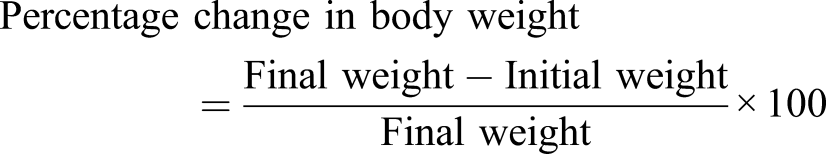Abstract
Background
Evidences are beginning to accrue that flavonoids, particularly phytoestrogens, could have beneficial effects against several age-related diseases linked to estrogen deficiency including postmenopausal osteoporosis.
Methods
In this study, the effect of chrysin on selected bone-remodeling markers in ovariectomized rats and its estrogen-like activity in silico were investigated.
Results
The data indicated that administration of chrysin at 50 mg/kg and 100 mg/kg for 6 weeks to OVX rats significantly (p < 0.05) prevented body weight gain and partially reverse uterine weight loss. In addition, treatment of OVX rats significantly (p < 0.01) increased femur dry weight, femur ash weight, bone ash calcium, and phosphorous levels in a dose-dependent manner. However, there was significant (p < 0.001) decline in serum estradiol level in all OVX rats compared to the sham-operated group. Interestingly, administration of chrysin significantly (p < 0.05) reversed the reduction of estradiol induced by ovariectomy compared to untreated OVX rats. Moreover, administration of chrysin to OVX rats significantly (p < 0.05) suppressed excessive elevation of bone-remodeling markers expression compared to untreated OVX rats. Similarly, molecular docking analysis revealed that chrysin interacts with both α and β estrogen receptors with exothermic binding energies of −229.83 kcal/Mol and −252.72 kcal/Mol, respectively, and also fits perfectly into the active site of both α and β estrogen receptors.
Conclusion
This study demonstrated that chrysin exhibits potential antiosteoporotic effects against bone loss in OVX rats through enhanced bone mineral contents and preventing excessive elevation of bone-remodeling markers and bone-resorbing cytokine.
Highlights
Chrysin prevents uterine weight loss and improves serum estradiol in OVX rats Chrysin prevents ovariectomy-induced excessive elevation of bone-remodeling markers Chrysin increases femur bone ash contents and improves Ca and P levels in OVX rats Chrysin shares structural similarities with estradiol and interacts with both ER-α and ER-β Chrysin exhibits almost same binding energy with estradiol on both estrogen receptors
Introduction
Osteoporosis is a metabolic bone disorder characterized by low bone mineral density (BMD) and impaired bone microarchitecture, leading to increased skeletal fragility and susceptibility to fracture in elderly women.1–4 The incidence rate of osteoporosis is one in three women and one in five men above 50 years of age. 5 Although, the etiology of osteoporosis is multifactorial; however, estrogen plays a crucial role in bone homeostasis and skeletal development through regulatory and direct effects on bone cells. 6 Thus, deficiency of estrogen at the onset of menopause in women has been shown to cause loss of its osteoprotective effect and consequently postmenopausal osteoporosis. During physiologic processes, bone tissue undergoes continuous remodeling mediated by coordinated activities of osteoclasts and osteoblasts.7,8 Therefore, an imbalance in this remodeling equilibrium by an increased rate of osteoclast-mediated bone resorption over osteoblast-mediated bone formation causes bone loss. 9 Bone-remodeling markers are either enzymes or by-products of active osteoblasts and osteoclasts expressed during phases of their development and are usually measured from serum or plasma to monitor the therapeutic efficacy of anti-resorptive and/or osteoanabolic agents. 10 These markers includes bone-specific alkaline phosphatase (BALP), osteocalcin (OC), carboxy- and amino-terminal propeptides of type 1 collagen (P1CP and P1NP), and receptor activator of nuclear factor-κB ligand (RANKL). 