Abstract
Background
Genetic polymorphism within the P1 isoenzyme of the Glutathione-S-Transferase (GST) family is found to modulate and alter the enzyme activity of GSTP1 protein and thus may result in a change of sensitivity to platinum-based chemotherapy. We investigated the relationship between GSTP1 Ile 105 Val polymorphisms and overall survival, treatment response, and for both hematological and non-hematological toxicity of advanced North Indian lung cancer patients undergoing platinum-based double chemotherapy.
Methods
The polymorphism of GSTP1 Ile 105 Val in North Indian lung cancer patients was assessed by polymerase chain reaction-restriction fragment length polymorphism. A total of 682 lung cancer patients were enrolled in the study, and it was observed that patients who were carrying both the mutant alleles (Val/Val) for the GSTP1 polymorphism showed a higher trend of median survival time (MST) as compared to the patients bearing the wild type of genotype (Ile/Ile) (MST = 8.30 vs. 7.47, p = 0.56). Based on toxicity profiling, we observed that lung cancer patients with the mutant genotype of GSTP1 (Val/Val) had an increased risk of leukopenia (OR = 2.41; 95% CI = 1.39-4.18, p = 0.001) as compared to subjects carrying both copies of the wild alleles (Ile/Ile). Our data suggested that patients with heterozygous genotype (Ile/Val) had a 2.14-fold increased risk of developing severe anemia (OR = 2.14, 95% CI = 0.97-4.62, p = 0.03). Our data also showed that in small cell lung carcinoma (SCLC) patients' polymorphism of GSTP1 was associated with thrombocytopenia (χ2 test = 7.32, p = 0.02).
Conclusions
Our results suggest that GSTP1 Ile105Val polymorphism could be a predictive biomarker for hematological toxicity, like leukopenia and anemia, but not thrombocytopenia or neutropenia
Introduction
Lung cancer (LC) is one of the most common tumors reported, and, more significantly, the morbidity and mortality rates from lung cancer are among the highest worldwide. 1 International Agency for Research on Cancer (IARC) has reported that greater than 50% of LC cases detected are in less developed countries.2,3 The 5-year survival rate of lung cancer is dismissed and evaluated to be 15% only, and further, the median survival time (MST) is less than 10 months. The standard treatment model for lung cancer is using a combination of platinum-based drugs like cisplatin and carboplatin and third-generation cytotoxic drugs. However, the treatment with platinum-based regimens is associated with adverse effects that lead to symptoms that might be distressing to the patient and prevent further therapies.4,5 Single nucleotide polymorphisms (SNPs) in genes involved in drug metabolism and transport, DNA repair, apoptosis, and inflammatory signals have been reported to have a profound effect on LC chemotherapy. 6
Glutathione-S-Transferase (GST) is a group of cytosolic enzymes that have been discovered to play an essential role in chemotherapeutic drug metabolism and detoxification. GSTP1 catalyzes the binding of cisplatin to glutathione so it can be converted to a water-soluble form and hence can be easily excreted. 7 The gene coding for the GSTP1 enzyme is found on chromosome 11q13 and is polymorphic. A non-synonymous A/G single nucleotide polymorphism at nucleotide position 313 in exon 5 results in the replacement of adenine to guanine (SNP’s ID:rs1695), which results in the substitution of the amino acid isoleucine to valine at position 105. This substitution in the GSTP1 protein encoded results in decreased enzyme activity, thermal stability, and substrate specificity, thus affecting the biotransformation reactions of chemotherapeutic drugs mediated by GSTP1. 8 The effect of the GSTP1 polymorphism can be seen during the downstream detoxification process of cisplatin and the formation of platinum-GSH conjugates. These conjugates may accumulate in the body, which then directly alter the response rates of the patients and may increase the risk of hematologic and gastrointestinal toxicity. 9 The previous research has suggested that the genetic variability of the GSTP1 IIe 105 Val genes may play a role in the treatment outcomes of LC patients. However, the results are inconclusive.10-12 This study was evaluated in LC patients receiving platinum-based chemotherapy to determine whether the genetic polymorphism of the GSTP1 gene affects the overall survival and the response towards chemotherapy. Furthermore, we also evaluated whether the GSTP1 SNP can assess the occurrence of toxicity in lung cancer patients.
