Abstract
In the present study, it is aimed to evaluate the effects of caffeic acid phenethyl ester (CAPE) against acute paw inflammation induced by carragenan (Carr) at macro and micro levels. Therefore, in this study, 1 hour after administering intraperitoneal of indomethacin (Ind) or CAPE (10 and 30 mg/kg body weight) to Sprague Dawley rats, Carr was injected intraplantarly into their right paws. The paw volumes of the rats were measured with a plethysmometer until the 4th hour. Also, X-ray and thermal camera images were taken to determine edema and temperature changes. At the end of the study, after the paw tissues and serums were taken, oxidative stress and inflammation status were determined using biochemical, molecular, and western blot techniques. In addition, lipid and protein profiles in paw tissue were determined using HPTLC and electrophoresis methods. The results depicted that a high dose of CAPE against Carr-induced inflammation may be almost as effective as Ind used as reference.
Introduction
Inflammation is the first reaction of the organism to ensure protection against injuries caused by various internal and external factors such as physical damage, chemical factors, microbial attack, and cancer.1–3 Mild levels of inflammation may occur during the day due to reasons such as stress, radiation, and intense physical activity in living beings. 4 Inflammation progression accelerates with the combined effect of the inherited and acquired immune system. It is accompanied by immune cells such as macrophages, dendritic cells, lymphocytes, and neutrophils. Moreover, various inflammatory mediators, including interferons, interleukins, and prostaglandins, are rapidly released into the blood in response to the inflammatory reaction and cause the acute inflammatory reaction to progress. 5 In this way, the uncontrolled progression of inflammation and its occurrence at high levels causes inflammatory diseases, leading to dysfunctions in various organs.3,6,7 In such cases, pharmacological intervention is required to reduce pain and increase life quality.4,7
Steroidal anti-inflammatory drugs (SAIDs) and nonsteroidal anti-inflammatory drugs (NSAIDs) are currently used against inflammation. 7 NSAIDs, including indomethacin (Ind), inhibit the activities of cyclooxygenase-1 (COX-1) and cyclooxygenase-2 (COX-2) enzymes, preventing the synthesis of thromboxanes and prostaglandins, and thus display antipyretic, analgesic, and anti-inflammatory effects.4,8 SAIDs contain glucocorticoid drugs with anti-inflammatory properties. They inhibit inflammatory genes by binding to glucocorticoid receptors as well as activating anti-inflammatory genes. 8 However, both SAIDs and NSAIDs have side effects that cause serious health problems. For instance, SAIDs cause obesity and osteoporosis, 8 while NSAIDs cause cardiovascular diseases, gastrointestinal ulcers, renal function disorders, and hepatotoxicity.6,7 Therefore, there has been an urgent need to discover new drugs that may be more effective and safer against inflammation. Naturally-derived compounds have entered the agenda of scientists as a promising strategy in this way.
Studies on polyphenols are increasing among natural compound groups.9,10 Caffeic acid phenethyl ester (CAPE), a phenolic acid derivative first identified in propolis, 11 contains catechol, which is a powerful antioxidant in its structure. 12 CAPE has been reported to have immunomodulatory, antiviral, antiproliferative, anti-cancer, chemopreventive, and wound healing properties, 11 and its use in traditional medicine has greatly increased in recent years. 13 On the other hand, the mechanisms of the reported features of CAPE have not been clearly elucidated. On the other hand, the effects of CAPE against λ-Carrageenan–induced paw edema have not been studied before.
Paw edema caused by intraplantar injection of carrageenan (Carr) which is a seaweed polysaccharide is a reliable method frequently used by researchers to evaluate the anti-inflammatory properties of drugs in acute inflammation.2,4,8,14 Immediately after subcutaneous administration, edema, hyperalgesia, and erythema occur in the paw with the effect of proinflammatory agents such as histamine, bradykinin, tachykinin, reactive oxygen, and nitrogen species.4,8 Furthermore, it is known that the increase in lipid peroxidation and the production of reactive oxygen species and the decrease in antioxidant enzyme activities contribute greatly to the development of inflammation. 6 In this model, inflammation reaches its highest levels within a period of 4 hours. 4
In the present study, the anti-inflammatory properties of CAPE against paw edema induced by Carr were investigated. For this purpose, temperature and humidity ratios of paws, paw volumes, lipid and total protein profile, cytokine and acute phase protein levels, and oxidative stress status were analyzed.
Material and Methods
Chemicals
Indomethacin (25 mg) used in the study was obtained from Deva Farma (Istanbul-Turkey). All other chemicals including caffeic acid phenethyl ester (≥97% (HPLC), powder, MW:284.31) and λ-Carrageenan were purchased from Sigma-Aldrich (St. Louis, MO, USA).
