Abstract
Objective
Shenfu injection (SFI) has been reported to have a protection against myocardial ischemia-reperfusion (MI/R) injury. However, the changes of adenosine receptors in MI/R postconditioning when pretreated with SFI are unclear.
Methods
Forty-five rats were randomly divided into sham group (sham), MI/R postconditioning group (MI/R-post), low-dose SFI group (1 mL/kg), middle-dose SFI group (2.5 mL/kg), and high-dose SFI group (5 mL/kg). In SFI groups, SFI was intravenously injected before reperfusion, and rats were treated with ischemic postconditioning after ischemia for 30 min. After 24 h of reperfusion, the levels of Ca2+ and cAMP in blood platelets were analyzed. Myocardial infarct volume and myocardial pathology were observed. The levels of adenosine receptor subtypes A1, A2b, and A3 in myocardium were analyzed using immunohistochemistry and Western blot. The oxidative stress–related indicators were also observed.
Results
Compared with the MI/R-post group, SFI ameliorated the MI/R injury by decreasing the myocardial infarct area, oxidative stress, and concentration of Ca2+ and cAMP (p < 0.01). Pretreatment with SFI enhanced the expression of adenosine receptors A1 and A2b in a dose manner compared with the MI/R-post group. In contrast, the levels of adenosine receptor A3 were increased after MI/R postconditioning compared with the sham group, and its expression continued to increase with the increase of SFI. Furthermore, the oxidative stress reduced with the concentrations of SFI.
Conclusion
These results demonstrated that pretreatment with SFI might regulate the expression of adenosine receptors to improve the MI/R postconditioning.
Keywords
Introduction
Heart failure is a complex clinical syndrome, which represents the final path of many heart diseases. More than 26 million people suffer from heart failure worldwide. 1 A common finding in failing hearts, myocardial ischemia (MI) is a self-propagating process and irreversibly impairs the cardiac function. 2 In order to prevent or reduce the cardiac ischemic burden, guideline-directed medical therapy is still the basic treatment strategy for patients with heart failure. In China, traditional Chinese herbal formulation is widely used in clinics because of its definite curative effect and few side effects. 3 Therefore, it is necessary to find a kind of traditional Chinese medicine to relieve the heart function of patients with heart failure.
Shenfu injection (SFI) is a Chinese medicine, and its formula is ginseng, which has ginsenosides as main bioactive components. 4 Yang et al. reported that 4 12 major ginsenosides in Shenfu Tang were determined using the UPLC-MS/MS method, and the contents of ginsenosides Re, Rc, Rb2, and Rb1 were more than 1000 μg/g. Therapeutic strategies to protect the ischemic myocardium have been studied extensively. Reperfusion is the definitive treatment for acute coronary syndromes, especially acute myocardial infarction; however, reperfusion has the potential to exacerbate lethal tissue injury, a process termed “reperfusion injury.” 5 Many previous studies have shown that SFI could improve myocardial damage,6–8 including myocardial ischemia-reperfusion (MI/R) injury. 7 Pretreatment with SFI can reduce the inflammation and apoptosis induced by lipopolysaccharide in rats with cardiac dysfunction. 6 Wang et al. revealed that SFI pretreatment activated eNOS in rats with MR/I injury. 7 The application of brief repetitive episodes of ischemia-reperfusion at the immediate onset of reperfusion, which has been termed “postconditioning,” reduces the extent of reperfusion injury.5,9 However, the effects of SFI on MI/R postconditioning are still unclear.