Materials and methods
Sample collection and study design
The current research included lung cancer patients who visited the author’s lung cancer clinic (Post Graduate Institute of Medical Education & Research (PGIMER), Chandigarh). The patients who were newly diagnosed with histologically or cytologically proven NSCLC and SCLC were enrolled for over 3 years. The Institutional ethics committee (i.e., Supplementary table 1) approved the study. All the participants signed an informed written consent form. The inclusion and exclusion criteria for the current study are shown in Supplementary table 2. At first, the patients were assessed at baseline for complete blood count (CBC), liver and renal function tests, contrast-enhanced computed tomography (CT) scan of thorax (which also includes upper abdomen), and chest radiograph. Demographic characteristics (including gender, age, performance status (PS), histology of the tumor, stage of disease, and patients smoking status) were recorded for all the subjects. CBC was done 10 days after the first chemotherapy cycle and that of the subsequent cycles if recommended. Before each chemotherapy, CBC, liver, and kidney function tests were also carried out and 3–4 weeks after the last chemotherapy cycle.
Chemotherapy regimen
For NSCLC patients, the standard regimen was a taxane–platinum combination administered on day one every 3 weeks. Single-agent docetaxel was considered for patients with advanced age or sub-optimal performance status deemed unfit for combination chemotherapy. Adenocarcinoma patients were given pemetrexed. Dosage of docetaxel, paclitaxel, cisplatin, and pemetrexed used was 75, 175, 60, and 500 mg/m2, respectively, per square meter (m2) of body surface area (BSA), while carboplatin was given at a dose calculated to produce an area under the concentration–time curve (AUC) of 5.0 mg per milliliter per minute. In SCLC, the standard chemotherapy regimen was irinotecan 100 mg/m2 plus cisplatin 60 mg/m2 each on day 1 of a 3-weekly cycle.
Before carrying out tumor response assessment, 4 cycles of chemotherapy were administered as is standard procedure at PGIMER. If there was unacceptable toxicity or clinic-radiological evidence of disease progression before the completion of four cycles, the tumor response was assessed, and, if necessary, chemotherapy was discontinued. Patients who demonstrated an objective response to treatment were given two additional cycles (i.e., maximum of six cycles). Response Evaluation Criteria in Solid Tumors (RECIST) criteria were used to evaluate tumor response. Adverse events (AEs) were recorded and classified according to the standard toxicity criteria (CTC) version 3.0. Toxicity was assessed for patients receiving three or more cycles of chemotherapy and all patients who received at least one chemotherapy cycle. In case of hematological toxicity, we have evaluated the following parameters like anemia, thrombocytopenia, neutropenia, and lymphopenia. PGIMER’s standard protocols were used to handle AEs. Patients with febrile neutropenia or grade 3 or higher gastrointestinal AEs were hospitalized because outpatient management was either not possible or was ineffective.
Further, any other AEs caused by chemotherapy were also recorded. Follow-up was done every 3 weeks during chemotherapy and then at 3-month intervals for the remaining study period. Overall survival (OS) was computed from the start of chemotherapy till death of the patient or the last follow-up date.
Genotyping of GSTP1 Ile 105 Val polymorphism
The DNA was extracted from 3-4 ml of the blood using a modified phenol-chloroform extraction procedure. The PCR-RFLP assay was used to determine the genotype of GSTP1 IIe 105 Val as previously reported 13 with slight modifications. For amplification of GSTP1 variants, the primer pairs used were 5′-AATACCATCCTGCGTCACCT-3′ and 5′-TGAGGGCACAAGAAGCCCCTT-3′. The 25 μl PCR mixture consists of 1X PCR buffer with 1.5 mM MgCl2, 0.5 μM each forward and reverse primer, 200 μM dNTPs, 100 μg/ml of bovine serum albumin (BSA) and 1U Taq polymerase (DNAzyme, Thermo Scientific), and 200 ng DNA. The following PCR conditions were used: 95°C for 5 min and 94°C for 30s (denaturation), 59°C for 45s (annealing) followed by 72°C for 30s (extension) for 29 cycles and the final extension for 5 min at 72°C. The PCR product of 568-bp was checked on 1.5% agarose gel. And then the PCR product was digested with BsmA1restriction enzyme (New England Biolabs, Ipswich MA) at 55°C. The PCR product was digested into fragments of 222 bp, 135 bp, 128 bp, and 83 bp, which represented mutant genotype (Val/Val), whereas the presence of five bands of 305 bp, 222 bp, 135 bp, 128 bp, and 83 bp was indicative of the heterozygous (Ile/Val) genotype, and presence of three bands of 305 bp, 135 bp, and 128 bp represented the wild genotype (Ile/Ile). Without prior knowledge genotyping of a 15% random sample of cases was carried out twice by different people to ensure quality control and reproducibility was 100%.