Ethics committee approval and animals used in the study
Ethics committee approval of the study was obtained from Atatürk University Experimental Animals Local Ethics Committee (Approval No: 2020/13/195). The 10–12-week-old male Sprague Dawley rats weighing 280–300 g used in the study were obtained from the Atatürk University Medical Experimental Practice and Research Centre and were cared for in this center. Their feeding was provided ad libitum with standard pellet feed and tap water. The ambient conditions which they sheltered in had 24 ± 1°C and 45 ± 5% humidity. In addition, animals were kept on a 12-h dark and 12-h light cycle. Before starting the experiment, animals were adapted to the environment for 7 days.
Design of the study
In the experiment, a total of 35 rats were divided into five groups, seven in each group. Doses of Carr, Ind, and CAPE were determined with reference to previous studies.7,14–16 The groups were designed as given below; 1. Control Group: One hour after administering intraperitoneal saline, physiological saline was injected intraplantarly into the right hind paw. 2. Carr + Ind Group: One hour after administering Ind (10 mg/kg body weight) intraperitoneally, 0.1 mL of 1% Carr was injected intraplantarly into the right hind paw.
7
3. Carr Group: One hour after administering intraperitoneal saline, 0.1 mL of 1% Carr was injected intraplantarly into the right hind paw.
14
4. Carr + CAPE 10 Group: One hour after administering CAPE (10 mg/kg body weight) intraperitoneally, 0.1 mL of 1% Carr was injected intraplantarly into the right hind paw.
15
5. Carr + CAPE 30 Group: One hour after administering CAPE (30 mg/kg body weight) intraperitoneally, 0.1 mL of 1% Carr was injected intraplantarly into the right hind paw.
16
Motility test
The motility test was designed based on a previous study. In this test, the movements of the rats were monitored for 5 min, and a score of 0–3 was given according to walking difficulties. Rats who could not walk were scored 0, those who walked with difficulty and avoided touching the floor with the fingers of the edematous paw were scored 1, those who walked with difficulty and touched the floor of the edematous paw fingers were given 2, and those who walked easily were given 3 points. 1
Measurement of paw volumes
The volume of paw edema was determined by plethysmometer (Ugo Basile, Italy) just before and 1, 2, 3, and 4 hours after carrageenan administration. Measurement results are given in microliters.
Taking thermal and X-ray images
Thermal images of the paws were taken by thermal imager (testo 885-2, Germany) just before the carrageenan administration and 1, 2, 3, and 4 hours after the administration. The images were analyzed digitally with the program of the camera, and the average temperature and humidity of the paws were determined.
At the 4th hour of the administration, animals were put under mild sevoflurane anesthesia and lateral radiographs of the paws were taken by X-ray (meX +100, Oberhausen-Germany).
Collection of samples
Immediately after the measurements at the 4th hour, the rats were decapitated under mild sevoflurane anesthesia and their blood and right paws were taken. Serums were separated by centrifuging the blood at 4000 r/min for 10 minutes. Serums and paw tissues were stored at −20°C until biochemical and molecular analysis.
Determination of the oxidative stress state of the paws
Paws were homogenized by means of a homogenizer (Tissue Lyser II, Qiagen, Netherlands). The tissues homogenized were diluted with the ratio of 1:15 with 1.15% KCl and homogenized again by means of a homogenizer. Homogenates were centrifuged at 1000 × g and +4°C for 15 min for superoxide dismutase (SOD), catalase (CAT), and malondialdehyde (MDA) measurements. In order to analyze glutathione peroxidase (GPx) activity and glutathione (GSH) levels, they were centrifuged at 9000 × g and +4°C for 20 minutes. MDA analyses were performed by the method of Placer, Cushman. 17 SOD, CAT, and GPx activities were determined by the methods developed by Sun, Oberley, 18 Aebi, 19 and Matkovics, 20 respectively. GSH levels were analyzed by Sedlak and Lindsay 21 method. Total protein amounts in paw tissues were determined by the method of Lowry, Rosebrough. 22
Determination of acute phase protein levels in serum
C-reactive protein (CRP) and albumin (ALB) levels in serum were analyzed according to the manufacturer’s instructions by means of commercial kits (Randox Pvt. Ltd., UK) to determine the level of acute phase response.
Hematological analysis
Hematological analyses (WBC: white blood cells, RBC: red blood cells, HGB: hemoglobin, PLT: platelet, NEU: neutrophil) of blood taken into anticoagulant tubes were performed with an automatic cell counter (Sysmex KX-21N, Japan).
Real-time PCR analysis
Primer sequences.

Sodium dodecyl sulfate polyacrylamide gel electrophoresis
Tissue samples were mixed 1:10 with 5% sodium dodecyl sulfate (SDS) and homogenized with TissueLyser LT (Qiagen, Hilden, Germany). Tissue homogenates were diluted at 1:5 with SDS-PAGE denaturing sample buffer and 10 μL of the mixture was loaded into each well. The electrophoresis was carried out in Tris-glycine-SDS running buffer at 200 V/gel for 70 min using 4% stacking and 10% resolving gel (Laemmli 1970). After completion of running, proteins were treated with Oriole fluorescent stain (Bio-Rad, Herkules, USA).