During myocardial ischemia, adenosine is intentionally released to improve local oxygen delivery by regulating blood flow. 10 It has been reported that ischemia and aging selectively modify the transcription of cardioprotective adenosine receptors, including specific receptor subtypes A1, A2a, A2b, and A3 receptors. 11 Sassi et al. found extracellular cAMP could reduce cAMP formation and hypertrophy in cardiomyocytes via activating the adenosine A1 receptor; at the same time, it delivered an antigibrotic signal to cardiac fibroblasts via activating the adenosine A2 receptor. 12 Further, a previous study showed the adenosine A2b receptor directly stimulated myocardial contractility in isolated adult mouse hearts. 13 In cardiac fibroblasts of patients with LV dysfunction, all adenosine receptor subtypes were overexpressed. 14 Although their roles in determining the phenotype after ischemia remain controversial, the cardioprotective activity is associated with each subtype.
In this study, we observed the protective effects and changes of adenosine receptors in rats with MI/R postconditioning when pretreated with SFI to explore its underlying mechanism.
Material and methods
Animals
All animal experiments followed the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH, publication Eighth Edition, 2011). And this work has been reviewed and approved by the Animal Care and Use Committee of Yantaishan Hospital (Approval No. 202013).
Forty-five Sprague Dawley male rats were obtained from Jinan Pengyue Experimental Animal Breeding Co., Ltd. (SCXK (Lu) 20190003), weighted 200–220 g and 8 weeks old. The rats were housed in the following environment: 22–26°C, 40–70% relative humidity, and 12 h light–dark cycles. All rats received food and water ad libitum.
Myocardium ischemia-reperfusion postconditioning
Rats were given aspirin (3 mg/kg, 1 mL/100 g) by gavage for three days before establishing injury. After aspirin administration, rats were fasted for 12 h. According to a previous study, 15 the MI/R injury model was constructed. Briefly, rats were anesthetized using 1.5% pentobarbital sodium (0.27 mL/100 g) and fixed in a spinal position. After skin preservation and sterilization, tracheal intubation was used to connect with a ventilator (tidal volume was 0.02 mL/g and respiratory rate was 50 times/min) to assist breathing. The thoracic cavity was opened in the 3 and 4 intercostal space to expose the heart. Then, the left anterior descending coronary artery was ligated with silk thread. The multifunctional ECG monitor (ECG—1250°C, Nihon Kohden, Japan) showed that ligation was successful, and the drugs were injected into the femoral vein. After ischemia for 30 min, the ligature was released for a 24-h reperfusion. After a 1-min reperfusion, the coronary blood flow was closed for 30 s, and then opened for 30 s, three times.
Animal groups
According to previous studies,7,16 we chose 1 mL/kg, 2.5 mL/kg, and 5 mL/kg SFI to study in this work. One week after adaptive feeding, rats were randomly divided into the sham group, MI/R postconditioning (MI/R-post) group, low-dose SFI (L-SFI) group (1 mL/kg, 1903120104, China Resources Sanjiu (Ya’an) Pharmaceutical Co., Ltd., China), medium-dose SFI (M-SFI) group (2.5 mL/kg), and high-dose SFI (H-SFI) group (5 mL/kg), with nine rats in each group. In sham group, rats were only threaded without the left anterior descending coronary artery. In SFI groups, SFI was intravenously injected 30 min before reperfusion, and rats were treated with ischemic postconditioning after ischemia for 30 min.
Determination of cytosolic Ca2+ in platelets
After 24 h of reperfusion, rats were anesthetized using 1.5% pentobarbital sodium. Blood was collected from the abdominal aorta, and platelet suspension was prepared by a platelet isolation kit (20190926, Solarbio, Beijing, China). The calcium fluorescent indicator Fura-2AM (20190929, Solarbio) was added and cultured at 37°C for 45 min in the dark. The concentration of Ca2+ in platelets was analyzed using a fluorescence spectrophotometer (BioTek Instruments, Inc. Synergy H1).
Determination of cyclic adenosine monophosphate level in platelet blood
Platelet-rich plasma was incubated at room temperature and washed twice with phosphate buffer saline (PBS). Then, the plasma was lysed with high-efficiency RIPA lysate (20190928, Solarbio) and centrifuged at 12,000 g for 5 min to collect supernatant. The cAMP levels were determined by a rat cAMP ELISA kit (201910, Jingsu Enzymic Biotechnology Co., Ltd., China).