11 Molecular docking is a cost-effective method used for identifying drug targets and has previously been used to elucidate the binding properties of estrogen receptors, mainly due to its capability to demonstrate the binding mechanism between a ligand and a target protein.12,13 Currently, phytoestrogens are receiving considerable interest and have been demonstrated to stimulate bone formation and inhibit bone resorption.14,15 Many natural bioactive flavonoids from diet are continuously recognized as an important life-style factors in the management of bone diseases and have demonstrated interesting estrogenic activity, 16 antioxidant, anti-inflammatory, anticancer, anti-diabetic, and antihypertensive effects. 17 Chrysin (5,7-dihydroxyflavone) is a natural flavonoid and a major component of some traditional medicinal herbs, 18 passion flowers, bee propolis, honey, and several fruits such as carrots and mushrooms. 19 Chrysin has been reported to possess anti-inflammatory effects, antioxidant activity, and regulate reproductive system and hormones. 20 A previous study has demonstrated chrysin promotes osteogenic differentiation of osteoblast cells via ERK/MAPK activation. 21 However, there was no information about the effect of chrysin on bone-remodeling markers in ovariectomized rats. Hence, this study investigates the effect of chrysin on selected bone-remodeling markers in OVX rats and its estrogen-like activity in silico.
Materials and methods
Chemicals and reagent kits
Chrysin (5,7-dihydroxyflavone) with molecular weight of 254.24 g/mol, with melting point of 284–286oC and 97% purity was purchased from Sigma Aldrich Chemical Co. Inc. (Milwaukee, WI, USA). Rat ELISA kits for RANKL, osteocalcin, and 17β-estradiol were purchased from Shanghai Coon Koon Biotech Co. Ltd (Shanghai, China). Rat alkaline phosphatase kit was purchased from Agappe Diagnostics Switzerland (Cham, Switzerland). All other chemicals and reagents used in this study were of analytical grade and purchased from local suppliers.
Surgical and wound dressing consumables
Ketamine and xylazine were purchased from the Popular Pharmaceuticals Ltd., Tongi Industrial Area, Bangladesh. Scalpel blades, gauze bandage, vicryl (4:0 and 2:0), 1-mL insulin syringe, cotton wool, Savlon, 70% ethanol, clipper, povidone iodine, surgical gloves, and scissors were either procured or provided by Veterinary Teaching Hospital, Ahmadu Bello University, Zaria.
Experimental animals
This study was conducted with approval from the Ethics Committee on Animal Use and Care of Ahmadu Bello University, Zaria, Nigeria. Twenty-five apparently healthy three-month-old female Wistar rats, weighing between 170 to 180g were purchased from Department of Pharmacology, Faculty of Pharmaceutical Sciences, Ahmadu Bello University, Zaria. The rats were kept in well-ventilated laboratory cages under standard conditions for 12 h of dark and light cycle at room temperature in the Department of Veterinary Pharmacology, Faculty of Veterinary Medicine, Ahmadu Bello University, Zaria. The rats were allowed to acclimatize to the surrounding environment for 2 weeks and provided access to standard laboratory diet (Growers Pelletized Vital Feeds, Bukuru, Plateau State) and water ad libitum prior to commencement of the study.
Induction of osteoporosis in experimental rats and chrysin treatment
The experimental rats were anesthetized intraperitonially using a combination of ketamine hydrochloride (50 mg/kg) and xylazine (12.5 mg/kg) and then subjected to either sham operation (n = 5) or ovariectomy (n = 20) under aseptic condition as described by a previous study. 22 In ovariectomy, both ovaries were completely removed from the animals. Sham operation was conducted in a similar manner as described in ovariectomy procedure, except ovaries were identified but not removed. After 2 weeks of post-surgical procedures and recovery of the animals, the rats were divided into five groups (n = 5) and treated daily for six (6) weeks by oral gavage as follows: Sham (control) + vehicle, OVX + vehicle, OVX +50 mg/kg chrysin, OVX +100 mg/kg, chrysin, and OVX + 5 mg/kg alendronate (standard drug). Alendronate (ALE) was used as the standard drug. After the last treatment, rats were fasted for 16 h, anesthetized; blood samples were collected through cardiac puncture and then autopsied for samples collection.
