Abstract
Objective
To explore the effects of hypoxic preconditioning neural stem cell (P-NSC) transplantation on rats with spinal cord injury (SCI).
Methods
After identification, the NSCs were treated with hypoxic preconditioning. The NSCs migration was detected by Transwell method. RT-qPCR was used to detect the mRNA levels of HIF-1α, CXCR4 in NSC. The secretion of representative neurotrophic factors (VEGF, HGF, and BDNF) was checked by Western blot. Forty-six SCI rats were randomly divided into three experimental groups: SCI group (PBS injection, n = 10); N-NSC group (NSC atmospheric normoxic pretreatment injection, n = 18); and P-NSC group (NSC ’s hypoxic preconditioning injection, n = 18). The sham operation group was also included (rats underwent laminectomy but not SCI, n = 10). The recovery of hindlimb motor function was evaluated by BBB score. The level of spinal cord inflammation (IL-1β, TNF-α, and IL-6) was determined by ELISA. Western blot was used to detect the content of TMIGD1 and TMIGD3 in spinal cord.
Results
Compared with the N-NSC group, the number of NSC-passing membranes in the P-NSC group increased with the increase of the culture time (p < 0.05). Compared with N-NSC, P-NSC had higher levels of VEGF, HGF, and BDNF after 1 week of culture (p < 0.05). The BBB score of the P-NSC group was significantly higher than that of the N-NSC group at 7 and 28 days (p < 0.05). Compared with the SCI group, the levels of TNF-α, IL-1β, and IL-6 were significantly reduced after NSC treatment, and the P-NSC group was lower than the N-NSC group (p < 0.05). Compared with the SCI group, the levels of TMIGD1 and TMIGD3 increased. Compared with the N-NSC group, and the levels of TMIGD1 and TMIGD3 increased in the P-NSC group (p < 0.05).
Conclusion
P-NSC administration could improve SCI injury, and the levels of TMIGD1 and TMIGD3.
Introduction
Spinal cord injury (SCI) is an unsolved medical problem, characterized by high morbidity and mortality. With the development of modern transportation and construction industry, the incidence of SCI is increasing annually. 1 SCI patients often show severe motor and sensory paralysis, and the prognosis is poor, resulting in a heavy family and social burden. 2 In the past few decades, cell transplantation has become a promising treatment for many diseases. Stem cell transplantation, including neural stem cells (NSC), is a promising therapy for SCI. 3
Stem cells are pluripotent and they have the ability to renew themselves and differentiate into specific cell lineages. In animal and preclinical studies, NSC transplantation has achieved good results in the treatment of craniocerebral trauma and SCI. 4 The mechanism of NSC treatment may be based on the principle of NSC trans differentiation to replace damaged cells, growth factor production, and the regulation of immune response. 5 In addition, NSC maintains the resting phenotype of astrocytes and inhibits astrocyte activation. 6 Activated astrocytes release too much inflammatory cytokines and increase the degree of SCI. These functions of NSC are essential for SCI repair. Therefore, stem cells have great potential for SCI regenerative therapy. 7
Hypoxic preconditioning is a powerful, endogenous and protective mechanism that has been identified in the body. 8 The ability of hypoxic pretreated stem cells to promote the efficacy of transplanted cells in certain diseases has been studied. 9 Hypoxic preconditioning has the advantage of improving the survival and function of stem cells in animal models of myocardial and cerebral infarction. 10 Potential mechanisms for time- and concentration-dependent hypoxic preconditioning include regulating intracellular transduction, increasing cell resistance to injury, up-regulating migration and differentiation, and enhancing growth factor secretion.
In this study, we explored the effects of hypoxia pretreated NSC transplantation on the SCI rats and observed the changes of transmembrane immunoglobulin domain-containing (TMIGD).
Materials and methods
Experimental animals
Ten-week-old male Sprague–Dawley (SD) rats were purchased from Shanghai Institute of Zoology, Chinese Academy of Sciences. Animal care and use procedures are based on the “National Institutes of Health Laboratory Animal Care and Use Guidelines” and approved by the Yantaishan Animal Care and Use Committee. During the study, rats were free to eat bulk food and drinking water.