2,3,5-triphenyltetrazole chloride staining
The hearts were taken out and washed with 0.9% NaCl and then frozen at −20°C for 10 min. One heart was cut into six sections in the cross-sectional direction and placed into 1% TTC solution (20190917, Solarbio). The sections were incubated at 37°C for 15 min in the dark. The results were analyzed using Image J 5.0 (National Institute of Health, Bethesda, MD, USA). Infarct area (%) = infarct area/total area × 100%.
Hematoxylin and eosin staining
The annular myocardial tissue in the middle of the left ventricular coronary surface was washed repeatedly with precooled phosphate buffer (pH = 7.4) and then fixed with 4% polyformaldehyde for 24 h. After paraffin embedding, the tissues were cut into 5-μm–thick sections. The sections were dewaxed with xylene and dehydrated with graded ethanol: 100% ethanol, 5 min; 95% ethanol, 2 min; 80% ethanol, 2 min; and 70% ethanol, 2 min. Hematoxylin (190704, Wuxi Jiangyuan Industry, China) was used to stain for 15 min. After differentiation for 30 s, the sections were soaked at 50°C for 5 min and stained with eosin for 40 s. After washing with ethanol and clearing with xylene, the sections were observed under a light microscope (DM1000 LED, Leica, Germany).
Immunohistochemistry
The methods of fixation, embedding, dewaxing, and dehydration were consistent with H&E staining. After washing with dH2O twice, each time for 5 min, the sections were immersed in 1x citrate solution (pH = 6.0; M053201, Mreda, Beijing, China) at 95–98°C for 10 min. After cooling at room temperature, the sections were washed with dH2O three times and incubated in 3% H2O2 for 10 min. After washing with 1x Tris-buffered saline with Tween@ 20 (TBST), the sections were sealed with 5% bovine serum albumin (BSA, A8010, Solarbio, Bejing, China) at room temperature for 30 min and then incubated with primary antibodies: anti-AODRA1 (bs-4235R, Beijing Bo’aosen Biotechnology Co., Ltd, China), anti-ADORA2B (bs-5900R), and anti-ADORA3 (bs-1225R) at 4°C overnight. After washing with PBST, the secondary antibody SP Kit (sp0023, Beijing Bo’aosen Biotechnology Co., Ltd.) was added and incubated at room temperature for 30 min. SignalStain@ DAB (100–400 μL) was used to observe the staining intensity for 1–10 min. After washing with dH2O, the sections were dehydrated with ethanol and cleared with xylene. Finally, the neutral resin was used for sealing.
Myeloperoxidase activity assay
The infarcted myocardium (40–50 mg) was collected in ice-cold MPO assay buffer (5 mL/mg) and then homogenized three times for 30 s at 30/s. After homogenization, the samples were measured using an MPO Fluorometric Activity Assay Kit (MAK069, MERCK). The fluorescein was measured every 15 min.
Malondialdehyde measurement
The MDA levels in myocardial tissues were measured using the spectrophotometric method. The tissues were homogenized with 10% tricholoroacetic acid and then heated with thiobarbituric acid. The absorbance was obtained at 532 nm using an MDA Assay (MAK085, MERCK).
Glutathione measurement
The infarcted myocardium (40–50 mg) was collected in ice-cold GSH assay buffer (5 mL/mg) and homogenized three times for 30 s at 30/s. After homogenization, the GSH levels in myocardial tissues were also measured using a GSH Assay (CS0260, MERCK) at 420 nm.