Measurement of body weight change
Body weight changes of the animals were determined weekly from day zero and continued for the entire 6-week duration of the study. Mean body weight of each group was recorded, and percentage changes in weights of the animals were calculated using the following formula as previously described
23
:
Samples collection and preparation
Blood was collected from each rat after mild anesthesia and allowed to clot for 30 min at room temperature and then centrifuged at 3000 r/min for 15 min; serum was carefully collected and stored at −200C to avoid contamination and loss of bioactivity until assayed for ALP activity, estradiol, RANKL, and osteocalcin levels. The right femur from each rat was excised, adherent tissues cleaned off, rinsed with deionized water, weighed, and dried in an oven and then dry weight was recorded prior to ashing for measurement of calcium and phosphorus contents. The uterus from each rat was carefully excised by dissection, adherent fat tissues cleaned off, and its weight was measured and recorded.
Measurement of serum estradiol, ALP activity, osteocalcin, and RANKL levels in OVX rats
The concentrations of estradiol were determined in serum samples using rat estradiol (E2) ELISA kits (Shanghai Coon Koon Biotech Co. Ltd., China) according to the manufacturer’s instruction. Bone-specific alkaline phosphatase (BALP) activity in serum samples was determined according to manufacturer’s instructions. The serum osteocalcin level was measured using rat osteocalcin ELISA kits (Shanghai Coon Koon Biotech Co. Ltd China) following the manufacturer’s protocols. The serum RANKL level was measured using rat RANKL ELISA kits (Shanghai Coon Koon Biotech Co. Ltd., China) according to the manufacturer’s protocols.
Measurement of femur bone anthropometry parameters in OVX rats
The length and weight (wet and dry) of each excised right femur was measured using a digital Vernier caliper and electronic weighing balance, respectively. The femur samples were dried in an oven at 100°C for 2 h, and the dry weights were recorded and expressed as g/100 g of body weight. The weights of empty silica crucibles were determined, and the weight of silica crucibles containing dried femur samples were also measured and then transferred to a muffle furnace for ashing at 550°C for 12 h. The crucible containing the femur ash was taken out and weighed. The weight of the empty silica crucible was subtracted from the weight of crucible containing the femur to determine the actual weight of the ash.
Digestion and measurement of femur bone ash calcium levels in OVX rats
The atomic absorption spectrophotometric method was used to determine the level of calcium in the femur bone. Each femur ash residue was dissolved in 10 mL of triple acids (nitric acid: perchloroacetic acid: sulfuric acid) in the ratio 3:2:1 and heated until it became colorless. The colorless solution was then transferred to 100-mL volumetric flasks, and volume was made to 50 mL with triple distilled water. Thereafter, 1 mL was taken and diluted to 10-mL volume with triple distilled water and used to determine femur bone ash’s calcium level using atomic absorption spectrophotometer at 422.7 nM. 24 Triplicate determinations were made for each sample, and the results were expressed as mg/g of the femur bone.
Measurement of femur bone ash phosphorous level in OVX rats
The femur bone ash’s phosphorous level was determined by the vanadomolybdophosphoric acid colorimetric method. The digested femur samples (500 μL) were pipetted into a 25-mL volumetric flask, and 5 mL of developing reagent (mixture of ammonium molybdate, ammonium metavanadate, and nitric acid) was added to it. The resulting mixture was diluted with distilled water, shaken, and allowed to develop color for 30 min. Thereafter, the absorbance of each sample was determined at 430 nM using colorimeter (SpectronicUnicamHeyios) in triplicate. Total phosphorous concentrations were recorded and expressed as mg/g of the femur bone.
