcosis (without oral involvement) survived. 45
In summary, we described an unusual presentation of oral cryptococcosis in a heart transplant recipient as part of a total of 3 examples in the literature. Knowledge of the clinicodemographic and ecodynamic aspects of oral cryptococcosis is important for the multidisciplinary team in decision making and surveillance.
Footnotes
Acknowledgements
This study was supported by Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES, Finance Code 001), Brazil. ACVPS, JAAA, SRO, NMP, and LSF are the recipients of fellowships. We also thank Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, #305077/2021-0). TAS is a research fellow of CNPq. We would like to extend our special thanks to Dr. Rafael P. F. Mendes, Dr. Marcelo A. P. Xavier, and Dr. Erika S. Pantuzzo for their help and support with the clinical information. Mrs. E. Greene provided English editing of the manuscript.
Authors’ Note
The data used to support the findings of this study are included within the article. Any other additional data are available from the corresponding author on request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, #305077/2021-0).
Ethical Approval
For this type of retrospective clinical report, formal consent is not required.
Informed Consent
No identifier information is included in the clinical report, and the study meets the waiver criteria for the institutional review board.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