Statistical analysis
The study was restricted to the North Indian population with complete information on gender, age, and smoking status. The goodness-of-fit Chi-square test was used to evaluate whether the cases followed Hardy-Weinberg equilibrium (HWE) (p 2 +2pq+q2 = 1; where p and q are the frequencies of wild and variant type, respectively). The odds of LC risk were examined using MedCalc Statistical Software version 14.8.1 (MedCalc Software, Ostend, Belgium). Descriptive data is typically presented as a mean [standard deviation (SD)], median, or number/percentages. Logistic regression analysis was performed for any toxicity, and the odds ratio (OR) with 95% confidence intervals (CIs) were calculated. Overall survival (OS) was computed for all subjects beginning with the first day of chemotherapy and ending with death or last follow-up. The log-rank, p-value, and MST were used to calculate the OS time using the univariate Kaplan-Meir method. The multivariate Cox regression analysis was used to assess the independent effect of the polymorphism on OS when adjusted for different variables affecting OS. A p-value of <0.05 was considered significant for all analyses.
Results
Patient characteristics and clinical predictors
Clinical predictor among cases.

Abbreviations: SD = Standard Deviation, n = total number of lung cancer cases or control subjects, a) p-values were derived from Pearson Chi – square test except age and pack-years; Student t-test was used for age and pack-years. All p-values are two–sided. p<0.05 was considered statistically significant.
Association of the GSTP1 Ile 105 Val polymorphism & clinic-pathological characteristics
The effect of GSTP1 variant SNPs on the various clinic-pathological features such as tumor stage, primary tumor extension, lymph node invasion, and distant metastasis was also calculated (Supplementary Table 3). The patients were stratified into stage III and stage IV patients. The subjects were also classified based on tumor extension into T3 and T4. Based on lymph node invasion, patients were split into Nx+N0+N1 and N2+N3+N4 groups. Further, to classify the patients on the distant metastatic status, they were stratified into M0 and M1 groups. Regarding clinical stage, tumor size, lymph node invasion, and distant metastasis, no interrelation was found between these clinic-pathological features and GSTP1 polymorphism in patients (Supplementary Table 3).
Association of GSTP1 IIe105Val polymorphism and chemotherapy response
The association between GSTP1 IIe 105 Val polymorphism and efficacy outcome of clinical benefit and the response rate were estimated using univariate logistic regression. The association of GSTP1 IIe 105 Val polymorphism with chemotherapy response is summarized in Supplementary Table 4. The patients were divided into good responders (exhibiting complete and partial response) and inadequate responders (those exhibiting stable and progressive disease). The frequency of patients possessing the mutant genotype was nearly identical in the group of responders. The results demonstrated a lack of significant difference in response towards treatment (p = 0.33). Thus, we can conclude that none of the GSTP1 IIe 105 Val genotypes could be used to predict chemotherapeutic response rate or the clinical benefit.
Survival analysis of GSTP1 genotype
Survival among patients of lung cancer categorized with GSTP1 Ile 105 Val genotype.
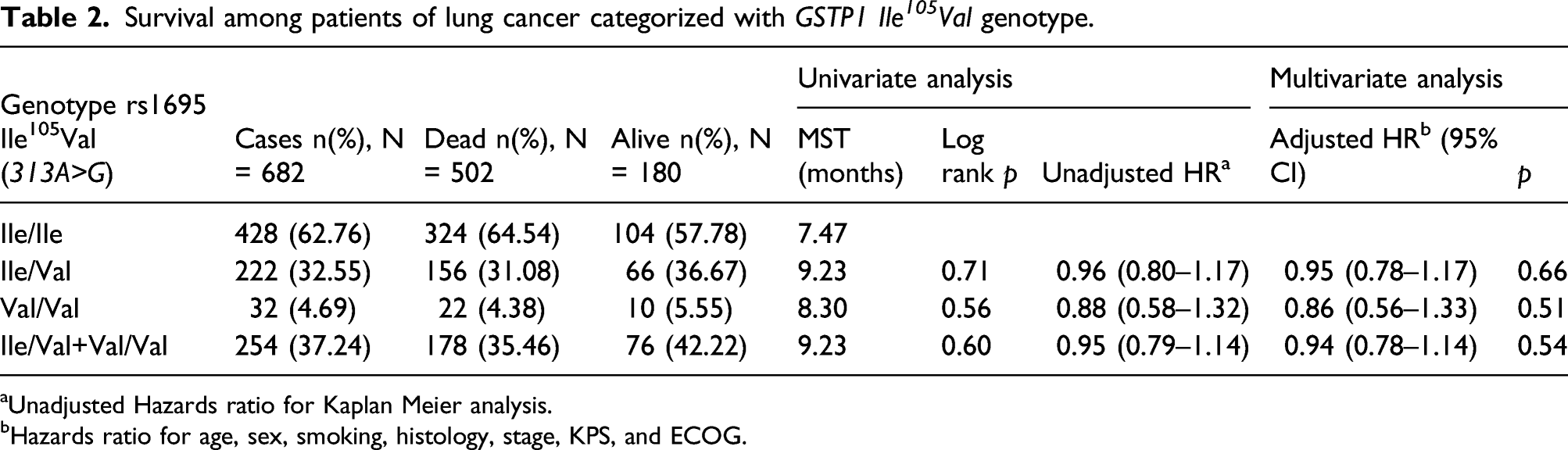
aUnadjusted Hazards ratio for Kaplan Meier analysis.
