Abstract
Resveratrol has been reported as an ideal medicine in the treatment of colorectal cancer. Meanwhile, cadmium could affect the occurrence and development of tumors in various ways. Epithelial–mesenchymal transition is a major progress regulated with colorectal cancer (CRC). We aimed to determine the effect and mechanism of resveratrol on the Cd-promoted EMT in CRC cells. First, we investigated the migration and invasion of CRC cells with or without the treatment of different concentrations of Cd in vitro by the transwell assay. Second, Western blot and RT-qPCR assay were used to detect the expressions of EMT-related markers (ZEB1, vimentin, E-cadherin, and N-cadherin) in Cd-exposed CRC cells. Subsequently, after treating with different concentrations of resveratrol, the migration and invasion of Cd-exposed CRC cells were detected again, as well as the expressions of EMT-related markers. Moreover, m6A-related RNAs in Cd-exposed CRC cells after treating with resveratrol were immunoprecipitated and validated by Me-RIP and RT-qPCR. These indicated that Cd promoted the migration and invasion of CRC cells. In addition, Cd up-regulated the expressions of N-cadherin, vimentin, and ZEB1, while it down-regulated that of E-cadherin in CRC cells. Resveratrol could reverse the Cd-promoted migration, invasion, and EMT procession by regulating the expression of ZEB1.
Keywords
Introduction
Colorectal cancer (CRC) is one of the most common malignant digestive tract tumors globally, and the mortality of CRC is second to lung cancer and liver cancer, which seriously endangers human health. 1 Despite the continuous innovation of treatment, the therapeutic effect and prognosis of CRC are still not satisfactory. Research findings indicated that most patients with CRC eventually experienced tumor metastasis and invasion.2,3 According to the current human understanding about CRC and conventional treatment methods, it is impossible to eliminate the tumors. Therefore, to clarify the key mechanism of tumor invasion and metastasis as well as establish corresponding blocking pathways to prevent metastasis and recurrence is the hope of the radical treatment of CRC.
Current studies have found that epithelial–mesenchymal transition (EMT) plays a crucial role in the procession of invasion and metastasis in various epithelial sources.4,5 During the progression of EMT, the epithelial cells of the primary tumor lose their cell polarity and become mesenchymal cells, which leads to the decrease of cell adhesion, the increase of invasiveness and migration, and the promotion of tumor cell metastasis to distant organs to form secondary tumors. EMT could promote the metastasis of various tumor cells, such as skin cancer, primary liver cancer, CRC, breast cancer, and malignant melanoma.6–10 On the other hand, the malignant progression of a tumor is affected by both genetic and environmental factors. Heavy metal pollution accounts for a large proportion of environmental pollution, especially cadmium (Cd). 6 More and more evidence indicated that long-term exposure to Cd will increase the risk of lung cancer, prostate cancer, and bladder cancer 11−13. A few experiments in vivo and in vitro also suggested that Cd affects the occurrence and development of tumors in various ways.14,15 Furthermore, some researchers suggested that Cd could promote the migratory and invasive ability of cancer cells,16−18 but whether these effects are related to EMT have not been fully elucidated.
Resveratrol (RES) is a natural polyphenolic compound, which is abundant in sources and widely exists in peanuts, grapes, and other plants. 19 Previous studies have shown that RES has a variety of biological effects, including antioxidant, anti-browning, anti-inflammatory, anti-pathogenic microorganisms, neuroprotection, and so on. 20 Cosín-Tomàs et al. 21 found RES functions as a candidate for the treatment of Alzheimer's disease via relieving the oxidative damage. In addition, at first, Jang et al. 22 found that RES plays an anti-tumor role on skin cancer of mice models, and it could reverse the occurrence, promotion, and progression of tumors. Recent studies have found that RES has an anti-tumor effect on many kinds of human tumor cells, including lung cancer, liver cancer, breast cancer, CRC, and so on through long-term basic research and clinical application.23−25 It is also worth mentioning that during the past years, resveratrol has been successfully used to reduce oxidative stress caused by excessive Cd exposure on the development of many cancers, suggesting that it might inhibit the effect of Cd. 26
In addition, due to the major location of RNAs in the expression of genes, N6-methyladenosine (m6A) modification is essential in regulating the progression of EMT and cancer development.27−29 There are many studies indicating that m6A could promote the development of tumors by increasing the level of suppressor genes and inhibiting the expressions of oncogenes.30,31 Zinc finger E-box binding homeobox 1 (ZEB1) is an important transcription factor in the proliferation and differentiation of cells. ZEB1 can recruit CTBP and BRG1 to suppress the transcription of CDH1 to regulate the procession of EMT. 32 However, whether m6A modification could affect the expression of ZEB1 in CRC cells and how RES and Cd affect the m6A modification, both need to be further explored. Hence, in the present study, we aimed to verify whether RES could reverse the effect of Cd on the migration and invasion of CRC, and the underlying mechanism.