The electrophoretograms were then visualized using the GelDoc XR gel documentation system (Bio-Rad, USA) and were analyzed by the ImageLab 5.2 software (Bio-Rad, USA).
Western blotting analysis
The soft tissues from each right paw were removed and homogenized in RIPA lysis buffer containing protease inhibitor cocktail and phenylmethylsulphonyl fluoride (PMSF) at 4°C. The homogenates were centrifuged at 12000 × g for 20 minutes and the supernatant was used for western blot analysis. Protein concentration was measured by PierceTM BCA Protein Assay Kit (Rockford, IL, USA) using bovine serum albumin (BSA) as standard. 30 μg of protein from the supernatant was dissolved in Laemmli sample buffer (Tris-HCl pH: 6.8, glycerol, sodium dodecyl sulfate, β-mercaptoethanol, bromophenol blue) and separated on 10% sodium dodecyl sulfate polyacrylamide gel (SDS-PAGE). Afterward, it was transferred to polyvinylidene fluoride (PVDF) membranes. Following transfer, membranes were blocked for 1.5 h with %5 BSA in phosphate-buffered saline with 0.1% Tween 20 (PBST). After blocking, membranes were washed 5 times in PBST for 5 min and each of which, mouse monoclonal primary antibodies β-actin (sc-47778), IL-1β (sc-52012), NF-κB p65 (sc-8008), intercellular adhesion molecule 1 (ICAM-1) (sc-8439) and TNF-α (sc-52746), was incubated at 4°C for overnight. After application of the primers, membranes were washed 5 times in PBST for 5 min and left for 1.5 h in the presence of goat anti-mouse IgG secondary antibody conjugated to horseradish peroxidase (1:2000 dilution, sc-2005). Protein bands were detected using enhanced chemiluminescence reagent ClarityTM Western ECL Substrate (Bio-Rad, Hercules, USA) and visualized with using ImageQuant LAS 500 (GE Healthcare Bio-Sciences AB, Uppsala, Sweden). Relative density of the bands was quantified with using Image J software (NIH, Bethesda, USA).
High performance thin layer chromatography (HPTLC)
An n-hexane/isopropanol (500 μL) mixture at a 3:2 ratio (v/v) was added to 1000 μl of tissue homogenates in Eppendorf tubes. After vigorous vortexing for 1 min, the tubes were centrifuged at 5000 g at +4°C for 5 min, and the hexane phases were collected. 24 A standard lipid mixture (cholesteryl oleate, triolein, cholesterol, and l-α-phosphatidylcholine) and tissue extracts were spotted on the high performance thin layer chromatography (HPTLC) plates and developed with an n-hexane/diethyl ether/acetic acid mixture (80:20:2) (v/v/v). Then, the entire plate was dipped in 10% CuSO4 (w/v), in 8% H3PO4 (v/v), and lipid classes on the plates were charred at 120°C. Tissue lipids were separated into the following classes: cholesterol esters (ColE), triacylglycerols (TAG), free cholesterol (fCol), and phospholipids (PL).
The HPTLC chromatograms were analyzed as described by Kaynar, Ileriturk, 25 and results were obtained as percentage of individual lipid class in the total lipid content.
Statistical analysis
In the study, one-way variance analysis (ANOVA) and Tukey’s multiple comparison test were performed using SPSS 20.0 software for all parameters, except for the motility test. Results are given as mean ± SD. Kruskal–Wallis test was used for motility scoring, and the results were expressed as median.
Results
Macro changes
Evaluation of changes in paw volume
The results of the paw volume measured before the carrageenan administration and at the 1st, 2nd, 3rd, and 4th hours of the administration, and paw images are shown in Figure 1. According to the results, it was seen that both doses of CAPE administration significantly reduced edema formation compared to the Carr group (p < .001). On the other hand, it was determined that the CAPE 30 group was more effective than the low dose (p < .05), and there was no significant difference between the CAPE 30 and Ind group. Effects of Carr, Ind, and CAPE on paw volume of rats. A: Control, B: Carr + Ind, C: Carr, D: Carr + CAPE 10, E: Carr + CAPE 30. Values are given as mean ± SD. ***p < .001, **p < .01, *p < .05 Carr versus Others; ###p < .001, ##p < .01, #p < .05 Control versus Others.
Radiographic evaluation of the paw
As can be seen in Figure 2, it was determined that soft tissue swelling occurred in the group with Carr administration compared to the control group; however, this swelling was significantly reduced with CAPE administration. Among the CAPE doses, it is noteworthy that the high dose is more effective. X-ray images confirm the results from the paw volume measurement. Radiographic examination of the effects of Ind and CAPE administrations against Carr-induced paw edema in rats. A: Control, B: Carr + Ind, C: Carr, D: Carr+CAPE 10, E: Carr+CAPE 30.