Comparative molecular docking of chrysin and β-estradiol to estrogen receptors
Protein–ligand docking study was conducted based on the crystal structure of human estrogen receptors alpha (ERα PDB ID: 5UFW) and beta (ERβ PDB ID: 2NV7), downloaded from the protein databank (www.rcsb.org/pdb). The 3D structure of chrysin was obtained from the PubChem database (PubChem ID: 5281607), while the structure of reference for endogenous estrogen (17β-estradiol) was drawn using ChemDraw Ultra v12.0.2 and the file was converted to PDB format. As standard control, 17β estradiol was docked with each receptor to compare docking energies with chrysin. Prior to docking, structures were prepared by removing all solvent molecules, co-crystallized ligands, and optimized to simulate physiological conditions using Chimera version 1.1. Different orientations of the ligands were searched and ranked based on their energy scores, and the most favorable binding interactions were determined using PatchDock server (www.bioinfo3d.cs.tau.il/PatchDock) as previously described. 25 The interaction was set as protein–small ligand, and 4.0 was selected as the clustering root mean square deviation (RMSD) of atomic position. This RMSD value aids selection of the most stable conformations by comparing between different ligands poses. The interaction of the docked complex was studied visually using Discovery Studio 2017 R2 Client (v17.2.0.16349).
Statistical analysis
Data analysis was performed using GraphPad Prism version 5.01 (GraphPad Software Inc., San Diego, CA, USA). One-way analysis of variance (ANOVA) was used, and data were expressed as mean ± SD (n = 5). Dunnett’s post-hoc multiple comparison test was used to determine the statistical difference among the various treatment groups for all parameters, and p < 0.05 was considered statistically significant.
Results
Effect of chrysin on body weight
Effect of chrysin on body weight gain in OVX rats.

Values are expressed as initial and final mean ± SD (n = 5). Body weight gain of various treatment groups (g) and expressed in percentage (%). ### Significant (p < 0.001) when compared to the sham group; ∗p < 0.05, ∗∗p < 0.01, ∗∗∗p < 0.001 Significant in comparison to the untreated OVX group. The treatments were carried out for 6 weeks.
Notes: BW: mean body weight; OVX: ovariectomized; CHR: chrysin; ALE: alendronate.
Effect of chrysin on uterine weight and estradiol level in OVX rats
The data obtained demonstrated significant (p < 0.001) reduction in uterine weight and serum estradiol concentration in OVX rats compared to the sham-operated group (Figure 1(a) and (b)). However, administration of 50 mg/kg and 100 mg/kg chrysin significantly (p < 0.01) reduced uterine weight loss and improved (p < 0.05) serum estradiol level, particularly the 100 mg/kg chrysin dose, compared to the untreated OVX group. Effect of chrysin on (a) uterine weight and (b) serum estrogen levels in ovariectomized rats. Values are expressed as means ± SD (n = 5). ### significant (p < 0.001) when compared to the sham group; ∗p < 0.05, ∗∗p < 0.01 significant in comparison to the untreated OVX group.
Effect of chrysin on serum BALP activity, osteocalcin and RANKL level in OVX rats
Ovariectomy indeed altered bone-remodeling markers as reflected by significant (p < 0.001) in bone-specific alkaline phosphatase (BALP) activity, osteocalcin, and RANKL levels in OVX rats compared to sham-operated rats (Figure 2(a)–(c)). This is an indication of rapid bone remodeling. Interestingly, treatment of chrysin to OVX rats revealed significant (p < 0.05) reduction in all these bone-remodeling parameters in a dose-dependent relation in comparison to untreated OVX rats (Figure 2(a)–(c)). These data further augment the beneficial effect of chrysin against bone loss induced by ovariectomy by prevention of rapid bone remodeling and bone resorption through inhibition of RANKL expression. Effect of chrysin on (a) serum bone alkaline phosphatase activity, (b) osteocalcin levels, and (c) receptor activator of nuclear factor-κB ligand levels in ovariectomized rats. Values are expressed as means ± SD (n = 5). ### significant (p < 0.001) when compared to the sham group; ∗p < 0.05, ∗∗p < 0.01 significant in comparison to the untreated OVX group.