Materials and methods
Materials and cell culture
The human CRC cell lines HT116 and SW480 were bought from the Institute of Biochemistry and Cell Biology of the Chinese Academy of Sciences (Shanghai, China). Cd and resveratrol were purchased from the Sigma-Aldrich (St Louis, MO, USA). The antibodies against E-cadherin, N-cadherin, ZEB1, and vimentin were purchased from Cell Signaling Technology. All reagents were stored according to the instruction of manufacturer.
All cells were cultured in RPMI-1640 medium (GIBCO) with 10% fetal bovine serum (FBS) and 1% penicillin–streptomycin solution (Invitrogen, Carlsbad, CA, USA) at 37°C in a moist atmosphere with 5% CO2 for the subsequent experiments.
Transwell assay for migration and invasion
HT116 and SW480 cells were pre-treated with or without different concentration of Cd alone, or Cd and RES. Cell culture transwell inserts (8-mm pore size; Falcon; BD Biosciences) were placed into the 48-well plates to generate the departed upper and lower chambers following 48 h of transfection. Furthermore, Matrigel (BD Biosciences) was used to pre-coat the upper chamber of the membrane. The cells in the upper chamber were then cultured for 1 h at 37°C for gelation. The membrane was hydrated with FBS 2 h prior. RPMI-1640 (600 μl) with 10% FBS and 1 × 105 cells/well was placed into the lower and upper chambers, separately. Each experiment was repeated 3 times. After 24 h of culturing, a microscope was used to count the numbers of invaded cells.
Quantitative real-time polymerase chain reaction (RT-qPCR)
The TRIzolTM reagent (Invitrogen; Thermo Fisher Scientific, Inc) was used to extract total RNA according to the instructions of the manufacturer. The concentration of RNA was measured using a Nanodrop 2000 spectrophotometer (Thermo, Fisher Scientific, Inc). Then, we used a Prime Script reagent kit (TaKaPa, Dalian, China) to transcribe the total RNA into cDNA. The PrimeScript RT reagent kit (Takara, Bio, Inc), transcriptase, Random 6mers, RNase inhibitor, Oligo dT primer, dNTP mixture, and reaction buffer were used to synthesize cDNAs.