NSC identification and culture
Pentobarbital sodium overdose (150 mg/kg, intraperitoneal injection) euthanized the rat. The bone marrow cavity was rinsed with Dulbecco’s modified Eagle’s medium. The NSC was centrifuged (200 × g, 5 min at 20°C) and resuspended in complete medium containing 10% fetal bovine serum and 1% penicillin–streptomycin. The identification of the third generation (P3) NSC was performed. The GFP expression in rats is driven by the chicken β-actin promoter and the cytomegalovirus enhancer CAG promoter, and NSCs in rats have been confirmed to be GFP positive (Supplementary material, S-Figure 1). The purity of NSC was evaluated by CD29/CD90 positive and CD31/CD45 negative staining (Supplementary material, S-Figure 2). Resuspend the NSC in PBS (1 × 107 cells/mL for verification test). The antibody was then incubated at room temperature for 15 min. All flow cytometry analyses were completed within 1 h using a flow cytometer (FAC500; Beckman Coulter, Inc., Brea, CA, USA).
The NSCs were subcultured in 1:4 dilution for hypoxic pretreatment, and then cultured until confluence.
Hypoxic pretreatment scheme
Before hypoxic pretreatment, fresh and complete culture medium was added for 24 h in fully controlled proOx-C-Ochamber (Thermo). A residual gas mixture of 5% carbon dioxide and balanced nitrogen is used to maintain the indoor O2 concentration at 5%. The cells in the atmospheric normoxia control group were only exposed to 21% O2 during the preparation.
Detecting neural stem cell migration by transwell method
Cells (5 × 104/well) were cultured in a transwell system with complete medium, and migration was observed after 6, 12, and 24 h at 37°C. NSC is divided into the following groups: N-NSC group and P-NSC group. The transwell method is performed according to the manufacturer’s procedures. At the end of different time points, the transwell insert was removed and the medium in the upper and lower chambers was aspirated. The cells were washed 3 times with 0.01 M PBS and fixed with 4% paraformaldehyde for 30 min at room temperature. After drying at room temperature, the non-migrated cells in the upper chamber were gently wiped with a cotton swab. The cells were stained with 0.1% crystal violet for 20 min at room temperature. The image was captured with an optical microscope at a magnification of 400 times. A total of five fields were randomly selected for cell counting. Repeat the experiment three times, thrice each time.
Measurement of HIF-1α and CXCR4 mRNA using transcription quantitative polymerase chain reaction (RT-qPCR)
NSCs were seeded in six-well plates at a density of 1 × 105 cells/well and cultured for 24 h. The adherent cells were digested with 0.25% trypsin (preheated to 37°C; Gibco; Thermo Fisher Scientific, Inc.) at 37°C for 2–3 min, and centrifuged at 150 × g for 5 min at 20°C to collect the cells. According to the manufacturer’s protocol (Nippon Gene Co., Ltd., Tokyo, Japan), total mRNA was extracted using ISOGEN. Reverse transcribe RNA samples into cDNA, specifically amplify NSC migration-related genes [Hypoxia inducible factor 1α (Hypoxia inducible factor 1α, HIF-1α) and CXC motif chemokine receptor 4, CXCR4)], and electrophoretic separation. According to the following temperature protocol, Generay Biotech RT kit (Generay Biotech Co., Ltd., Shanghai, China) was used to reverse transcription. A total of 1 µg of total RNA sample to cDNA in a volume of 20 µl: 37°C for 15 min at 85 Leave at five°C for 5 s and at 4ºC for 5 min. PCR was performed using the Stratagene mx3000P real-time PCR system (Agilent Technologies, Inc., Santa Clara, CA, USA) according to the manufacturer’s protocol using Maxima ™ SYBR-Green/ROX qPCR master mix (Thermo Fisher Scientific, Inc.). The thermal cycling conditions were: 95°C for 5 min, 40 cycles, 95°C for 10 s, and 60°C for 34 s, respectively. PCR products were separated on 1.2% agarose gel and visualized using ethidium bromide staining. The resulting gel image was analyzed using an AlphaImager gel analysis system (AlphaImager 2000; ProteinSimple, San Jose, California, USA). The 2-ΔΔCq method was used to calculate the mRNA expression of the target gene, and the mRNA expression of the target gene was normalized to GAPDH. Repeat the experiment three times in triplicate.