Evaluation of paw temperature and moisture percentages
Images taken with a thermal camera are given in Figure 3(a)–(e)/I and temperature analyses are given in Figure 3/II. It is seen from both images and temperature analysis that Carr administration caused inflammation and significantly increased paw temperatures compared to the control group (p < .001). However, it was determined that CAPE significantly decreased paw temperatures by alleviating inflammation compared to the Carr group (p < .001). Among the CAPE doses, it was found that high dose administration was more effective (p < .01). Also, Ind, which was used as an anti-inflammatory reference drug, was more effective than the CAPE group given at a dose of 30 mg/kg (p < .01).
Based on the humidity rates given in Figure 3/III, it was determined that the humidity rate reduced with the increasing temperature in the Carr group (p < .001). Still, due to the CAPE and Ind administrations reducing the paw temperatures, it was observed that the humidity rates increased compared to the Carr group. It was determined that Ind caused more moisture increase compared to CAPE groups, and among the CAPE doses, the highest effect was found in high dose administration (p < .001).
Evaluation of lipid profile in paw tissue by HPTLC method
Lipid profile results analyzed in paw tissue by HPTLC method are presented in Figure 4 and Table 2. Accordingly, it was observed that lipid destruction started in tissues with the inflammation caused by Carr and the TAG level in the total tissue lipid composition was 70% in the control group, while it decreased up to 64% in the group given Carr. On the other hand, although it was observed that Ind and 10 mg/kg CAPE injection decreased lipolysis numerically, it was determined that 30 mg/kg CAPE showed the statistically most significant effect. In this group, the TAG level in the total tissue lipid composition was found to be 69%. Similar to TAG, it was observed that phospholipid degradation was the most in the CAR group, while the least was in the group treated with 30 mg/kg CAPE. Effects of Carr, Ind, and CAPE on the lipid profile of rat paw tissues. ColE: Cholesterol ester, TAG: Triacyl glycerol, fCol: Free cholesterol, PL: Phospholipid. Effects of Carr, Ind, and CAPE on the lipid profile of rat paw tissues. ColE: Cholesterol ester; TAG: Triacyl glycerol; fCol: Free cholesterol; PL: Phospholipid. Values are given as mean ± SD. ***p < .001, **p < .01, *p < .05 Carr versus Others; ###p < .001, ##p < .01, #p < .05 Control versus Others.