The RT-qPCR was performed on a 7500 Fast Real-Time PCR System (Applied Biosystems; Thermo Fisher Scientific, Inc) using a SYBR Premix Ex Taq II kit (Takara Bio, Beijing, China). In addition, GAPDH was used to normalize the expressions of EMT-related markers. The cycling conditions were 95°C for 30 s (initial denaturation), followed by 95°C for 5 s and 60°C for 34 s, 40 cycles. The 2−ΔΔCq method was used to present the results. 33
The sequences of PCR primer were as follows:
ZEB1 (forward) 5’-ACCTCTTCACAGGTTGCTCCT-3’ and
(reverse) 5’-AGTGCAGGAGCTGAGAGTCA-3’;
N-cadherin (forward) 5’-TGTTTGACTATGAAGGCAGTGG-3’ and
(reverse) 5’-TCAGTCATCACCTCCACCAT-3’;
E-cadherin (forward) 5’-GGCCTTAGAGGTGGGTGACT-3’ and
(reverse) 5’-AATGGCAGGAATTTGCAATC-3’;
Vimentin (forward) 5’-AATGACCGCTTCGCCAAC-3’ and
(reverse) 5’-CCGCATCTCCTCCTCGTAG-3’;
GAPDH (forward) 5’-GAAGGCTGGGGCTCATTTG-3’ and
(reverse) 5’-GGGCCATCCACAGTCTTC-3’;
Western blot for detecting the expressions of EMT-related markers
The HT116 and SW480 cells in the logarithmic phase were treated with or without different concentrations of Cd alone or Cd and resveratrol, respectively. The sediment of cells was collected to lyse the cells by using RIPA for extracting the total proteins. The concentration of protein was determined by the BCA assay, and the protein was separated by SDA-PAGE gel according to the molecular weight of the target gene. Then, the protein was transferred onto the PVDF membrane by electroporation. Next, the membrane was blocked with 5% non-fat milk for 2 h at room temperature, followed by incubation with the first antibody (diluted 1000–2000 times according to the instructions) in a shaker at 4°C overnight. On the second day, we used TBST to wash the membrane for 10 min, 4 times. And then, we added the second antibody to culture the membrane for 1 h, washed the membrane for 10 min, 3 times. The target gene was detected with an enhanced chemiluminescence (ECL) kit, and the band image was analyzed using QuantityOne software. Each experiment was repeated 3 times.
Methylated RNA immunoprecipitation
The Dynabeads® mRNA purification kit (Ambion #61006, USA) was used to purify total RNA twice to obtain mRNAs, and then DNase (Promega M6101, USA) was used to remove genomic DNA. The mRNA (about 100 nucleotides long) was divided into two parts, one for IP and the other for IP control (input), and IP was performed according to the method reported by Dan Dominissini et al. 34 In short, 5 μg mRNA and 20 μg m6A antibodies (Synaptic Systems, Germany) were incubated in 500 μl IPP buffer (150 mM NaCl, 0.1% NP-40, and 10 mM Tris-HCl, pH = 7.4) at 4°C for 2 h. The above mRNA–m6A mixture and protein A beads were incubated at 4°C for 2 h. After fully eluting the mRNA-m6A-beads mixtures, the IPP buffer containing 0.5 mg/ml m6A was used to elute the mRNA bound to the beads and precipitated with alcohol. Then, we dissolved the precipitated mRNA containing m6A modification into RNA-free water for the subsequent research. Further enrichment was calculated by qPCR, and the corresponding m6A enrichment in each sample was calculated by normalizing to the input.
Statistical analysis
SPSS 17.0 software was used to analyze statistically (IBM Corp, Armonk, NY, USA). Data were emerged as the mean ± standard deviation of three repeated experiments. For comparisons between two groups, the two independent samples’ t-tests were used. For comparisons between multiple groups, analysis of variance (ANOVA) was used. p < 0.05 was considered to indicate a statistically significant difference.
Results
Cd could promote the migration and invasion of CRC cells in vitro
First, to determine the roles of Cd on the migration and invasion of CRC cells, the transwell assay was performed to detect the migratory and invasive ability after HT116 and SW480 cells were treated with different concentrations Cd (0, 1, 5, and 10 μM). As shown in Figure 1, the migratory (Figure 1(a), p < 0.05) and invasive (Figure 1(b), p < 0.05) ability was significantly increased in Cd-exposed HT116 and SW480 cells compared to the control group (0 μM) and the low-concentration group (1 μM). Cd promoted the migration and invasion of CRC cells. After treatment of different concentrations of Cd, the transwell assay was used to detect the (a) migration and (b) invasion of CRC cells. **p < 0.01. Notes: CRC: colorectal cancer; Cd: cadmium.