Measurement of VEGF, HGFM, and BDNF in cells, TMIGD1 and TMIGD3 in rats using Western blotting.
Cell lysates and the spinal cord were collected, and the protein concentration was measured using Pierce BCA protein detection kit (Thermo Scientific, Rockford, IL, USA). Western blot analysis was performed on equal amounts of protein according to standard procedures. The main antibodies used are anti-VEGF (ab32152, Abcam), anti-HGF (MA5-14160, Thermo Fisher Scientific) and anti-BDNF (ab108319, Abcam), anti-TMIGD1 (PA5-38057, Thermo Fisher Scientific), and anti-TMIGD3 (PAb128, GenScript). After incubation with appropriate secondary antibodies, the resulting protein bands were observed by ECL (Beyotime, Shanghai, China). The absorbance value of the target protein was obtained through Gel-Pro Analyzer version 4.0 software (Media Cybernetics, Silver Spring, MD, USA).
Animal models of spinal cord injury and cell transplantation
All rats were treated in accordance with local guidelines on ethical use of animals and NIH guidelines. After anesthesia by intraperitoneal injection of 3.6% chloral hydrate (1 mL/100 g body weight), the skin was cut open to expose the thoracic column, and T9 laminectomy was performed. A strong impact of 250 kdyn (kdyns) resulted in contusion. SCI rats were randomly divided into three experimental groups: SCI group (PBS injection, n = 10); N-NSC group (NSC atmospheric oxygen pretreatment injection, n = 18); and P-NSC group (NSC hypoxia pretreatment injection, n = 18). The sham operation group was also included, sham operation group (sham operated rats received laminectomy without SCI, n = 10). After injury, a total of 105 cells were divided into two doses and transplanted into the injured spinal cord. Each injection was made by Hamilton syringe from the epicenter of the lesion to the head side 2 mm, to the tail side 2 mm. After the operation, the animals were recovered on 37ºC heating pad. The bladders of the rats were squeezed 3 times daily after surgery until spontaneous urination was restored.
Behavior analysis
Using Basso, Beattie, and Bresnahan (BBB) motor scores from 0 (complete paralysis) to 21 (normal exercise) to assess the hindlimb motor function. All animals were placed in an open place and observed by two inspectors who did not know the identity of the animals. The test time is not less than 3 min, and a rat who has stopped exercising for more than 1 min is placed in the center of the open area again for retesting. BBB test was conducted 1 day, 7 days, and 28 days after transplantation.
Detection of TNF-α, IL-1β, and IL-6 levels in the spinal cord by ELISA.
Seventy-two hours after cell transplantation, a total of 20 rats were sacrificed by pentobarbital sodium overdose (150 mg/kg, intraperitoneal injection), and the lamina was quickly opened. Harvest 3 mm above and below the damaged spinal cord (n = 5). The spinal cord tissue collected from each group was weighed and immediately homogenized in 1 mL of saline at 4°C. The homogenate was centrifuged at 2000 × g for 15 min at 4°C. The spinal cord supernatant was collected, and the levels of TNF-α and IL-6 were measured using an ELISA kit. The absorbance was measured at 450 nm according to the standard curve to calculate the sample content using CurveExpert 1.3 software and plotted in the dose response curve.
Statistical analysis
The data is expressed as the mean SD of the results from three independent experiments. SPSS version 19.0 (SPSS Inc., Chicago, Illinois, USA) was used for statistical analysis. One way ANOVA was used for multiple comparisons. p < 0.05 was defined as statistically significant.