bHazards ratio for age, sex, smoking, histology, stage, KPS, and ECOG.
Association of GSTP1 IIe 105 Val polymorphism on overall survival according to tumor histology.

aUnadjusted Hazards ratio for Kaplan Meier analysis.
bHazards ratio for age, sex, smoking, histology, stage, KPS, and ECOG.

Kaplan Meier curves illustrating association between overall survival in different genotypes of GSTP1 Ile 105 Val polymorphism in ADCC subjects. (Note: 0 represents wild type genotype, i.e., Ile/Ile, 1 represents heterozygous genotype, i.e., Ile/Val and 2 represents homozygous for mutant genotype, i.e., Val/Val).
Association of GSTP1 IIe 105 Val polymorphism with chemotherapy regimens and OS
Association of GSTP1 IIe 105 Val on overall survival according to chemotherapeutic regimen
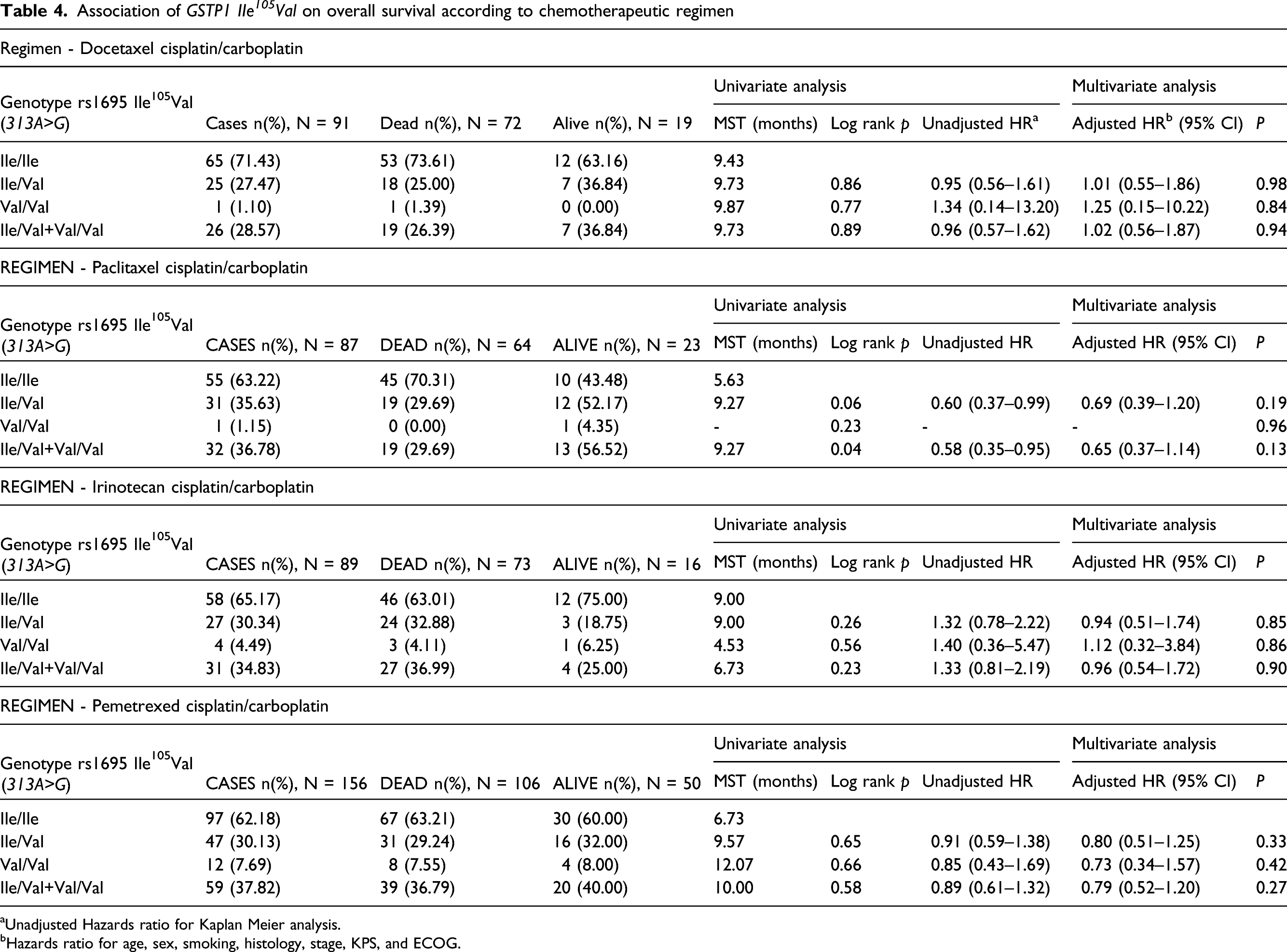
aUnadjusted Hazards ratio for Kaplan Meier analysis.