Western blot
Myocardial tissues were dissolved in cold RIPA buffer containing protease and phosphatase inhibitors (R0010, Solarbio) for 15 min and then centrifuged at 12,000 g and 4°C for 25 min. Total protein was extracted by a total protein extraction kit (bc3640-50t; Solarbio). 40 μg protein was separated by 10% SDS-PAGE (Bio-Rad laboratories, Inc.), then transferred to a PVDF membrane (EMD, Millipore), and blocked with 5% skim milk at 5°C for 1 h. The proteins were incubated with primary antibodies: anti-AODRA1 (bs-4235R, Bioss, bioon.com.cn), anti-ADORA2B (bs-5900R, Bioss), anti-ADORA3 (bs-1225R, Bioss), anti-iNOS (bs-0162R, Bioss), and GAPDH (bsm-33033M, Bioss) at 4°C overnight. After washing with PBST, the secondary antibody SP kit (sp0023, Bioss) was incubated at room temperature for 60 min. ECL chemiluminescence reagent (C05-07004, Bioss, Beijing, China) was used to observe the protein bands.
Statistical analysis
Statistical analysis was carried out by SPSS 19.0 software and Image J version 5 (National Institutes of Health, USA), and the results were described as mean ± standard deviation (X ± SD). One-way analysis of variance was used for data analysis among groups, and Tukey’s test was used for subsequent analysis. p < 0.05 represents statistical difference.
Results
SFI reduced the infarct area of myocardial ischemia
The effect of SFI on the infarct area of myocardial ischemia is shown in Figure 1. In the MI/R-post group, there was a clear infarct area compared with the sham group (p < 0.01). After SFI injection, the infarct area decreased compared with the MI/R-post group. The infarct area decreased with the increase of SFI. The TTC staining of myocardial tissue in each group: (a) TTC staining and (b) The infarct area. Compared with sham group, **p < .01; compared with MI/R-post group, ##p < .01; and compared with L-SFI group, &p < .05. Notes: TTC: 2,3,5-triphenyltetrazole chloride; L-SFI: low-dose SFI.
SFI reduced the levels of cytosolic Ca2+ and cAMP