Results
Hypoxic pretreatment increases neural stem cell migration
Transwell experiment (Figure 1) and the numbers of migrated cells (Table 1) showed that hypoxic pretreatment increased NSC migration rate. When NSC was cultured in transwell system for 6 h, only a small amount of NSC in N-NSC group passed through 8 μm membrane. Compared with the N-NSC group, the number of NSC passing through the membrane in the P-NSC group increased (p < 0.05). With the increase of culture time, the number of cells passing through the membrane in the two groups increased significantly, and the number of cells in the P-NSC group increased more than that in the N-NSC group after 12 h of culture (p < 0.05). After 24 h of culture, the number of cells passing through the membrane increased in both groups, but the difference was not statistically significant (p > 0.10) The NSC migration was measured by transwell assay, and the numbers of migrated cells were analyzed at 6 h, 12 h, and 24 h. All data were presented as the mean ± standard deviation (SD). The t test was used to analyze the difference between groups. The numbers of migrated cells were significantly increased at 6 h and 12 h in P-NSC group compared with the N-NSC group. P-NSC: hypoxic preconditioning neural stem cell; N-NSC: normoxic pretreatment neural stem cell. Numbers of migrated cells. The NSC migration was measured by transwell assay, and the numbers of migrated cells were analyzed at 6 h, 12 h, 24 h. All data were presented as the mean ± standard deviation (SD). The t test was used to analyze the difference between groups. The numbers of migrated cells were significantly increased at 6 h, 12 h in P-NSC group compared with the N-NSC group. P-NSC: hypoxic preconditioning neural stem cell; N-NSC: normoxic pretreatment neural stem cell. SCI: Spinal Cord Injury.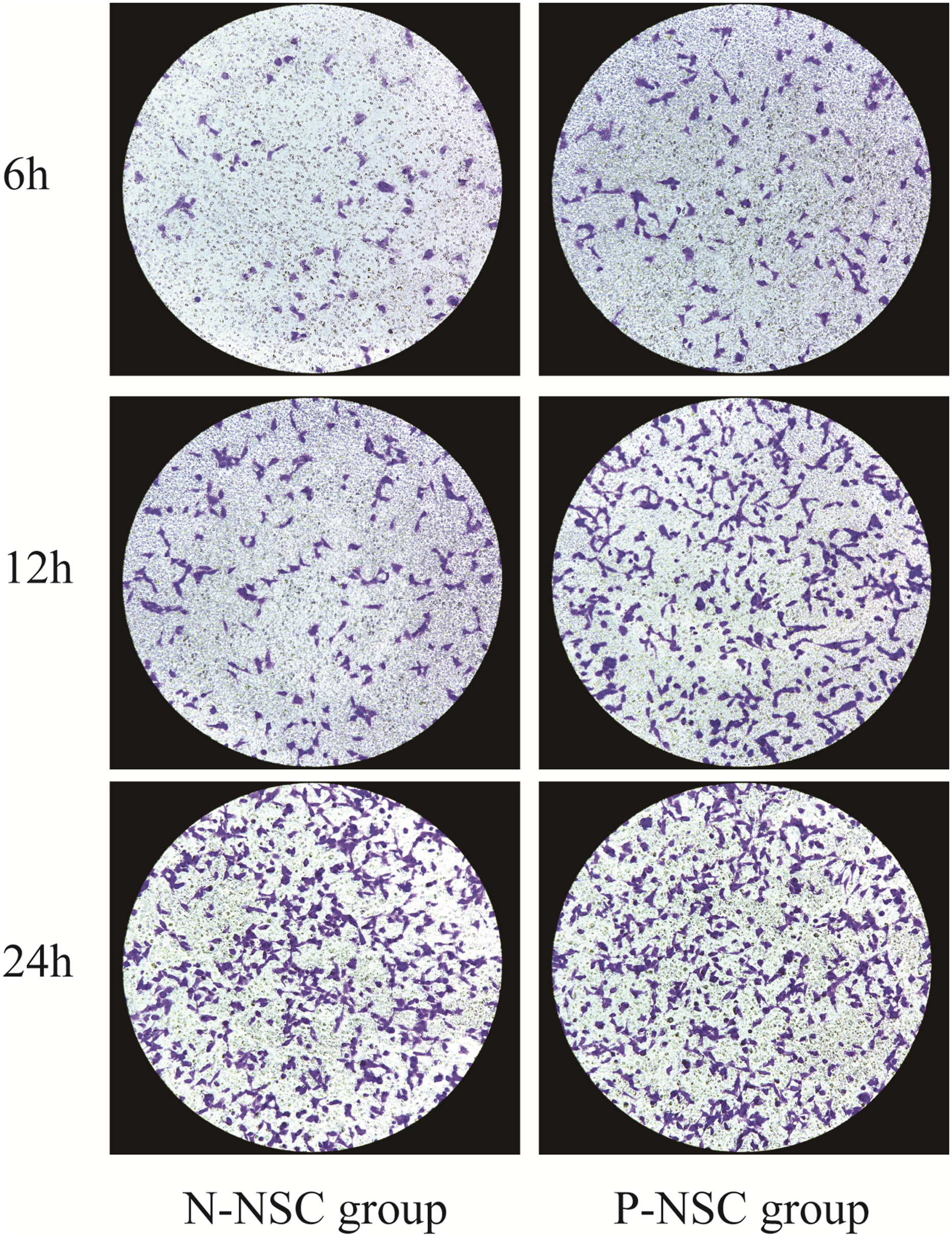

Hypoxia pretreatment increases the expression of HIF-1α and CXCR4 mRNA in neural stem cell
Expression of HIF-1 α and CXCR4 mRNA was analyzed by RT-.