bHazards ratio for age, sex, smoking, histology, stage, KPS, and ECOG.
Association of GSTP1 IIe 105 Val polymorphism with gender and OS
Applying the univariate analysis, the subjects possessing the heterozygous and combined genotype of GSTP1 IIe 105 Val polymorphism in case of both males (MST = 9.00 vs. 7.33 and MST = 8.30 vs. 7.33, respectively) and females (MST = 13.70 vs. 7.80 and MST = 12.07 vs. 7.80, respectively) correlated with a higher overall survival but was not significant (Supplementary Table 5).
Association of GSTP1 IIe 105 Val on overall survival according to clinico-pathological features
We have also assessed the role of clinic-pathological characteristics concerning GSTP1 IIe 105 Val polymorphism and its impact on the OS of LC subjects. There were, however, no statistically significant correlations found. The associations between GSTP1 IIe 105 Val genotype and clinic-pathological prognostic factors are summarized in Supplementary Table 6.
Association between GSTP1 IIe 105 Val Genotypes and tumor size and its correlation with chemotherapeutic regimens, TNM staging, and histological types
We have also assessed the association between the GSTP1 polymorphism in relation with tumor size and its correlation with chemotherapeutic regimens, TNM staging, and histological types as shown in supplementary tables 7-9. The supplementary table 7 shows that significant differences were observed for patients carrying mutant genotype for GSTP1 polymorphism and having T4 tumor size in comparison to patients having T3 tumors and administered with Pemetrexed + cis/carboplatin (χ2 = 8.85, p = 0.012). When we evaluated tumor size and stage of tumors (Supplementary table 8) it was clearly evident that patients with stage IV tumors had a significant correlation with tumor size as compared to patients with stage III tumors (χ2 = 8.80, p = 0.012). Similarly significant differences were also observed between tumor size when stage III tumors were compared with stage II tumors (χ2 = 7.67, p = 0.02).
Association Between GSTP1 IIe 105 Val Genotypes and Hematological Toxicities
One of the current outcomes of this study was evaluating the toxicity development in LC patients undergoing platinum-based doublet chemotherapy. The odds ratios (OR) and 95% CI were calculated using univariate logistic regression analysis to estimate the risk of developing different toxicity grades after treatment in LC patients. We also used the multivariate regression model to calculate the adjusted ORs, with the dependent variable being the toxicity grades. In the multivariate model, gender, age, performance status, and polymorphism were included as covariates and were thus evaluated for a potential relationship or confounding impacts. We conducted two types of analysis to determine the effect of GSTP1 polymorphism on different toxicity grades. In the first analysis, we compared the presence of any toxicity grade (grades 1–4) versus the absence of toxicity (grade 0), and in the second analysis, we compared the subjects having intermediate/severe toxicity (grades 2–4) versus no/low-grade toxicity (0–1) to comprehensively determine whether the GSTP1 polymorphism was associated with any severe toxicities.
Hematological Toxicities (grades 0 vs. 1-4: grades 0-1 vs. 2-4: grades 0-2 vs. 3–4) associated with GSTP1 IIe 105 Val polymorphisms in multivariate analysis.

OR1 Crude odds ratio, 95% CI: 95% confidence interval, AOR2 Adjusted Odds ratio data were calculated by unconditional logistic regression and adjusted for age, gender, performance status, and type of treatment regimen. A p value < 0.05 was considered statistically significant.
