Abstract
Introduction
Melanoma, formed by the malignant transformation of normal melanocytes, is a common type of skin cancer. 1 This disease is characterized by a high metastatic potential. 2 Epidemiological studies show that the incidence of melanoma is extremely high in geriatric populations, and the development of melanoma can be affected by multiple factors such as genetic background, pigmentation status, and ultraviolet radiation.3–6 According to the WHO classification, melanoma is divided into nine distinct types, and these subtypes can be distinguished by their epidemiology, histologic morphology, and genomic features. 7 Currently, a variety of therapeutic approaches are available for melanoma, including surgery, chemotherapy, and immunotherapy.8–10 These therapies have effectively improved the overall survival of melanoma patients at an early stage and thus are beneficial for them.11,12 But for those with advanced melanoma, lesion metastasis is incurred, resulting in treatment failure and a poor prognosis.13,14 Therefore, it is urgently needed to explore a novel therapeutic strategy for melanoma treatment.
Taraxacum officinale (TO), also known as dandelion, is a kind of herbaceous perennial plants belonging to the family Compositae. 15 Its therapeutic value was first mentioned in China, and its main use was for acute mastitis or liver and spleen ailments. 16 Nowadays, it is widely used as an herbal remedy in Asia, Europe, and North America. 16 Increasing evidence indicates that TO extracts possess important therapeutic value for their diverse biological properties against various diseases.17–19 Taraxasterol (TX), a pentacyclic triterpene, is one of the main active constituents isolated from TO. 20 A growing number of studies have reported that TX exhibits a wide range of biological activities such as anti-oxidative, anti-inflammatory, and neuro-protective effects.21–23 Recently, TX has been demonstrated to be a potential drug candidate for treatment of some types of cancers.24–26 However, the specific role of TX in melanoma remains unclear.
In this study, we aimed at exploration of the effect of TX on melanoma cell viability, apoptosis, migration, invasion, and epithelial-mesenchymal transition (EMT) as well as the underlying mechanisms.
Materials and methods
Agents
TX (CAS No. 1059-14-9) was purchased from Sigma (St. Louis, MO, USA), and DMSO was used to prepare the TX solution according to the manufacturer’s instructions.27,28 Antibodies against cleaved-PARP and cleaved-caspase-3 were purchased from Abcam (Shanghai, China). Antibodies against E-cadherin, α-catenin, N-cadherin, vimentin, p-PI3K, PI3K, p-Akt, and Akt were purchased from Santa Cruz Biotechnology (Santa Cruz, CA, USA). NAC was purchased from BestBio (Shanghai, China) and its concentration used in this study was based on previous studies.29,30
Cell lines and cell culture
Human melanoma cell lines A375 and SK-MEL-28 were purchased from American Type Culture Collection (ATCC, Manassas, VA, USA). All cell lines were cultured in Dulbecco’s modified Eagle’s medium (DMEM; Sigma, St. Louis, MO, USA) containing 10% fetal bovine serum (FBS; Sigma) and 1% penicillin/streptomycin, followed by incubation at 37°C in a humidified atmosphere with 5% CO2.
CCK-8 assay
The potential cytotoxic effect of TX on cells was evaluated by CCK-8 assays as described previously. 31 In brief, cells were seeded in a 96-well plate at a density of 5 × 103 cells/well and treated with various concentrations of TX (0, 2.5, 5, 10, 15, or 20 μg/mL). After incubation for 24, 48, or 72 h, CCK-8 solution was added to each well and the cells were further cultured at 37°C for 4 h. The absorbance was measured at a wavelength of 450 nm using a microplate reader.