Expression of HIF-1 α and CXCR4 mRNA in cells was analyzed using RT-qPCR at 24 h. All data were presented as the mean ± standard deviation (SD). The t test was used to analyze the difference between groups. For HIF-1 α and CXCR4 mRNA levels, there were significant differences between the P-NSC group and N-NSC group. P-NSC: hypoxic preconditioning neural stem cell; N-NSC: normoxic pretreatment neural stem cell.
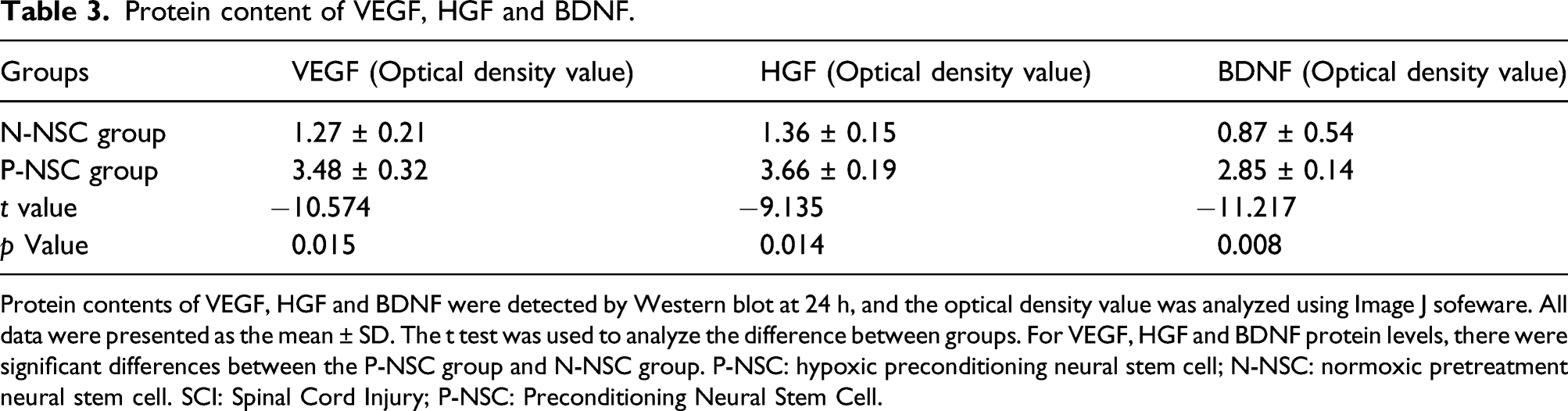
Hypoxic preconditioning can increase the regenerative neurotrophic factor in neural stem cell
In this study, we examined the secretion of representative neurotrophic factors by Western blot (Figure 2), and the optical density value of each protein was showed in Table 3. We found that NSC expressed detectable levels of several neurotrophic factors under normoxic conditions, including VEGF, HGF and BDNF. Compared with N-NSC, P-NSC had higher levels of VEGF, HGF, and BDNF after 1 week of culture (p < 0.05). Protein contents of VEGF, HGF and BDNF were detected by Western blot at 24 h, and the optical density value was analyzed using Image J sofeware. All data were presented as the mean ± SD. The t test was used to analyze the difference between groups. For VEGF, HGF and BDNF protein levels, there were significant differences between the P-NSC group and N-NSC group. P-NSC: hypoxic preconditioning neural stem cell; N-NSC: normoxic pretreatment neural stem cell. Protein content of VEGF, HGF and BDNF. Protein contents of VEGF, HGF and BDNF were detected by Western blot at 24 h, and the optical density value was analyzed using Image J sofeware. All data were presented as the mean ± SD. The t test was used to analyze the difference between groups. For VEGF, HGF and BDNF protein levels, there were significant differences between the P-NSC group and N-NSC group. P-NSC: hypoxic preconditioning neural stem cell; N-NSC: normoxic pretreatment neural stem cell. SCI: Spinal Cord Injury; P-NSC: Preconditioning Neural Stem Cell.