We further analyzed hematological toxicity, especially leukopenia, by comparing patients with no or low-grade toxicity (grades 0–1) to those with intermediate or high grades (grades 2–4). As shown in Table 5, LC patients with the Val/Val (mutant) genotype had an increased risk of leukopenia (OR = 2.41; 95% CI = 1.39-4.18, p = 0.001) as compared to subjects carrying both copies of the wild alleles (Ile/Ile) after adjusting for confounding factors like age, gender, performance status, and chemotherapeutic regimen. No such association was found in the grade 0 versus 1-4 category both in univariate and multivariate groups. Univariate and multivariate analysis showed no statistically significant association between the GSTP1 polymorphism and other hematological toxicities such as thrombocytopenia and neutropenia (data not
We also investigated the role of the GSTP1 polymorphism and hematological toxicities after stratification based upon different histological subtypes of lung cancer. As shown in Table 5, in the case of SCLC patients, the polymorphism of GSTP1 was associated with thrombocytopenia (χ2 test = 7.32, p = 0.02) where the minor allele was predictive for more toxicity. The univariate analysis revealed that SCLC patients carrying mutant genotype had a 9-fold risk for developing thrombocytopenia (OR = 9.14, 95% CI = 1.39-60.17, p = 0.02), and after adjusting for confounding factors, there was still a slight increase in toxicity was observed (OR = 9.75, 95% CI = 1.33-71.52, p = 0.01). Further, on combining both the heterozygous and mutant genotypes and comparing it with the wild genotype, the polymorphism of GSTP1 was found to be associated with thrombocytopenia in SCLC subjects both in univariate and multivariate models (OR = 4.98, 95% CI = 1.50-16.50 p = 0.008). No other relationship between hematological, gastrointestinal and, nephrotoxicity was seen in ADCC and SQCC patients (data not shown).
Discussion
Genetic and environmental factors play a profound role in the development and advancement of LC. On the contrary, the precise mechanism of both these factors is not well elucidated. 14 Data does suggest that polymorphisms in genes that regulate cell signaling, metabolism, DNA synthesis, and response pathways might explain the interindividual differences of drug response, drug resistance, and toxicity. 15 The standard first-line regimen for lung cancer patients is platinum-based doublet chemotherapy; however, the effectiveness of platinum-based chemotherapy has been found to vary among individual patients. 16 Therefore, understanding and subjugating the inter-individual variation towards drug response, sensitivity, and toxicity shall play a vital role in choosing the most effective line of treatment for patients with lung cancer. In light of the current therapeutic scenario used for lung cancer treatment, we endeavored to appraise whether GSTP1 polymorphism influenced survival rates or whether they were associated with toxicity or clinical response to platinum-based chemotherapy.
According to published data, the GSTP1 IIe 105 Val variant may be relevant to the effectiveness of platinum-based chemotherapy in an LC patient. Our data suggest that patients carrying the wild-type alleles for the GSTP1 IIe 105 Val had an abysmal survival time than patients carrying either a single or both mutant alleles for the GSTP1 gene. When stratified based on histological cell types of lung cancer, it was visible that ADCC patients had a better prognosis. According to the findings of a study conducted by Booton et al., 10 the GSTP1 IIe 105 Val polymorphism was associated with NSCLC patient’s survival. A Chinese study has also reported that GSTP1 IIe 105 Val also affects the clinical outcome of NSCLC patients. 17
Similarly, Deng et al. 18 concluded that NSCLC patients harboring the mutant genotype of GSTP1 IIe 105 Val polymorphism had convalescent recurrence-free survival. Jia et al. 19 found that the advanced NSCLC patients with mutant genotype (Val/Val) had improved OS and clinical response. However, some studies that looked into the relationship between GSTP1 IIe 105 Val polymorphism and the prognosis of advanced NSCLC have reported inconclusive results.5,11,12,17,18,20-22 It was observed that Asian lung cancer patients who carried the Val allele of the GSTP1 IIe 105 Val polymorphism had a better survival time and clinical response than patients with the wild-type genotype.5,11,20-22 At the same time, others have reported that the Val allele of GSTP1 IIe 105 Val was associated with poor survival and clinical response.17,18 On the contrary, some studies have reported inconsistent results; a study conducted in the Chinese population by Lu et al. 23 and the Turkish population by Ada et al. 11 reported that GSTP1 IIe 105 Val polymorphism was not associated with a better OS in patients with advanced NSCLC. Furthermore, it has been established that individuals having the mutant form of the GSTP1 gene express a decreased ability to detoxify the drug metabolites, promoting the OS of NSCLC subjects.5,18,21-23