Evaluation of protein profile in paw tissue by SDS-PAGE method
When the tissue protein profile was examined, it was seen that the amount of tissue protein increased by 1.15 times due to Carr-induced edema (Figure 5 vs Table 3). While this increase reduced to 1.05 after indomethacin injection, it reduced to 1.10 and 1.04 after 10 and 30 mg/kg CAPE injection, respectively. Effects of Carr, Ind, and CAPE on total protein profile of paw tissues of rats. Effects of Carr, Ind, and CAPE on total protein profile of paw tissues of rats. TP: Total protein. Values are given as mean ± SD. ***p < .001, **p < .01, *p < .05 Carr versus Others; ###p < .001, ##p < .01, #p < .05 Control versus Others.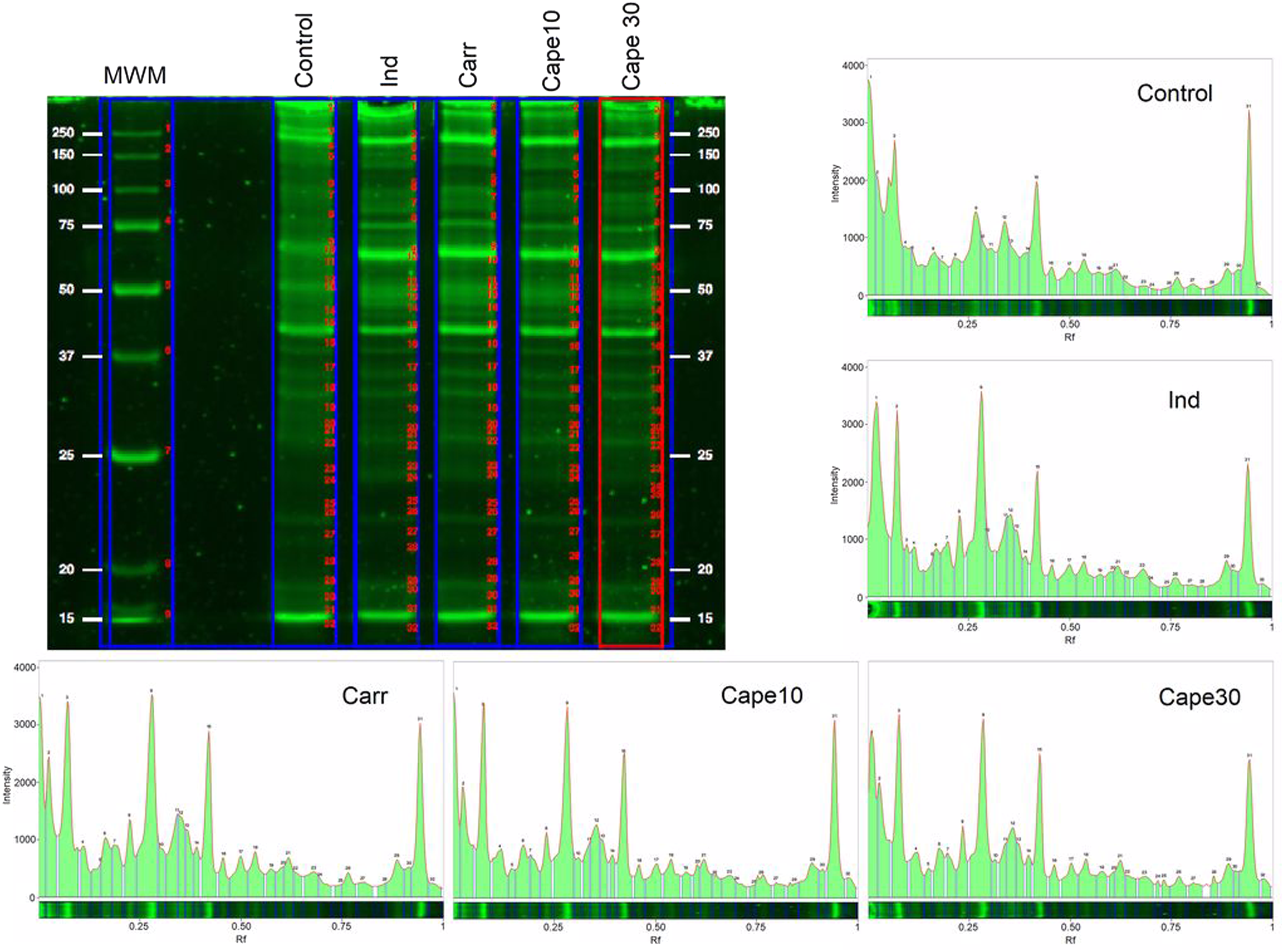

Motility evaluation
In the current study, the motility test was applied to the rats at the highest level of inflammation and graded from 1 to 3 according to the disruptions in walking. According to the observations made, the median of the Carr group was the group with the highest malfunction with a value of 1 ± 0.24 (p < .01). There was no statistically significant difference between the CAPE groups and the reference drug. The results of the motility test are summarized in Figure 6. Effects of Carr, Ind, and CAPE administrations on motility in rats. Values are given as median ±SD ***p < .001, **p < .01, *p < .05 Carr versus Others; ###p < .001, ##p < .01, #p < .05 Control versus Others.
Micro changes
Evaluation of hematological analysis and MPx expression results
Effects of Ind and CAPE administrations on hematological parameters against Carr-induced paw edema in rats.
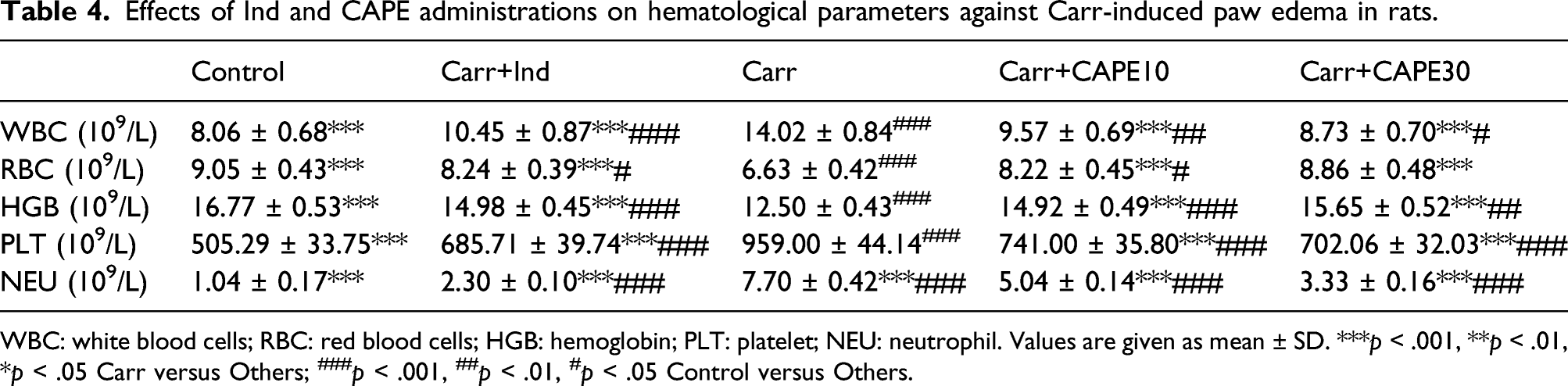
WBC: white blood cells; RBC: red blood cells; HGB: hemoglobin; PLT: platelet; NEU: neutrophil. Values are given as mean ± SD. ***p < .001, **p < .01, *p < .05 Carr versus Others; ###p < .001, ##p < .01, #p < .05 Control versus Others.