Effect of chrysin on femur bone anthropometric parameters in OVX rats
The femur length did not differ significantly (p > 0.05) in all the experimental rats (Figure 3(a)–(d)), although femur ash weight, wet weight, and dry weight were significantly (p < 0.001) decreased in OVX rats compared to the sham group. Conversely, these altered anthropometric parameters were significantly (p < 0.05) restored in a dose-dependent manner when OVX rats were treated with different doses of chrysin (Figure 3(a)–(d)). This is an indication of potential mitigation effect of chrysin against bone loss in OVX rats. Effect of chrysin on femur bone anthropometric parameters in ovariectomized rats. (a) femur length, (b) femur ash weight, (c) femur wet weight, and d) femur dry weight. Values are expressed as means ± SD (n = 5). ### significant (p < 0.001) when compared to the sham group; ∗p < 0.05, ∗∗p < 0.01, ∗∗∗p < 0.001 significant in comparison to the untreated OVX group.
Effect of chrysin on femur bone ash calcium and phosphorous level
The femur bone ash’s calcium and phosphorous contents were significantly (p < 0.001) decreased in OVX rats in comparison to the sham-operated group (Figure 4(a) and (b)). However, treatment of OVX rats with 100 mg/kg of chrysin dose significantly (p < 0.05) prevented the depletion of bone ash calcium and phosphorus levels compared to untreated OVX rats (Figure 4(a) and (b)). This also indicated that chrysin could prevent bone loss via prevention of demineralization in the femur bone of OVX rats. Effect of chrysin on (a) femur bone ash calcium and (b) phosphorous level values are expressed as means ± SD (n = 5). ### significant (p < 0.001) when compared to the sham group; ∗p < 0.05, ∗∗p < 0.01, ∗∗∗p < 0.001 significant in comparison to the untreated OVX group.
Structural chemistry of chrysin and 17β-estradiol
The structural chemistry of chrysin revealed a ubiquitous fifteen-carbon flavone backbone consisting of two benzene rings (A and B) linked through a heterocyclic pyran ring C (Figure 5(a)). Also chrysin has a characteristic C2 to C3 double bond in a carbonyl-containing C-ring lacking oxygenation. Similarly, ring B is coplanar with ring A and C owing to conjugation. Whereas 17β-estradiol (estrogen) is an estrane steroid containing two hydroxyl groups at C3 and 17β position, a methyl group at C13 and three double bonds in ring A (Figure 5(b)). Structure of (a) chrysin downloaded from the PubChem database and (b) β-estradiol drawn using ChemDraw Ultra v12.0.2.
Molecular docking interactions at the binding site of estrogen receptors
About 600 ligands’ poses within the binding pocket of each estrogen receptor were generated for both chrysin and 17β-estradiol, and the best docked conformation of these compounds within the binding site of each receptor are presented in Figure 6(a)–(d)). Thus, both ligands (chrysin and 17β-estradiol) occupy similar positions on the binding site of α and β estrogen receptors. The docked complexes were ranked based on geometric shape complementary score due to optimal fit with a wide interface area and less stearic clashes. Lowest energy-docked pose of (a) chrysin with ER-α, (b) β-estradiol with ER-α, (c) chrysin with ER- β, and (d) β-estradiol with ER-β. ER= estrogen receptor.
Binding energy of the docked complexes
Binding energy of chrysin and β-estradiol on estrogen receptors.