Our study also investigated the impact of GSTP1 polymorphism and response concerning the treatment outcome of LC patients who underwent platinum-based chemotherapy. Our findings reported that none of the GSTP1 IIe 105 Val genotypes could significantly predict chemotherapeutic response rate or clinical benefit. Our findings reported that none of the GSTP1 IIe 105 Val genotypes could significantly predict chemotherapeutic response rate or clinical benefit. Data published so far has indicated that the variant allele of the GSTP1 is linked to the effectiveness and clinical response of platinum-based chemotherapy in LC patients.5,17-19,22,24-26 Booton et al. 11 observed that the GSTP1 IIe 105 Val polymorphism was significantly associated with the clinical response of NSCLC patients. In a Chinese study, Liu et al. 17 in a Chinese study reported that the GSTP1 IIe 105 Val polymorphism affects the clinical outcome of lung cancer patients who have undergone platinum-based chemotherapy. Similarly, other studies conducted in Chinese lung cancer patients have also observed that GSTP1 polymorphism is correlated with chemotherapy response.5,22 A meta-analysis study has reported that the polymorphism in the GSTP1 predicts the response to platinum-based chemotherapy in East-Asian LC subjects. 12 In Caucasian subjects, especially in Greek lung cancer patients, Vlachogeorgos et al. 27 found that the GSTP1 polymorphism may predict the treatment response in patients with advanced NSCLC. However, on the contrary, some reports have conferred no link between the GSTP1 IIe 105 Val polymorphism and response. GSTP1 polymorphism was not associated with treatment outcome in advanced NSCLC by Kalikaki et al. 28 and Ada et al. 11 In a recent meta-analysis study, the GSTP1 IIe 105 Val variant allele was an excellent clinical predictor and had better response rates in Asian lung cancer patients who have undergone platinum-based chemotherapy than Caucasian lung cancer subjects. The inconsistent results in different populations might be due to differences in ethnicities, source of patients, and sample size. Lung cancer is a complex disease with diverse affecting, the anti-cancer therapeutic outcome of platinum-based chemotherapy. It has been reported that GSTP1 mediates the detoxification of platinum-based chemotherapeutic agents and plays an essential role in both intrinsic and acquired resistance to platinum components. Some studies have reported that platinum resistance is not associated with GSTP1. 29 One plausible reason for not being able to exemplify the relationship between the expression of GSTP1 and platinum drugs at any time might be due to the polymorphism of the GSTP1 gene. A single nucleotide substitution (A>G) at position 313 results in replacing isoleucine with valine, resulting in the reduction of the enzyme activity.
Our study observed that the responses to cisplatin-based chemotherapy of lung cancer patients who were homozygous/heterozygous for the variant allele of GSTP1 were similar to the responses observed in patients harboring the wild type of genotype of the GSTP1 gene. Data has suggested that GSTP1 develops resistance to different drugs like etoposide, adriamycin, cisplatin, and carboplatin. 30 Therefore, the favorable outcome exerted by the variant allele of GSTP1 IIe 105 Val cannot be directly linked to a specific drug concerned with treatment response. It might be possible that the response for a tumor to cisplatin might also be attributed to different drug combinations. Another reason that the results obtained here are different from other studies conducted in different tumors; like breast, colon, and leukemia, maybe that the sample size of patients was different, different types of tumors, different resistance mechanisms which might be dominant, and finally, type of chemotherapy regimens applied.
This study also examined whether polymorphisms in the GSTP1 IIe 105 Val were linked with increased toxicity among platinum-based chemotherapy-treated lung cancer patients. We found that patients with the single allele of GSTP1 IIe 105 Val were significantly more likely to develop grade 3 or 4 hematologic toxicity, especially severe anemia toxicity (OR = 2.14, p = 0.03). Our data also revealed that lung cancer patients with the mutant genotype for GSTP1 IIe 105 Val had a 2.5-fold risk of leukopenia (p = 0.001) compared to patients carrying wild genotype. Based on the histological classification, SCLC patients harboring the mutant alleles for GSTP1 IIe 105 Val had more severe hematological adverse effects, especially thrombocytopenia (OR = 9.75, p = 0.01).