Effects of Carr, Ind and CAPE administrations on MPx activities in paw tissue of rats. MPx: Myeloperoxidase. Values are given as mean ± SD. ***p < .001, **p < .01, *p < .05 Carr versus Others; ###p < .001, ##p < .01, #p < .05 Control versus Others.
Evaluation of oxidative and antioxidant markers in paw tissue
The effects of Carr, CAPE, and Ind administrations on oxidative and antioxidant markers in paw tissue are given in Figure 8(a)–(e). The results showed that Carr administration increased MDA levels by causing lipid peroxidation. In addition, it was determined that the levels of enzymatic and non-enzymatic antioxidants in the paw tissue decreased compared to the control group with Carr administration (p < .001). Also, Ind and different CAPE doses were found to induce SOD, CAT, and GPx activities and increase GSH levels. Accordingly, it was observed that the formation of MDA decreased following the reduction of lipid peroxidation. It was determined that the CAPE 30 dose was more effective in the comparison of MDA levels although there was no statistically significant difference in the levels of antioxidant markers between the doses of CAPE.
RT-PCR analysis results of mRNA transcript levels demonstrated that Nrf2 and HO-1 expressions were suppressed by Carr administration. Different CAPE doses and the reference drug activated these genes and provided an important protection against oxidative stress. Moreover, it was observed that high dose of CAPE triggered Nrf2 and HO-1 expression more than low dose (p < .001) (Figure 9(a) and (b)).
Evaluation of inflammatory markers in paw tissue
Expression levels of important inflammatory markers, NF-κB, IL-1β, TNF-α, iNOS, COX-2, STAT3, and MAPK were evaluated in paw tissue by RT-PCR. The data obtained showed that Carr administration significantly increased the mRNA transcript levels of MAPK, STAT3, NF-κB, IL-1β, TNF-α, iNOS and COX-2 compared to the control group. It was determined that the Ind, NSAID agent, decreased the expression levels of all other markers, especially COX-2, compared to the Carr group (p < .001). While CAPE administration significantly decreased the expression of target genes compared to the Carr group, the expression levels of STAT3, TNF-α, andCOX-2 did not make a significant difference between the CAPE30 and Ind group. mRNA transcript levels are presented in Figure10(a)–(g).
In addition to the RT-PCR analysis results of NF-κB, IL-1β, and TNF-α, protein analyses were performed by western blot. According to the results, it was determined that Carr administration to the paw caused an increase in protein levels of NF-κB, IL-1β, and TNF-α compared to the control group, however, CAPE decreased the levels of these pro-inflammatory cytokines depending on the dose. There was no significant difference in NF-κB, IL-1β, and TNF-α protein levels between the CAPE 30 group and the Ind group. All results are shown in Figure 11.
Evaluation of ICAM-1 protein levels in paw tissue by western blot method
It was observed that Carr administration to the paw caused an increase in ICAM protein levels compared to the control group (p < .001). It was determined that Ind and CAPE administrations decreased ICAM levels in paw tissue compared to Carr group, and high dose of CAPE was more effective than low dose (p < .001). All results are shown in Figure 11.
Evaluation of acute phase protein levels in paw tissue
Results of albumin and CRP, which are acute phase proteins, are given in Figure 12. Accordingly, it was determined that Carr administration significantly increased the CRP levels, which is a positive acute phase protein, and decreased the levels of albumin, which is a negative acute phase protein, compared to the control group (p < .001). It was observed that increased CRP levels with Carr administration decreased in a dose-dependent manner after CAPE administration, while albumin levels increased (p < .01). It was determined that there was no statistically significant difference between Ind and CAPE 30.
Discussion
Nonsteroidal anti-inflammatory drugs have a very rapid and significant curative effect against inflammation. Therefore, they are widely used for the treatment of various inflammatory diseases. However, using these drugs for a long time may cause various toxicities. 3 For this reason, researchers work hard to develop herbal medicines that can be used as an alternative to NSAIDs. In this study, the anti-inflammatory effects of CAPE against Carr-induced paw edema, which is a standard experimental model of acute inflammation, were evaluated. CAPE significantly reduced Carr-induced paw edema.