P-NSC improves the motor function of hind limbs after spinal cord injury
The recovery of motor function of hind limbs was evaluated by BBB score, and the scores were showed in Figure 3 and Table 4. SCI can reduce BBB score. With the passage of time, BBB of all groups showed a gradual increase trend. The application of NSC increased BBB score (p < 0.05). The BBB score of P-NSC group was higher than that of N-NSC group at 7 and 28 days (p < 0.05). The BBB scores were obtained by 1, 7, and 28 days after SCI. All data were presented as the mean ± standard deviation (SD). The ANOVA was used to analyze the difference among groups. Sham operation group (sham operated rats received laminectomy without SCI, n = 10); SCI group (PBS injection, n = 10); N-NSC group (NSC atmospheric oxygen pretreatment injection, n = 18); P-NSC group (NSC hypoxia pretreatment injection, n = 18). The application of NSC increased BBB score. The BBB score of P-NSC group was higher than that of N-NSC group. BBB score. The BBB scores were obtained by 1, 7, 28 days after SCI. All data were presented as the mean ± standard deviation (SD). The ANOVA was used to analyze the difference among groups. Sham operation group (sham operated rats received laminectomy without SCI, n = 10); SCI group (PBS injection, n = 10); N-NSC group (NSC atmospheric oxygen pretreatment injection, n = 18); P-NSC group (NSC hypoxia pretreatment injection, n = 18). The application of NSC increased BBB score. The BBB score of P-NSC group was higher than that of N-NSC group. SCI: Spinal Cord Injury; P-NSC: Preconditioning Neural Stem Cell.

Preconditioning neural stem cell inhibits inflammatory response factors in spinal cord
Detection of inflammation level.

The levels of IL-1β, TNF-α, and IL-6 in spinal cord were analyzed by ELISA. All data were presented as the mean ± standard deviation (SD). The ANOVA was used to analyze the difference among groups. Sham operation group (sham operated rats received laminectomy without SCI, n = 10); SCI group (PBS injection, n = 10); N-NSC group (NSC atmospheric oxygen pretreatment injection, n = 18); P-NSC group (NSC hypoxia pretreatment injection, n = 18). The levels of TNF-α, IL-1 β and IL-6 decreased after NSC treatment, and P-NSC group was lower than N-NSC group.
Influence of TMIGD1 and TMIGD3 in spinal cord
In addition, the content of TMIGD1 and TMIGD3 was detected by Western blot (Figure 4, and the optical density value of each protein was showed in Table 6. The content of TMIGD1 and TMIGD3 were lower than that in sham operation group (p < 0.05). Compared with SCI group, the contents of TMIGD1 and TMIGD3 increased, the contents of TMIGD1 and TMIGD3 increased in P-NSC group compared with N-NSC group (p < 0.05). The levels of TMIGD1 and TMIGD3 were analyzed by Western blot, and the optical density value was analyzed using Image J software. All data were presented as the mean ± standard deviation (SD). The ANOVA was used to analyze the difference among groups. Sham operation group (sham operated rats received laminectomy without SCI, n = 10); SCI group (PBS injection, n = 10); N-NSC group (NSC atmospheric oxygen pretreatment injection, n = 18); P-NSC group (NSC hypoxia pretreatment injection, n = 18). The contents of TMIGD1 and TMIGD3 increased after NSC treatment, and the P-NSC group higher than N-NSC group. Content of TMIGD1 and TMIGD3. SCI: Spinal Cord Injury; P-NSC: Preconditioning Neural Stem Cell. The levels of TMIGD1 and TMIGD3 were analyzed by Western blot, and the optical density value was analyzed using Image J software. All data were presented as the mean ± standard deviation (SD). The ANOVA was used to analyze the difference among groups. Sham operation group (sham operated rats received laminectomy without SCI, n = 10); SCI group (PBS injection, n = 10); N-NSC group (NSC atmospheric oxygen pretreatment injection, n = 18); P-NSC group (NSC hypoxia pretreatment injection, n = 18). The contents of TMIGD1 and TMIGD3 increased after NSC treatment, and the P-NSC group higher than N-NSC group.