Compared with the sham group, the levels of Ca2+ and cAMP in platelets markedly rose in the MI/R-post group (p < 0.05, Figures 2(a) and (b)). The concentrations of Ca2+ and cAMP were reduced after different doses of SFI treatment. Compared with the MI/R-post group, there was a significant difference in both the M-SFI and H-SFI groups (p < 0.05, Figures 2(a) and (b)). The changes of myocardial pathology in each group were observed in Figure 2(c). In the sham group, there was no infarction. In the MI/R-post group, the tissue was loose with a large amount of infiltration inflammatory cells, and there was a large infarction area. In the SFI groups, the degree of lesions was significantly reduced compared with the MI/R-post group. Effects of SFI on levels of Ca2+ and cAMP in platelets and on changes of myocardial pathology. (a) Ca2+ concentration; (b) cAMP concentration; (c) myocardial pathology. Compared with sham group, **p < .01; compared with MI/R-post group, #p < .05, ##p < .01; compared with L-SFI group, & p < .05, && p < .01; and compared with M-SFI group, ^p < .05. Notes: SFI: Shenfu injection; M-SFI: medium-dose SFI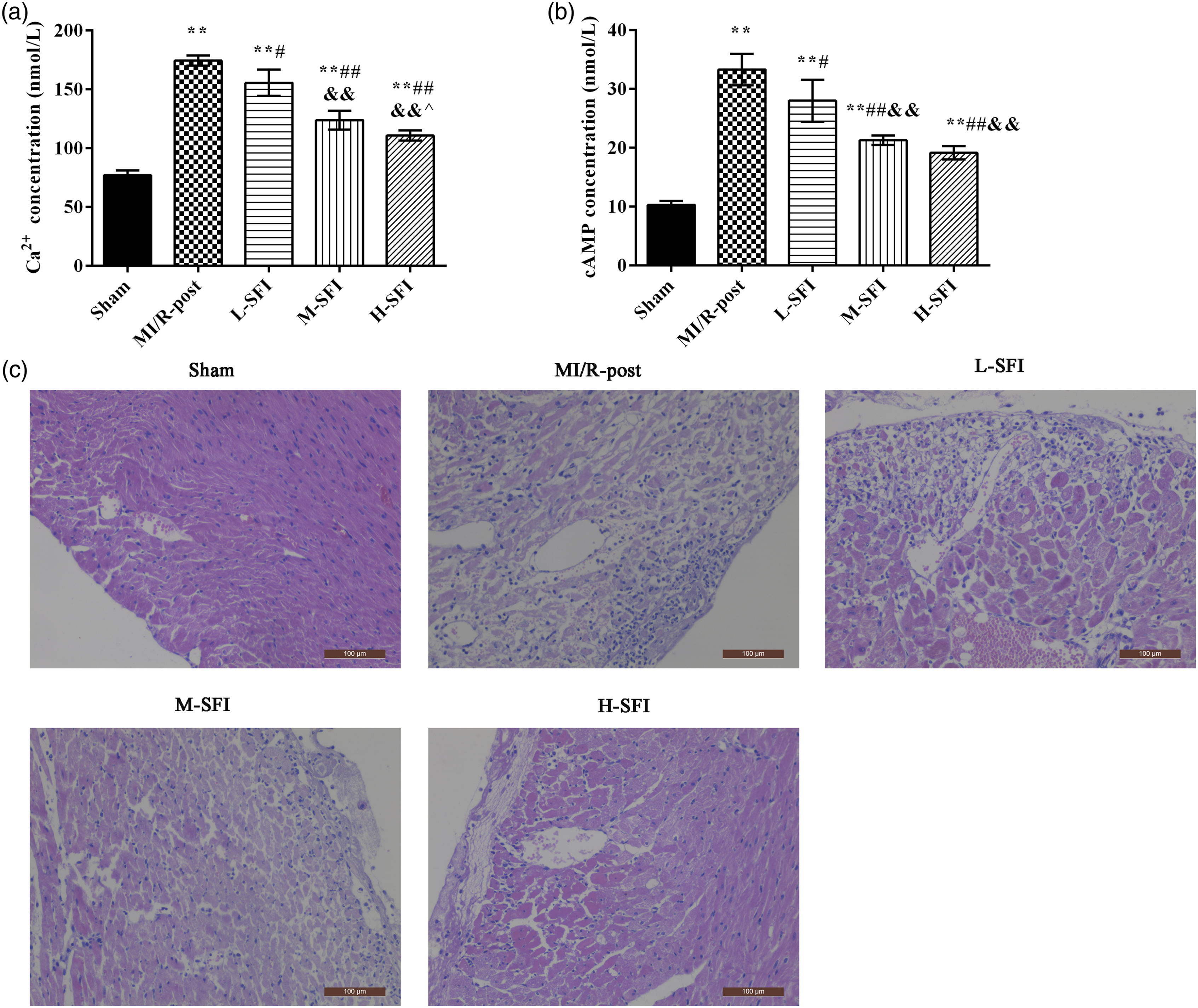
SFI increased the expression of adenosine A1 receptor in myocardium
The expression of the adenosine A1 receptor in myocardium was analyzed using immunohistochemistry (Figure 3(a)) and Western blot (Figure 3(b)), separately. The expression of the adenosine A1 receptor was located in the cytoplasm of cardiomyocytes. Compared with the sham group, the expression of the adenosine A1 receptor in the MI/R-post group was significantly decreased (p < 0.01). Compared with the MI/R-post group, the adenosine A1 receptor expression in each SFI group was significantly increased (p < 0.01). Contrasted with the L-SFI group, the expression of the adenosine A1 receptor was obviously increased in the H-SFI group (p < 0.01). Effect of SFI on expression of the adenosine A1 receptor in myocardial tissue. (a) Immunohistochemistry