The first change observed in the study was obtained by measuring the paw volume at 1st, 2nd, 3rd, and 4th hours. While large swelling occurred in the Carr group compared to the control group, it was observed that Ind or different doses of CAPE reduced the swelling caused by Carr. These differences can also be seen in the images taken of the paw volume presented graphically. Additionally, the radiographic images of the paw clearly reflect the differences in edema size. Previous studies have shown that paw volume increases with Carr administration, whereas various natural metabolites may reduce swelling by alleviating the formation of edema.2,26
Another aspect in which the degree of inflammation was evaluated in the present study was the measurement of paw temperatures. It is a fact that the temperature increases especially in the inflammatory region in case of inflammation.27–29 In the literature, we have not encountered a method in which thermal images of paw temperatures are taken and temperatures are calculated in Carr-induced paw edema. In this study, the temperatures of the paws were monitored with a thermal camera at certain intervals before and after the administration, and the average temperature values of the paws were recorded with the program of the camera. According to the results, it was determined that the paw temperature increased gradually with the Carr administration; on the other hand, it diminished after increasing to a certain degree with the Ind and CAPE administrations. These results provide important evidence for the anti-inflammatory properties of CAPE in the paw edema model.
Production of excessive amounts of reactive oxygen species in cells causes oxidative stress. 30 In case that oxidative stress is out of control, it can have dangerous consequences for cells. 6 One of these consequences is inflammation. 31 Researchers have reported that increased free radical production at the inflammation site is one of the triggering causes of tissue damage caused by various inflammatory disorders. 2 It is considered that the severity of many inflammatory diseases can be reduced by controlling oxidative stress. In this study, the effects of CAPE on oxidative stress in case of acute inflammation caused by Carr were investigated. For this purpose, lipid peroxidation levels, which is an important marker of oxidative stress in paw tissue, 6 were measured. The results showed that, similar to the literature, Carr administration caused an increase in lipid peroxidation. 32 However, it is thought that CAPE prevents the formation of free radicals by displaying antioxidant properties due to the hydroxyl groups in its structure and thus decreases the lipid peroxidation levels.
Body has enzymatic and non-enzymatic antioxidant defense mechanisms that play an important role in balancing oxidative stress. 33 Nevertheless, the suppression of these mechanisms by various factors or the excessive increase in ROS production cause this balance to be deteriorated. 34 Reactivation of these mechanisms provides an important protection against oxidative stress. In studies on toxicity induced in various tissues by different toxic agents, it has been reported that CAPE increases enzymatic and non-enzymatic antioxidant levels, alleviates oxidative stress, and thus protects tissues from the destructive effects of these toxic agents.35–37 In our study, it was determined that SOD, CAT, and GPx enzymes suppressed by Carr, as well as GSH, the levels of which, we observed their significant decreases, increased significantly with CAPE administration. SOD is responsible for the conversion of the superoxide radical to hydrogen peroxide. Hydrogen peroxide, which is an extremely toxic compound for cells, is broken down into water and molecular oxygen by CAT and GPx. 38 In the study, it is thought that CAPE increases SOD activity by scavenging superoxide radicals, and accordingly, there is an increase in CAT and GPx levels. Also, it can be said that GSH regenerates and lipid peroxidation decreases depending on the decreasing ROS.
The HO-1 enzyme, which catalyzes heme metabolism, is one of the important antioxidant defense mechanisms in the body. HO-1 expression is regulated by the Nrf-2 pathway. It is known that providing Nrf2 activation contributes significantly to anti-inflammation by up-regulating HO-1 expression. 39 Therefore, in this study, the anti-inflammatory effects of CAPE against Carr-induced paw edema were evaluated through the Nrf2 pathway. The results obtained suggest that CAPE up-regulates HO-1 levels with the increase in Nrf2 expression, thus contributing significantly to the alleviation of inflammation. In line with our results, it has been reported in previous studies that various natural compounds trigger HO-1 activity by increasing Nrf2 levels.40,41
During the acute inflammatory response, immune cells such as neutrophils are activated. 42 In particular, infiltration of neutrophils into the inflammation area has an important role in providing inactivation of antigens. However, the excessive occurrence of these events and the increase in MPO levels, which are secreted from the azurophilic granules of neutrophils and reflect the tissue neutrophil content, cause exacerbation of tissue damage.43,44 Mohsin and Kurup 45 reported in their study that inhibition of MPO activity can prevent neutrophil infiltration and have a significant effect against inflammation. In the present study, it was observed that MPO expression was triggered and inflammation was exacerbated in connection with the increase in WBC and NEU numbers with Carr administration. It was determined that platelets also contribute to the inflammatory process by interacting with leukocytes, and in our study, Carr administration significantly increased PLT numbers. 46 It was also seen that with the administration of CAPE, there was a significant decrease in the number of WBC, NEU, and PLT depending on the dose, and this alleviated the inflammation caused by Carr.
Intercellular adhesion molecules (ICAMs) are cell surface glycoproteins expressed in a wide variety of cell types with different gene regulation and effector functions. Due to its wide distribution and specific regulation, ICAM-1 is the most extensively studied of the five ICAM molecules. 47 It has been reported that ICAM-1 may play a role in leukocyte transmigration in the second phase of inflammation and thus activate MPO by causing neutrophil immunoreactivity. 48 In line with previous studies, it was observed in our study that there were increases in ICAM-1 protein levels in correlation with increased MPO activity after Carr administration.48,49 However, it was determined that ICAM-1 levels decreased with CAPE administration, and thus, MPO activity was suppressed.