Discussion
In the past few decades, extensive research has been conducted on stem cell therapy of SCI, and new possibilities have been created for regenerative medicine. 11 Stem cells may provide an inexhaustible source of neurons and glia, and exert neuroprotective effects on host tissues. Several kinds of stem cells are considered to be useful for the treatment of SCI, including embryonic stem cells, glial cells, Schwann cells, and NSC. 12 Because NSCs are pluripotent and plastic, they are attractive cells for regenerative medicine, especially for neuroprotection and nerve repair therapy.13–15 In this study, hypoxic preconditioning could increase the migration of NSC, and improved the SCI in rats. These results were consistent with previous studies. In previous studies, transplantation of hypoxic preconditioned NSC benefited functional recovery induced by SCI, 16 and hypoxic preconditioning improved bone mesenchymal stem cells migration and ameliorated SCI in rats. 6 In addition, hypoxic preconditioning could enhance the therapeutic effects of human umbilical mesenchymal stem cells for SCI in rats. 17
Studies have reported that in adult rat models with severe ischemic stroke, NSC transplantation with physical and chemical pretreatment can increase the expression of growth factors (including BDNF, GDNF, and VEGF), and NSC transplantation with physical and chemical pretreatment.18–20 Consistent with these reports, our in vitro studies indicated that locally delivered P-NSC could increase the VEGF, HGF, and BDNF expression, which might be related to the recovery in the damaged spinal cord.
Previous studies on migration-related pathways have shown that hypoxic preconditioning may improve cells migration by promoting the expression of HIF-1α and CXCR4.21–23 The downstream gene CXCR4 of HIF-1 and its ligand stromal cell-derived factor 1 are considered to be the key factors for stem cells migration. NSC’s homing ability is affected by cell culture conditions. If the cells are cultured under normoxic conditions for 24 h, the migration and homing ability of NSC will be significantly reduced. 24 At the same time, the number of adhesion molecules on the surface of NSC, including CXCR4, gradually decreased, and the ability to respond to chemokines also decreased. The results of this study also indicated that hypoxic preconditioning induces adaptive metabolic changes in NSC by regulating migration-related genes (HIF-1α and CXCR4), thereby improving their migration ability.
TMIGD1 plays important roles in regulating cell–cell adhesion, cell proliferation, and cell cycle. Rahimi et al. 25 found that the cell adhesion molecule TMIGD1 binds to moesin and regulates tubulin acetylation and cell migration. TMIGD3, as a suppressor of NF-κB, inhibited osteosarcoma cells migration to improve osteosarcoma progression. 26 In our study, the TMIGD1 and TMIGD3 were increased after transplantation of P-NSC when compared with the N-NSC group, suggesting that hypoxic preconditioning might regulate NSC migration to improve SCI progression.
Although the transplantation of neural stem cell types substantially promotes the functional recovery of SCI, there are several limitations that may impede its clinical translation, such as limited accessibility and postmitotic feature of primary cell types precluding the production of sufficient amounts of cells required for the transplantation.
Conclusion
In conclusion, P-NSC transplantation provides an effective method to improve SCI treatment. Hypoxic preconditioning could improve NSC’s migration and enhance its potential to treat SCI.
Supplemental Material
sj-pdf-1-het-10.1177_09603271211066587 – Supplemental Material for Hypoxic preconditioning neural stem cell transplantation promotes spinal cord injury in rats by affecting transmembrane immunoglobulin domain-containing
Supplemental Material, sj-pdf-1-het-10.1177_09603271211066587 for Hypoxic preconditioning neural stem cell transplantation promotes spinal cord injury in rats by affecting transmembrane immunoglobulin domain-containing by Xiaoguang Fan, Hongchun Wei, Juan Du, Xiuguo Lu and Leishen Wang in Human & Experimental Toxicology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Consent for publication
Written informed consent for publication was obtained from all participants.
Ethics declarations
All procedures performed in studies involving animals were in accordance with the ethical standards of the Yantaishan Hospital.
Availability of data and materials
Please contact the corresponding author for data requests.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