Cd could promote the expressions of EMT-related markers in CRC cells
Second, to explore the functions of Cd on the development of EMT, the expressions of a few representative EMT-related markers were detected by the Western blot analysis. The results are shown in Figure 2; after treating with different concentrations of Cd, the levels of ZEB1, vimentin, the mesenchymal markers N-cadherin, and the epithelial marker E-cadherin were regulated at the high-concentration groups (5 and 10 μM) (Figure 2(a), p < 0.05). Compared with the control group and low-concentration group (5 and 10 μM), the levels of ZEB1, vimentin, and N-cadherin have different degrees of increase, while the expression of E-cadherin was reduced nearly about 75% (Figure 2(b), p < 0.05). Increasing the concentration of Cd enhanced the up-regulation of ZEB1, N-cadherin, and vimentin, as well as the down-regulation of E-cadherin in Cd-exposed HT116 and SW480 cells. Cd regulated the expression of EMT-related proteins. **p < 0.01. (a, b). The expression levels of EMT-related proteins including ZEB1, E-cadherin, N-cadherin, and vimentin in CRC cells were evaluated by Western blot.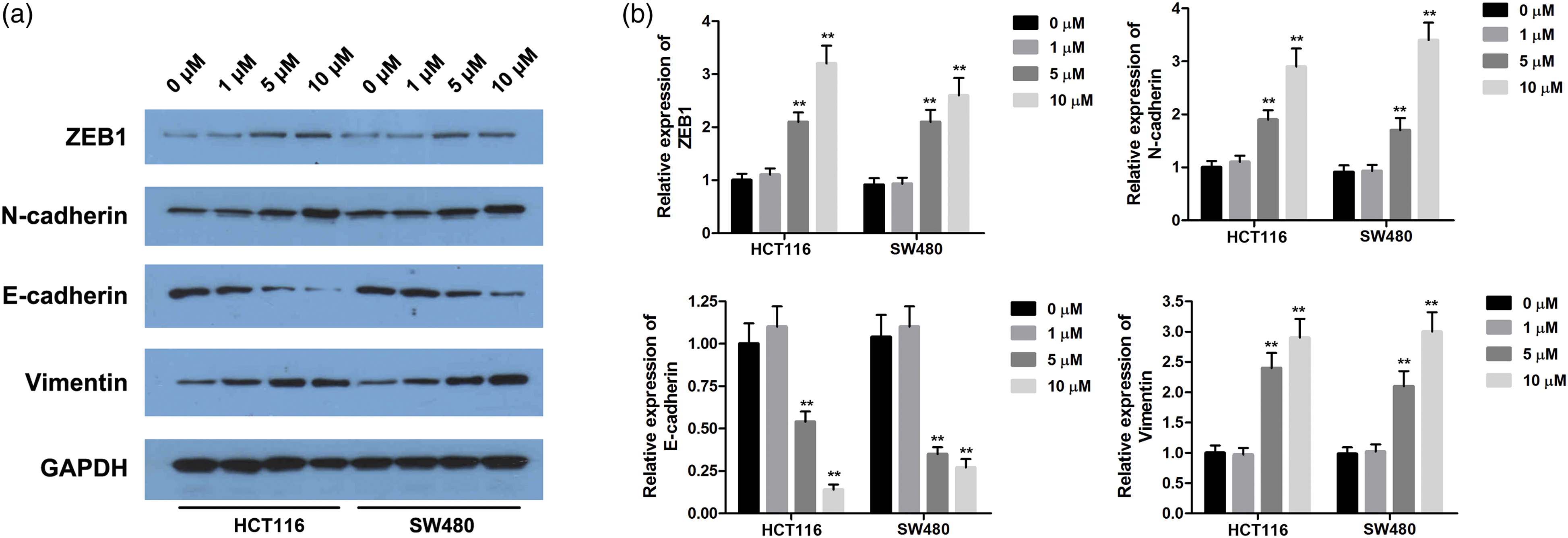
RES could reverse the Cd-promoted effects on the migration and invasion of CRC cells
Next, to further determine the effects of resveratrol on the migration and invasion of Cd-exposed CRC cells, this study used the transwell assay to validate the changes of migratory and invasive ability after treating with resveratrol. After culturing the HT116 and SW480 cells in the transwell chamber for 24 h, compared with the control group, the migration and invasion of CRC cells in the Cd-exposed group were significantly increased. Furthermore, compared with the Cd-exposed group, the number of migrated or invaded cells in the groups co-treated with Cd and resveratrol reduced. The changed degree of the migratory and invasive ability of HT116 and SW480 cells in the groups with co-treatment with Cd and resveratrol increased as the concentration of resveratrol rose (Figure 3, p < 0.05). RES could reverse the promoted effects of Cd on the migration and invasion of CRC cells. After treatment of different concentrations of Cd or Cd and RES, the transwell assay was used to detect the (a) migration and (b) invasion of CRC cells. *p < 0.05, **p < 0.01. Notes: CRC: colorectal cancer; Cd: cadmium; RES: resveratrol.