Cell apoptosis assay
Cells (1 × 106) were grown in a 6-well plate and treated with different concentrations of TX for 24 h. Subsequently, cells were collected, washed in cold PBS, and incubated with Annexin V-FITC/PI (BD Bioscience, San Diego, CA, USA) for 10 min in the dark, followed by analysis with flow cytometry (BD Bioscience). 32
Western blot
Following treatment with 0, 2.5, 5, or 10 μg/mL TX for 24 h, cells (1 × 106) were lysed in RIPA lysis buffer for extraction of total protein. The protein concentration was determined by a BCA protein assay kit (Pierce, Rockford, IL, USA) as previously described. 33 An equal amount of protein was separated by 12% SDS-PAGE and transferred onto PVDF membranes (Millipore, Billerica, MA, USA). The membranes were blocked with 5% non-fat milk and incubated overnight at 4°C with primary antibodies against cleaved-PARP (ab225715), cleaved-caspase-3 (ab2302), E-cadherin (sc-8426), α-catenin (sc-9988), N-cadherin (sc-8424), vimentin (sc-373717), p-PI3K (sc-376112), PI3K (sc-365290), p-Akt (sc-514032), Akt (sc-5298), and GAPDH (sc-365062). Then the membranes were washed three times with TBS buffer and incubated with HRP-conjugated secondary antibodies for 2 h. Protein blots were visualized by an enhanced chemiluminescence detection reagent (Pierce). Protein intensity was quantified by the ImageJ software.
Transwell assay
Cell migration and invasion were measured using transwell chambers (with Matrigel coated for cell invasion). 34 In brief, 5 × 104 cells were planted onto the upper chamber containing serum-free medium while culture medium containing 10% FBS was added to the lower chamber. After incubation for 24 h, cells migrating or invading to the lower surface of the insert were fixed and stained with 0.1% crystal violet. Cells from five random fields were counted under a microscope.
Determination of intracellular reactive oxygen species
Intracellular reactive oxygen species (ROS) was detected using an ROS assay kit (Beyotime) as previously described. 35 Briefly, cells (1 × 105/well) were seeded in a 6-well plate and cultured for 24 h. After washing with PBS, cells were incubated with DCFH-DA for 30 min in the dark. Subsequently, cells were washed and DCFH-DA florescence was measured by using flow cytometry.
Statistical analysis
Data were collected from at least three-independent experiments and shown as means ± standard deviation (SD). Statistical analysis was performed by GraphPad Prism 8.0 software. The Student’s t-test or one-way ANOVA was used to compare the differences between different groups. p < .05 was considered statistically significant.
Results
Effects of TX on melanoma cell viability
First, we evaluated the cytotoxic effect of TX on melanoma cells. A375 and SK-MEL-28 cells were exposed to TX at different concentrations (0, 2.5, 5, 10, 15, or 20 μg/mL) for 24, 48, or 72 h, and cell viability was measured using CCK-8 assays. The results showed that treatment with TX below 10 μg/mL exhibited no cytotoxic effects on either A375 or SK-MEL-28 cells while exposure to TX above 10 μg/mL remarkably decreased A375 and SK-MEL-28 cell viability in a dose- and time-dependent manner (Figure 1(a) and (b)). Effects of TX on melanoma cell viability. (A, B) A375 and SK-MEL-28 cells were treated with various concentrations of TX for different time, followed by detection of cell viability using CCK-8 assays. Notes: *p < .05. TX: Taraxasterol.
TX induces melanoma cell apoptosis
To confirm whether TX-induced inhibition of melanoma cell viability was due to induction of apoptosis, we examined the effect of TX on melanoma cell apoptosis. A375 and SK-MEL-28 cells were treated with different concentrations of TX and then their apoptosis rate was determined by using flow cytometry. As shown in Figure 2(a) and (b), apoptotic A375 and SK-MEL-28 cells were significantly increased after treatment with TX above 10 μg/mL. To further verify the apoptotic effect induced by TX, the expression of apoptosis-associated proteins was assessed by using western blot. The results showed that the protein expression of cleaved-PARP and cleaved-caspase-3 was markedly increased in TX-treated A375 and SK-MEL-28 cells in comparison with the control cells (Figure 2(c) and (d). TX induces melanoma cell apoptosis. (A, B) A375 and SK-MEL-28 cell apoptosis was determined by using flow cytometry after treatment with different concentrations of TX. (C, D) Expression levels of cleaved-PARP and cleaved-caspase-3 in A375 and SK-MEL-28 cells were detected by western blot. Notes: *p < .05. TX: Taraxasterol.