and (b) Western blot. Compared with sham group, **p < .01; compared with MI/R-post group, #p < .05, ##p < .01; compared with L-SFI group, & p < .05, && p < .01; and compared with M-SFI group, ^p < .05, ^^p < .01. Notes: SFI: Shenfu injection; L-SFI: low-dose SFI; M-SFI: medium-dose SFI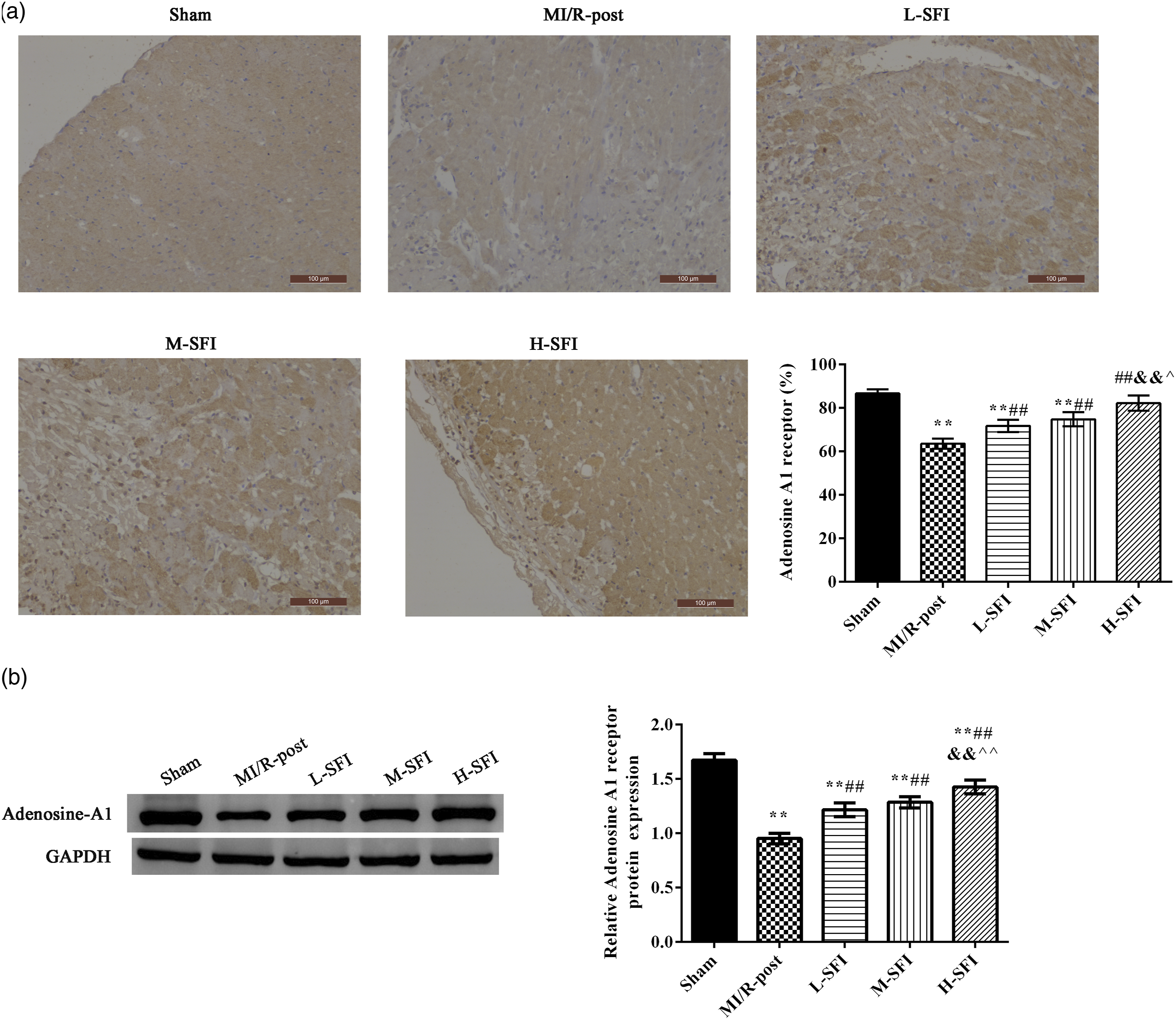
SFI increased the expression of adenosine A2b receptor in myocardium
The expression of the adenosine A2b receptor in myocardium was analyzed using immunohistochemistry (Figure 4(a)) and Western blot (Figure 4(b)), separately. The expression of the adenosine A2b receptor was similar with the expression of the adenosine A1 receptor. After MI/R postconditioning, the expression of the adenosine A2b receptor was obviously decreased when compared with the sham group (p < 0.01). Compared with the MI/R-post group, the levels of the adenosine A2b receptor were increased after SFI injection in a dose manner. The level of the adenosine A2b receptor was significantly higher in the M-SFI or H-SFI group when compared with the L-SFI group (p < 0.01). Effect of SFI on expression of the adenosine A2b receptor in myocardial tissue. (a) Immunohistochemistry and (b) Western blot. Compared with sham group, **p < .01; compared with MI/R-post group, #p<.05, ##p<.01; compared with L-SFI group, &p < .05, &&p < .01; and compared with M-SFI group, ^p < .05, ^^p < .01. Notes: SFI: Shenfu injection; M-SFI: medium-dose SFI; L-SFI: low-dose SFI.
SFI increased the expression of adenosine A3 receptor in myocardium
In addition, the expression of the adenosine A3 receptor was analyzed by immunohistochemistry (Figure 5(a)) and Western blot (Figure 5(b)). Interestingly, the expression of the adenosine A3 receptor was increased after MI/R postconditioning compared with the sham group (p < 0.01). After different doses of SFI injection, the levels of the adenosine A3 receptor were raised in a dose manner. Compared with the MI/R-post group, the expression of the adenosine A3 