RES could suppress the Cd-increased expressions of EMT-related markers in CRC cells
Then, to examine the regulation ability of resveratrol on the levels of EMT-related markers, the expressions of ZEB1, vimentin, E-cadherin, and N-cadherin were detected by Western blot. The results showed that the expressions of ZEB1, vimentin, and E-cadherin were significantly up-regulated in CRC cells exposed to Cd compared with the control group, while N-cadherin down-regulated. Moreover, in contrast to the Cd-exposed group and the low-concentration resveratrol group (10 μM), the expressions of ZEB1, vimentin, and N-cadherin in the Cd-exposed CRC cells were remarkably decreased in the high-concentration resveratrol group (100 and 200 μM), while the expressions of E-cadherin increased. Resveratrol and Cd both had no effects on the expression of GAPDH in HT116 and SW480 cells (Figure 4, p < 0.05). RES-inhibited Cd increased the expression levels of EMT-related proteins in CRC cells (a, b). The expression levels of EMT-related proteins including ZEB1, E-cadherin, N-cadherin, and vimentin in CRC cells were evaluated by Western blot. *p < 0.05, **p < 0.01. Notes: CRC: colorectal cancer; Cd: cadmium; EMT: epithelial–mesenchymal transition; RES: resveratrol.
RES could reverse the Cd-effects on the m6A modification of ZEB1 in CRC cells
Finally, to further explore how resveratrol regulated the Cd-regulated development of EMT, this study performed RT-qPCR and Me-RIP assays to determine the underlying mechanism. From the data, we found the m6A content in total RNA of HT116 and SW480 cells remarkably increased in the Cd–DMSO group compared to the control group. However, the elevation caused by Cd was reduced after adding with resveratrol (Figure 5(a), p < 0.05). Meanwhile, depending on the Me-RIP assay, the result showed that resveratrol could suppress the m6A modification of ZEB1 which was increased by Cd (Figure 5(b), p < 0.05). After treating with dactinomycin D, the expression of ZEB1 at the mRNA level was significantly up-regulated by treatment with Cd compared with the control group. On the other side, the up-regulated degree of the expression of ZEB1 at the mRNA level was reduced at 15 h after treating with resveratrol (Figure 5(c), p < 0.05). RES could inhibit the Cd-promoted m6A modification of ZEB1 in CRC cell (a). The m6A content of total RNA in CRC cells after treatment of Cd or RES were detected using a kit. (b). qPCR was used to evaluate the expression of ZEB1 after Me-RIP. (c) After treatment of actinomycin D, qPCR was used to evaluate the mRNA expression of ZEB1 at the different time points. **p < 0.01. Notes: CRC: colorectal cancer; Cd: cadmium; RES: resveratrol.
Discussion
In the present study, we found that RES could reverse the effects of Cd on the migration and invasion of CRC cells through regulating EMT. The results indicated that Cd could promote the migration and invasion of CRC cells and the development of EMT, while RES could inhibit this function of Cd. We also demonstrated that RES could reverse the Cd effects on the m6A modification of ZEB1 in CRC cells. This is the first study to explore the roles of resveratrol about the effect of Cd on the CRC cells, at least to the best of our knowledge.
More and more studies have shown that colorectal cancer is caused by genetic factors and environmental factors. Heavy metals play key roles in environmental pollution, especially As and Cd.11−13 Cd is an environmental carcinogen that we already know. It mainly comes from the smelting release of related industrial production, fossil fuel combustion, metal refining, and smoking. It can be seen that the risk of human exposure to Cd has existed for a long time. Worryingly, some studies have indicated that Cd is closely related to the occurrence of a variety of tumors.16−18 However, reports about the effects of Cd on the migration and invasion of CRC cells and related mechanisms are limited. In the present study, through transwell, Western blot, and qRT-PCR assay, it has been found that Cd could promote the migration and invasion of CRC cells by promoting EMT.