TX inhibits melanoma cell migration and invasion
The effect of TX on melanoma cell migration and invasion was evaluated using transwell chambers. A375 and SK-MEL-28 cells were exposed to TX at different concentrations and then cell migration and invasion were detected by transwell assays. The results showed that TX significantly attenuated the migratory and invasive abilities of A375 cells in comparison with the control cells (Figure 3A and B). Similar results were obtained for SK-MEL-28 cells (Figure 3C and 3D). TX inhibits melanoma cell migration and invasion. A375 and SK-MEL-28 cells were treated with TX at different concentrations. Transwell assays were performed to measure A375 (A, B) and SK-MEL-28 (C, D) cell migration and invasion. Notes: *p < .05. TX: Taraxasterol.
TX reverses the EMT process of melanoma cells
The EMT process is a crucial mechanism involved in regulation of cell migration and invasion.36,37 Thus, EMT-associated markers were assessed to investigate the mechanism underlying TX-inhibited migration and invasion of melanoma cells. The western blot analysis showed that TX treatment increased the protein expression of E-cadherin and α-catenin but decreased the protein expression of N-cadherin and vimentin in a dose-dependent manner in A375 and SK-MEL-28 cells (Figure 4(a) and (b)). These results suggested that TX could reverse the EMT process of melanoma cells in vitro. TX reverses the EMT process of melanoma cells. A375 and SK-MEL-28 cells were exposed to different concentrations of TX, followed by detection of the protein expression of E-cadherin, α-catenin, N-cadherin, and vimentin in A375 (A) and SK-MEL-28 (B) cells by using western blot. Notes: *p < .05. TX: Taraxasterol.
TX inhibits activity of the PI3K/Akt pathway in melanoma cells
Increasing studies have demonstrated the important role of the PI3K/Akt signaling in the progression of various cancers including melanoma.38–40 To explore whether this pathway is associated with TX-inhibited melanoma progression, we detected the effect of TX on the expression of proteins related to the PI3K/Akt pathway by using western blot. As shown in Figure 5(a) and (b), TX treatment significantly reduced the expression levels of p-PI3K and p-Akt proteins in A375 and SK-MEL-28 cells in a dose-dependent manner. TX inhibits activity of the PI3K/Akt pathway in melanoma cells. A375 (A) and SK-MEL-28 (B) cells were treated with TX at different concentrations, followed by detection of the relative protein levels of p-PI3K/PI3K and p-Akt/Akt by using western blot. Notes: *p < .05. TX: Taraxasterol.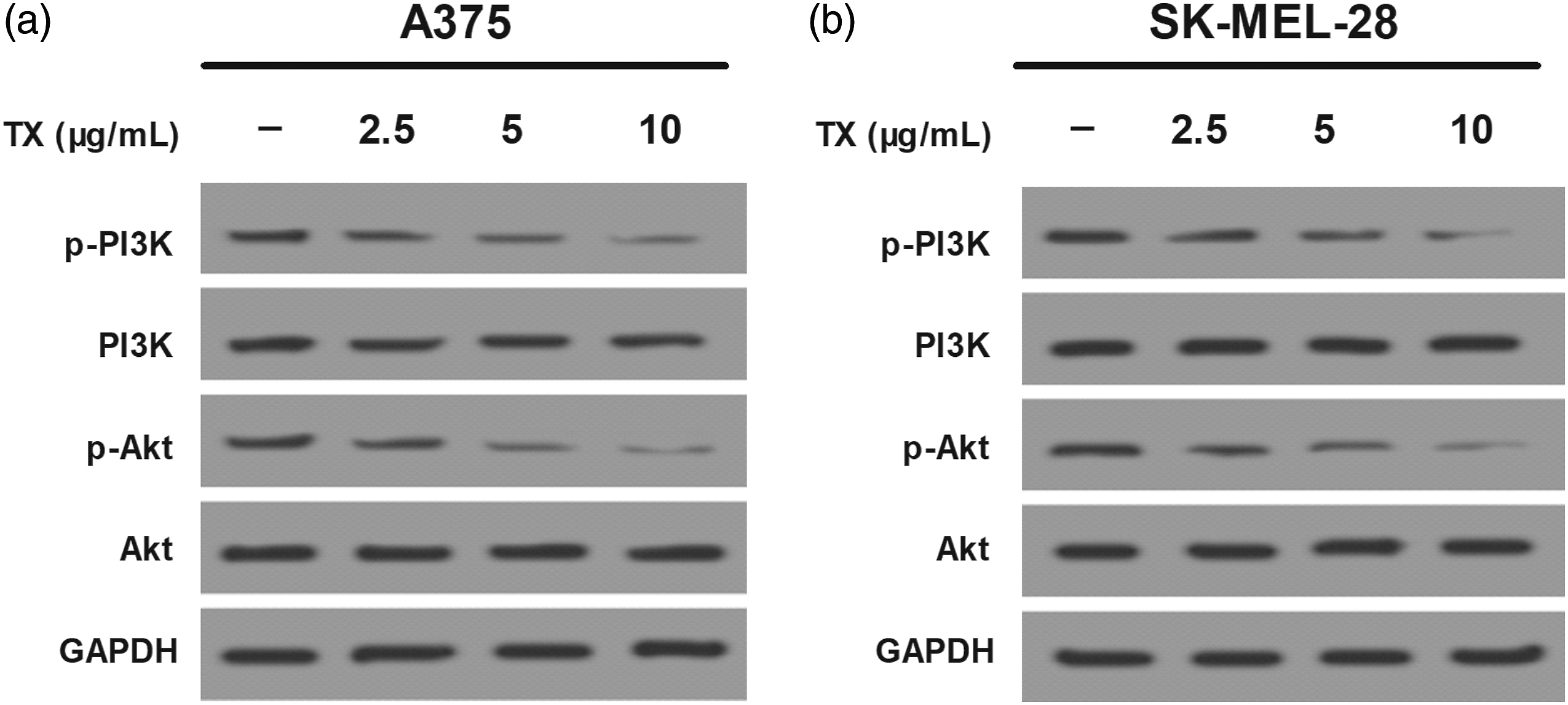
TX-induced ROS inactivates the PI3K/Akt pathway in melanoma cells