Bonding interactions between estrogen receptors and the ligands
Bonding interactions between amino acid residues on the binding site of both α and β estrogen receptors and the ligands (chrysin and β-estradiol) revealed non-conventional interactions involving the aromatic pi bond, hydrophobic side chain, and unfavorable clashes (Figure 7(a)–(d)). The amino acid residues on the binding pocket of ER-α is Glu353 forming a hydrogen bond with 5-OH of chrysin, whereas Leu346, 525, 428, 384, 349, Met388, 528, Arg394, Phe425, Ile424, and His524 are bonded through van der Waals forces (Figure 7(a)). Similarly, there was pi-sulfur contact between the aromatic π bond of chrysin and sulfur side chain of Met343. In addition, the hydrophobic side chain of Ala350, Leu387, 391, Met421 are in pi-alkyl contact with the aromatic π bond of chrysin, but the aromatic side chain of Phe404 was having π–π interaction with the aromatic ring of chrysin. However, Arg394 on the binding pocket of ER-α was hydrogen bonded to 3-OH of β-estradiol (Figure 7(b)). Leu428, 384, 349, Phe425, Gly521, Met421, 528, and 343 formed a van der Waals interaction, whereas alkyl side chains of Leu391, 387, 346, Met388, Ile424, and Ala350 350 are in π-alkyl contact with the aromatic π bond of β-estradiol. Leu525 was in an alkyl interaction with the methyl group (C18) of β-estradiol, whereas Phe404 was in π–π contact. Thus, there was an unfavorable clash (stearic hindrance) between Glu353 and His524 residues of ER-α and β-estradiol. Conversely, for ER-β binding, chrysin (Figure 7(c)) was bound via van der Waals forces with Met336, 340, 295, Leu301, 380, Glu305, Arg346, Phe356, 377, Ile376, 373, His475, and Gly472. Furthermore, alkyl side chains of Leu298, 343, 339, Ala302 form π-alkyl bond with the aromatic ring of chrysin even though there was stearic hindrance between Leu476 and chrysin. Glu305, Trp335, Thr299, Met336, 295, 340, Leu476, 380, 343, His475, Gly472, Ile376, and Arg346 were involved in the van der Waals interaction with β-estradiol, while alkyl side chains of Ala302, Phe356, 377, Ile373, Leu298, 339, 301, and Met340 form π-alkyl bonds with its aromatic ring (Figure 7(d)). Bonding interactions and amino acids around the active site of ER-α with (a) chrysin, (b) β-estradiol, (c) ER-β chrysin (d) β-estradiol. ER= estrogen receptor.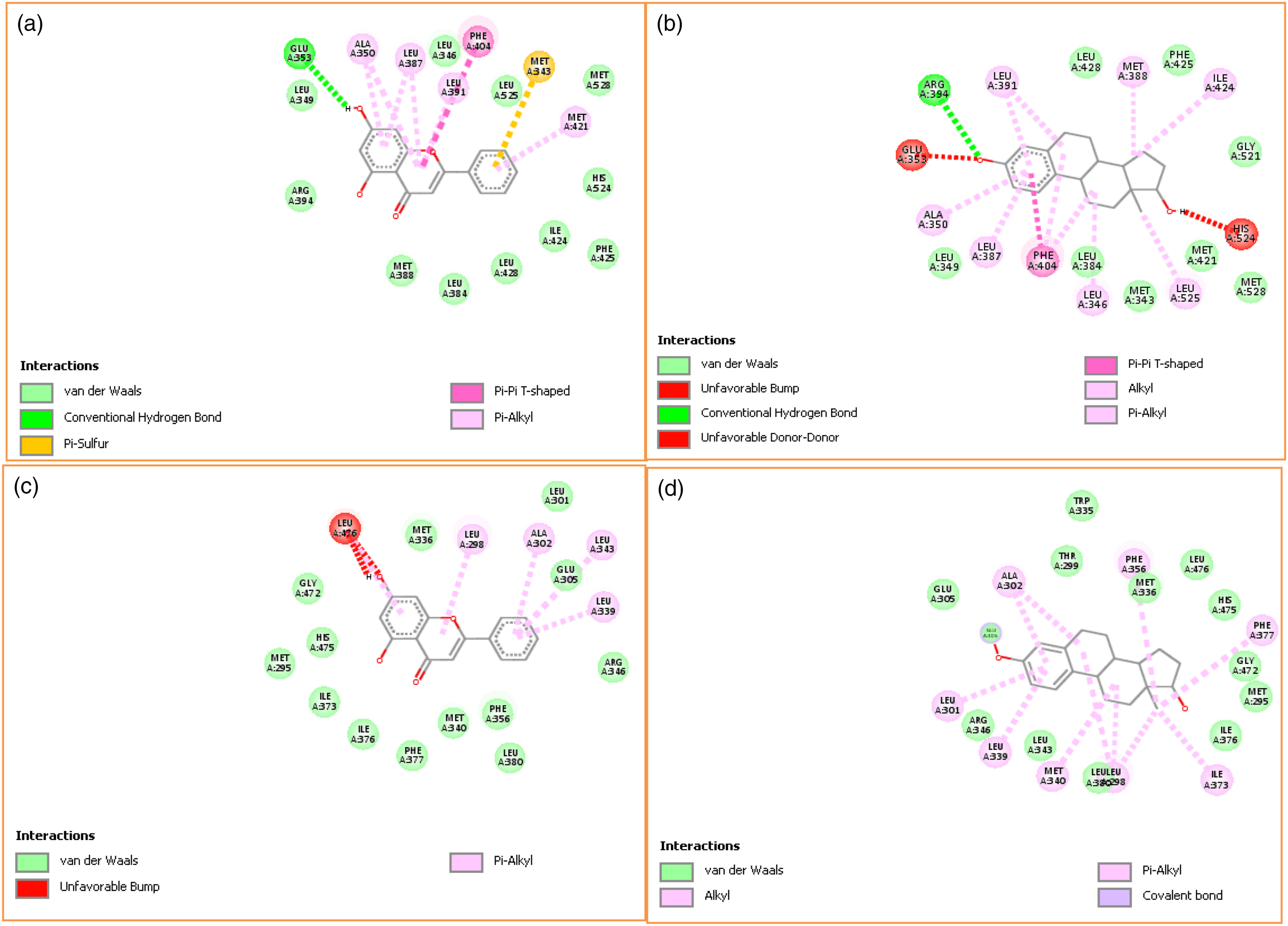
Discussion
Ovariectomized rats have been reported to mimic the physiological features of osteoporosis in postmenopausal women such as weight gain, decrease in serum estrogen level, increased oxidative stress, rapid bone turnover, and subsequently low bone mineral density.26,27 This is because estrogen is directly involved in energy metabolism and its receptors are also expressed in visceral and subcutaneous adipocytes. 28 Chrysin belong to the GRAS (generally regarded as safe) group of compounds and a previous report on toxicological evaluation of chrysin in rats revealed its LD50 as 4350 mg/kg. 29 In addition, another study involving volunteers that received oral doses of chrysin ranging from 300 to 625 mg does not reveal any toxicity. 