Up to now, RES has been found to suppress the migration and invasion of many kinds of cancer cells, such as pancreatic cancer, breast cancer, and liver cancer.24,35,36 Studies have shown that resveratrol plays an anti-cancer role through various cell signaling pathways to block the adhesion, invasion, and metastasis of cancer cells. 37 Qing et al. suggested that different concentrations of RES can inhibit the proliferation of colorectal cancer cells. 38 However, the mechanism of RES on migration and invasion of CRC cells is still unclear. We used different concentrations of RES to treat the Cd-exposed CRC cells to detect the changes of the migratory and invasive ability. The result showed that RES could reverse the Cd-promoted effects on the migration and invasion of CRC cells, which indicated that RES relieves the progress of CRC by inhibiting the metastasis of CRC cells.
EMT plays an important role and is currently one of the hotspots in the research on the mechanism of malignant tumor development.32,39 Therefore, it is necessary to understand the malignant progression of the tumor and its correlation with EMT. Xu et al. 32 confirmed that RES reverses doxorubicin resistance via inhibiting EMT through the PTEN/Akt signaling pathway in gastric cancer. The loss of the expressions and functions of E-cadherin and other epithelial markers as well as the overexpression of N-cadherin, vimentin, and other mesenchymal cell markers are the molecular features of EMT.40,41 It has been proposed in the research that the induction of Cd will destroy the connection between cells mediated by E-cadherin protein, which will lead to the further development of tumors. 42 Furthermore, Li et al. found that RES can inhibit the invasion and metastasis of tumor cells by blocking the occurrence of EMT in tumor cells in pancreatic cancer. 35 Previous reports showed that chronic exposure to As could promote the malignant progression of colorectal cancer cell lines HT-29 through the EMT mechanism. 35 Nevertheless, whether and how Cd and resveratrol affected the progression of EMT in CRC cells are still unclear. In this study, through the Western blot assay, we found that Cd could up-regulate the expressions of ZEB1, N-cadherin, and vimentin while down-regulating the expression of E-cadherin. Moreover, resveratrol also could reverse Cd-effect on CRC cells. These results implied that RES inhibits the migration and invasion of CRC cells through regulating the process of EMT, which may be the mechanism of RES on CRC.
Furthermore, to further explore the specific mechanism of RES, we analyzed the role of RES in m6A modification of ZEB1. It is well known that the expression of m6A can regulate the RNA methylation landscape in human cells. There are more and more researches about biological functions of the m6A modification in mRNA during the past years. m6A has been reported to significantly affect the regulating the expressions of genes and are associated with various human diseases including cancers.43,44 Chen et al. found METTL3 could increase the m6A modification of SOCS2 to regulate the proliferation of hepatocellular carcinoma cells. 45 Wang et al. 46 suggested that knockdown the m6A regulators will promote the proliferation of cervical cancer cells. Depending on the latest research, Lin et al. indicated that the m6A modification of mRNAs could increase the EMT of tumor cells. 29 Thus, we detected the mRNA expression of ZEB1 and the content of m6A in CRC cells through qRT-PCR and Me-RIP assays. The data indicated that the m6A modification of mRNAs could increase the expression of ZEB1 in CRC cells. Meanwhile, the result also indicated that RES could reverse the effects of Cd on the m6A modification of ZEB1in CRC cells. These results further implied that RES regulates the process of EMT via m6A modification, which may be the mechanism of RES alleviating the occurrence and development of CRC.
Conclusion
In summary, we demonstrated that RES plays an important role in reversing the effects of Cd on CRC cells. RES could inhibit the migration and invasion of CRC cells which are promoted by Cd in vitro through regulating the m6A modification of ZEB1 and the expressions of the EMT-related markers. These results suggested that RES might be a novel target for the treatment of CRC.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Key Projects of Natural Science Research in Anhui Universities (KJ2018A0932).
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