It is well known that the PI3K/Akt pathway could be regulated by ROS during cancer progression.41,42 Thus, we determined whether the inhibitory effect of TX on the PI3K/Akt pathway was associated with its oxidative stress. As shown in Figure 6(a) and (b), TX treatment increased the generation of ROS in A375 and SK-MEL-28 cells. Besides, treatment with the ROS scavenger NAC (5 mM) markedly rescued TX-induced down-regulation of p-PI3K and p-Akt in A375 and SK-MEL-28 cells (Figure 6C and 6D). These findings suggested that TX-regulated inactivation of the PI3K/Akt pathway was dependent on ROS generation. Taraxasterol-induced ROS inactivates the PI3K/Akt pathway in melanoma cells. (A, B) ROS levels in A375 and SK-MEL-28 cells were measured using flow cytometry. (C, D) The relative protein levels of p-PI3K/PI3K and p-Akt/Akt in A375 and SK-MEL-28 cells were determined by using western blot in the presence or absence of NAC (5 mM). Notes: *p < .05.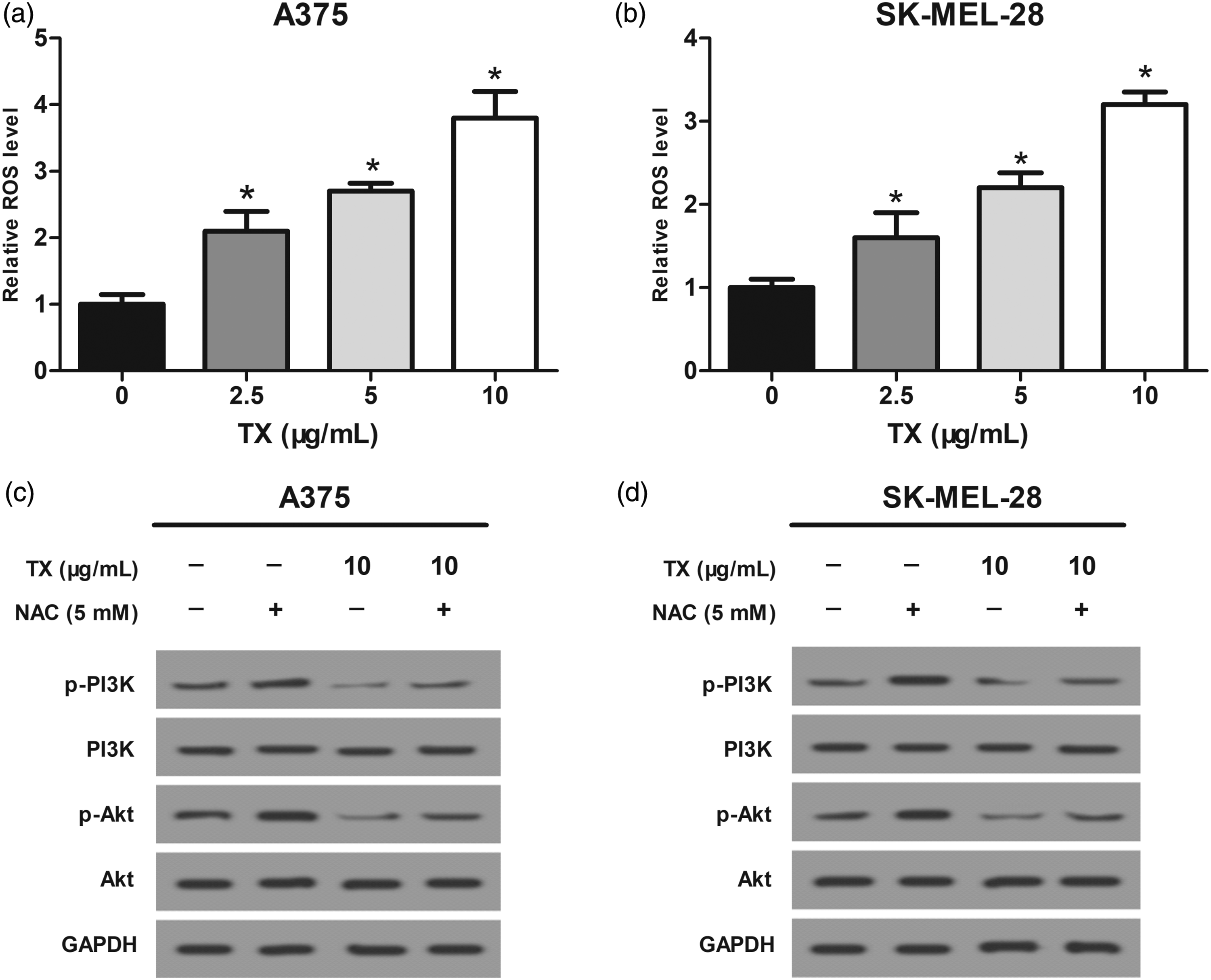
Discussion
With an annual growth rate of 3%–5%, melanoma has become one of the fastest growing malignant tumors, requiring a special attention from public health. 43 So far, there have been multiple therapeutic approaches for melanoma.8–10 However, these therapies are not effective enough especially for metastatic melanoma.13,14 Therefore, exploration of a novel therapeutic strategy is in desperate need.