30 The present study demonstrated excessive weight gain in OVX rats when compared with sham-operated rats. This study is consistent with previous reports that demonstrated body weight gain, fat redistribution, and marked decrease of estrogen levels in menopausal women.31,32 Interestingly, treatment of chrysin to OVX rats significantly suppressed the weight gain in a dose-dependent manner. A previous study also indicated the anti-obesity effect of chrysin via inhibition of pancreatic lipase (PL) activity and reduction of adiposity index and triacylglycerol levels in the oral fat tolerance test. 33 In addition, high incidence of bone loss in menopausal women is directly linked to the key role played by endogenous estrogen in skeletal development and bone homeostasis because deficiency of estrogen leads to a negative shift in bone-remodeling equilibrium.34,35 Estrogen is mainly synthesized by ovaries during reproductive and premenopausal period and has been implicated in uterine growth and development.36–38 The present study revealed drastic reduction in uterine weight and estradiol level in OVX rats in contrast to sham-operated rats. Conversely, chrysin markedly ameliorated uterine weight loss and improve serum estradiol level in OVX rats. Thus, our data demonstrated that chrysin might possess phytoestrogen-like activity and could be beneficial against bone loss in OVX rats. A previous study reported high responsiveness of uterus to estradiol level.39,40 In addition, several studies reported direct linkage between consumption of diet rich in phytoestrogens and low incidence of estrogen deficiency–related disease in elderly women. 41 For instance, herbal extracts (isoflavones) administered to OVX rats showed significant increase in estradiol levels. 42 Moreover, anthropometric parameters were significantly decreased in OVX rats compared to sham-operated rats, except the femur length which did not differ in all the experimental groups. In contrast to the sham group, femur ash calcium and phosphorus levels were substantially reduced as shown in a previous study which reported that ovariectomy prevents intestinal calcium re-absorption and induced rapid bone demineralization.43,44 However, administration of chrysin to OVX rats restored these anthropometry parameters and improved femur bone ash calcium and phosphorous levels in a dose-dependent manner. Biochemical markers of bone remodeling are effective for diagnosis and monitoring of osteoporosis severity. For instance, bone-specific alkaline phosphatase (BALP) is involved in the calcification of bone matrix, while osteocalcin plays a vital role in bone mineralization due to its high affinity for calcium.45,46 The coupling reaction between the RANK receptor expressed by osteoclasts and RANK ligand secreted by osteoblasts enhances