receptor was notably increased in the SFI groups (p < 0.01). Effect of Shenfu injection on expression of the adenosine A3 receptor in myocardial tissue. (a) Immunohistochemistry and (b) Western blot. Compared with sham group, **p < .01; compared with MI/R-post group, #p < .05, ##p < .01; and compared with low-dose SFI group, & p < .05, && p < .01.
SFI reduced the oxidative stress in myocardium
The changes of oxidative stress–related factors, MDA, MPO, GSH, and iNOS were observed in myocardium of each group (Figure 6). The MDA levels (Figure 6(a)), MPO activities (Figure 6(b)), and iNOS expression (Figure 6(d)) were significantly increased after MI/R postconditioning compared with the sham group. With the increase of SFI administration, their levels were decreased. The changes of GSH in myocardium showed an opposite trend compared with above indexes (Figure 6(c)). In addition, the antioxidant effects of a high dose of SFI were better than those of a low dose of SFI (p < 0.05). Effect of SFI on the levels of oxidative stress in myocardial tissue. (a) MDA level, (b) MPO activity, (c) GSH level, and (d) iNOS expression. Compared with the sham group, **p < .01; compared with MI/R-post group, #p < .05, ##p < .01; compared with L-SFI group, &p < .05, && p < .01; and compared with M-SFI group, ^^p < .01. Notes: MDA: malondialdehyde; MPO: myeloperoxidase; GSH: glutathione; M-SFI: medium-dose SFI.
Discussion
Ischemic heart disease in humans is a complex disorder caused by or associated with known cardiovascular risk factors, including hypertension, diabetes, atherosclerosis, and heart failure. 9 In these diseases, the pathological processes are associated with fundamental molecular alterations that can potentially affect the development of ischemia-reperfusion injury itself and responses to cardioprotective interventions. 17 The aim of this study is to show the potential of developing cardioprotective drugs on the basis of endogenous cardioprotection by postconditioning.