NF-κB, the main regulatory transcription factor, plays an important role in the exacerbation of inflammatory diseases.39,50 The main relationship of NF-κB with inflammation is based on the synthesis of pro-inflammatory mediators. 51 NF-κB is activated by translocation into the nucleus and activates inflammation-related genes such as iNOS, COX-2, TNF-α, and IL-1β. 52 Therefore, it was thought that by preventing the activation of NF-κB, the genes of iNOS, COX-2, TNF-α, and IL-1β could be suppressed and a significant decrease in the severity of inflammatory diseases could occur. 53 Our results confirmed this hypothesis. In fact, it was observed that the levels of iNOS, COX-2, TNF-α, and IL-1β increased along with NF-κBafterCarr administration was applied to the paws. It triggered the development of edema by causing inflammation in the paw. On the other hand, it was observed that Ind, a COX-2 inhibitor, suppressed both NF-κB levels and other inflammatory mediators. Similar to Ind, CAPE showed anti-inflammatory properties by controlling NF-κB expression. In previous studies, it was reported that NF-κB and inflammatory cytokines which increased with Carr administration were suppressed by giving various herbal product sand relieved inflammation.54–56
An important upstream component of NF-κB is the MAPK proteins consisting of p38. 53 Recently, researchers have shown great interest in MAPK inhibition.57,58 The activation of MAPKs causes an increase in the expression of genes that cause various biological responses, including pro-inflammatory factors. 53 Therefore, it was considered that suppression of MAPK expressions might have an important effect on alleviating inflammatory reactions. For this purpose, the effects of CAPE against Carr-induced paw edema were evaluated over MAPK expression. The results were promising. CAPE was found to lower the increased MAPK expression levels with Carr, similar to the Ind group, and significantly reduce the severity of inflammation. In previous studies, it has been reported that by inhibiting MAPK activity, NF-κB can be suppressed and significantly alleviate inflammation.59,60
STAT3, which is a member of the STAT family and a transcription factor, plays an important role in various pathophysiological conditions, including inflammation. 61 STAT3 increases the transcription of inflammatory genes that are at the target of NF-κB by translocating into the nucleus after forming a complex with NF-κB. 62 In our study, the fact that the Carr administration increases the levels of inflammatory mediators in parallel with the expression levels of NF-κB and STAT3 coincides with this information. In previous studies, it has been reported that various toxic substances trigger the inflammatory process in different tissues by increasing the expression of STAT3.63,64 However, inflammation was found to be significantly suppressed with the decrease in STAT3 levels in the paw tissues of rats given CAPE.
The acute phase response is the reaction to the impairment of local or systemic homeostasis caused by various conditions such as infection, tissue damage, trauma, or surgery. A few hours after the situation that causes acute phase response, protein synthesis in the liver changes greatly, causing some proteins to increase and some to decrease. Among these, CRP increasing (positive acute phase protein) and ALB decreasing (negative acute phase protein) are acute phase proteins. 65 In our study, similar to the literature, it was observed that while the serum of rats treated with Carr was increased in CRP levels, ALB levels were decreased.5,43,45 However, it was determined that CAPE administration alleviated tissue damage and brought the levels of acute phase proteins closer to the control group levels.
In the study, the relationship between the development of edema and tissue total protein and lipid profile was investigated. According to the results of the lipid profile analyzed by HPTLC method, it was determined that lipid destruction occurred due to Carr-induced inflammation, and thus, TAG and phospholipid levels were significantly reduced. This is in line with the statement reported in a previous study that NF-κB can activate endothelial lipase in case of inflammation and this can show triglyceride lipase and phospholipase activity. 66 In addition, it was observed that there was a remarkable increase in the amount of total protein in the paw due to edema and dehydration caused by Carr. However, it was determined that CAPE decreased the total protein amount and lipid breakdown. We have not come across a sample study evaluating these methods from this point of view.
Conclusion
Taken together, it was determined that CAPE suppresses the expression of inflammatory mediators by reducing the number of immune cells against Carr-induced acute inflammation in micro-analyses and thus attenuates inflammation. Various macro analyses conducted in the study also presented that CAPE has an anti-inflammatory effect, resulting in a significant reduction in paw edema.
Footnotes
Author contributions
Halil Sezgin Semis: Conceptualization, methodology, formal analysis, investigation; Cihan Gur: Writing—review and editing, data curation, methodology; Mustafa Ileriturk: Data curation, methodology, conceptualization; Ozgur Kaynar: Data curation, methodology; Fatih Mehmet Kandemir: Data curation, methodology, writing—original draft.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Ethics committee approval of the study was obtained from Atatürk University Experimental Animals Local Ethics Committee (Approval No: 2020/13/195).