the maturation and activity of osteoclast cells thus, increasing bone resorption.47–49 Excessive bone resorption in postmenopausal osteoporosis is characterized by elevated serum ALP activity, osteocalcin, and RANKL levels in the estrogen-deficient rat model.50–52 Interestingly, chrysin significantly suppressed the increased of BALP activity, osteocalcin, and RANKL levels in OVX rats when compared to untreated OVX rats. In a previous study, polysaccharides from Morinda officinalis reduced deterioration of trabecular microarchitecture and excessive expression of biochemical bone turnover markers in OVX rats. 53 These data indicated the beneficial effect of chrysin against excessive bone remodeling and inhibition of bone-resorption induced by ovariectomy. The extent of binding of the ligand to its target protein is characterized by the binding constant (Ka) used interchangeably with the inhibition constant (Ki), an indicator of strength of interaction that enhances binding affinity. 54 Molecular docking analysis revealed the presence of carbonyl carbon and methyl group in chrysin and β-estradiol, respectively, which could possibly enhance and strengthen the interactions between chrysin and estrogen receptors. A previous study demonstrated chrysin binds to the active site of SAM-dependent methyl transferase with strong hydrophobic and hydrogen-bonding interactions. 55 The present study showed that chrysin docked strongly to ER-β than ER-α unlike β-estradiol which docked strongly to both ERs. This implies that both ERs confer affinity to β-estradiol–binding locus only at slightly differential degrees due to differential expression profiles of the ERs across tissues and cell types. 56 In addition, somewhat similar amino acid residues on the binding pocket of each receptor were involved in the binding of chrysin as well as β-estradiol. Also, other studies have shown that metabolic function of both estrogen receptors are equally distributed between cortical and trabecular bone and also phytochemicals in herbal supplements can bind to human estrogen receptors where they may exhibit selective estrogen receptor modulation.57,58
Conclusion
In conclusion, this study demonstrated that chrysin exhibits potential anti-osteoporotic effect in OVX rats through improvement of bone mineral contents and reduction of excessive alteration of bone-remodeling markers. Comparative molecular docking analysis revealed that chrysin could enhance the sensitivity and function of estrogen receptors. Altogether, this study suggests that chrysin possesses phytoestrogen-like activity and could be beneficial against postmenopausal osteoporosis.
Footnotes
Acknowledgments
The authors are grateful to Department of Biochemistry and Veterinary Teaching Hospital, Faculty of Veterinary Medicine, Ahmadu Bello University, Zaria, Nigeria, for providing facilities and consumables to carry out this research work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.