TX is an active component extracted from TO which exerts a variety of pharmacological effects. The functions of TX involve in many aspects such as anti-oxidative and anti-inflammatory action. Recent studies showed that TX displayed its anti-tumor activity against some types of cancers. For example, Bao et al. reported that TX suppressed growth and induced apoptosis in liver cancer cells by regulating the expression of Hint1. 24 Chen et al. demonstrated that TX inhibited gastric cancer cell growth via suppression of EGFR/Akt1 signaling and suggested TX as a novel therapeutic agent for treatment of gastric cancer. 25 Similarly, Tang et al. showed that TX targeted RNF31 to inhibit colorectal cancer cell proliferation. 26 All these observations have confirmed the anti-tumor effect of TX on cancer progression. Consistently, this study found that TX induced melanoma cell apoptosis. Furthermore, exposure to TX inhibited melanoma cell migration and invasion. As regards to regulation of cell migration and invasion, the EMT process is a crucial mechanism. During the EMT process, a critical step is the decrease in the expression of E-cadherin and α-catenin, which is important for formation of intercellular junctions and is accompanied simultaneously by an increase in the expression of N-cadherin and vimentin. 44 In this study, we found that TX treatment increased the protein expression of E-cadherin and α-catenin but decreased the protein expression of N-cadherin and vimentin in melanoma cells, suggesting the reversal effect of TX on the EMT process. In accordance with our findings, Zhu’s study demonstrated that TX showed a suppressive effect on EMT in papillary thyroid cancer cells. 45 These results further verified the role of TX as a tumor suppressor.
The molecular mechanisms underlying the anti-cancer activity of TX in melanoma are complex and associated with diverse factors. Numerous studies have reported that the PI3K/Akt pathway plays an indispensable role in melanoma progression. For instance, Long et al. displayed that suppression of the PI3K/Akt pathway led to enhanced apoptosis of melanoma cells. 46 Additionally, Cao et al. demonstrated that the metastasis of melanoma could be inhibited via blockage of the PI3K/Akt pathway. 47 Furthermore, phosphorylation of PI3K/Akt could activate transcription of some important molecules and subsequently promote the intracellular lipid synthesis of tumor cells. 48 In this study, we investigated the association between TX and the PI3K/Akt pathway. The results showed that TX reduced the protein expression of p-PI3K and p-Akt; thus, inhibiting activity of the PI3K/Akt pathway in melanoma cells. These results provided a possible mechanism responsible for the effects of TX on melanoma.
ROS plays an essential role in various biological processes. 49 Notably, ROS could exert a dynamical effect on the tumor microenvironment with different concentrations. 49 Previous studies have reported the regulatory role of ROS in activation of the PI3K/Akt pathway during cancer progression.41,42 However, the association between ROS and the PI3K/Akt pathway following TX treatment has not been illustrated. This study showed that TX treatment increased the levels of ROS in melanoma cells. To further investigate the link between ROS and the PI3K/Akt pathway, the ROS scavenger NAC was used for experiments. NAC is known for its ability of clearing the intracellular ROS. 50 Our study showed that treatment with NAC significantly rescued TX-induced down-regulation of p-PI3K and p-Akt in melanoma cells. These findings suggested that TX-regulated inactivation of the PI3K/Akt pathway was dependent on ROS generation.
In conclusion, our study showed that TX induced apoptosis and inhibited migration and invasion of melanoma cells. TX also reversed the EMT process in melanoma cells. Besides, TX suppressed activity of the PI3K/Akt pathway in melanoma cells. We also found that TX increased the levels of ROS in melanoma cells, and the ROS scavenger NAC rescued TX-induced down-regulation of p-PI3K and p-Akt in melanoma cells. Taken together, TX induced ROS accumulation followed by inactivation of the PI3K/Akt pathway and subsequently attenuated melanoma progression, suggesting that TX may be a potential candidate for treatment of melanoma.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