The Inter-individual differences of platinum-based toxicity might be because of alterations in the GSTP1 proteins, which reduces the transport capacity for cisplatin in patients with mutant genotype (Val/Val). 31 GSTP1 is a phase II detoxification enzyme that primarily functions in the detoxification of platinum-based compounds. The polymorphism in the GSTP1 gene affects the enzyme’s thermal stability and its conjugation capacity. GSTP1 polymorphism results in GSTP1 modification, which results in GSTP1’s altered ability to detoxify chemotherapeutic agents and modulates drug responses. 32 So, patients with mutant genotype have less enzyme activity, and hence the ability to detoxify the chemotherapeutic agent used is less. Hence such patients retain more of the drug and might show higher toxicity than subjects with the wild genotype. It has been documented that those individuals carrying both the mutant alleles for the GSTP1 IIe 105 Val polymorphism are more susceptible to inflammatory symptoms. 33 The decrease in the activity of GSTP1 in lung cancer subjects harboring the heterozygous or mutant genotype may result in high exposure to platinum-based compounds and thus proportional increase towards drug toxicity because of reduced GSTP1 mediated catalyzed reactions, thus leading to reduced metabolism of platinum-based drugs.
The association of GSTP1 IIe 105 Val genotypes with the clinical outcomes of platinum-based chemotherapy is still controversial. Deng et al. 18 reported that individuals with mutant or heterozygous genotypes had a lower risk of anemia. According to Ma et al., 34 patients with breast cancer who have the GSTP1 Ile/Val or Val/Val genotype were more likely to have an increased incidence of toxicity. According to Kim et al., 35 patients with the GSTP1 wild-type genotype had higher grade 3 or 4 hematological toxicity rates than patients with the mutant GSTP1 genotype. According to our findings, patients with the GSTP1 Val/Val genotype were more likely to have extreme anemia, leukopenia, and thrombocytopenia toxicity.
In summary, our study of North Indian lung cancer patients provides information about the role of GSTP1 polymorphisms in outcome after platinum-based doublet therapy of LC. The current study’s findings support the general hypothesis that subjects expressing the variant form of the GSTP1 enzyme have a reduced capacity to detoxify platinum-based chemotherapeutic drugs, resulting in lower clearance and poor efficacy. Despite the inadequate response, the decreased enzyme activity of GSTP1 also shows a propensity towards severe toxicity. The current study has some deficiencies, such as a weak relationship between toxicity and genotypes, resulting from limited sample size biases and lower toxicity occurrence. Some patients in this study had their procedures/treatment postponed because they could not handle the side effects of platinum-based therapy. We are also aware that histology and varied cisplatin-based chemotherapy treatment might stymie us to reach a firm conclusion, even though the findings were adjusted for confounding factors. Therefore, it becomes imperative to have multiple centers to conduct such studies with many cases. Our study has several strengths, such as many patients who were receiving platinum-based chemotherapy were enrolled, and all the patients were treated in the same hospital. Second, the recruitment of patients and the clinical parameters data collection was carried out independently without knowledge of the GSTP1 polymorphism.
Our general recommendations suggest that GSTP1 genotypes may play an essential role in the pharmacogenetic basis for variability in severe toxicity following platinum therapy treatment in advanced NSCLC patients. However, in vivo, functional studies should be conducted in the future, emphasizing the biological basis of these findings. The pharmacokinetics has to be evaluated to understand the response and toxicity. Better-designed studies with a large set of patients and evaluation of toxicity and end points have to be undertaken to consider taking this polymorphism into the area of personalized medicine.
Supplemental Material
sj-pdf-1-het-10.1177_09603271211059496 – Supplemental Material for GSTP1 Ile 105 Val polymorphism among North Indian lung cancer patients treated using monotherapy and poly-pharmacy
Supplemental Material, sj-pdf-1-het-10.1177_09603271211059496 for GSTP1 Ile 105 Val polymorphism among North Indian lung cancer patients treated using monotherapy and poly-pharmacy by Harleen Kaur Walia, Navneet Singh and Siddharth Sharma in Human & Experimental Toxicology
Footnotes
Acknowledgments
We would like to express our appreciation/gratitude to all of the subjects that took part in this research. The authors thanks to Department of Pulmonary Medicine, Post Graduate Institute of Medical Education and Research (PGIMER), Chandigarh, India for providing samples and Thapar Institute of Engineering and Technology, Patiala, Punjab for providing the required infrastructure to carry out the research work.
Author’s Contribution
HKW performed experiments; SS conceived and designed the research; HKW, NS, and SS analyzed data; NS provided the samples and clinical data for analysis. SS and HKW wrote the paper; and all authors revised and approved the paper.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
“All procedures performed in studies involving human participants were carried out in compliance with the institutional and/or national research committee’s ethical guidelines, as well as the 1964 Helsinki declaration and its corresponding modifications or equivalent ethical standards.”
Informed consent
“Individual participants in the study were asked to provide informed consent.”
Disclosure
Apart from those disclosed, the authors have no other financial involvement or related affiliations with any organization or individual with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. There was no writing assistance used in the development of this manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