Compared with MI/R postconditioning, our study showed that SFI injection reduced the degree of myocardial infarction and oxidative stress, and improved the histopathological morphology before reperfusion after ischemia. These results were consistent with previous studies,6,7,16 which showed SFI treatment attenuated myocardial injury. For myocardial injury, SFI could decrease the secretion of proinflammatory factors, such as interleukin (IL)-1β and tumor necrosis factor (TNF)-α, and inhibit the expression of apoptotic proteins, such as caspase-3, caspase-9, and Bax.6,18 The protective mechanism may be related to the main active components ginsenosides in SFI. Clinically, routine medicine combined with SFI was used to improve the left ventricular ejection fraction and cardiac function for patients with chronic heart failure, a mechanism which was related to inhibition of TNF-α and the nuclear factor-kappa B (NF-κB) pathway. 19 However, SFI contains a variety of ingredients, and their roles are not single, but multifaceted. This suggests the mechanism of SFI in improving myocardial injury is complex. Adenosine receptors have important roles in regulating myocyte contractility, and myocardial metabolism, and response to hypoxic injury. 10 Fretwell and Dickenson 20 showed large-conductance Ca2+-activated potassium channels (BKCa channels), which were involved in the adenosine A1 receptor–induced pharmacological postconditioning in H9C2 cells with hypoxia and reoxygenation. BKCa channels have been implicated in protection against MI/R damage, 21 which abundantly express on the plasma membrane of vascular smooth muscle cells, whereas cardiomyocytes suffered from Ca2+ overload in MI/R injury. 22 The adenosine A2b receptor could stimulate the production of cAMP through coupling to Gs proteins, which is related to inflammatory responses and vascular structure and function. 23 Given the possibility that local metabolic regulation through adenosine signaling coordinates ischemia injury, we evaluated the effect of SFI on changes of myocardial adenosine receptors in rats with MI/R postconditioning. The results showed the adenosine A1 and A2b receptors expression increased after SFI administration compared with the MI/R postconditioning. However, the expression of adenosine A3 receptors increased after MI/R postconditioning, and its expression continued to increase after SFI administration. This difference among adenosine receptors indicates their different roles in MI/R postconditioning. Furthermore, these data showed us the complex mechanisms of SFI.
Historically, excessive oxidative stress in myocardial tissues is mainly caused by MI/R, which aggravates myocardial injury.24,25 It is reported that MPO, MDA, GSH, and iNOS are major representatives for oxidative stress.26,27 Consistent with previous studies, our data showed the levels of MPO, MDA, and iNOS were increased, but the levels of GSH were decreased in ischemic myocardium. Interestingly, the excessive oxidative stress was suppressed after SFI administration in MI/R-induced myocardium damage, suggesting the antioxidation of SFI.
In this study, we found the effects of SFI on the changes of adenosine receptors in myocardial tissues, but the underlying mechanisms are still not clear. For example, how the SFI implements the protection by regulating adenosine receptors in MI/R postconditioning. In addition, Wilkinson et al. 28 found that agonism of the adenosine A2b receptor resulted in increased intracellular cAMP while agonism of the adenosine A1 receptor inhibited cAMP modulation in renal fibroblast cell line NRK-49F. In MI/R postconditioning, the abovementioned facts are also not clear. These prompt us to continue research and exploration.
Conclusions
In light of our data, SFI reduced the infarct size and oxidative stress, improved the myocardial pathological damage, and increased the expression of myocardial adenosine A1, A2b, and A3 receptors in rats with MI/R postconditioning.
Footnotes
Authors contributions
Jie Wang and Xiaohuan Wang carried out the experimental work and the data collection and interpretation. Weiping Wan and Yuanying Guo participated in the design and coordination of experimental work, and acquisition of data. Yanfang Cui, Wenbo Liu, and Fangming Guo participated in the study design, data collection, analysis of data, and preparation of the article. All authors read and approved the final article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
